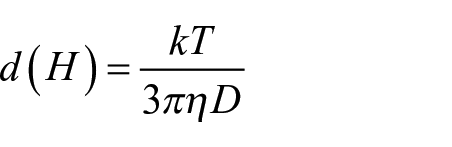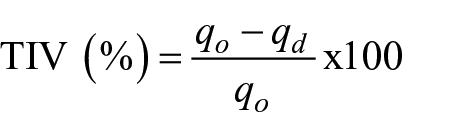Abstract
The concept of leveraging the optical properties of cerium oxide into viscose fibers was demonstrated in this study. The process of dispersing cerium oxide particles uniformly into viscose dope prior to viscose fiber spinning was studied in detail. Cerium oxide powder was dispersed in the chosen media using mechanical treatments such as ball milling and ultrasonication. These experiments were carried out at two extreme pH values to replicate the viscose spinning bath (acidic pH 5) and viscose dope (alkaline pH 13). The dispersion was characterized for particle size and surface charge properties. A combination of the mechanical treatments synergistically reduced the cerium oxide particle size in acidic and alkaline pH from 1300 to 290 nm and from 1040 to 280 nm, respectively. Three surfactants of different ionic natures were evaluated for stabilizing the cerium oxide dispersions. The effects on the particle size of cerium oxide in two dispersion environments, that is, aqueous and polymeric media, were studied through conventional as well as novel visual characterization techniques. The phosphate ether-based anionic surfactant was found to significantly reduce the dispersed particle size and effectively stabilize the dispersion better, which was validated through a pressure buildup monitoring system during viscose fiber spinning. The surfactant-stabilized cerium oxide dispersion was used to get functional infrared reflecting viscose fibers. To characterize this effect, a direct visual evaluation was done through an infrared camera which indicated a significant improvement of infrared reflectivity and a 2.5–3°C surface temperature buildup of the cerium-oxide viscose fibers in comparison to regular viscose fibers. The infrared reflective property of cerium oxide imparts a thermal insulation effect which was found to increase to a thermal insulating value of 90 in the case of cerium oxide viscose fibers compared with a thermal insulating value of 79 in regular viscose fibers, as measured by the KESF Thermolabo II instrument. This study can very well be extended to many fiber-making processes where an understanding of interfacial interactions of additives with cellulose is critical to impart functionality to viscose fibers.
Introduction
Viscose fibers are derived sustainably from natural sources. They are wet spun and made from cellulose derived from wood pulp. The conventional viscose fiber manufacturing process involves four critical operations, namely, steeping, which is the purification and mercerization step, aging, xanthation, and spinning. Dissolution of pulp from wood takes place in two stages: (1) alkali cellulose is made by mixing wood pulp with caustic soda and (2) formation of cellulose xanthate on reaction with carbon disulfide. 1 The sodium cellulose xanthate is then dissolved in a weak alkaline caustic soda solution to form a spinnable solution called viscose dope. The viscose dope is then extruded through a spinneret with fine holes into a sulfuric acid spin-bath. Aftertreatment processes like washing, desulphurization, bleaching, and soft finish application are carried out to finally complete viscose fiber manufacturing.2–4 Over the years viscose fibers have become popular due to unique properties such as good absorbency, high softness, good dyeability, and drapability. Hitherto, their applications are predominantly in apparel and medical hygiene products. However, the possibility of using them in functional textiles and technical applications is immense if the fibers can be engineered as required. This has given rise to increased research and development (R&D) efforts to develop functional viscose fibers. The conventional approach to imparting functionalities to viscose in the fabric stage is seen to have limitations, mostly in terms of durability of the imparted effect. Incorporation of additives into the polymer solution prior to spinning results in longer lasting effects. The key challenge during this incorporation process is getting a uniform dispersion of additives in the highly alkaline viscose solution that does not affect the fiber manufacturing process adversely. This study describes the addition of cerium oxide (CeO2) particles into viscose dope to obtain functional infrared (IR) reflecting viscose fibers.
CeO2 is an oxide of the rare-earth metal cerium belonging to the lanthanide series having two oxidation states: +3 and +4. 5 It is an abundantly available rare-earth metal used prolifically in various engineering and biological applications. 6 It exhibits catalytic properties7,8 and is widely used in solid-oxide fuel cells. 9 Recently it has gained popularity in biomedical applications due to its antioxidant properties. 10 CeO2 nanopowder is a good ultraviolet absorbent. 11 It is also known to enhance the far infrared properties of mineral compounds.12,13 This study provides new insights into understanding and improving the dispersion of CeO2 additive to impart thermal insulating properties to viscose fiber attributed to the infrared reflecting behavior of CeO2.
Dispersion is a unit operation widely used in paints, dyestuffs, fibers and textiles, pharmaceuticals, food, coatings, cosmetics, and detergent industries. It can be defined as the process in which aggregates and agglomerates of a particulate matter are broken down into finer particles via mechanical action. Milling, high-shear mixing, and ultrasonication are commonly used for particle size reduction. On milling, the mechanical force applied on the material by the grinding media breaks up the agglomerates. Subsequently, the finer particles are stabilized with the help of surfactants into a stable dispersion. 14
The particle size of CeO2 was reduced via ball milling (BM), ultrasonication (US), and a combination of both. The effect of anionic, cationic, and non-ionic surfactants on the dispersion stability in acidic and alkaline pH was investigated. In addition, dispersion of CeO2 in viscose solution was studied via high-magnification images. Fibers were spun from viscose dope containing CeO2 particles and processability was assessed via monitoring the pressure buildup during spinning. The key findings from the study can be extended to studying different additive–cellulose dope systems. This would help bridge the gap in understanding interfacial interactions and facilitate development of functional viscose fibers.
Materials and Methods
CeO2 powder (purity > 99%) was purchased from Saral Chem at Mumbai, India and used in experiments as received. Surfactants used for this study were a fatty alcohol ethoxylate-based non-ionic surfactant of HLB 11.8 (Unitop Chemicals Pvt. Ltd), a polyoxyethylene alkylphenol ether phosphate-based anionic surfactant of HLB 12 (Unitop Chemicals Pvt. Ltd) and a cationic quaternary ammonium-based surfactant, that is, cetyl trimethyl ammonium bromide (CTAB) (purity > 98%) (Sigma Aldrich) of HLB 10. The three surfactants were used as stabilizing agents for uniform dispersion of CeO2 particles. The pH adjustments were done using stock solution of sodium hydroxide (NaOH), purchased from Merck, in de-ionized water, by EXL water purification, at 5% (wt/vol) concentration.
BM of CeO2 Dry Powder
The CeO2 dry powder was finely ground using an XQM Ball Mill (Changsha Tianchuang Powder Technology Pvt. Ltd., China). The planetary ball mill has four jars that are arranged on a sun wheel. The jars contain SS balls, 4.75 and 7.3 mm in diameter, for grinding; 200 g of CeO2 powder was filled in a jar containing 578.8 g SS balls and ball milled at a speed of 200 rpm for runtime intervals of 30, 60, and 90 min to reduce the particle sizes.
US of CeO2 Particles in an Aqueous Dispersion
The CeO2 powder (as such or ball milled) was added to de-ionized water at 0.5% (wt/vol) to prepare the acidic (pH 5) dispersion and at 0.5% (wt/vol) NaOH stock solution to prepare high pH (pH 13) dispersion. It was stirred for 15 min on a magnetic stirrer. The dispersion was subsequently ultrasonicated using probe type ultrasonic processor (UPS 500). The frequency was set at 30 kHz and the power at 500 W with a runtime of 60 min (9 s on–9 s off cycle) throughout all the experiments. US of untreated and ball milled (denoted as BMUS henceforth in the paper) CeO2 dry powder samples with and without the use of surfactant was carried out.
Stabilization of CeO2 Particles With Surfactant in an Aqueous Dispersion
An amount of 0.015 molar solutions of each surfactant, that is, phosphate ether-based surfactant (Unitop RE-610), alcohol ethoxylate-based surfactant (Unitop Hydol-2370), and cetyl trimethyl ammonium bromide (CTAB), was prepared in de-ionized water. This solution was stirred for 15 min using a magnetic stirrer. CeO2 particle dispersions of concentration 0.5% (wt/vol) in iacidic/alkaline media were added to the surfactant solution dropwise under continuous stirring. These dispersions were then assessed through particle size and zeta potential measurements.
Particle Size Measurement
The particle size measurements of CeO2 dispersions were carried out on a Malvern Zetasizer Nano ZS. In this instrument the particle size in a dispersion system is measured by the dynamic light scattering (DLS) principle. DLS measures the random movement of the particles known as the Brownian motion and this relates to the size of the particles using the Stokes–Einstein equation:
where D is the translational diffusion coefficient, d(H) is the hydrodynamic diameter, k is Boltzmann’s constant, T is the absolute temperature, and η is the viscosity.
CeO2 dispersions prepared at 0.5% (wt/vol) in acidic and alkaline medias were measured for particle size by this instrument.
BET Surface Area
The surface area (SBET) of the powder sample was estimated by BET (Brunauer–Emmett–Teller) method. The SBET was determined by nitrogen adsorption at −196°C by the surface area analyzer Nova 1000e (Quantachrome). The sample was degassed under vacuum at 1000°C for 2 h before measurement.
Zeta Potential Measurement
The magnitude of the electrostatic or charge repulsion/attraction between particles is one of the fundamental parameters known to affect stability. This was measured by the Malvern Zeta Sizer Nano ZS. The dispersion samples at 0.5% (wt/vol) in acidic and alkali were used for measuring the zeta potential values.
Characterization of CeO2 Dispersion in a Cellulosic Medium
The agglomeration behavior of the CeO2 dispersions prepared by combination of BM and US were visually characterized by an in-house-built high-magnification microscope–camera system, a Nikon Model Eclipse E200. The dispersion was added to viscose dope, having a viscosity of the order of 300–500 Pa.s and a pH of 13, and mixed thoroughly. The weighed amount of this dispersion in viscose dope was then placed on glass slides under the microscope and images were captured with the help of the software Imagingsource.
Spinning of Viscose Fibers From Dope Dispersed With CeO2
To understand the impact of the quality of CeO2 dispersion on the fiber manufacturing process, ball milled, and ultrasonicated CeO2 dispersion (with and without surfactant) were added to viscose dope. As seen in Figure 1, CeO2 dispersion was added to the viscose dope and mixed thoroughly. Viscose fibers were spun from the dope on a lab scale spinning line (600 g/h throughput) using a spinneret of hole diameter 60 µm. Monitoring of back pressure buildup with time, using an inline pressure transducer, during fiber spinning was carried out Any agglomeration of particles would lead to choking of spinneret holes, which in turn would result in higher pressure buildup with time. Control experiments without addition of CeO2 in viscose dope were also carried out.
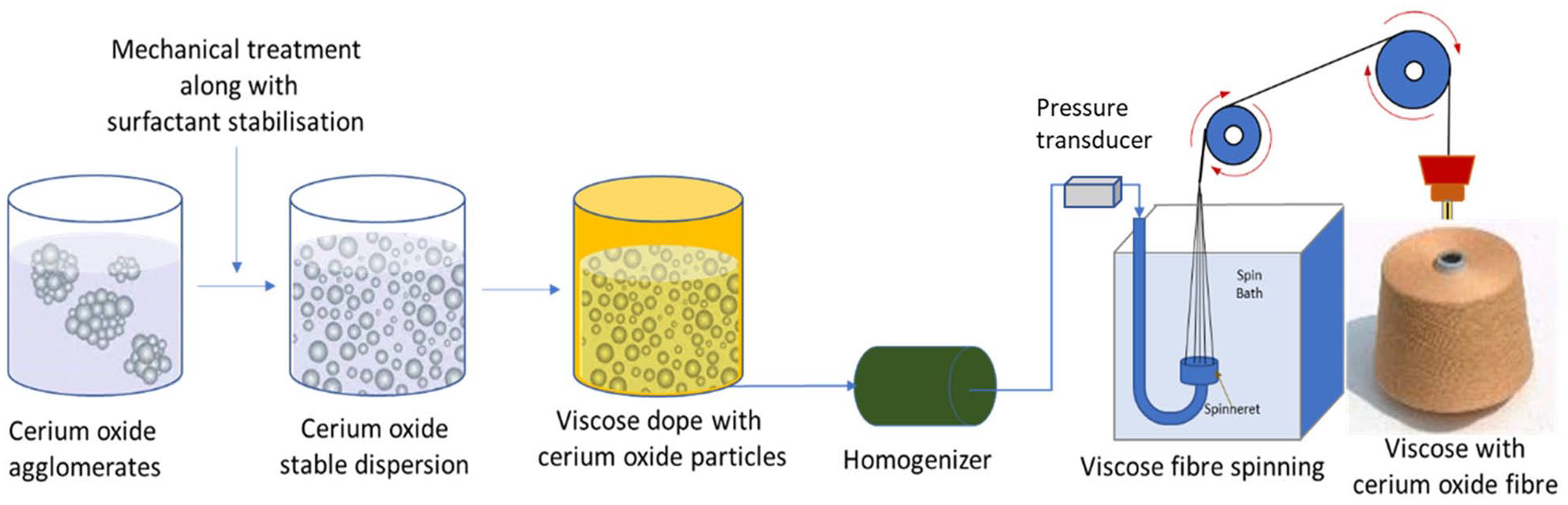
Schematic representation of cerium oxide dispersion preparation and its addition into viscose dope to spin viscose with cerium oxide fibers.
Tensile Properties of Dope Dispersed CeO2-Viscose Fibers
To evaluate the initial fiber performance properties of dope dispersed CeO2 viscose fibers, their tensile properties (e.g. denier, tenacity, and elongation) were measured on Vibroscop-400 and Vibrodyn-400 instruments (Lenzing Instruments). Fibers were conditioned at 25 ± 2°C and 65% relative humidity (RH) was set for 24 h before testing.
Characterization of Functional CeO2-Viscose Fibers
The thermal insulation characteristics of functional CeO2 viscose fibers were measured by IR radiation reflectivity and thermal insulating value (TIV). To measure the IR reflectivity the viscose fibers were made into non-woven sheets of size 16 cm × 16 cm and GSM 585 by a hand sheet former machine.
The fiber sheets were then placed under an IR lamp and exposed to IR energy. The extent of IR reflectivity was visually captured through an IR camera (Microepsilon thermoIMAGER TIM 40, with optical resolution of 382 × 288 pixels and a spectral range of 8–14 µm). The IR camera converted the IR data into an electronic image that showed the apparent surface temperature of the viscose fiber sheet being measured.
The second method was in terms of measuring the TIV which was measured by the KESF Thermolabo II instrument. The heated device was maintained at 31°C and the room temperature was maintained at 21°C. The heat flux was determined by measuring the energy required to maintain a set temperature in a heated device when it is covered by the viscose fiber sheet. The energy required when the heated device is not covered by the fiber sheet was also noted. TIV was calculated using the following formula:
where qo is the heat flux of the uncovered heated device and qd is the heat flux of the covered heated device by the viscose fiber sheet.
Results and Discussion
Particle Size Studies of CeO2
The average particle size and particle size distribution (PSD) are critical parameters for any material processing and final product quality as they influence the surface area and porosity. Particle size controls the surface area available for interactions between an aqueous solution and solid surface. This parameter plays a key role in understanding the physical stability, rate of settling, and agglomeration tendencies. When the particles are lower in size and uniformly dispersed, the settling tendencies will be slow with lower tendencies of agglomeration.
Table 1 shows the effect of mechanical treatments and pH on the particle size of CeO2 dispersions and Table 2 shows its distribution data. It is observed from the tables that the average particle size is lower in high pH medium, whereas it is higher in the low pH medium. The effect of independent mechanical treatments like US and BM on the particle size is contrasting in both extreme pH media (Table 1). US and BM reduce the particle size at pH 13 but increase it at pH 5. This phenomenon can be explained by the two different stable oxidation states of ceria. Although the most common oxidation state of ceria is Ce3+, it also shows its presence in the Ce4+state. The CeO2 particles are known to show adsorption and desorption behaviors due to the interfacial interactions in the dispersed phase. 15 This happens mainly due to the adsorption of co-ions or counter-ions. The interchange of the two oxidation states of ceria, namely, Ce3+ to Ce4+ in dispersed form leads to a change in the interfacial tensions and agglomeration tendencies. In the high pH medium, the oxidation of Ce3+ to Ce4+ is preferred, whereas in the lower pH media, the reduction of Ce is higher. 16 Due to the higher oxidation state of CeO2 at high pH medium, there are greater chances of electrostatic repulsion. This reduces the average particle size at pH 13 as shown in Table 1. As also reported in the literature the particle size reduction is primarily due to the electrostatic repulsion tendencies at high pH, whereas the agglomeration takes place due to the coulombic and van der Waal forces of attraction. 17
Particle size values of the mechanically treated cerium oxide dispersion at two extreme pHs.
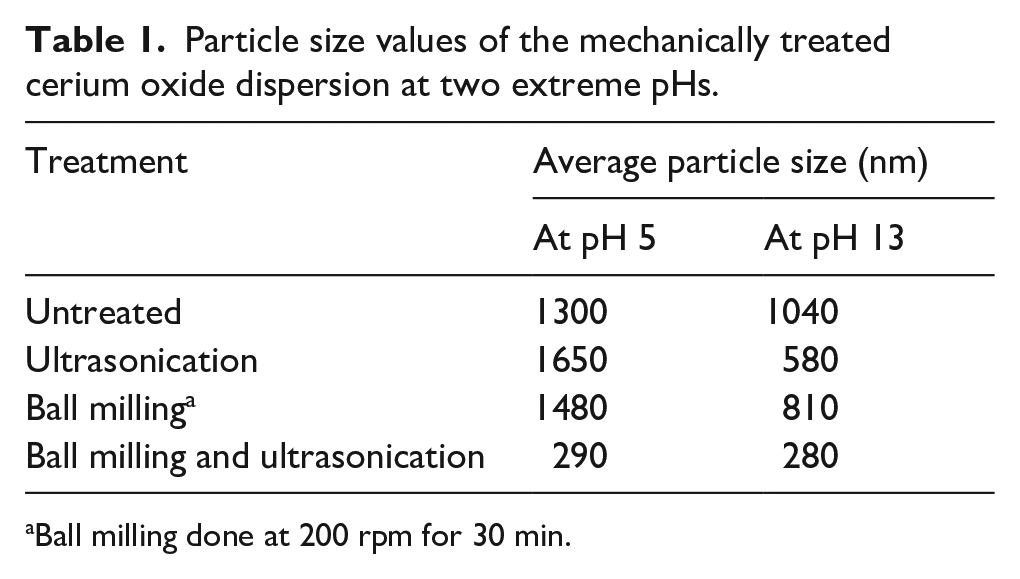
Ball milling done at 200 rpm for 30 min.
Particle size distribution of the mechanically treated cerium oxide dispersion at two extreme pH.
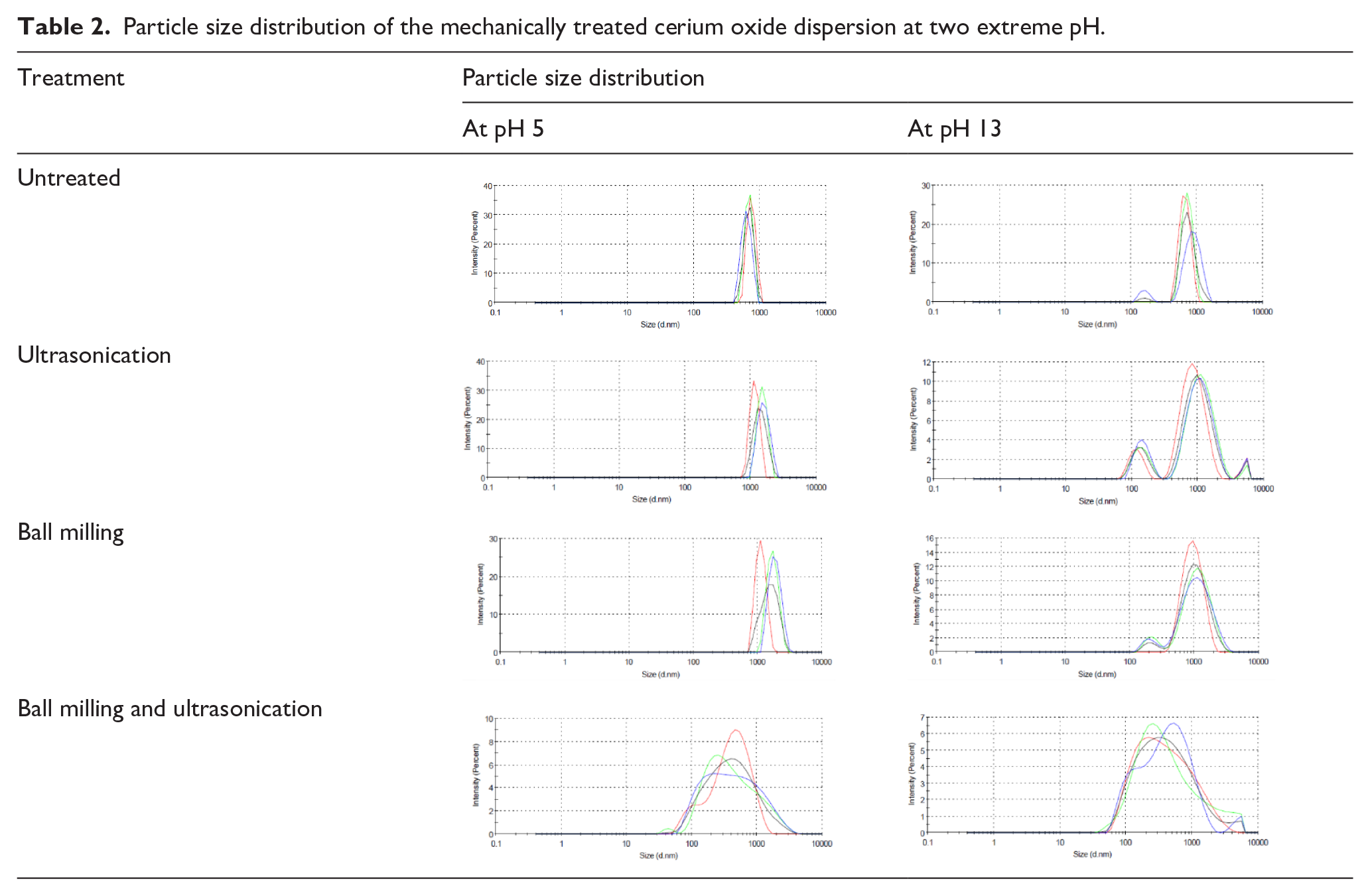
It is also observed from the tables that the mechanical treatments affect the particle size and its distribution. The increase in average particle size from 1300 to 1650 nm post US and 1480 nm post BM at low pH can be explained by the re-agglomeration tendencies of CeO2 and lower repulsion tendencies of the particles. The US treatment after dry BM broke the soft agglomerates and reduced the particle size further (Table 1). We can see the synergistic effect of BM and US which reduced the average particle size to 290 and 280 nm at low and high pH, respectively. Ball-milling ground the particles to some extent and US broke down the soft agglomerates to get a lower average particle size in aqueous media.
The PSD (Table 2) is broader as we try to decrease the particle size beyond certain limits. This is primarily due to the reagglomeration tendencies of the particles. This observation has also been reported in the literature where reagglomeration of particles broadens the PSD. 18
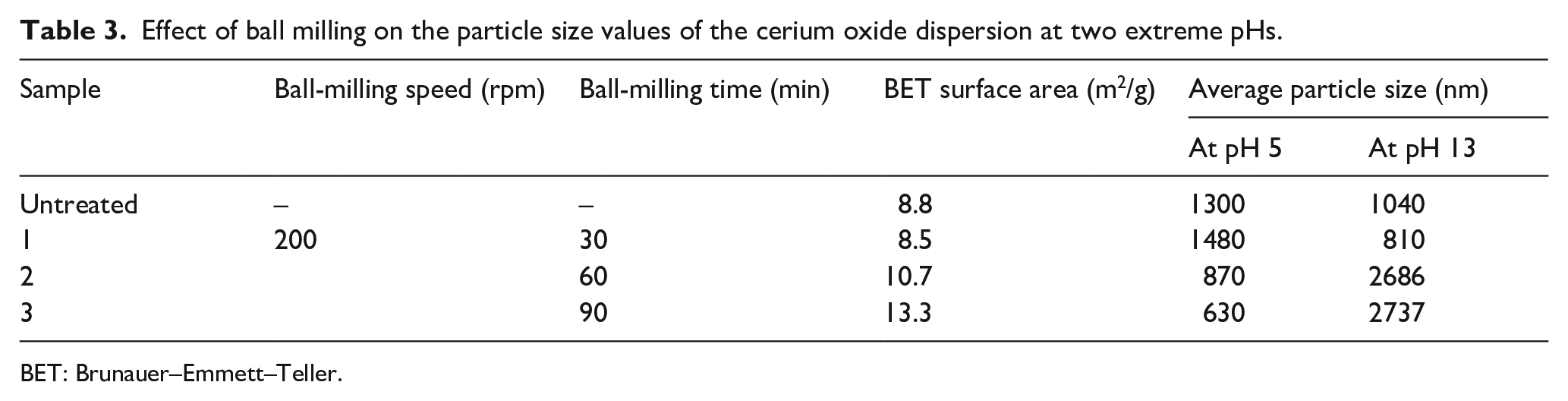
Effect of BM Conditions
To understand the effect of BM effectively, the CeO2 particles were ball milled for three different time intervals, that is, 30, 60, and 90 min at a speed of 200 rpm, as seen in Table 3. As the BM runtime was increased from 30 to 90 min the BET surface area increased. Although the increase was not very significant it indicates a trend of increased pore size and surface area. The values of BET surface area indicate that the particles are in the sub-micron range and need further grinding to get nanosized particles. In the aqueous dispersion at low pH the BM runtime interval of 30 min causes reagglomeration and increases the particle size. As the runtime is increased, there is a reduction in particle size of up to 630 nm. At high pH, the particles due to electrostatic repulsion reduce to 810 nm, but further milling causes an increase in particle size. This is mainly due to reduced BM efficiencies at higher time intervals. The particle size reduction rates decrease, which in turn reduces the electrostatic repulsion drastically, causing reagglomeration of ceria particles.
Effect of ball milling on the particle size values of the cerium oxide dispersion at two extreme pHs.
BET: Brunauer–Emmett–Teller.
This observation has been reconfirmed by the literature and prior studies show that the particle size reduction rates decrease with decreasing particle size. 19 This is attributed to a reduction in the inertial and hydrodynamic forces and an increase of particle aggregate strength with reduction in particle size.18,20,21 In practice, this means that the ball-milling efficiency is reduced drastically at particle sizes smaller than 500 nm. If a further size reduction is required, then the time for milling needs to be increased but at a risk of contamination and unwanted phase changes.22,23
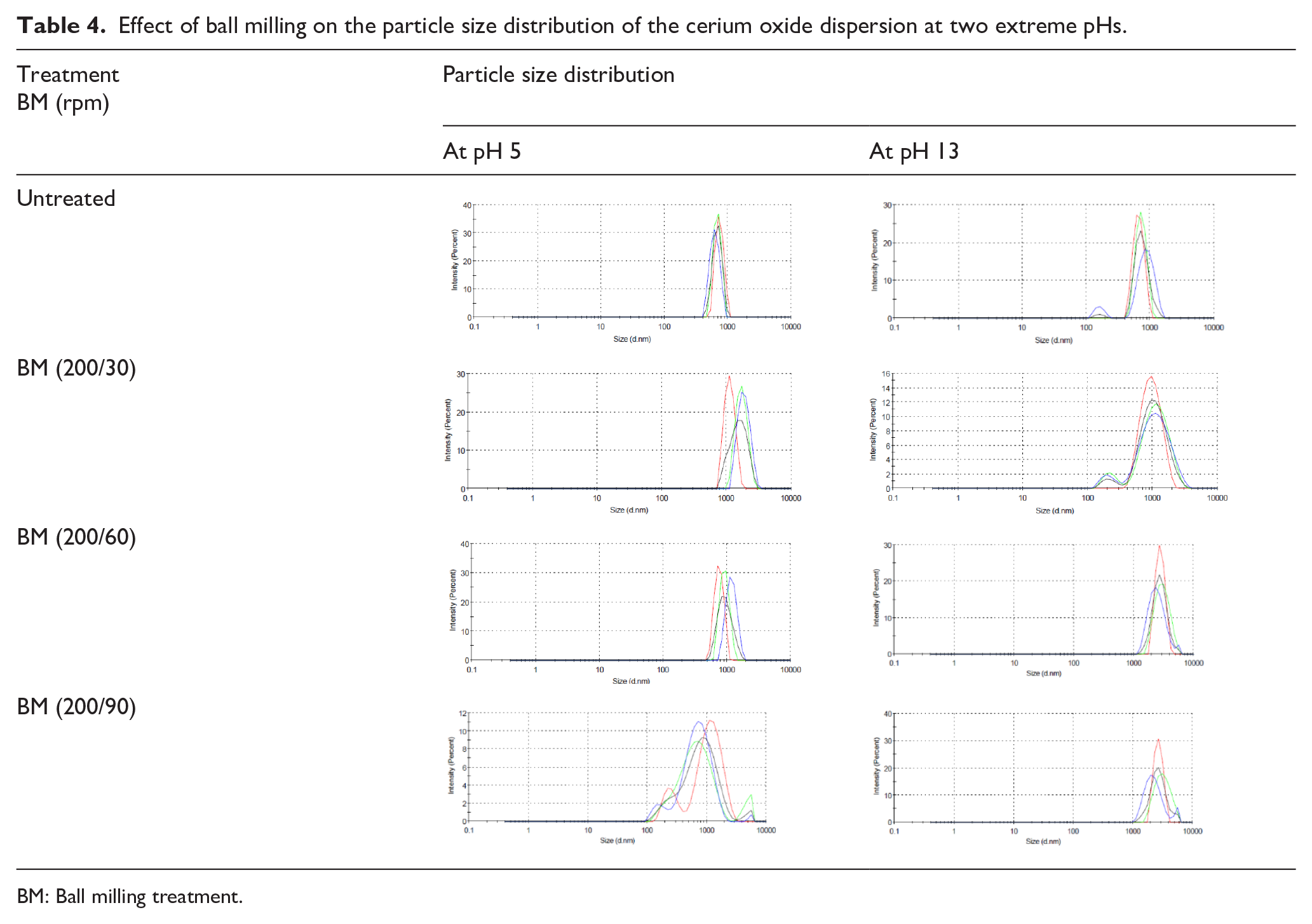
The PSD data in Table 4 show the trend of BM efficiencies reducing at higher runtime intervals. At an rpm of 200, the PSD is broader at a runtime interval of 90 min at both pHs. This is due to reagglomeration tendencies and lower BM efficiencies at higher runtime intervals. These observations are in accordance with several literature reports on BM efficiencies reducing with reducing particle size. Due to this limitation of BM, it was decided to evaluate the synergistic effect of combining BM and US treatments restricted to a BM speed of 200 rpm for a runtime of 30 min.
Effect of ball milling on the particle size distribution of the cerium oxide dispersion at two extreme pHs.
BM: Ball milling treatment.
Surfactant Stabilization of Ceria Particles
Surfactants affect the stability of a dispersion by changing the electrical repulsive energy or steric repulsive energy between particles by adsorption. We have described a technique to stabilize the dispersion via adsorbing surfactants on the CeO2 particle surfaces. Three categories of surfactants, as shown in Figure 2, were used to stabilize the CeO2 dispersion.
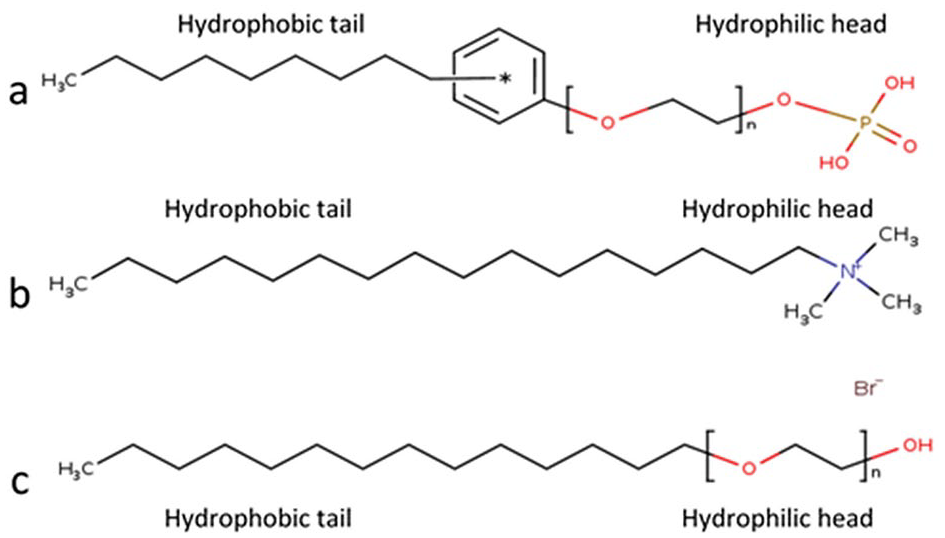
Structure of surfactants: (a) anionic-based polyoxyethylene alkylphenol ether phosphate, (b) cationic-based cetyl trimethyl ammonium bromide (CTAB), and (c) non-ionic-based polyoxyethylene alkyl ether.
As seen in Tables 5 and 6 the phosphate-based anionic surfactant was effective in getting the lowest particle sizes of CeO2 post BM and US in both pH media. The alcohol ethoxylate-based non-ionic surfactant does not stabilize the dispersion effectively and reduce the particle size due to lower adsorption of the surfactant on ceria particles. In the case of CTAB surfactant the interfacial interactions between the surfactant and ceria surface do not provide the stabilization and separation of CeO2 particles. In all the three cases the lowest particle sizes have been obtained by combining the BM and US treatments. This validates our earlier observation. Interparticle repulsions between colloidal particles are necessary to overcome the van der Waals attractive forces of interaction. In a dispersion that is stable, the maximum attractive forces between particles should be just enough to allow any other agitation to easily break all particle–particle bonds. 18 The significant effect of pH remains in the case where non-ionic surfactant is used for stabilization, where relatively lower particle sizes are obtained at a higher pH medium.
Stabilization of ceria dispersion at different mechanical treated conditions in two extreme pH media.
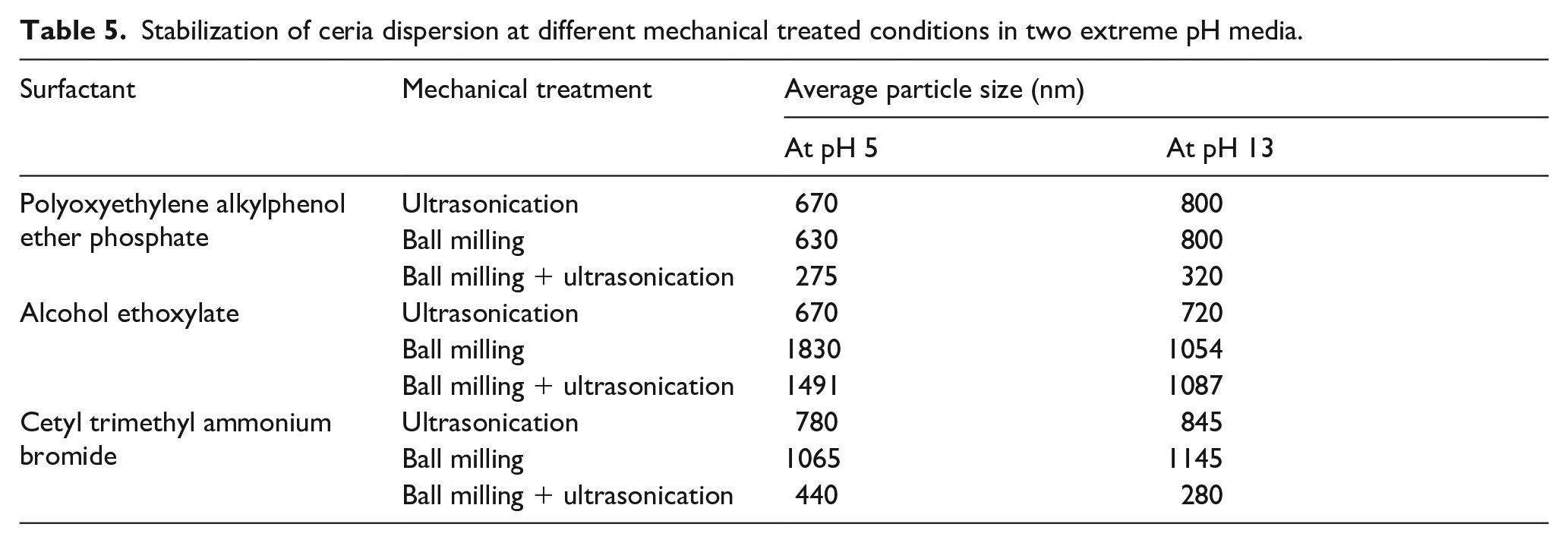
Particle size distribution data of the stabilization of ceria dispersion at different mechanical treated conditions in two extreme pH media.
In the case of metal oxide particles, the high surface density of hydroxyl groups is generally targeted as the adsorption sites for surfactants or the polymer dispersants. High cover-up of the surface is commonly achieved through electrostatic interactions (e.g. phosphates and carboxylates), acid–base reactions (e.g. carboxylic acids), or hydrogen-bonding structures (e.g. ethers, alcohols and acrylamides).24,25 Multiple anchor groups of the surfactants provide a strong interaction and increase coverage on the particles. 26 The phosphate ether-based anionic surfactant anchors itself on the CeO2 particle, keeping them well separated, reducing the average particle size. Typically, it has been reported that anionic, predominantly carboxylic acid-based polyelectrolytes have a stronger interaction with surfaces of metal oxides. The ceria particles have been effectively stabilized by polyacrylic acid (PAA) at pH values between pH 6 and pH 9, the reason being that the negatively charged carboxylate groups on the PAA interact very well with the weakly positively charged hydroxyl groups on the ceria surface.27,28 Another study reports the efficient dispersion in water by combining polyether chains with a strongly adsorbing head group, for example, phosphonated polyethlene glycol (PEG) for dispersions of CeO2. 27 Thus, the stabilization of CeO2 dispersion with anionic surfactant is in accordance with that reported in the literature.
Zeta Potential Studies of Ceria
Colloidal particles under the effect of electric field exhibit electrokinetic effects. These effects usually have a great consequence for the electrical charges on the surface of colloidal particles. The magnitude of the net surface charge is called the zeta potential. The zeta potential depends on several factors such as the velocity of the moving particle under the influence of electric field as well as the viscosity of the dispersion media. Smaller particles tend to move faster in a less viscous media in contrast to larger ones, and hence undergo faster diffusion and higher stability. Zeta potential measurements, therefore, are carried out to evaluate particle’s stability in a colloidal dispersion.
As seen in Table 7 the zeta potential values significantly increase with an increase in the pH. Also, the ball-milled, ultrasonicated samples lead to higher zeta potential values. The magnitude of the zeta potential indicates the degree of electrostatic repulsion between co-ions in a colloidal dispersion. Particles with high zeta potential values indicate stability and less susceptibility to agglomeration. This can be attributed to lower particle sizes as observed earlier in Table 1. Values higher than ±30 mV indicate higher particle–particle repulsion, indicating a stable dispersion. The effect of BM and US improved the stability of dispersion significantly as seen by the higher absolute zeta potential values at both pHs.
Zeta potential values of the mechanically treated cerium oxide dispersion under two extreme pH conditions.
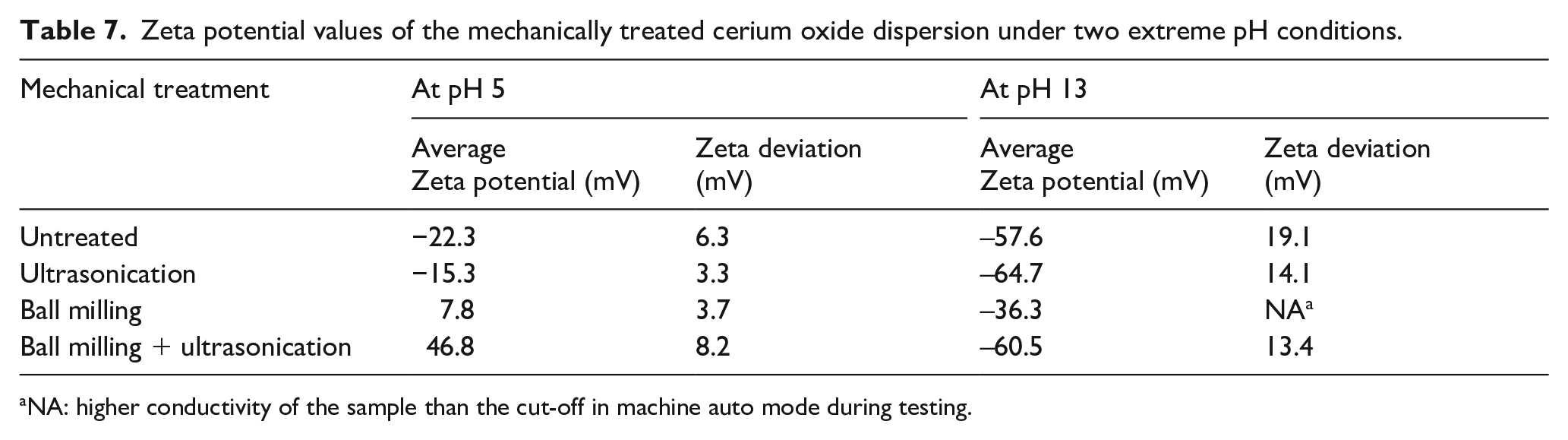
NA: higher conductivity of the sample than the cut-off in machine auto mode during testing.
The occurrence of more positive zeta potential at acidic pH implies columbic interaction between nanoceria and other ionic species within the solution, whereas negative zeta potential values at basic pH imply strong electrostatic repulsion, as also reported in the literature. 22
As seen in Table 8, the effect of BM alone gives lower average zeta potential values in general. This is an indication of the instability and lower particle–particle repulsion tendencies. The zeta potential values are relatively higher at a 60-min runtime interval compared with the other runtime intervals indicating higher stability, but as per the particle size characterization the particles are bigger and the distribution is broader. For surfactant stabilization experiments a BM speed of 200 rpm for a runtime of 30 min was considered.
Effect of ball milling on the zeta potential values of the cerium oxide dispersion at two extreme pH.
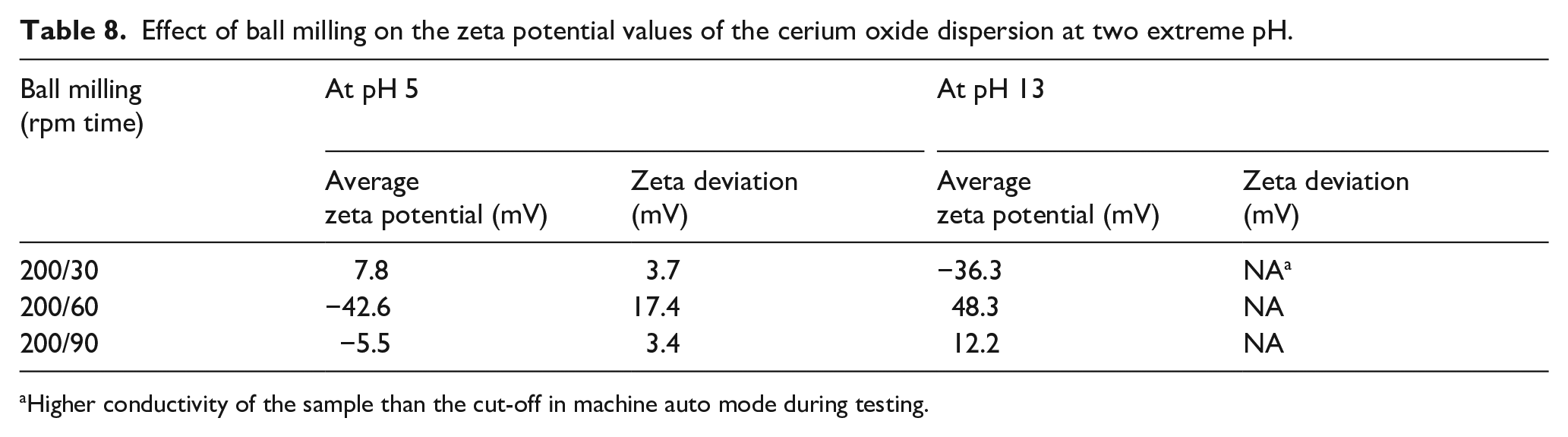
Higher conductivity of the sample than the cut-off in machine auto mode during testing.
As seen in Table 9, the high negative zeta potential values on phosphate ether surfactant stabilization indicate greater electrostatic repulsion due to the high adsorption of this surfactant on ceria particles. This is due to the effective interaction between negatively charged carboxylate groups and positively charged ceria surface. Alcohol ethoxylate-based surfactant does not reflect the stability of dispersion through zeta potential characterization due to its non-ionic nature. In the case of CTAB the zeta potential charges are positive in both the low and high pH media. The lower molecular weight of CTAB compared with phosphate ether surfactant may lead to lower steric repulsion tendencies. This is indicated by lower absolute values in the zeta potential in the case of CTAB.
Zeta potential values of the surfactant stabilized mechanically treated ceria dispersions.
CTAB: cetyl trimethyl ammonium bromide.
NA: higher conductivity of the sample than the cut-off in machine auto mode during testing.
Characterization of CeO2 Dispersion in Viscose Dope
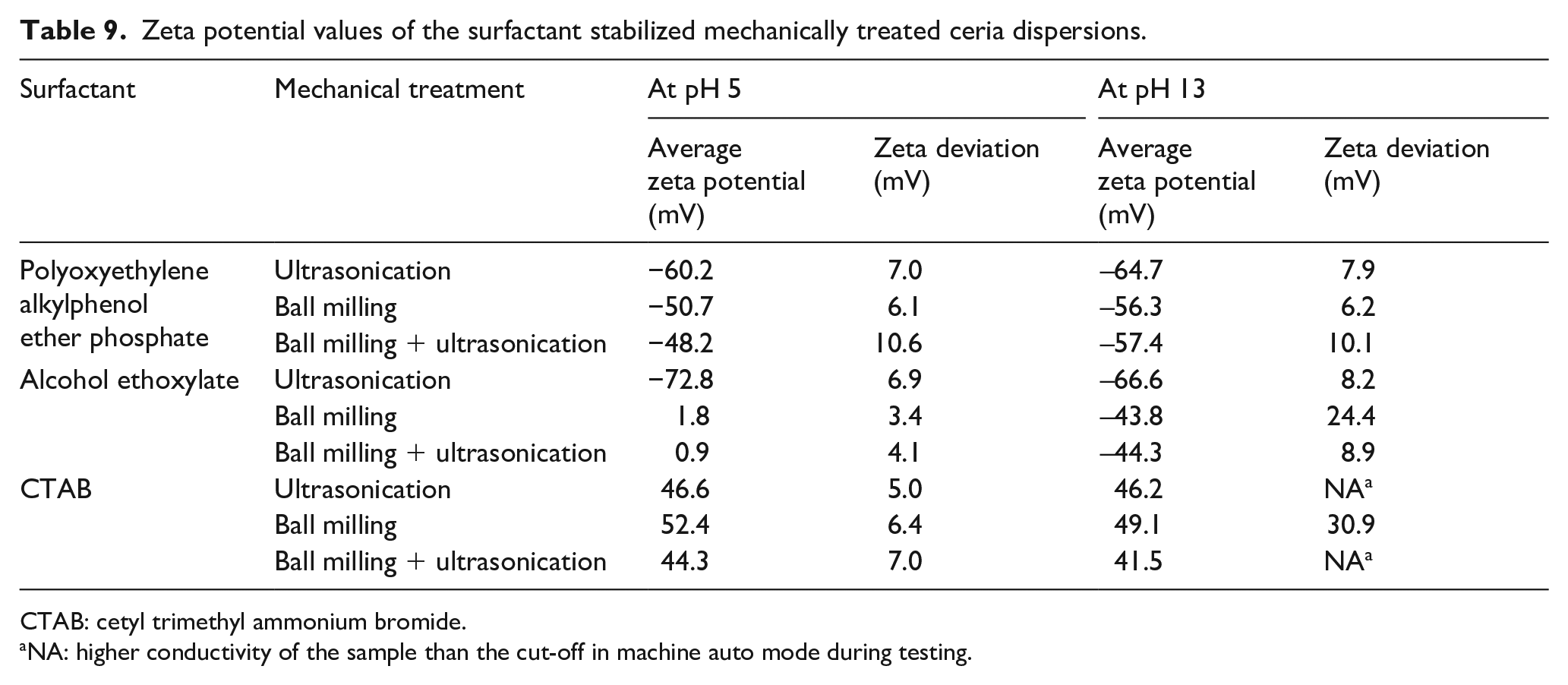
The behavior of ceria dispersions in high pH media gives an idea about its behavior in high pH conditions, which exist in viscose dope. But in the case of dope, along with high pH, the viscosity and cellulosic polymeric interactions also come into play. In order to understand this better, at the first level, visual characterization of the particle dispersions was carried out through a high-magnification microscope–camera system. The CeO2 particles in the dispersion form were added to the viscose dope to see its behavior in the viscous polymeric medium. Figure 3 shows the high-magnification images of viscose dope containing untreated and mechanically treated CeO2 dispersions.
Microscopic images of cerium oxide particle dispersion in dope through an in-house-developed high magnification camera system. (a) Untreated cerium oxide, (b) ultrasonicated cerium oxide, (c) ball milled cerium oxide, and (d) ball milled and ultrasonicated.
These images qualitatively indicate that a combination of BM and US of CeO2 resulted in a finer and more uniform dispersion in viscose dope. This technique provides a better understanding regarding the dispersion behavior in actual process conditions, which are otherwise difficult to characterize in the viscous polymeric medium.
Effect of CeO2 Dispersion With Surfactant on Spinning Performance and Fiber Properties of Viscose
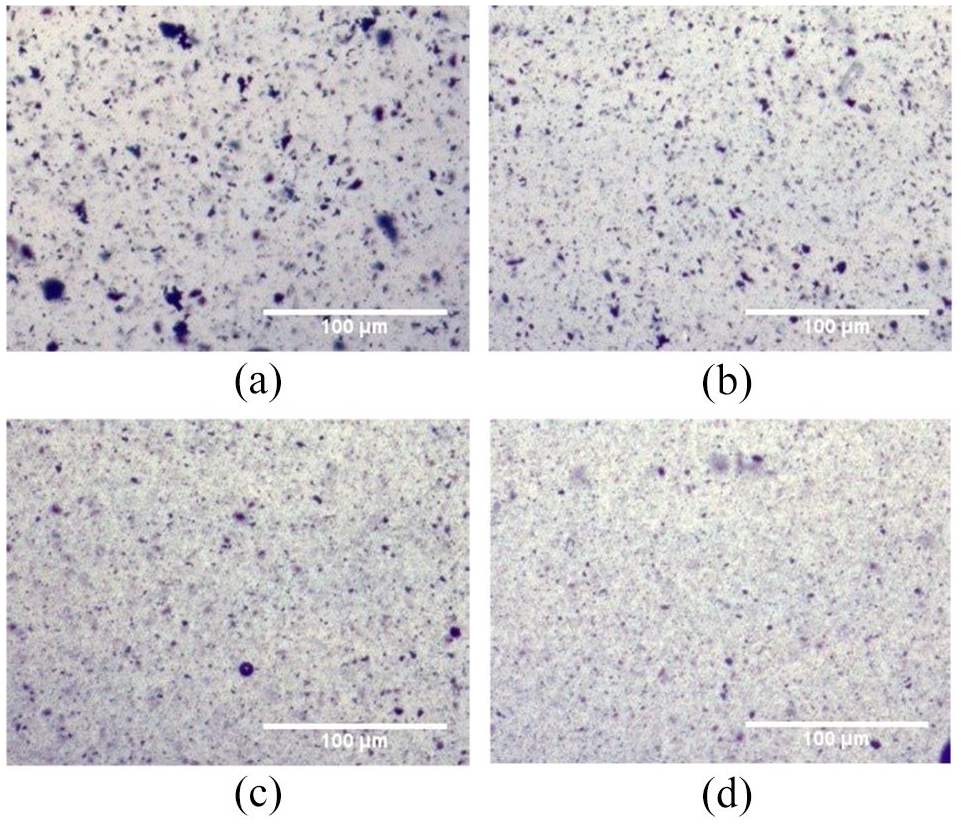
Once it was established that the phosphate ether-based surfactant reduced the average particle size in aqueous condition under both the pH media, its effect on spinning performance of viscose fibers was ascertained. An indirect method of monitoring the back pressure buildup during real-time spinning was adopted. The pressure buildup was plotted against the duration of spinning to get an idea of the pressure rise trend (Figure 4).
Real-time spinning performance of pressure buildup over time of viscose dope and BMUS CeO2 dispersion, with and without phosphate ether surfactant.
As seen in Figure 4 the viscose dope without any CeO2 particles does not increase the pressure buildup over time, but the BMUS CeO2 particles dispersed into the viscose dope increased the pressure by ~30% in a matter of 30 min. However, when the phosphate ether surfactant was added into the BMUS CeO2 dispersion and mixed into viscose dope, it reduced the pressure buildup and the pressure increased by only ~15% in 30 min. Although particle size studies show a lower average particle size in both BMUS CeO2 dispersions with and without surfactant, the situation is different in a dynamic spinning system. Without the use of surfactant, the particles agglomerate with time, leading to faster choking of the filter at the outlet of the spinneret, leading to back pressure buildup. The extent of agglomeration was not as severe with the use of surfactant as it possibly kept the particles well separated from each other and delayed the choking of the spinneret, increasing productivity. The optimized mechanical conditions of CeO2 particles along with stabilization of dispersion with the surfactant can be effectively used to reduce the average particle size, lowering agglomeration, and hence improving processability during the spinning of viscose fibers.
As seen in Table 10, the fiber tensile properties have been measured to validate the particle size and processability. There is an increase in denier and a drop in tenacity of the BMUS CeO2 viscose fiber due to agglomeration of CeO2 particles in dope, resulting in spinneret choking. The choking leads to fiber faults and lower fiber strength. In the case of BMUS CeO2–surfactant–viscose the denier is similar to viscose fiber without CeO2 and the reduction in tenacity is also lower. This is indicative of the improvement in processability on stabilization of CeO2 particles in dope with phosphate ether-based surfactant.
Fiber physical properties.

BMUS: ball milling and ultrasonication.
Functionality of CeO2-Viscose Fiber
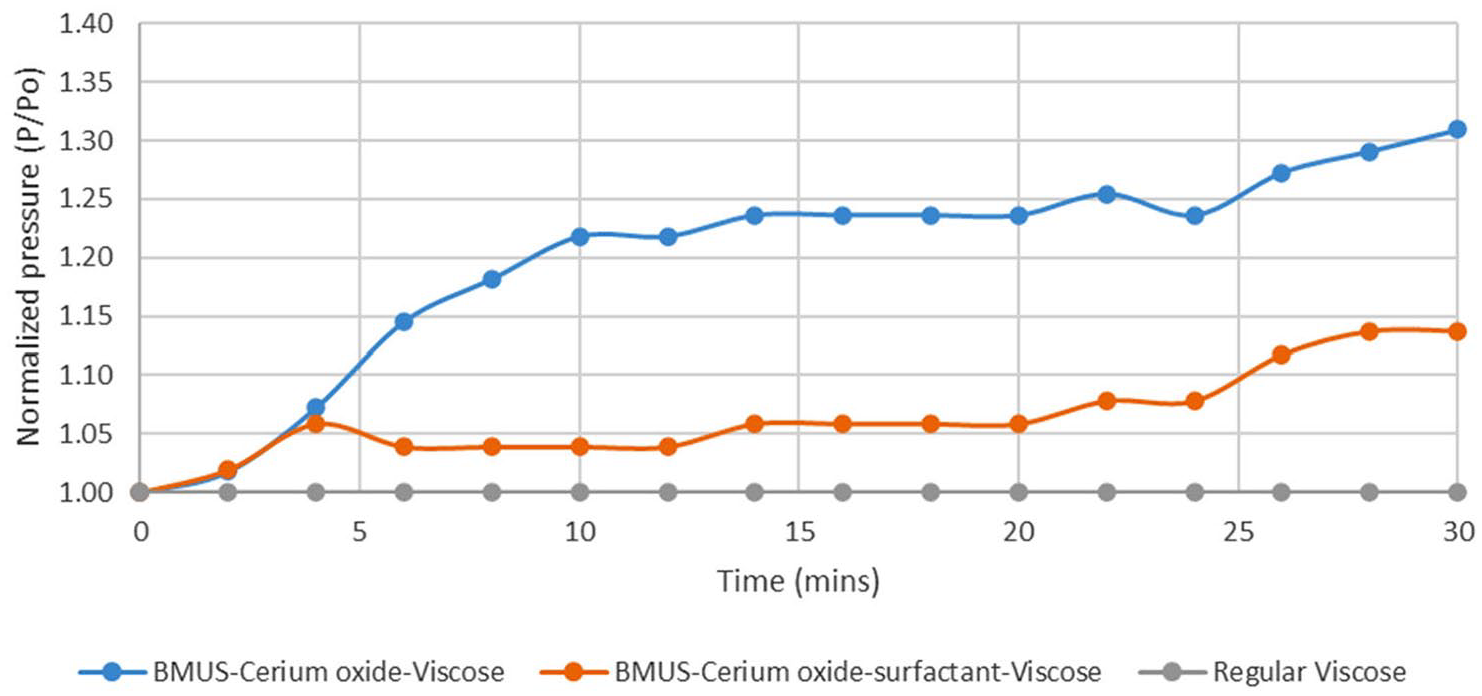
The CeO2-viscose fiber and regular viscose fiber sheets were exposed to IR energy emitted by the IR lamp. The thermal IR camera detected the thermal energy or heat emitted by the fiber sheets into electronic signals. The electronic images were color mapped to indicate the surface temperature increase of the viscose fiber sheets.
The viscose fiber non-woven sheets when exposed to an IR heating source partially reflected, absorbed, and transmitted the IR radiation based on their characteristic properties. The viscose fiber sheet incorporated with CeO2 reflected a higher amount of radiation, as indicated by the dark orange and red regions in Figure 5, indicating an increase in the surface temperature. The regular viscose fiber sheet without CeO2 did not reflect much IR radiation, as seen by the lighter yellow-colored regions, and showed a lower surface temperature. There was a significant temperature increase of CeO2 viscose fibers of almost 2.4°C. This clearly shows a significant surface temperature rise effect of CeO2 imparted to viscose fibers.
IR camera images of regular viscose fiber non-woven sheet without cerium oxide and with cerium oxide.
The TIV of regular viscose fiber and CeO2 viscose fiber was measured by the Kawabhata instrument and the readings obtained are as shown in Figure 6. As per the TIV formula, the lower the flux required to maintain the heating device at the set temperature, the higher the TIV of the fiber sheet covering the heated device. The CeO2 viscose fiber showed an incremental TIV value of 90% compared with the regular viscose fiber TIV value of 79%. This was due to the IR reflectivity of CeO2 thereby imparting the thermal insulating effect to CeO2-viscose fibers.

Thermal insulating values of regular viscose fiber and cerium oxide viscose fiber.
Conclusion
There is a significant effect of pH on the CeO2 dispersion stability. The particle interactions are mainly due to the electrostatic repulsion tendencies at high pH. There is a major effect of adsorption and desorption of ions of ceria in aqueous/polymeric medias on dispersion stability. The combination of first BM of the dry CeO2 powder followed by US of its dispersion reduced the CeO2 particle size significantly. The phosphate ether-based anionic surfactant was found to reduce the particle size by up to ~600 nm by the electrostatic and steric repulsion of the phosphate groups keeping the CeO2 particles well separated and thereby enhancing the stability of dispersion. The back pressure buildup reduced significantly when the surfactant was used, leading to better fiber tensile properties. The CeO2 viscose fibers reflected the IR radiation which increased the surface temperature by nearly 2.4°C compared with viscose fibers without CeO2. The CeO2 viscose fibers showed a significantly higher average TIV value of 90 compared with a value for regular viscose fibers of 79. Using this concept of higher IR reflectivity the functional CeO2 viscose fibers can be used for thermal insulating applications for winter protective clothing, quilts, and so on. The experiments demonstrated here have established the importance of studying CeO2 particle–particle interactions and the effect of pH and the environment, that is, aqueous or viscose polymer, on the dispersion. Functional viscose fibers can be made through addition of different additives after studying and optimizing their dispersion behavior prior to spinning. The insights from the study can be used for further evaluation of particle–medium interactions in wet spun functional fiber development and manufacturing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.