Abstract
A mixture of minerals consisting of calcite, dolomite, talc and quartz, known to have infrared (IR) reflecting properties, was mechanically treated and dispersed in aqueous medium and incorporated in viscose solution to be physically entrapped into the fibre structure. The process of incorporation of the mineral mixture in viscose dope involved steps of slurry making by suspending and grinding the particles in aqueous medium followed by mixing the slurry with the viscose dope, ready for spinning. The slurry preparation and grinding were carried out under different mechanical conditions, such as ball milling and ultrasonication. The evaluation of particle size of slurry was carried out under two pH conditions, first at the inherent pH (neutral pH 7) and at high pH (alkaline pH 13) equivalent to the pH of viscose dope. Furthermore, for stable slurry making, different surfactants were used. The phosphate ether-based anionic surfactant was found to effectively stabilize the dispersion better under the pH conditions used. The effect of stable slurry on good spinning was validated through an inline pressure gauge during viscose fibre spinning. To assess the IR reflecting property of viscose fibre, a direct visual evaluation was done through an IR camera, which indicated a significant increase of ~2°C surface temperature of the IR-Viscose Staple Fibre (VSF) in comparison with control VSF.
Keywords
Introduction
Viscose fibre is regenerated from sustainable natural sources of cellulose. The conventional viscose fibre manufacturing process involves operations such as steeping, aging, xanthation and spinning. Dissolution of pulp from wood takes place by mixing wood pulp with caustic soda and its reaction with carbon disulfide. 1 The sodium cellulose xanthate thus formed is then dissolved in a weak alkaline caustic soda solution to form viscose dope. The dope is then extruded through a spinneret with fine micron-sized holes into a highly acidic spin bath. After treatment, processes such as washing, desulphurization, bleaching and soft finish application are then carried out to complete viscose fibre manufacture.2–4 Viscose fibres are versatile fibres with similar comfort properties to natural fibres. They are popular due to properties such as good absorbency, drapability and softness. Because of these properties, their applications are predominantly in apparels and medical hygiene products. The manufacture of functional viscose fibres would open doors to more possibilities and applications in functional textiles. One of the ways of imparting functionality to viscose is addition of additives into the dope stage prior to spinning. The key challenge during this incorporation process is getting a uniform dispersion of additives in the highly alkaline viscose solution that does not affect fibre manufacturing adversely. This study describes the addition of mixed mineral particles into viscose dope to obtain infrared (IR) reflecting viscose fibres.
Application of mineral particles on textiles is used to impart multifunctional properties.5,6 Coating of specific minerals on textiles is one of the ways to impart specific properties, including protection from UV radiation, 7 soft feel, 8 flame resistance and flame retardancy, 9 antibacterial properties 10 and water and oil repellency. 11 The major challenge with using the methodology of coating inorganic mineral micron/nano particles is lack of compatibility, which has restricted their use in textiles. 6 Mineral particles such as silica and clay have been used for functionalization of melt-spun synthetic fibres to improve their thermal stability and UV blocking properties. 12 IR reflective properties of minerals or ceramic particles have promising applications, such as IR therapy and body thermoregulation.13–15 The reflected IR energy is absorbed by the molecule’s vibrational bonds into the human cells. This leads to the expansion of blood vessels, thereby improving blood circulation.13,15 Leveraging and regulating this IR emitting property of the functional textile surface can open doors to new opportunities and applications in sportswear and medical and even smart textiles. The IR emissivity of such mixed mineral compounds also leads to heat generation and warming effects in the textile product. 16
The process of dispersion is critical to many industrial applications such as cosmetics, pharmaceuticals, detergents, food and textiles. The dispersion process consists of breaking down particulate agglomerates and their stabilization. Milling or grinding and ultrasonication (US) are commonly used techniques for breaking down the agglomerates and particle size reduction. On grinding, the mechanical shear forces applied on the particles by the grinding media break up the agglomerates. Subsequently, the smaller sized particles are stabilized, that is, kept separate with the help of surfactants in aqueous dispersion. 17 The dispersion is then mixed homogenously into viscose solution and extruded through fine spinneret holes into an acidic spin bath.
In this study, experiments are carried out to incorporate a mixture of minerals into viscose fibres and challenges associated with it. The process of incorporation of the mineral mixture in viscose dope involved steps of slurry making by suspending and grinding the particles in aqueous medium followed by mixing the slurry with the viscose dope, ready for spinning. Particle size of mineral particles in the aqueous medium was reduced via ball milling (BM), US and a combination of both, that is, ball milling and ultrasonication (BMUS). The effect of different ionic surfactants on the dispersion stability under different solution pH was investigated to simulate the alkaline viscose (pH 13). Understanding surfactant adsorption on such mineral surfaces is critical as it affects the size, morphology, coating thickness, coating weight, surface roughness and so on. The dispersion uniformity of minerals improves significantly with the use of anionic and cationic surfactants compared with the ones without surfactant. 18 The surfactant adsorption behaviour19–21 and amount are decided based on the chemical composition of minerals, its surface charge properties, water chemistry (pH condition) and surfactant chemistry.22–25 The surface charge properties of minerals change based on dissociation of ions in aqueous medium. 26 The surfactants contribute to altering the interfacial tension, improving the wettability and foaming tendency.22,27 This enhances the surfactant adsorption on the particle surfaces and leads to particle–particle separation through electrostatic and steric interactions.
To understand the impact of type of surfactant on the particle dispersion in the dope stage, spinning was carried out in a laboratory-scale setup equipped with an inline pressure gauge. Agglomeration tendencies and the instability of particles in the dope, if any, would lead to significant choking of spinneret holes, resulting in increased pressure build-up during spinning with time.
This study on formulating a stable dispersion of mixed minerals and adding it uniformly to viscose dope can be applicable to different categories of additives to develop multifunctional viscose fibres.
Materials and Methods
The mixed mineral powder was purchased from Astrra Chemicals, Chennai, India, and used in the experiments as received. The mixed mineral composition varies widely, and a typical composition in the pre-mix was used in the study containing Calcite (∼50%), Talc (∼40%), Dolomite (∼5%) and Quartz (∼5%). The surfactants used in this study were a fatty alcohol ethoxylate-based non-ionic surfactant of HLB 11.8 (Unitop Chemicals Pvt. Ltd., Mumbai, India), a polyoxyethylene alkylphenol ether phosphate-based anionic surfactant of HLB 12 (Unitop Chemicals Pvt. Ltd., Mumbai, India) and a cationic quaternary ammonium-based surfactant, that is, cetyl trimethyl ammonium bromide (CTAB) (purity > 98%) (Sigma Aldrich,Bengaluru, India) of HLB 10. The three surfactants were used as stabilizing agents for uniform dispersion of mineral particles. The pH adjustments were done using stock solution of sodium hydroxide (NaOH), purchased from Merck, Mumbai, India in de-ionized water, by EXL water purification, of 5% (wt/vol) concentration.
Ball Milling of Mixture of Minerals Powder
The mixture of minerals was finely ground using XQM Ball Mill (Changsha Tianchuang Powder Technology Pvt. Ltd., China). The planetary ball mill has four jars, which are arranged on a sun wheel. About 200 g of mixed mineral powder was filled in a jar containing 578.8 g stainless steel balls of 4.75 mm diameter and 7.30 mm diameter. The ball mill was run at speeds of 200 rpm and 400 rpm for time intervals of 30, 60, 90, 120, 240 and 360 min.
Ultrasonication of Mixture of Minerals in Aqueous Dispersion
The mixture of minerals (as such or ball milled) was added to de-ionized water at 0.5% (wt/vol) to prepare the neutral pH (pH 7) dispersion and at 0.5% (wt/vol) NaOH stock solution to prepare high pH (pH 13) dispersion. It was stirred for 15 min on a magnetic stirrer. The dispersion was subsequently ultrasonicated using probe type Ultrasonic Processor (UPS 500). The machine set frequency was 30 kHz and the power was 500 W, with a run time of 60 min (9 s on–9 s off cycle) throughout all the experiments. US of untreated and ball milled mineral mixture (henceforth BMUS) with and without the use of surfactant was carried out.
Stabilization of Mineral Mixture Particles with Surfactant in Aqueous Dispersion
A 0.015-molar solution of each surfactant, that is, phosphate ether-based surfactant (PAP), alcohol ethoxylate-based surfactant (AE) and CTAB, was prepared in de-ionized water. This solution was stirred for 15 min using a magnetic stirrer. Mixtures of mineral dispersions of concentration 0.5% (wt/vol) in acidic/alkaline media were added to the surfactant solution drop wise under continuous stirring. These dispersions were then assessed through particle size and zeta potential measurements.
Particle Size Measurement
The particle size measurements of mineral mixture dispersions were carried out on Malvern Zetasizer Nano ZS. In this instrument, the particle size in a dispersion system is measured by Dynamic Light Scattering (DLS) principle. DLS measures the random movement of the particles known as the Brownian motion and this relates to the size of the particles by using Stokes–Einstein equation:
where D is translational diffusion coefficient, d(H) is hydrodynamic diameter, k is Boltzmann’s constant, T is absolute temperature and η is viscosity.
Mineral mixture dispersions prepared at 0.5% (wt/vol) in acidic and alkaline media were measured for particle size by this instrument.
BET Surface Area
The surface area (SBET) of the mixed mineral powder was estimated using the BET (Brunauer–Emmett–Teller) method. The SBET was determined by nitrogen adsorption at −196 °C by the surface area analyzer Nova 1000e (Quantachrome, Aditya Birla facility in Mumbai). The sample was degassed under vacuum at 1000 °C for 2 h before the measurement.
Zeta Potential Measurement
The magnitude of the electrostatic or charge repulsion/attraction between particles is one of the fundamental parameters known to affect stability. This was measured by the Malvern Zeta Sizer Nano ZS. The dispersions at 0.5% (wt/vol) in acidic and alkali were used for measuring zeta potential values.
Spinning of Viscose Fibres from Dope Dispersed with Mineral Mixture
Viscose solution was made and used at an in-house facility. To understand the impact of mineral mixture dispersion on the fibre manufacturing process, ball milled and ultrasonicated mineral mixture dispersion (with and without surfactant) were added to viscose solution. As seen in Figure 1, the mineral mixture dispersion was added into viscose solution and mixed thoroughly. Viscose fibres were spun from the dope on a laboratory-scale spinning line (600 g/h throughput) using a spinneret of hole diameter 60 µm. Monitoring of back pressure build-up with time, using an inline pressure transducer, during fibre spinning was carried out. Any agglomeration of particles would lead to the choking of spinneret holes, which in turn would result in higher pressure build-up with time. Control Viscose Staple Fibre (VSF) spinning, without addition of mineral mixture in viscose solution, was also carried out for comparison with IR-VSF.
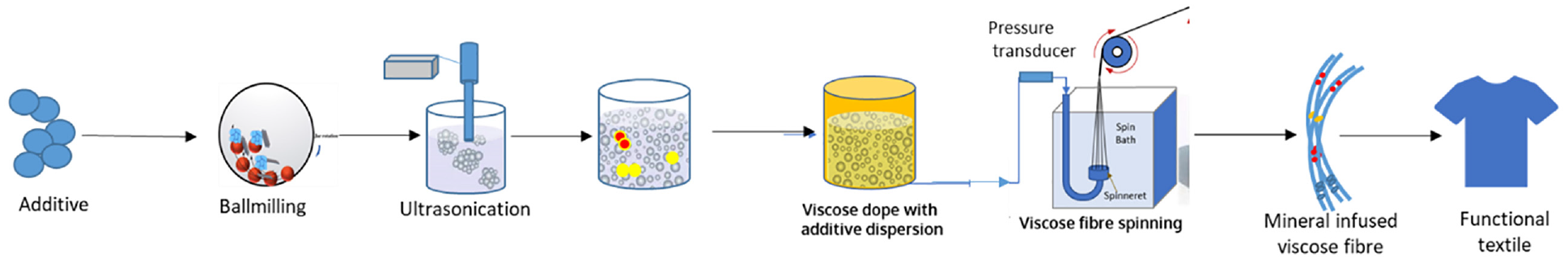
Schematic representation of mineral mixture dispersion preparation and its addition into viscose solution to spin IR-VSF.
Characterization of IR-VSF
The IR reflecting characteristics of IR-VSF were measured by thermal mapping from IR radiation reflectivity through an IR camera. To measure the IR reflectivity, the viscose fibres were made into non-woven sheets of size 16 cm × 16 cm and GSM 585 by a hand sheet former machine. The wet-laid web manufacturing process used in the sheet former machine is derived from the papermaking process. The steps involved are (1) dispersion of fibres; (2) deposition of fibres on the screen; (3) removal of water from the web by suction, gravity, or a pressure difference developed by water column; (4) consolidation; (5) drying of the web. The fibre sheets were then placed under an IR lamp (wavelength range of 2–10 µ) for 3 min, and the extent of IR reflectivity, indicated by extent of surface temperature increase, was visually captured through an IR camera (Microepsilon thermoIMAGER TIM 40, optical resolution of 382 × 288 pixels and a spectral range of 8–14 µ).
Results and Discussion
Effect of Mechanical Treatments and Surfactant on Slurry Properties
Final product quality requires efficient material processing, which is dependent on critical parameters such as average particle size and particle size distribution (PSD) as they influence surface area and porosity. The surface area available for interactions between aqueous solution and solid surface is determined by particle size. Particle size is one of the parameters which critically impacts physical stability of dispersion, rate of settling of particles and agglomeration tendencies. In a dispersion when the particles are lower in size and uniformly dispersed, the settling tendencies will be slow with lower tendencies of agglomeration. 17
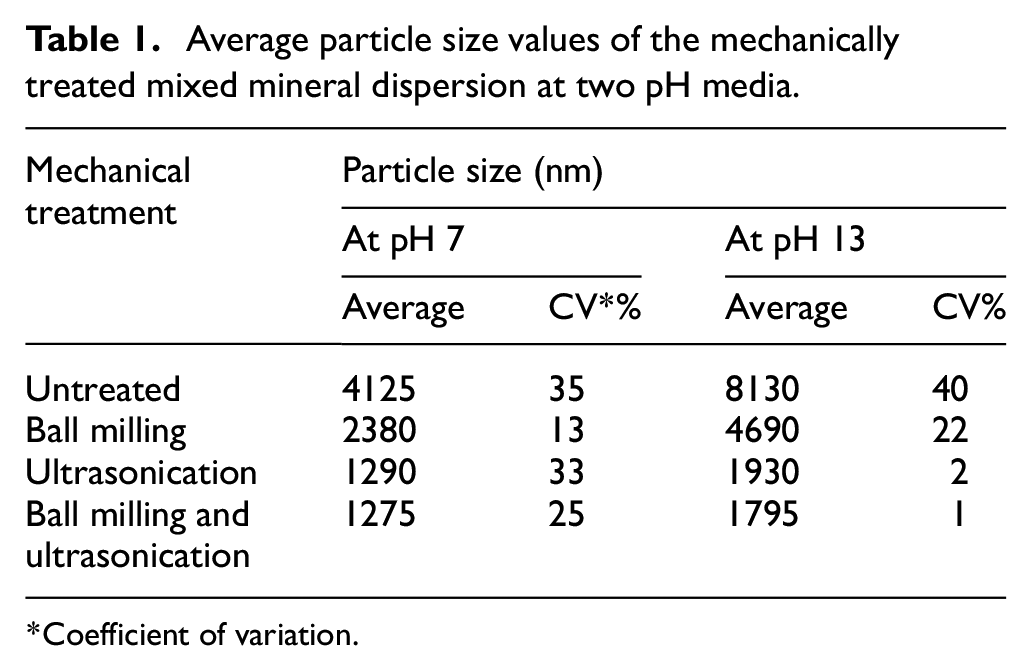
Table 1 shows the effect of mechanical treatments and pH on the particle size of mineral mixture dispersion. As observed from the table, average particle size was higher at high pH, whereas it was comparatively lower in the neutral pH. In a dispersion which is stable, the maximum attractive forces between particles should be just enough to allow any other agitation to easily break all particle–particle bonds. 28 Independent treatments like BMUS reduced the particle size at both pH. There was a synergistic effect and further reduction in particle size when the mixed minerals were subjected to both BM and US.
Average particle size values of the mechanically treated mixed mineral dispersion at two pH media.
Coefficient of variation.
In the mixture, magnesium silicate hydroxide is hydrophobic due to the presence of oxide surfaces. There are hydrophilic groups such as SiOH and MgOH present on its surface; however, these groups are pH dependent.29,30 The nature of the charge on the mineral surface can be decided based on dissociation tendencies or adsorption behaviour of the ions. At neutral pH, the surfaces of the silicate are negatively charged, while the surfaces of the carbonate are positively charged. 31 The magnitude of the charge on silica surface increases with the increase in pH of the dispersion. 32 This leads to higher agglomeration tendencies between the oppositely charged particles in aqueous medium.
It was observed from the tables that the mechanical treatments affect the particle size and its distribution (Table 1 & Table A – Supplementary information). BM ground the particles to some extent and US broke down the soft agglomerates to get a lower average particle size in aqueous media. There was a synergistic effect of the US treatment after dry BM, which reduced the particle size further. After BM, the particle size was significantly reduced by 50%, and post US, it further reduced by 75%. Another observation of increase in average particle size at high pH in all cases can be explained by the re-agglomeration tendencies of mineral particles with an increase in pH. Negatively charged Si surfaces become more negative with an increase in pH, which leads to lower electrostatic repulsion tendencies of the particles. 31 With an increase in pH, the particles agglomerate due to Van der Waals forces of attraction and other columbic forces, leading to increase in particle size.
The particle size distribution (Table A – Supplementary information) is broader as we try to decrease the particle size by a combination of BM and US. This is primarily due to the re-agglomeration tendencies of the particles. This observation has also been reported in the literature where re-agglomeration of particles broadens the particle size distribution. The impact created by the milling of mineral particles breaks down the agglomerates but at the same time causes re-agglomeration of particles. 28 The mineral BMUS particles had a broader particle size distribution at high pH because of lowering of electrostatic repulsion tendencies and re-agglomeration of particles due to high shear milling and US.
Effect of Ball-Milling Conditions
The effect of BM was analysed by varying the parameters like speed and run time. The mixed mineral powder was ball milled at two different speeds, that is, 200 rpm and 400 rpm for 30 min. As seen in Table 2, an increase in the BM speed increased the BET surface area by 4 times. This indicates an increase in pore volume and surface area of mineral particles, with the reason being that, as the pore volume increases, the number of pores (for nitrogen sites) increases, which facilitates an increase in surface area.
Effect of ball-milling speed on BET surface area.

BET: Brunauer–Emmett–Teller.
Coefficient of variation.
At a higher BM speed, the run time was increased from 30 to 360 min to see its effect on any further increase in surface area. Up to 120 min, there was no significant increase in surface area; however, there was a marginal increase in surface area to 22.2 ± 1.3 m2/gm after 240 min. As the run time was increased beyond 240 min, there was again no further increase in surface area. This is primarily due to the reduction in the rate of particle size decrease after a certain particle size. This observation has been reconfirmed by studies showing that the particle size reduction rates decrease with decreasing particle size. 33 This is attributed to a reduction in the inertial and hydrodynamic forces and an increase in particle aggregate strength with a reduction in particle size.28,34,35 In practice, this means that the BM efficiency is reduced drastically at particle sizes smaller than 600 nm. If a further size reduction is required, then the time or speed for milling needs to be increased but at a risk of contamination and unwanted phase changes.36,37 These surface area values also indicate that the particles are in the sub-micron range and would need further grinding to get nano-sized particles.
As seen in Table 2, a lower particle size was achieved with an increase in BM speed from 200 to 400 rpm in both pH mediums. This was due to a reduction in particle size due to high grinding speed, which resulted in a lower particle size in aqueous dispersion as well as post US. However, the particles were not of uniform size in the dispersion at high pH, as seen by the multiple peaks in the particle size distribution graphs in Table B – Supplementary information. This was mainly due to re-agglomeration behaviour at high pH and lowering of electrostatic repulsion tendencies of mineral particles. There was a need to stabilize the particles and maintain the particle–particle separation in dispersion through adsorption of a layer of surfactant molecules.
Effect of Nature of Surfactant on Slurry Properties
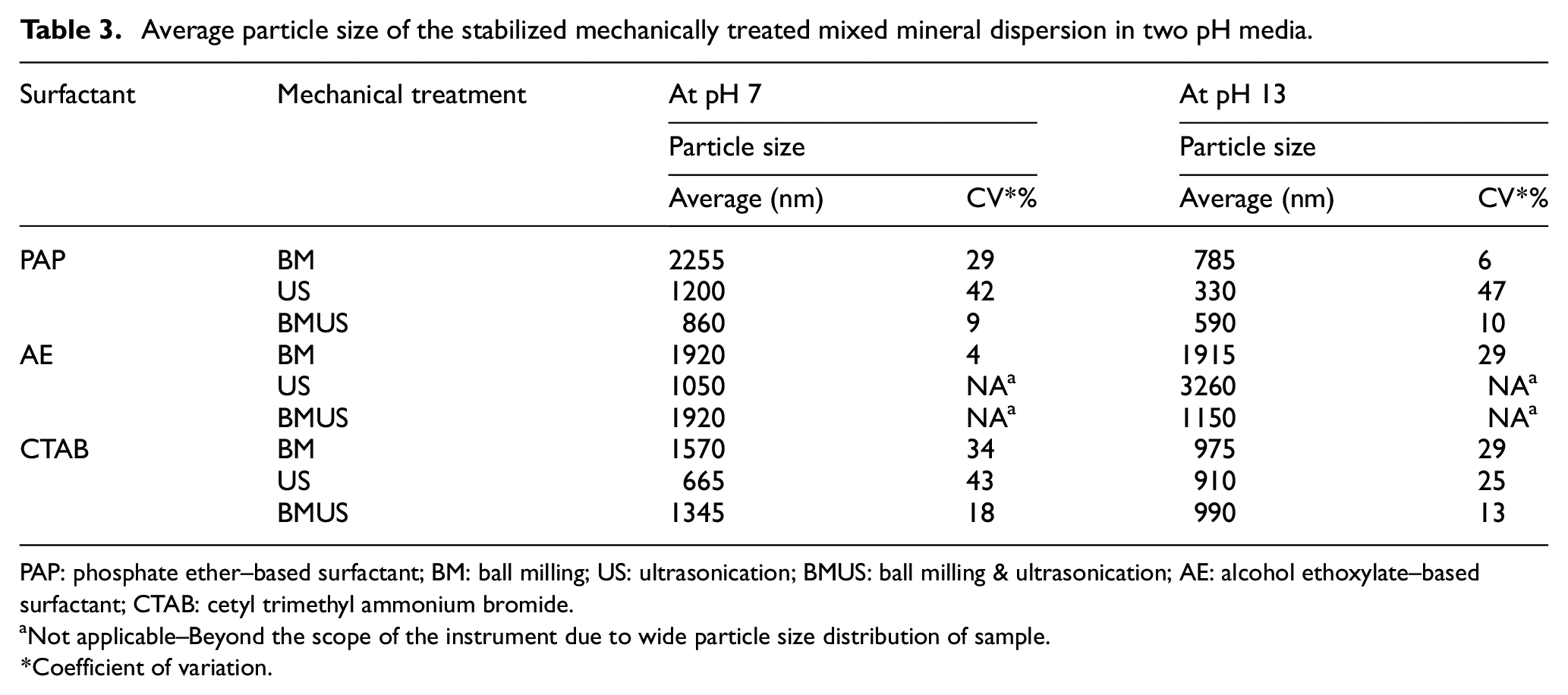
We have described a technique to stabilize the mixed mineral dispersion by evaluating three categories of surfactants, that is, anionic, cationic and non-ionic. Surfactants are a group of chemical compounds, which create micelles in aqueous solution. They get adsorbed at the interface of the particles and increase the stability of a dispersion through electrostatic or steric repulsion. They are classified into ionic and non-ionic surfactants depending on the charge at their hydrophilic head group. As seen in Table 3, the phosphate-based anionic surfactant was effective in getting the lowest particle sizes of mixed mineral post BM and US at both pH values. This means that anionic surfactants would be effective in stabilizing the mineral dispersion at inherent pH as well as the highly alkaline pH of viscose solution. In the mixture, the calcium and silica ions interact in the aqueous medium and are dependent on the pH of the medium. The divalent Ca2+ cations form a bridge between the negatively charged silica and the phosphate-based anionic surfactant.23,38 This Ca2+ cation bridge decreases the cationic surfactant adsorption on silica surfaces.39,40
Average particle size of the stabilized mechanically treated mixed mineral dispersion in two pH media.
PAP: phosphate ether–based surfactant; BM: ball milling; US: ultrasonication; BMUS: ball milling & ultrasonication; AE: alcohol ethoxylate–based surfactant; CTAB: cetyl trimethyl ammonium bromide.
Not applicable–Beyond the scope of the instrument due to wide particle size distribution of sample.
Coefficient of variation.
As seen by the average particle size values in Table 3, the AE surfactant does not effectively stabilize the dispersion to reduce the particle size of mixed mineral particles as there is no interaction with the mineral particles. There is an adsorption of CTAB surfactant at negatively charged silica surfaces to some extent at high pH as Si gets more negatively charged at high pH. However, it was observed that PAP surfactant interaction stabilizes the particles most effectively and reduces the particle size. This phenomenon can be explained through the interaction of AE surfactant with Ca2+ ions. When the Ca2+ ions form a bridge with the Si– surface, it reduces the adsorption with CTAB surfactant. However, the PAP surfactant gets readily adsorbed, increasing the electrostatic repulsion tendencies. In all the three cases, the lowest particle sizes have been obtained by combining the BM and US treatments. There was not much difference in particle size with and without PAP surfactant on BMUS mixed mineral particles, as seen in Table 3. However, the stabilization effect was clearly seen when the surfactant-based mixed minerals were dispersed in viscose dope and extruded through the spinneret to spin mineral infused viscose fibres, which is further explained in the section ‘Effect of Surfactant on Dispersion of Mineral Particles in Viscose Dope’.
Stability of Mixed Mineral Dispersion
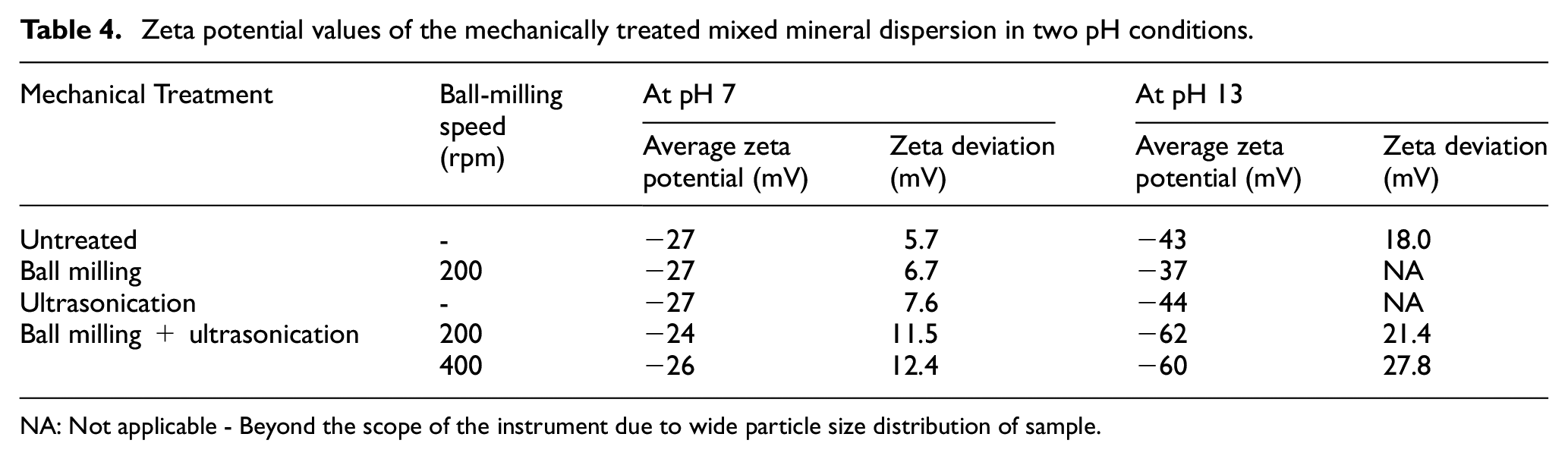
Stability of the formulation is indicated by the zeta potential values of the particles in the dispersion. The zeta potential is assessed by subjecting the particles in a dispersion to an electric field and evaluating the electrokinetic properties of the particles. It is dependent on the velocity of the moving particle under the influence of the electric field as well as the viscosity of the dispersion media. Smaller particles tend to move faster in a less viscous media in contrast to larger ones and hence undergo faster diffusion and higher stability. The magnitude of the surface charge on the slipping plane of the particle which repels the other surface and keeps the particles well separated is of utmost importance to evaluate the stability of dispersion. 41 As seen in Table 4, the zeta potential values were higher at high pH with BMUS mixed mineral particles. There is no significant change in zeta potential values with independent treatments of BM & US in both pH media. This is an indication of instability and lower particle–particle repulsion tendencies even with a reduction in particle size. Here, according to the observation, higher zeta potential values indicated stability and less susceptibility to agglomeration beyond a certain particle size at high pH.
Zeta potential values of the mechanically treated mixed mineral dispersion in two pH conditions.
NA: Not applicable - Beyond the scope of the instrument due to wide particle size distribution of sample.
As seen in Table 5, the high absolute zeta potential values with PAP surfactant indicate greater adsorption of this surfactant and higher electrostatic repulsion between mixed mineral particles. This is due to the effective interaction between negatively charged carboxylate groups and positively charged Ca ions. CTAB also increases the surface charge and stability of the dispersion due to its adsorption with the negatively charged silica ions. AE surfactant does not reflect the stability of dispersion through zeta potential characterization due to its non-ionic nature.
Zeta potential values of the stabilized mechanically treated mixed mineral dispersion in two pH mediums.
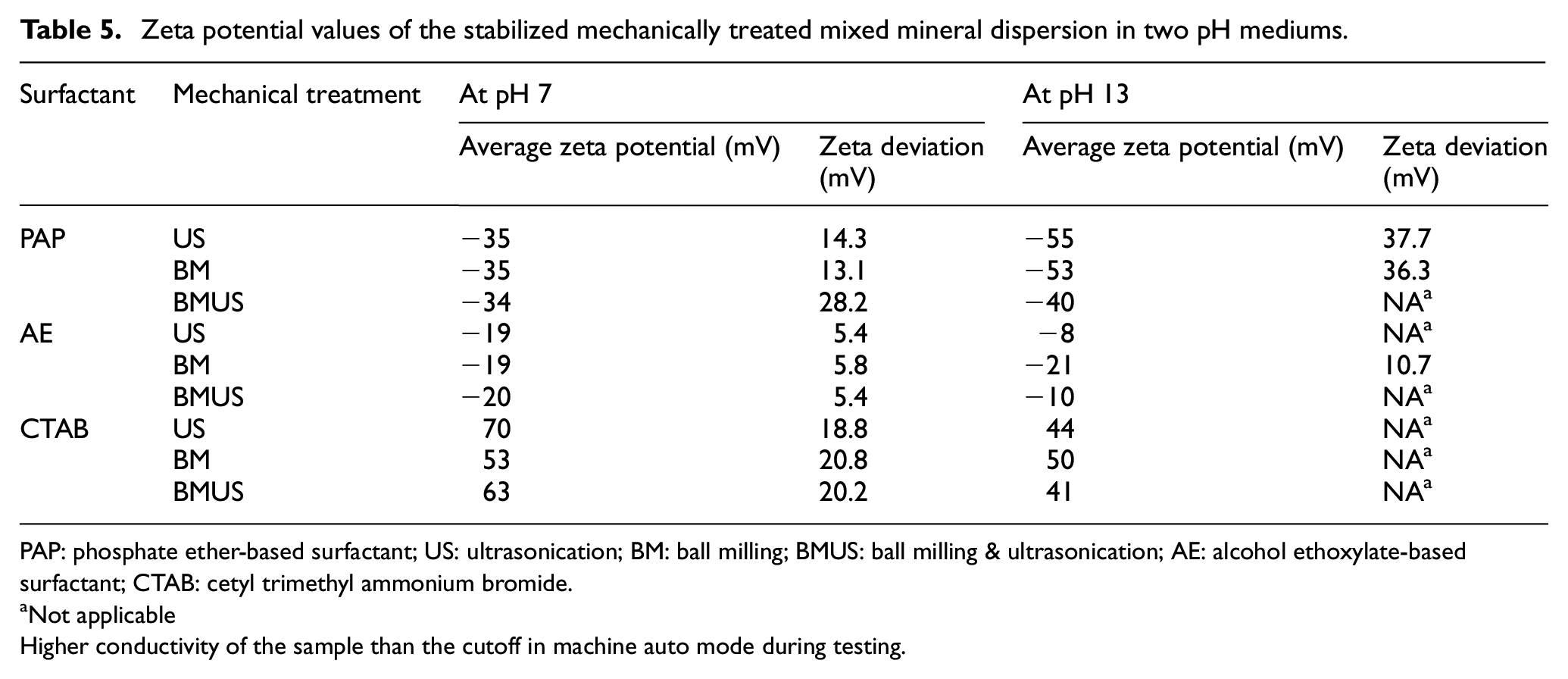
PAP: phosphate ether-based surfactant; US: ultrasonication; BM: ball milling; BMUS: ball milling & ultrasonication; AE: alcohol ethoxylate-based surfactant; CTAB: cetyl trimethyl ammonium bromide.
Not applicable
Higher conductivity of the sample than the cutoff in machine auto mode during testing.
Effect of Surfactant on Dispersion of Mineral Particles in Viscose Dope
Conventional methods of measuring particle size used for slurry are not useful for measuring the same in viscose dope, due to higher viscosity. Towards this, an indirect method has been used, wherein the dope is extruded through spinneret holes in a laboratory-scale wet spinning setup and a pressure gauge indicates pressure build-up with time. An excessive poor dispersion of mineral particles will lead to choking of the spinneret holes with time. The rate of pressure build-up gives a good idea of quality of particle dispersion in the dope (Figure 2).
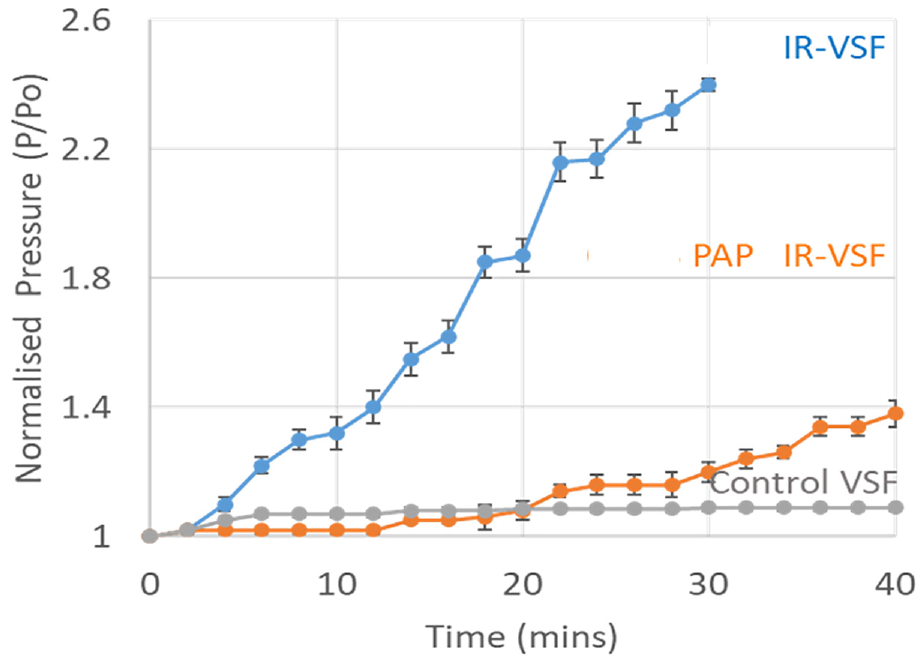
Pressure build-up during spinning of IR-VSF with and without PAP surfactant vs Control VSF.
As observed in Figure 2, in the case of control viscose spinning, i.e. spinning of viscose dope without any minerals, there was no significant pressure rise in 40 minutes. However the pressure rate significantly increased by 1.2 kg/cm2 during BMUS mixed mineral dispersed viscose spinning. However, when the PAP surfactant was added into the BMUS mixed mineral dispersion and mixed into viscose dope, to spin PAP IR-VSF, the pressure rise increased by only 0.2 kg/cm2 in 40 min. Although the particle size studies show lower average particle size in both BMUS mixed mineral dispersion with and without surfactant, the situation is different in a dynamic spinning system. Without the use of surfactant, the particles agglomerate with time, leading to faster choking of the filter at the outlet of the spinneret as indicated by the back pressure build-up. The extent of agglomeration was not as severe with the use of surfactant as it possibly kept the particles well separated from each other and delayed the choking of the spinneret. The optimized mechanical conditions of mixed mineral particles along with stabilization of dispersion with the surfactant can be effectively used to reduce the average particle size, lowering agglomeration and hence improve processability during spinning.
IR Reflectivity of IR-VSF
The IR-VSF & control VSF fibre sheets were exposed to IR radiation emitted by the IR lamp, and surface temperatures were measured using an IR camera. There was a significant surface temperature increase of mineral infused viscose fibres of almost 2 °C. Visually, the IR-VSF reflects higher IR radiation, as indicated by dark orange and red regions in the Figure 3. The control viscose fibre sheet did not reflect significant IR radiation, as seen by the lighter, yellow-coloured regions, and showed a lower surface temperature.
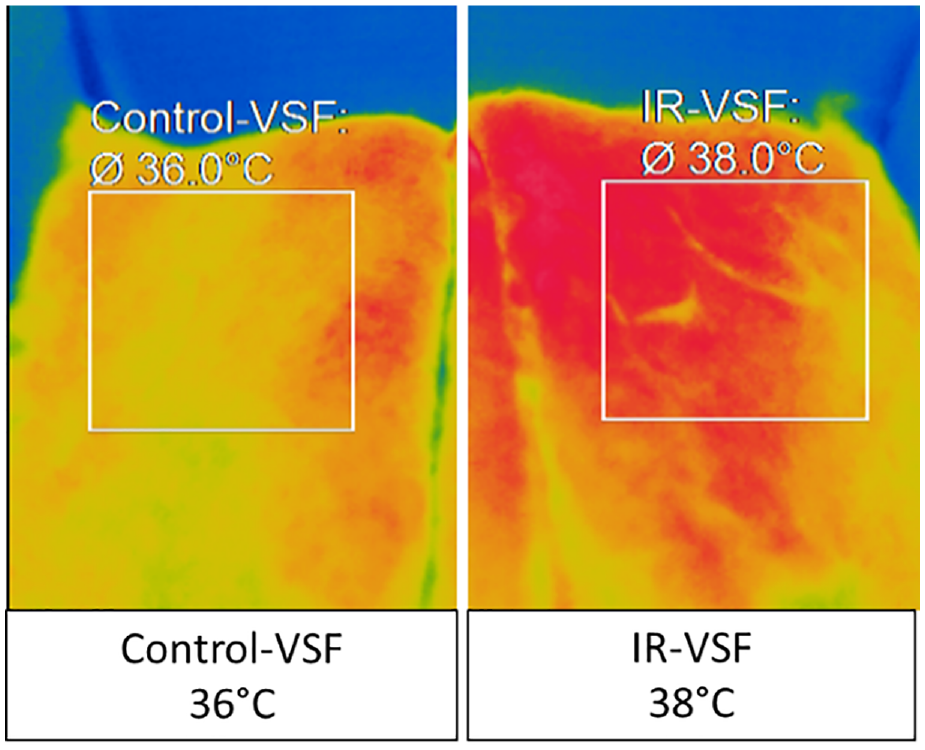
IR camera images of control VSF and IR-VSF non-woven.
Conclusion
The studies have shown some good insights on challenges to incorporating mineral additives into viscose fibres. There is a significant effect of pH on the mixed mineral dispersion stability. The agglomeration tendencies are mainly due to particle interactions leading to lower electrostatic repulsion at high pH. The combination of first ball milling the mixed mineral powder followed by US of its dispersion reduced the particle size significantly. The stability of dispersion is majorly impacted by the nature of the surfactant used. There is a major effect of adsorption and desorption of Ca ions on Si surfaces to enable adsorption of PAP surfactant for improved dispersion stability. This surfactant was found to reduce the particle size by the electrostatic and steric repulsion keeping the mineral particles well separated, thereby enhancing the stability of the dispersion. A good and stable dispersion of lower particle sizes in slurry also leads to good dispersion in dope as evidenced by lower pressure build-up during spinning. The IR-VSF is shown to reflect IR radiation better as indicated by increased surface temperature by nearly 2°C compared with control VSF. The experiments have established the challenges to incorporating mineral additives in viscose fibres and ways to overcome them, leading to desired final attribute in the fibres. Functional viscose fibres can be made through addition of different additives after studying and optimizing their behaviour of dispersions prior to spinning. The insights from this study can be used for further evaluation of particle–medium interactions in the wet-spun functional fibre development and manufacturing.
Supplemental Material
sj-docx-1-aat-10.1177_24723444221147975 – Supplemental material for Studies on Incorporating Infrared Reflecting Minerals into Viscose Fibres
Supplemental material, sj-docx-1-aat-10.1177_24723444221147975 for Studies on Incorporating Infrared Reflecting Minerals into Viscose Fibres by Esha Sharma, Shrikant Ralebhat, Dhirendra Singh, Gurudatt Krishnamurthy, Sunil Bhagwat and Ravindra V. Adivarekar in AATCC Journal of Research
Footnotes
Acknowledgements
The authors acknowledge the Analytical Science and Technology laboratory at Aditya Birla Science & Technology Company Pvt. Ltd.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

