Abstract
Background
The purpose was to report mid-term to long-term clinical outcomes in a multicentre series of patients who received stemless total shoulder arthroplasty (TSA). The hypothesis was that stemless TSA would be a safe and effective treatment with satisfactory clinical outcomes.
Methods
Authors retrospectively reviewed records of 62 stemless TSAs implanted between March 2013 and December 2014. Six were excluded because they had fractures or muscular impairment, which left 56: primary osteoarthritis (n = 49), rheumatoid arthritis (n = 4), avascular necrosis (n = 1), or glenoid dysplasia (n = 2). Outcomes were absolute Constant Score (CS), age-/sex-adjusted CS, and the American Shoulder and Elbow Surgeons (ASES) score. Proportions of patients that achieved substantial clinical benefits for absolute CS and ASES scores were determined.
Results
Of the 56 patients, 8 (14%) died (unrelated to TSA), 5 (9%) were lost to follow-up, and 2 (4%) refused participation. One patient was reoperated for infection with implant removal (excluded from analysis), and one for periprosthetic fracture without implant removal. At 7.6 ± 0.5 years (range 6.8-9.3), the remaining 40 patients, aged 71.0 ± 8.5 years, achieved net improvements of 40.7 ± 15.8 (CS), 62%±23% (age-/sex-adjusted CS), and 59.7 ± 16.4 (ASES). Of patients with complete absolute CS (n = 37) and ASES score (n = 28), respectively, 33 (89%) and 27 (96%) achieved substantial clinical benefits.
Conclusions
Stemless TSA yields improvements in functional outcomes at mid-term to long-term that exceed the substantial clinical benefits of the absolute CS and ASES score at a mean follow-up of 7.6 years. Although the findings of this study revealed low complications and revision rates, more studies are needed to confirm long-term benefits of stemless TSA.
Level of evidence
IV, case series.
Keywords
Introduction
Total shoulder arthroplasty (TSA) is an effective treatment for end-stage osteoarthritis, posttraumatic sequelae, and other shoulder pathologies. 1 Conventional anatomic TSA aims to reproduce glenohumeral kinematics with a stemmed humeral component and pegged and/or keeled glenoid components that restore anatomy and centre-of-rotation. Stemmed humeral components remain the gold standard, 2 though over the past decades, manufacturers have shortened stems to reduce risks of malpositioning, loosening and periprosthetic fractures. 3 This led to development of stemless humeral components that restore proximal humeral anatomy without reaming the humeral diaphysis. 1
Since their introduction in 2004, stemless humeral components gained increasing popularity, 2 as theoretical advantages over their stemmed counterparts include improved accuracy of anatomic reconstruction and bone preservation, with reduced rates of periprosthetic humeral shaft fractures, stress shielding, surgical time, and blood loss.3–6 Moreover, stemless components are easier to revise because they preserve the humeral canal.3–6 Finally, comparative studies report no significant differences in functional outcomes between stemless and stemmed TSA.3,4,6–8
There are few studies and registries that report mid-term to long-term outcomes after stemless TSA, and information on functional outcomes and implant longevity are therefore lacking.2,4 The purpose of this study was to report mid-term to long-term clinical outcomes in a multicentre series of patients who received stemless TSA. The hypothesis was that stemless TSA would be a safe and effective treatment with satisfactory mid-term to long-term clinical outcomes.
Materials and Methods
Cohort
The authors retrospectively reviewed the records of 62 patients who received stemless TSA (Easytech® Anatomical, FX Solutions, Viriat, France) by 5 surgeons (TA, TL, HW, AG, and EB) between March 2013 and December 2014. Of the 62 patients, 56 met inclusion criteria which were treatment for primary osteoarthritis (n = 49), rheumatoid arthritis (n = 4), avascular necrosis (n = 1), and glenoid dysplasia (n = 2). Six patients met exclusion criteria (fracture, n = 4; muscular impairment, n = 2) and were excluded from the analysis. This left an initial cohort of 56 patients (14 men and 42 women) aged 70.1 ± 10.2 years (range 45-93 years) with a body mass index (BMI) of 29.4 ± 4.9 kg/m2 (range 23.1-44.4 kg/m2) available for analysis. Patients originally scheduled for stemless TSA, but who had weak bone during surgery, received a stemmed component. 9
Implant Design
The humeral assembly consists of an anchoring base (Ø30, Ø34, or Ø38 mm), morse taper, optional spacer and a centered or eccentric head (Figure 1A). Primary stability is granted by a central retaining peg and 5 peripheral fir tree-shaped pegs, and secondary stability is granted by a layer of plasma-sprayed titanium coated with plasma-sprayed hydroxyapatite (Ti + HA). The humeral head (Ø39, Ø43, Ø46, or Ø50 mm) is either centered or eccentric, and connects to the humeral anchoring base with a morse taper.

Graphical presentation of (A) humeral assembly and (B) glenoid assembly of the stemless TSA system. Abbreviation: TSA, total shoulder arthroplasty.
The humeral assembly is compatible with both all-polyethylene (sizes of XS, S, M, or L) or metal-backed (Ø20 mm) glenoid components. The all-polyethylene components are made of ultrahigh molecular weight polyethylene (UHMWPE), whereas the metal-backed glenoid component consists of a UHMWPE disc net-shape molded within a pegged titanium alloy shell coated with a layer of plasma-sprayed titanium and plasma-sprayed hydroxyapatite (Ti + HA) (Figure 1B).
Surgical Technique
All patients were operated under general anesthesia in a beach chair position. The surgical approach was superolateral in 36 patients (64%; by 2 surgeons) and deltopectoral in 20 patients (36%; by 3 surgeons) (Table 1). The subscapularis was detached in all cases, and based on surgeon preference, reattached by a trans-osseous technique (80%) or suture anchor technique (20%). The surgical technique was otherwise identical for all patients as described hereafter.
Intraoperative Characteristics.

Humeral Preparation
The humeral head osteotomy was made along the head-neck junction using an oscillating saw, after removal of osteophytes, with a retroversion of 20° relative to the humeral transepicondylar axis (a cutting guide is available to assist in measuring retroversion relative to the forearm axis). Using a threaded K-wire as a guide, the size of the humeral anchoring base was determined, aiming to obtain adequate peripheral support, after which it was impacted to be flush within the resected surface using a cannulated impactor. The humeral head covered the cortical rim and extended above the greater tuberosity (5-8 mm).
Glenoid Preparation
The glenoid was exposed by either an anterior, superior, or inferior retractor, after which the glenoid labrum was excised, and osteophytes removed for exposure of bony anatomy. For all-polyethylene glenoid components, the glenoid was reamed with a canulated reamer. The peg holes were then created using a peg drilling-guide placed over a K-wire. A trial component was then inserted to perform a mobility test, and upon verification, the final glenoid implant was cemented in place. When using a 3-pegged or 4-pegged glenoid component, the central peg was not cemented.
For the metal-backed glenoid, a K-wire guide was positioned perpendicular to the glenoid surface in accordance with the native anatomy in the proximodistal plane. The glenoid surface was reamed until the reamer could not progress further. Upon verification of the position and angle with a trial component, the glenoid component was impacted into the glenoid.
Rehabilitation
Shoulders were immobilized in a sling, in either internal or neutral rotation, for 4 to 6 weeks depending on the surgical approach and surgeon assessment. Passive range of motion exercises were initiated on the first postoperative day. Active range of motion exercises started after 4 to 6 weeks, under supervision of physiotherapists.
Clinical Outcomes Scores
The primary functional outcomes were the absolute constant score (CS),10,11 age-/sex-adjusted CS,12,13 and the American Shoulder and Elbow Surgeons (ASES)14,15 score. Functional outcome scores were recorded preoperatively and postoperatively at a minimum follow-up of 6.8 years. The proportions of patients who achieved a satisfactory outcome after TSA were based on the substantial clinical benefit, as proposed for the absolute CS (net improvement ≥19.1) and ASES score (net improvement ≥31.5). 16 In addition, complications related to the TSA as well as need for revision were recorded.
Informed Consent
This study was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki, and approved by the institutional review board (Ref#: COS-RGDS-2022-03-009-NOURISSAT-G). All participants provided informed consent for the use of their data.
Statistical Analysis
The cohort characteristics were summarized using descriptive statistics. For continuous variables, the mean, standard deviation (SD), and range were reported, and for categorical variables the number and proportion were reported. Boxplots were used to visually compare functional outcomes (preoperative, postoperative and net-improvement) among patients: (1) different indications for surgery and (2) surgical approach. Finally, Shapiro–Wilk tests were used to assess the normality of distributions. Differences between normally distributed groups were assessed using the student's t test, whereas differences between skewed groups were evaluated using the Wilcoxon rank sum test (Mann–Whitney U test). A P-value <.05 was considered statistically significant. All analyses were performed using R version 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Of the initial cohort of 56 patients, 8 (14%) died from causes unrelated to the TSA, 5 (9%) were lost to follow-up, and 2 (4%) refused that their data be used in the analysis (Figure 2 and Table 2). Two patients required reoperation; 1 for an infection that required revision of the humeral head (excluded from the analysis due to component revision), and 1 for a periprosthetic fracture that required humeral osteosynthesis without implant removal (included in the analysis). This left a study cohort of 40 patients (17 men and 33 women) aged 71.0 ± 8.5 years (range 50-86 years) with a BMI of 30.1 ± 5.4 kg/m2 (range 23.1-44.4 kg/m2).

Flowchart of the case series.
Cohort Demographics.

Abbreviations: BMI, body mass index; OA, osteoarthritis; SD, standard deviation.
At a mean follow-up of 7.6 ± 0.5 years (range 6.8-9.3 years), net improvements were 40.7 ± 15.8 for the absolute CS, 62%±23% for the age-/sex-adjusted CS, and 59.7 ± 16.4 for the ASES score (Table 3). Of the 37 patients who had complete records for absolute CS, 33 (89%) achieved a substantial clinical benefit (net improvement ≥19.1). Of the 28 patients who had complete records for ASES score, 27 (96%) achieved a substantial clinical benefit (net improvement ≥31.5).
Outcomes of the Study Cohort.

*Complete records for 37 patients.
Complete records for 28 patients (one surgeon did not collect ASES scores).
Abbreviations: ASES, American Shoulder and Elbow Surgeons; BMI, body mass index; CS, constant score.
Patients treated for primary OA, compared to secondary OA, had significantly better preoperative absolute CS (30.2 ± 6.7 vs 23.5 ± 6.9; P < .05). Patients operated by a superolateral compared to deltopectoral approach had better postoperative absolute CS (76.3 ± 16.0 vs 64.4 ± 18.7, P < .05) and age-/sex-adjusted CS (114 ± 24% vs 94 ± 27%, P < .05) (Figure 3). Comparison of clinical outcomes at both timepoints revealed no significant differences in absolute CS, age-/sex-adjusted CS, or ASES score (Table 4). Multivariable analyses were not performed due to the small sample size, so it is not possible to ascertain whether the aforementioned associations were direct or confounded.

Comparison of clinical outcomes by (A) indications for surgery and (B) surgical approach.
Comparison of Clinical Outcomes Scores at Early and Long-Term Follow-Up.

Abbreviations: ASES, American Shoulder and Elbow Surgeons; CS, constant score; SD, standard deviation.
Discussion
The main finding of the present study was that, at a follow-up of 6.8 to 9.3 years after stemless TSA, the mean improvements in functional outcomes exceeded the substantial clinical benefits of the absolute CS and ASES score. Moreover, when considering patients with complete clinical records, 98% and 96% of respondents achieved substantial clinical benefits respectively for absolute CS and ASES score. Finally, only two complications were reported, of which one required revision surgery. These findings therefore support the hypothesis that stemless TSA is a safe and effective treatment with satisfactory mid-term to long-term outcomes.
A recent systematic review and meta-analysis 17 on four clinical studies comparing CS scores between stemless (n = 358) versus stemmed TSA (n = 229), found no difference in net improvement (range 11.60-55.2 vs 40.1-60.00; standard mean difference (SMD), −0.57 (confidence interval [CI −1.59-0.45]) or in postoperative scores (range 65.5-101.5 vs 65.7-100.2; SMD, 1.25 (confidence interval −1.41-3.91). Nixon et al 18 retrospectively compared postoperative ASES scores between stemless (n = 115) versus stemmed TSA (n = 59); at 12 months ASES was significantly higher in the stemless versus stemmed cohort (91.48 vs 86.34; P = .01), whereas at 24 months no significant difference was observed (89.05 vs 90.25; P = .54). These results suggest similar clinical outcome scores for stemless versus stemmed TSA at 24 months, but also highlight the need for long-term comparative studies.
Restoration of proximal humeral anatomy has a direct influence on functional outcomes of TSA. 19 Stemmed humeral components have set inclination angles, and though modern designs have different inclination options, restoring proximal humeral anatomy remains challenging in some patients. 20 Stemless humeral components allow surgeons greater freedom to personalize the inclination angle, but have a reduced bone-implant interface and hence greater risks of component loosening. A variety of methods are available to achieve metaphyseal fixation, which include pegs and/or fins with grit-blasted surfaces or porous coatings to enhance bone ingrowth using titanium or hydroxyapatite. 3 Stemless designs therefore rely on adequate humeral metaphyseal bone stock for primary fixation to promote osseous ingrowth for long-term fixation.
There remains a lack of evidence to support the utility and safety of stemless TSA. 4 In the present study, of the two complications reported, none were related to component loosening, as the only case requiring implant removal was due to infection. Moreover, there were no clinical symptoms that suggest any osteolysis, stress shielding, and aseptic loosening, but it should be noted that there was no radiographic evidence to support this finding. A recent systematic review revealed similar aggregate complication rates of stemless versus stemmed components. 4 Moreover, registries only recently started to compare revision rates, and reported rates of 3.8% to 4.7% for stemless TSA versus 4.2% to 8.8% for stemmed TSA, at 6 to 8 years.21–23
Little is known about the effect of patient factors on clinical and functional outcomes of stemless TSA. 6 In the present study, comparison of functional outcomes revealed that postoperative scores and net improvements in absolute and age-/sex-adjusted CS were greater in patients operated by a superolateral versus a deltopectoral approach. The deltopectoral approach requires release of the subscapularis, and subsequent repair once the arthroplasty is completed. Subscapularis dysfunction after TSA could lead to shoulder instability, pain, and decreased shoulder function. 24 Due to the small sample size, it was not possible to investigate effects of glenoid design, subscapularis function, and other confounders on functional outcomes. Therefore, studies specifically designed to determine these effects on functional outcomes are needed to confirm the aforementioned observations. Finally, it is noteworthy that differences among the two groups were more pronounced for CS compared to differences in ASES score. One explanation might be that the postoperative ASES score has a more substantial ceiling effect compared to the CS.25,26
The present study revealed encouraging results when compared to previous reports on mid-term to long-term clinical outcomes of stemless TSA (Table 5)7,8,27–32; but these findings should be interpreted with the following limitations in mind. First, this is a multicentre retrospective analysis of functional outcomes after TSA at a follow-up of 6.8 to 9.3 years. Therefore, pooling of demographics, surgical data, and functional outcomes could be subject to high levels of heterogeneity—different surgeons, approaches, and glenoid designs. Moreover, all patients scheduled for TSA during the inclusion period were considered for stemless components, but as bone quality were assessed intra-operatively, 9 a final decision was made during surgery. Finally, information on intra-operative conversions from stemless to stemmed components were not available. Second, the purpose of this study was to report the mid-term to long-term outcomes in a series of patients who received stemless TSA. Therefore, by design this study is likely not powered to draw any definitive conclusions from the comparisons made in terms of indications for surgery, glenoid, or humeral component design, and choice of surgical approach. Third, there were no preoperative or postoperative radiographs available to assess implant position or signs of osteolysis. In addition, no range of motion or strength data were reported. Finally, the study did not have a control group for comparison, and also did not report.
Comparison of Absolute and Age-/Sex-Adjusted CS to Published Reports on Stemless TSA at Mid-Term to Long-Term Follow-Up.
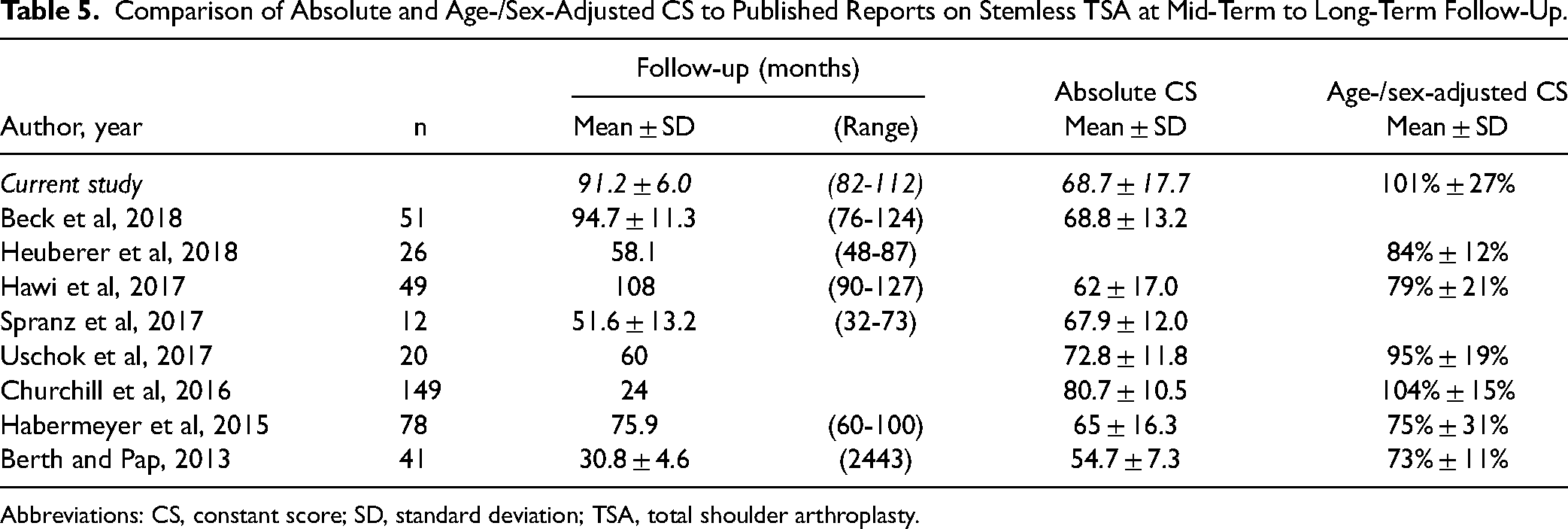
Abbreviations: CS, constant score; SD, standard deviation; TSA, total shoulder arthroplasty.
Conclusion
Stemless TSA yields improvements in functional outcomes scores at mid-term to long-term that exceed the substantial clinical benefits of the absolute CS and ASES score at a mean follow-up of 7.6 years. Although the findings of this study revealed low complication and revision rates, more studies are needed to confirm the long-term benefits of stemless TSA.
Footnotes
Acknowledgements
The authors would like to thank Sonia Dubreuil for assistance with the collection of postoperative outcome scores.
Author Contribution Statement
TA, TL, HW, AG, EB, were involved in study design, interpretation of findings, and manuscript editing. JHM performed statistical analysis and manuscript writing. MS contributed to interpretation of findings and manuscript writing. GN was involved in procurement of IRB approval and manuscript editing. All authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TL and HW: This author receives royalties from FX Solutions and will receive fees from FX Solutions for data collection in relation to this study, but no other financial payments or other benefits from any commercial entity related to the subject of this article. TA, AG and EB: This author will receive fees from FX Solutions for data collection in relation to this study, but no other financial payments or other benefits from any commercial entity related to the subject of this article. JHM and MS: This author is an employee of ReSurg SA which received fees from FX Solutions for statistical analysis and manuscript preparation for this study. GN: This author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by FX Solutions.
