Abstract
Introduction
Accurate restoration of the humeral joint line during shoulder arthroplasty is critical to maximizing outcomes. However, clinical studies show that both resurfacing and stemmed arthroplasty systems routinely do not restore the articular surface accurately, even by experienced surgeons. The purpose of this study was to evaluate surgeons’ ability to recreate the 3-dimensional geometry of the proximal humerus using a newly developed arthroplasty system.
Methods
The difference in articular surface location before and after arthroplasty was measured using a new nonspherical arthroplasty system which uses a calibrated multiplanar osteotomy technique matching bone removal thickness to implant thickness. Eight human cadaver specimens were measured at 9 separate point locations over the articular surface from a fixed reference point on the lateral humerus. We repeated the measurements after bone preparation with the implant trial. Articular surface points >3.0 mm from their original location were considered outliers, based on literature.
Results
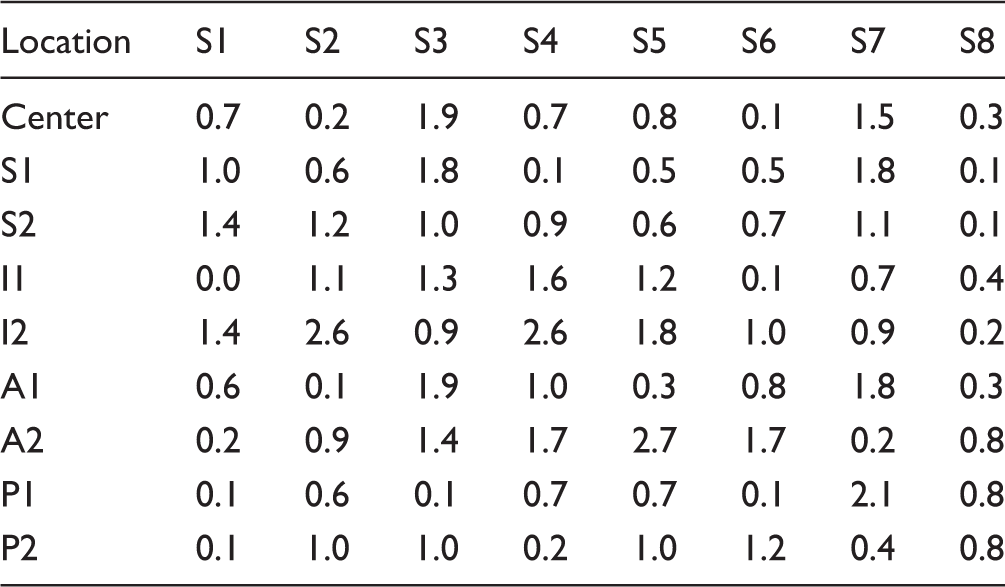
Average absolute deviation of each point on the articular surface measured 0.9 ± 0.7 mm (range: 0.0–2.7 mm) from preoperative to postoperative articular surface location. All (72/72) points measured were below the 3.0 mm outlier threshold from their original location.
Conclusion
Surgeons using the new system, ie, using a nonspherical head design and a calibrated multiplanar osteotomy surgical technique, were able to restore the joint line accurately with minimal (<1.0 mm) change from preop, with no outliers. Arthroplasty systems matching bone removal directly to implant thickness potentially may improve the reproducibility of arthroplasy and improve shoulder kinematics compared to traditional systems.
Keywords
Introduction
Anatomic restoration of the glenohumeral joint during shoulder arthroplasty is of primary importance for maximizing outcomes. Improvements in prosthetic design have increased our ability to replicate the location of the articular surface by allowing a stemmed arthroplasty to adjust for factors such as articular surface inclination, offset, and retroversion. Numerous studies link worse clinical outcomes and implant failure to nonanatomic implant placement,1–3 including complications such as subscapularis failure, stiffness, glenoid failure, and late supraspinatus tear. Often nonanatomic implantation results in an imbalance of the rotator cuff and capsule leading to poor shoulder mechanics and outcomes.
Third- and fourth-generation stemmed prostheses with multiple offset and inclination options have shown in computer models to have better restoration of anatomic relationships than older fixed designs but are still always limited in some fashion by the placement of the humeral stem. In addition, the head-neck taper junction may lead to malalignment, as the taper placement often does not coincide with the center of rotation (COR). Variations in head thickness also contribute to soft tissue tension, motion, and stability. Stemless (or canal-sparing humeral arthroplasty) and resurfacing designs have the theoretical advantage of greater ability to fit to the articular surface, but clinical evidence has shown that resurfacing implant placement is even less accurate than stemmed devices in maintaining the joint line and COR, mostly due to overstuffing. 4 Few total shoulder systems have been designed to have the surgeon resect a fixed thickness of bone, which corresponds to the specific thickness of the final implant to potentially reduce overstuffing.
In contrast, total knee arthroplasty fundamentals teach maintaining the joint line for optimum function, and the implant systems—both implants and instruments—are designed to facilitate this. It is proposed that by designing a shoulder arthroplasty system with similar principles and creating implants with thickness that correspond to bone resection, it may lead to more accurate joint line restoration and potentially better clinical outcomes, implant longevity, and reduced cost to the healthcare system.
The purpose of this study was to determine if a new anatomically designed implant and instrument system would facilitate accurate implant placement and articular surface restoration in cadaveric specimens, allowing surgeons to perform humeral arthroplasty reconstruction with minimal change in the location of the articular surface. It was hypothesized that implantation of an original, nonspherical humeral head implant would result in no significant differences in the joint line compared to the native anatomy.
Materials and Methods
In order to improve upon both prosthetic shape and implant placement accuracy, a new type of prosthesis was engineered with the goal of more accurate replication of normal anatomical relationships in 3 distinct areas: (1) prosthesis shape, (2) prosthesis location, and (3) prosthesis height (or joint line height). The Catalyst CSR Shoulder System (Catalyst OrthoScience, Naples, FL) utilizes an elliptical humeral head to replicate the normal nonspherical shape of the humerus (Figure 1). The articular surface shape is elliptical rather than spherical,
5
with a radius of curvature in the anterior–posterior axis less than the radius of curvature in the superior–inferior axis. The head is seated upon the proximal end of the humerus without utilizing a canal stem, which allows optimal implant placement with complete freedom with respect to inclination, version, and medial/posterior offset from the humeral canal. There are 7 humeral head implants to choose from, with size diameters ranging from 42.8 to 56.0 mm in approximately 2.3mm increments. There is only 1 head height per diameter. The implant respects the relationship of the rotator cuff insertion to prosthetic margin. To protect the rotator cuff from iatrogenic injury, the implant has a recessed superior margin to keep both the implant and the saw blade 3–5 mm away from the supraspinatus fibers.
Articulating (a) and bone-facing (b) sides of the humerus implant. Notice the nonspherical shape and 4 planes to the implant underside, made with a chamfer-cut technique.
In addition to implant design, substantial efforts went toward developing a surgical technique and surgical instruments in an attempt to more consistently reproduce the precise height and location of the articular surface. In contrast to the single large osteotomy used traditionally, this surgical technique utilizes a sequence of bone cutting instruments with multiple small precise osteotomies that match the thickness of bone removed to the thickness of the implant. In concept, this multiplanar osteotomy technique would reduce overstuffing, reestablish the proper joint line, and maintain the precise location of the center of the articular surface.
Five different fellowship trained shoulder surgeons participated in performing the surgical procedure on cadaveric specimens. A total of 8 fresh human shoulder cadaver specimens (age range: 64–69 years) were used in the analysis. Each specimen was placed onto a specimen holder and a standard deltopectoral approach to the humerus was made. A subscapularis tenotomy leaving 1 cm of tissue laterally on the subscapularis was performed, and the humeral head was dislocated anteriorly using standard shoulder retractors.
A total of 9 measurements were taken on the native humeral head after exposure, and then those same 9 measurements were then repeated after bone cuts were made and placement of the humeral trials. The measurements were made by measuring the distance between 9 different predetermined locations on the humeral head against a fixed reference point on the lateral aspect of the humerus.
The 9 selected points on the humeral head were as follows: (1) Center of the head, (2) 1 cm Superior to the center [S1], (3) 2 cm Superior to the center [S2], (4) 1 cm Inferior to the center [I1], (5) 2 cm Inferior to the center [I2], (6) 1 cm Anterior to the center [A1], (7) 2 cm Anterior to the center [A2], (8) 1 cm Posterior to the center [P1], and (9) 2 cm Posterior to the center [P2] (Figure 2).
The 9 reference points on the humeral head.
The center of the articular surface of the humeral head was located by measuring the superior–inferior and anterior–posterior axes and locating the midpoint. The fixed reference point was then determined by drilling a 3.2-mm pin through the center of the humeral head, perpendicular to the articular surface, and then bicortically such that the drill created an exit hole on the lateral aspect of the humerus (Figure 3). The exit hole location was then used as the fixed reference point. The k-wire pin was removed from the bone while the measurements were then taken.
A 3.2-mm wire was drilled bicortically through the humerus to create the reference location on the lateral aspect of the humerus.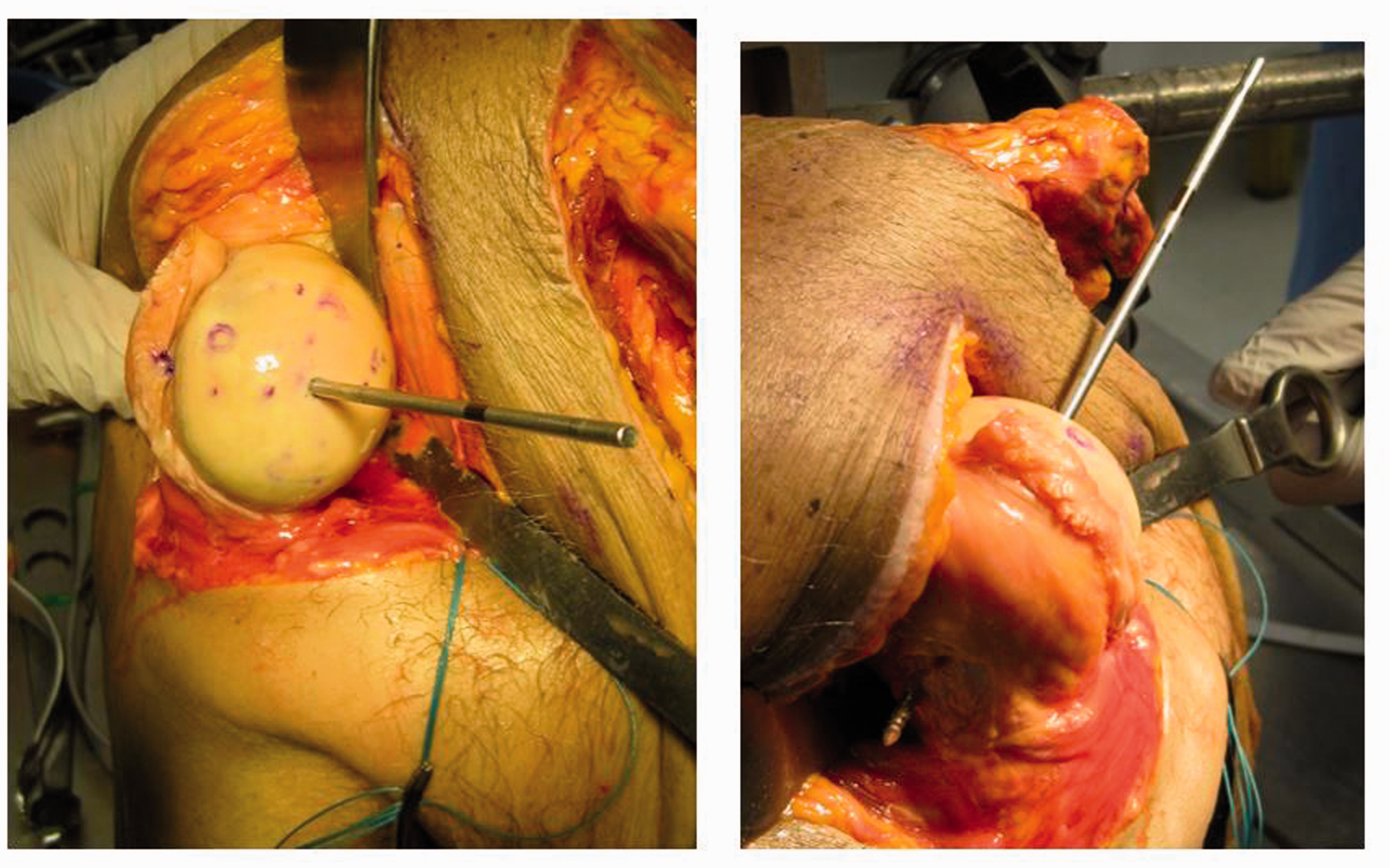
A set of digital point calipers (Fowler, Newton, MA) was used to measure the distance from the reference point to each of the individual 9 reference point on the humeral head (Figure 4). These same measurements were then repeated on the selected humeral trial (Figure 5) after performing the surgical procedure using the standard instruments and according to the surgical technique guide. The sequence of bone cuts involved in humeral preparation are as follows: anterior and posterior osteotomy with a cutting guide, superior and inferior osteotomy with a second cutting guide, drilling 4 parallel holes for the implant pegs, and inserting a trial implant (Figure 6). The humeral trial that most closely matched the peripheral contour of the cortical bone was selected as the proper size. The final location of the implant will remain in the center of the articular surface because the first cutting guide attaches to the central pin, fixing its location, and subsequent steps are referenced along the pin’s axis.
Measurements being made on the native head. Measurements being made on a humeral trial. Humeral head preparation steps (a) anterior and posterior osteotomy, (b) superior and inferior osteotomy, and (c) drilling holes for implant pegs.


The absolute difference between the preoperative and postoperative distance measurement of each surface point from the fixed reference point was calculated, and statistical analysis was performed using paired t-tests with a statistical significance level of P ≤ .05. The measurement error was determined by calculating the average intraobserver error at repeated locations.
Results
Absolute Changes in Joint Line Location at Each Measured Point (mm).
Discussion
The results of this study demonstrate that surgeons can perform shoulder arthroplasty reproducibly with minimal change in the location of the articular surface when using a multiplanar osteotomy surgical technique that is referenced by the center point location rather than the humeral canal. Five surgeons, each without significant prior experience with this system but with significant arthroplasty experience, were all able to maintain each of the 9 points on the humerus to under 3.0 mm deviation in all specimens, with an average change in location below 1.0 mm.
This is the first study we are aware of that used multiple points on the humeral head relative to a fixed reference point on the humerus to measure the location of the proximal articular surface and the amount it changes after arthroplasty reconstruction. The advantages of this method are its ability to measure the accuracy of implant placement in 3 dimensions rather than just on a 2-dimensional plot or radiograph and its technical simplicity, which would make future studies easy to perform in this manner. The limitations are that this technique is unable to calculate for change in inclination or COR for each specimen, and that there is not a direct comparison of this measurement method to other previous reported literature. In addition, by using just 1 reference point on the lateral humerus, the implant in theory could be rotated in space anywhere relative to the fixed point and still result in an acceptable measurement. However, the cutting blocks are attached to and maintain the orientation of the initial central guide pin, and the final implant location is fixed along this axis. In addition, though minor, there is a slight error in measurements using handheld point calipers.
Numerous authors have demonstrated that accurate implant placement is crucial for restoring normal glenoid kinematics and motion5–8 and others have shown worsening clinical outcomes and higher rates of pain and implant loosening when implants were not placed anatomically.1–3 This is such an important concept that it essentially was the primary inspiration for creating the implant under investigation in this study.
Hertel et al. 9 evaluated proximal humerus geometry and came to the following 4 conclusions, each of which were incorporated into the design of the implant utilized in this study. First, the implant placement should respect the supraspinatus insertion. The implant design has a recessed superior margin to remain 3 to 5 mm away from the most medial fibers of the supraspinatus. Second, Hertel et al. concluded that a constant head inclination is adequate. The surgical technique utilizes a standard 135° inclination reference instrument, but the surgeon has unlimited ability to change if necessary. Third, only 1 head height per radius is required, and the system follows this. Fourth, the capability for adjustment for medial offset is mandatory. By not having a stem, medial or posterior offset is fully adjustable. In addition, the system utilizes a nonspherical, elliptical humeral head which more closely matches the anatomy of the proximal humerus.9–11
Other authors have shown good results restoring normal anatomy with stemmed devices on radiographic analysis of cadavers. 12 The creation of stemmed implants with variable inclination and offset has improved computer models 13 compared to previous studies, 14 with the exception of scenarios with extreme offset. However, in vivo data from living patient studies have not been as enthusiastic. Alolabi et al. 4 evaluated the postoperative radiographs of 125 stemmed arthroplasties and measured the humeral head COR change after surgery compared to preoperative, using a validated best-fit circle method of 3 nonarticular humeral head landmarks. In that study, 31% of stemmed arthroplasties had a change in COR >3.0 mm, a deviation they considered clinically significant based on biomechanical studies. This 3.0-mm COR deviation was the basis for our benchmark of 3.0 mm of articular surface change considered an outlier in the present study.
In theory, resurfacing implants and implants without a canal stem should have better implant placement than stemmed implants; however in that same study, 4 the ability to restore COR was even worse for resurfacing prostheses, with 65% of all implants measured as outliers postoperatively. Mechlenburg et al. 15 also evaluated resurfacing implant radiographs in the Danish arthroplasty registry and determined that traditional resurfacing implants overstuff the shoulder joint. We propose that the worse COR restoration is due to 2 factors, both shortcomings in implant design and technique.
First, though previous resurfacing design can theoretically improve implant placement, they currently offer little help to the surgeon control for implant height. Depth of reaming is variable, not calibrated, and not correlated with implant size, leading to a common finding of overstuffing after surgery. Second, the use of spherical prostheses forces the surgeon to choose between matching the superior–inferior humerus size, leading to overhang of the implant, or matching anteroposterior, leading to undersizing in the coronal plane. The nonspherical head shape may make implant selection more simple and may be a significant contributing factor to the positive results in this study.
Malpositioned implants can have significant effects. Implants placed inferiorly effectively raise the greater tuberosity and predispose the patient to impingement. 8 Implants placed too high can put stress on the rotator cuff as the supraspinatus tendon has to traverse around the superior margin of the implant. Franta et al. 3 reported in their study of 282 unsatisfactory shoulder arthroplasties, 64% had component malpositioning. Denard et al. 1 reported in follow-up of total shoulder arthroplasty in young patients, when the humeral head was not positioned anatomically, the glenoid was 6.6 times more likely to require removal. Although not studied in this article, malpositioning of the glenoid component can also lead to problems. Using finite element analysis, Hopkins et al. 16 reported that glenoid malalignment placed superior, inferior, or in retroversion increased the probability of mechanical failure of the cement mantle.
Each of the multiple surgeons involved in this study demonstrated the ability to accurately reproduce the anatomy without any significant outliers and without a large learning curve. The limitations of the study include the measurement technique using handheld digital calipers rather than CT scanning. In addition, the specimens used had mild or no arthritic changes, and restoration of anatomy could prove more challenging if significant deformity was present. The effect of cement thickness under the final implant also was not factored into this study.
In addition, this study focused solely on the humerus. Implantation of a glenoid prosthesis also has similar potential to alter the joint line and lead to overstuffing. This can independently put increased tension on the rotator cuff, leading to subscapularis rupture and also alter joint mechanics. Thicker glenoid implants are more likely to lateralize the joint line. 17 Further clinical studies are warranted to validate the findings of this study.
Conclusion
Using a nonspherical implant design and multiplanar osteotomy technique, surgeons were able reconstruct proximal humerus geometry with high accuracy, reproducibility, and precision, even without experience on the system. While these cadaver data represent a small sample, arthroplasty systems matching bone removal directly to implant thickness may reduce overstuffing, allow improved range of motion, and prove to have significant advantages over traditional variable surgeon-dependent bone removal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven S Goldberg is a paid consultant, stock and stock options in Catalyst OrthoScience. Ephraim Akyuz is a paid consultant. Theodore Blaine is a Stock and stock options in Catalyst OrthoScience.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven S Goldberg is a paid consultant, stock and stock options in Catalyst OrthoScience. Ephraim Akyuz is a paid consultant. Theodore Blaine is a Stock and stock options in Catalyst OrthoScience.
