Abstract
Background:
This technique video reviews anatomic total shoulder arthroplasty (TSA) with a stemless humeral component using a representative case example.
Indications:
Stemless TSA is indicated for patients with symptomatic glenohumeral arthritis typically younger than 65 years of age. Stemless TSA may be used for other glenohumeral degenerative conditions such as avascular necrosis or posttraumatic arthropathy, provided acceptable humeral bone quality is confirmed intraoperatively using a thumb-press test. Poor bone quality is the primary contraindication.
Technique Description:
Preoperatively, computed tomography is typically used for planning, and intraoperative navigation through magnetic resonance imaging is also useful. The procedure’s critical elements include beach chair positioning, standard deltopectoral approach with lesser tuberosity osteotomy (LTO), freehand proximal humeral osteotomy, perpendicular proximal humeral exposure and subsequent preparation over a central guidewire, stemless implant and humeral head sizing, perpendicular glenoid exposure and implant placement, and final humeral implantation with LTO repair/fixation. A phased rehabilitation protocol includes 6-week sling immobilization to protect the subscapularis/LTO repair and return to normal activities between 3 and 6 months postoperatively.
Discussion/Conclusion:
Stemless TSA shows promising early and mid-term outcomes with complication rates, including humeral loosening rates, similar to standard stemmed components. Potential complications include neurovascular injury, infection, glenoid or humeral component loosening, and rotator cuff failure.
This is a visual representation of the abstract.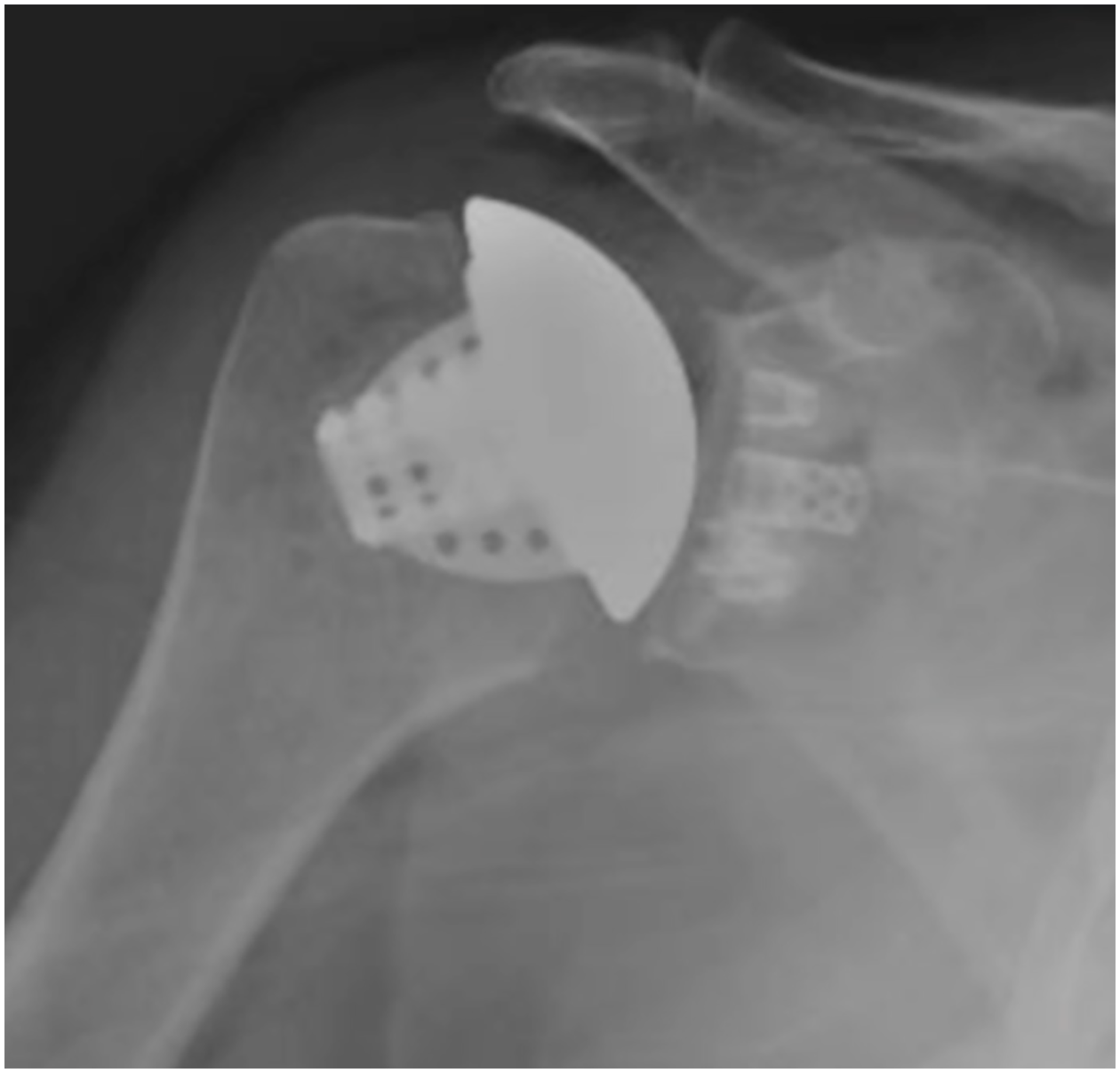
Keywords
Video Transcript
This is a video technique for an anatomic total shoulder arthroplasty (TSA) using a stemless humeral component as performed by Dr Stephen Brockmeier at the University of Virginia. Our disclosures are listed.
We will review the following items:
•Patient history, physical examination, and imaging workup and preoperative planning;
•Patient positioning and preparation;
•Surgical technique;
•Postoperative management strategies and Return to Activities guidelines/criteria; and
•Published outcomes and a concise review of the current literature available on this topic.
The patient is a 61-year-old left hand–dominant woman with a history of degenerative arthritis and worsening right shoulder pain and functional limitations after an injury 8 months prior. She has not improved despite the implementation of conservative management strategies, including an ultrasound-guided injection, and presents for evaluation for joint replacement surgery. This patient is physiologically young, active, and is employed in a labor-intensive occupation. Examination demonstrates restriction in joint motion but intact rotator cuff strength.
Preoperative imaging includes plain radiographs (anteroposterior, Grashey, and axillary views) that demonstrate the absence of glenohumeral joint space, an inferior humeral osteophyte, and mild posterior glenoid bone loss. The patient presented with a prior magnetic resonance imaging (MRI) scan which confirmed these findings and demonstrated an intact rotator cuff. In the presence of the MRI scan, further computed tomographic (CT) imaging was deferred in this patient; however, we commonly do obtain CT imaging for presurgical planning purposes and intraoperative navigation.
Preoperative planning for a patient undergoing anatomic TSA includes both patient-based and pathology-based considerations. Patient age; activity level; overall health; bone density, bone deformity, and joint alignment as demonstrated on advanced imaging such as CT or MRI; and soft-tissue integrity are all critical considerations. In physiologically “young” patients, we have transitioned to using a stemless humeral component for essentially all cases over the past few years. We estimate approximately 70% of anatomic TSA procedures use a stemless component at the current time. Although a CT scan was not obtained in this particular case, it does provide a number of advantages, especially in cases with significant glenoid bone deformity as can be seen in this case example image. The combination of allowing for suitable preoperative templating and planning for component size and positioning, as well as intraoperative navigation and guidance for precise execution of this plan is a significant technical advantage. Outcomes data regarding the use of intraoperative navigation are emerging.
Patient positioning for this procedure is in the standard beach chair position, at between 30° and 60° based on patient habitus as well as surgeon and anesthesia preference. All bony prominences are well padded. We maintain the head in a neutral position with secure alignment of the cervical spine and torso. The knees are bent to avoid prolonged traction to the sciatic nerve. A pneumatic arm positioner is used throughout the procedure to assist with extremity positioning and exposure.
This surgical technique video will demonstrate an anatomic TSA with emphasis specifically on the utilization and placement of a stemless humeral component. As can be seen here, a standard deltopectoral approach has been completed with humeral exposure. Deep retractors have been placed and the subscapularis has been detached using a lesser tuberosity osteotomy. All humeral releases have been completed, including deltoid mobilization and anterior and inferior capsular release to beyond the 6 o’clock position, and humeral osteophytes have been removed. A freehand humeral osteotomy is carried out with an oscillating saw at 30° of retroversion, with appropriate visualization and protection of the rotator cuff tendon attachments. We target an approximate 135° neck angle for the osteotomy; however, this is not rigidly required in the setting of a stemless component. Next, a sizing guide is used to determine the circumference and placement of a central guidewire. Of note, the absence of an intramedullary stem provides the advantages of a technically easier and more efficient humeral preparation, a more anatomic reconstruction using the patient’s individual anatomy, and obviates the need for the use of eccentric heads in this system. The placement of the central guidewire is a critical step as seen here, as the remaining preparation steps all reference this guidewire. For this system, the next step is the determination of the implant depth to be used, which is referenced off of the central pin in the guide. We generally recommend erring to the shorter level of depth to limit the risk of perforation or compromise the metaphyseal cortical bone. Next, a central reamer is used as seen here, followed by a punch to prepare the humeral metaphysis for the implant.
After reaming, it is recommended that the surgeon use the “thumb test” to ensure adequate metaphyseal bone structure and density to support a stemless implant. If the surgeon’s thumb easily pushes into the bone with this maneuver, then consideration should be given to audibling to a stemmed implant. Placement of the punch can position the 3 fins as determined by the surgeon. We prefer to position them as seen here to take advantage of the harder bone in the humeral calcar and region of the bicipital groove.
Once the broach has been impacted into place, attention is turned to glenoid preparation and implantation. Standard glenoid exposure is performed, and we then prepare the glenoid over a central guidewire as seen here. This glenoid component is hybrid in nature with the advantage of a central cage and 3 peripheral pegs. After glenoid preparation is complete, the implant is opened and the cage is packed with bone from the proximal humeral head. Cement is placed on the component and then pressurized into the peripheral sockets. Of note, the central cage is designed for ingrowth, and thus, cement is not used for this socket. The glenoid is then impacted into place with care taken to ensure perpendicular impaction, which is critical. Excess cement is removed. Here, our final glenoid component is viewed.
Next, the humerus is again exposed and head sizing is carried out. As previously delineated, concentric heads are used in a stemless system, with the most attention centered on appropriate head circumference and width. Any excess residual osteophytes are removed. We generally recommend erring toward the smallest appropriate head width as determined by trialing intraoperatively. The trial broach is removed at this time to reveal our final humeral bone preparation; note the limited amount of bone removal required. At this time, the arm is internally rotated and sutures are placed for our lesser tuberosity osteotomy (LTO) fixation. Three drill holes are created in the bicipital groove, and #5 high-strength sutures are placed for subsequent LTO fixation. The sutures are passed into the area of humeral prep and then subsequently linked to the humeral component as will be demonstrated. Here is our final implant; this is impacted into position, partially, and then the sutures are individually linked to the implant before final placement. A similar fixation technique can be used for other subscapularis management techniques, such as a subscapularis peel, based on surgeon preference. Note the purchase of this implant in the patient’s bone, which is secure and anatomic. The previously sized humeral head component is now placed and impacted into position. The joint is reduced and appropriate tensioning and balance are demonstrated with anterior-posterior play to the front and back of the glenoid component, no instability, and appropriate retensioning of the rotator cuff. Next, our subscapularis and LTO repair is carried out with 3 cerclage sutures placed in a modified Mason-Allen technique around the lesser tuberosity fragment and anatomic reduction of this fragment before tensioning. Here, the final repair is demonstrated with excellent stability and range of motion. Retractors are removed and a layered closure follows.
Our postoperative radiographs demonstrate anatomic joint reconstruction and suitable alignment in both plains.
We have moved toward the outpatient setting for most shoulder replacement procedures in appropriate patients. Postoperative rehabilitation involves sling immobilization for 6 weeks; a supervised physical therapy program is initiated immediately with a graduated program. During the first phase, protective range is used to protect the subscapularis repair. Active motion is initiated at 6 weeks with subsequent strength progression with a return to low-demand sports at 3 months and moderate-demand and high-demand sports between 4 and 6 months.
The overall rate of complications after shoulder arthroplasty has been reported to be around 15%, with severe complications such as neurovascular injury and infection being a very small subset of these. Long-term complications such as loosening of the glenoid component have been reported as a predominant mode of failure in anatomic TSA. Of note, humeral component loosening is an uncommon complication of traditional stemmed TSA. Importantly, studies to date of stemless TSA have not demonstrated any increased risk of humeral loosening or failure.
Reported outcomes for stemless anatomic TSA have been quite encouraging, with comparable results to standard stemmed TSA at early follow-up in the United States and mid-term and long-term follow-up in Europe and elsewhere. Some early data suggest additional potential benefits for stemless TSA may include shorter operative times and decreased blood loss. Some surgeons anecdotally have suggested stemless TSA may lead to less perioperative pain and potentially favorably impact successful return to functional activities.
Our references are listed. Thank you.
Footnotes
Submitted October 16, 2020; accepted December 29, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.F.B. is a consultant for Exactech, Arthrex, Zimmer Biomet, and Workforce Rx; a speaker for Exactech, Arthrex, and Zimmer Biomet; receives clinical trial and/or research funding from Zimmer Biomet and Arthrex; and receives publishing royalties from Springer. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
