Abstract
Background
During shoulder arthroplasty with substantial bone and soft tissue loss, reverse shoulder arthroplasty (RSA) with a tumor prosthesis may restore function, reduce pain, and improve implant fixation.
Methods
Thirteen adult patients undergoing RSA using a tumor prosthesis system were retrospectively reviewed. Preoperative visual analog score (VAS), single assessment numeric evaluation (SANE), American Shoulder and Elbow Surgeons (ASES) score, simple shoulder test (SST), and forward flexion were compared to latest follow up. Postoperative radiographs and complications were recorded.
Results
Mean age at surgery was 68.4 years. Eight patients had undergone at least 1 prior operation on the indicated shoulder. Six patients required wide excision of proximal humerus tumor. At mean of 34 months postoperatively, significant improvements were noted in VAS (P = .03) and ASES score (P = .04). Active forward elevation was 81.1 degrees. For all patients, postoperative radiographs demonstrated satisfactory alignment. Complications occurred in 38% of patients, with 31% requiring reoperation.
Conclusion
In cases of failed shoulder arthroplasty with excessive bone and soft tissue loss or substantial tumor burden, RSA with a tumor prosthesis can reduce pain levels and improve functional outcomes. However, forward elevation remains limited, and postoperative complications are a concern.
Keywords
Introduction
Over the past decade, the use of reverse shoulder arthroplasty (RSA) has increased, while the indications for its use have continued to expand. 1 Initially indicated primarily for management of rotator cuff tear arthropathy, 2 RSA is increasingly used for the treatment of acute, complex proximal humerus fractures with rotator cuff deficiency, proximal humeral malunions or nonunions, inflammatory arthritis, revision shoulder arthroplasty, or tumors of the proximal humerus.2–6 In the settings of revision arthroplasty with substantial proximal humeral bone loss, proximal humerus fractures with severe comminution, or extensive tumor burden, soft tissue stabilizers and their bony attachments are frequently disrupted. Additionally, surgeons are faced with the challenge of achieving appropriate implant fixation due to limitations in the host bone.7–9 To restore function in cases when proximal bone stock is limited, various tumor prostheses have been developed, incorporating a modular design and a long humeral stem to allow for flexibility during component insertion and improved implant stability at the bone-implant interface. 10
Tumor prostheses have demonstrated promising outcomes during limb salvage procedures of the shoulder,7–9 and their use is favored over the use of osteoarticular allografts or allograft-prosthesis composite reconstruction due to concerns of nonunion and graft failure.11–15 However, investigations have also reported high rates of postoperative complications following tumor prosthesis implantation, including infection, component loosening, instability, and periprosthetic fractures.4,9,11 RSA may improve joint instability and shoulder function. 5 While limitations and complications have been previously described, few studies to date have evaluated functional outcomes following RSA tumor prosthesis implantation, especially in the setting of revision arthroplasty or tumor reconstruction. The purpose of this study is to report patient reported outcomes (PROs), active range of motion (ROM), and the incidence and timing of complications in patients treated using 1 of 2 shoulder tumor endoprosthetic systems.
Methods
Patient Selection
Following Institutional Review Board approval, a retrospective case series was conducted for patients treated with revision arthroplasty or tumor reconstruction using 1 of 2 tumor prosthesis systems (Comprehensive Segmental Revision System, Zimmer Biomet; Exactech Equinoxe Humeral Reconstruction Prosthesis, Exactech, Inc.). Inclusion criteria consisted of patients aged 18 to 89 undergoing shoulder arthroplasty with 1 of the 2 specific tumor implant systems between January 2013 and October 2019. Specific indications for the use of the tumor prosthesis were proximal humeral bone loss past the level of the medial calcar or bone loss that would otherwise require allograft to achieve appropriate deltoid tension. Exclusion criteria were patients younger than 18 or older than 89 years at the time of surgery, and patients treated using another nonsegmental RSA implant system. The selection of the 2 specific tumor implant systems in this investigation was based on surgeon preference and the specific implant design which provides humeral lateralization. Potential patients for inclusion were identified using validated current procedural terminology (CPT) corresponding to “repair, revision, and/or reconstruction procedures on the shoulder” (CPT 23412) and “introduction or removal procedures on the shoulder” (CPT 23350).
Patient Assessment
For patients meeting inclusion criteria, electronic medical charts were reviewed, and the following demographic data was recorded: patient age, sex, hand dominance, laterality and number and type of prior operations. Prior to surgery, all patients were evaluated by the senior author in the office. The following data was recorded: indications for the use of an RSA tumor prosthesis, active forward elevation, pain level (assessed using visual analog score [VAS]), and subjective functional status using single assessment numeric evaluation (SANE), American Shoulder and Elbow Surgeons (ASES), and simple shoulder test (SST) scores. Operative reports were assessed to extract the following data: estimated blood loss (EBL), implant system used, concomitant procedures performed, and soft tissue reconstruction. The deltoid insertion was preserved, and a soft tissue reconstruction of the rotator cuff and deltoid musculature was performed whenever possible. The technique for soft tissue repair was consistent regardless of whether the patient required a tumor resection. In all cases, cement was used for fixation of the Exactech prostheses, while the Biomet stem was implanted via either a cemented or a cementless press-fit technique. For patients requiring wide proximal humeral resection due to substantial tumor burden, a fellowship-trained orthopedic oncologist assisted in the operation to perform tumor debridement. Postoperatively, radiographs were obtained in the postanesthesia care unit with the shoulder immobilized in an abduction sling. Motion was limited to the elbow and wrist for 4 weeks following surgery. After 4 weeks, active assisted range of motion was permitted. At 8 weeks, full range of motion exercises were incorporated, and patients were advanced to muscle strengthening based on standardized physical therapy protocols.
Patients were instructed to follow up in clinic at 2 weeks, 6 weeks, 3 months, 6 months, and then annually for monitoring. During each follow up appointment, the incidence of any surgery-related complications was recorded, while active forward flexion was recorded beginning at 6 weeks postoperatively. Radiographs were taken at 2 weeks, 6 months, and annually postoperatively to evaluate for implant alignment and signs of component loosening. PROs (VAS, SANE, ASES, and SST scores) were obtained at final patient follow up.
Postoperative forward flexion and PROs were calculated and compared to preoperative values using a 2-tailed T-test. Statistical significance was set at a P-value < .05 was considered significant. Complications and reoperations were described for each individual patient. Statistical analyses were performed using Microsoft Excel (Microsoft Excel for Office 365).
Results
Patient Demographics
A total of 13 shoulders in 13 patients were identified, consisting of 10 females and 3 males (Table 1). Mean age at time of surgery was 68.4 years (range, 39-88 years). The left shoulder was the operative shoulder in 54% of patients (n = 7 of 13), while the dominant arm was involved in 38% of patients (n = 5 of 13). Sixty-two percent (n = 8 of 13) had undergone at least 1 prior operation to the indicated shoulder, with an average of 2.25 (range, 1-4) prior operations. The Exactech tumor prosthesis was used in 10 patients while 3 patients received the Biomet tumor prosthesis.
Preoperative Patient Demographics.
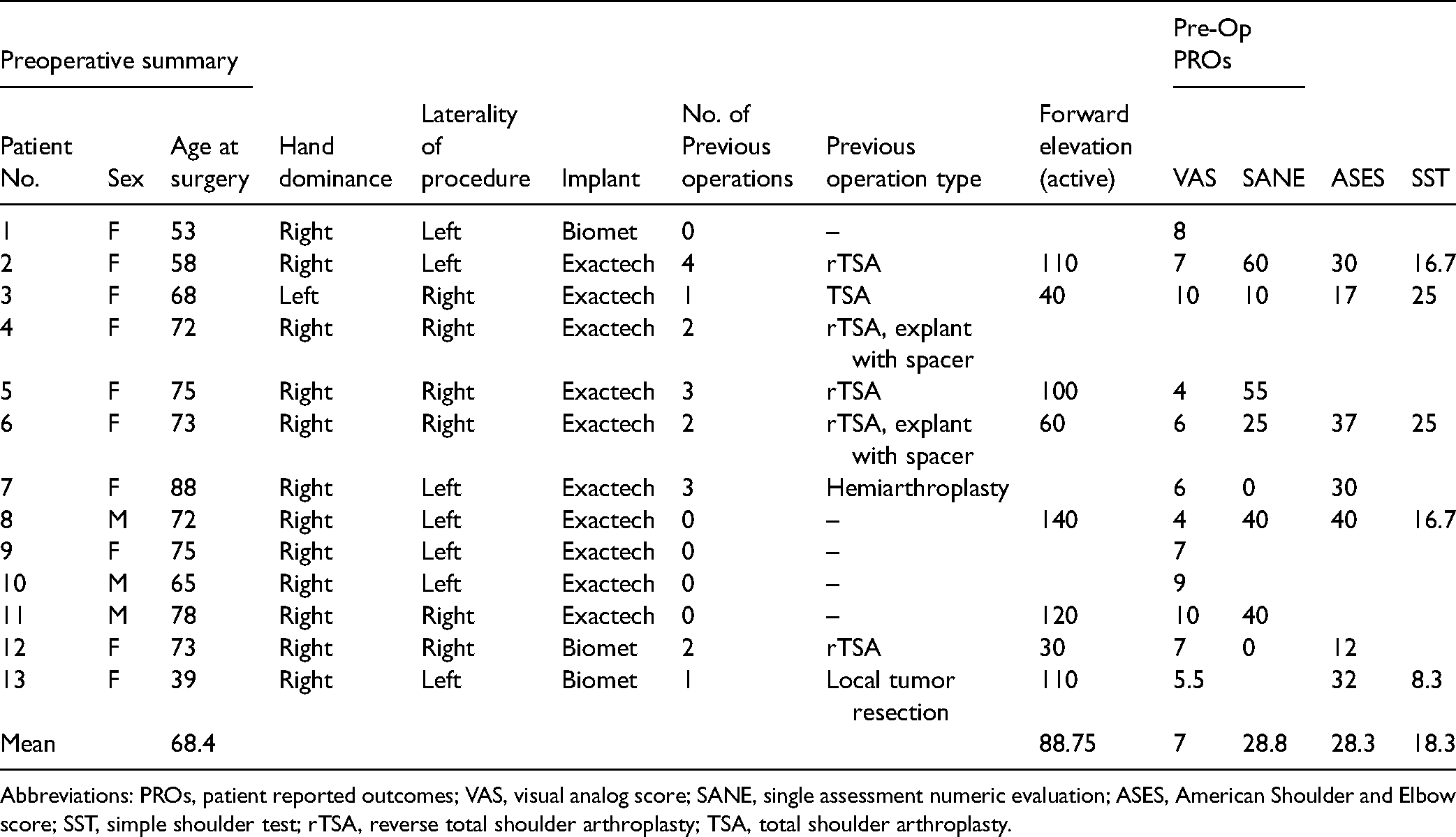
Abbreviations: PROs, patient reported outcomes; VAS, visual analog score; SANE, single assessment numeric evaluation; ASES, American Shoulder and Elbow score; SST, simple shoulder test; rTSA, reverse total shoulder arthroplasty; TSA, total shoulder arthroplasty.
Preoperative Evaluation
Preoperatively, VAS scores were available for 12 patients with an average pain score of 7.0 (range, 4-10). SANE scores were available for 8 patients with an average score of 28.8 (range, 0-60), ASES scores were available for 7 patients with an average score of 28.3 (range 12-40), and SST scores were recorded for 5 patients with an average score of 18.3% (range, 8.3%-25%) (Table 1). Forward elevation was measured in 8 patients, with an average measurement of 89° (range, 30-140°).
Operative Findings
Operative indications for each patient and concomitant procedures are listed in Table 2. All patients underwent arthroplasty under general anesthesia. The average EBL for patients was 277 mL (range, 50-650 mL). The average length of reconstruction was 117 mm (range, 59-189 mm). The deltoid insertion was preserved in 8 patients, and repair of the rotator cuff and/or deltoid musculature was conducted in 9 patients. No intraoperative complications were reported. Six patients who presented preoperatively with malignant tumors to the proximal humerus underwent wide tumor excision prior to insertion of the tumor prosthesis. Immediate postoperative radiographs demonstrated appropriate alignment of the tumor prosthesis in all patients with no suspicion for intraoperative fracture in any of the shoulders.
Intraoperative Findings.

Abbreviations: TSA, total shoulder arthroplasty; rTSA, reverse total shoulder arthroplasty; RCT, rotator cuff tear; RCC, renal cell carcinoma.
Postoperative Evaluation
At the time of final follow-up, 3 patients had died from conditions unrelated to their shoulder operation and were unable to be assessed beyond 2 weeks postoperatively (n = 2 patients) or 6 weeks postoperatively (n = 1 patient). All 3 patients had received a tumor prosthesis for tumor involvement of the proximal humerus. Nine patients returned for routine clinical and radiographic follow-up at a mean of 35 months postoperatively (range, 12-69 months) following surgery. One patient was unable to return to clinic for evaluation beyond 6 weeks postoperatively although PROs at 19 months postoperatively were recorded via telephone.
Active forward flexion at 6 weeks, 3 months, 6 months, and final follow-up are recorded in Table 3. Of the 6 patients with both preoperative and postoperative measurements, active forward elevation was found to increase by a mean of 4°at final follow-up with no significant relationship between preoperative measurements and postoperative outcomes (P = 1.0). Only 2 patients demonstrated any improvement in active forward elevation at final follow-up. During postoperative rehabilitation, active forward elevation was noted to significantly improve between 6 weeks and 3 months postoperatively (P = .02), with no significant change observed beyond 3 months. Furthermore, no significant association was found between soft tissue repair and postoperative forward elevation (P = .34).
Postoperative Forward Flexion Active Range of Motion.

Individual results of PROs are reported in Table 4. At final follow-up, when compared to preoperative values, mean VAS, SANE, SST, and ASES improved by 1.9 points, 18.6%, 12.5%, and 16.9 points, respectively. Improvement in VAS (P = .03) was both statistically and clinically significant while improvement in ASES (P = .04) was statistically significant only, and improvements in SANE (P = .18) and SST (P = .16) were not significant. 16
Postoperative Outcomes.
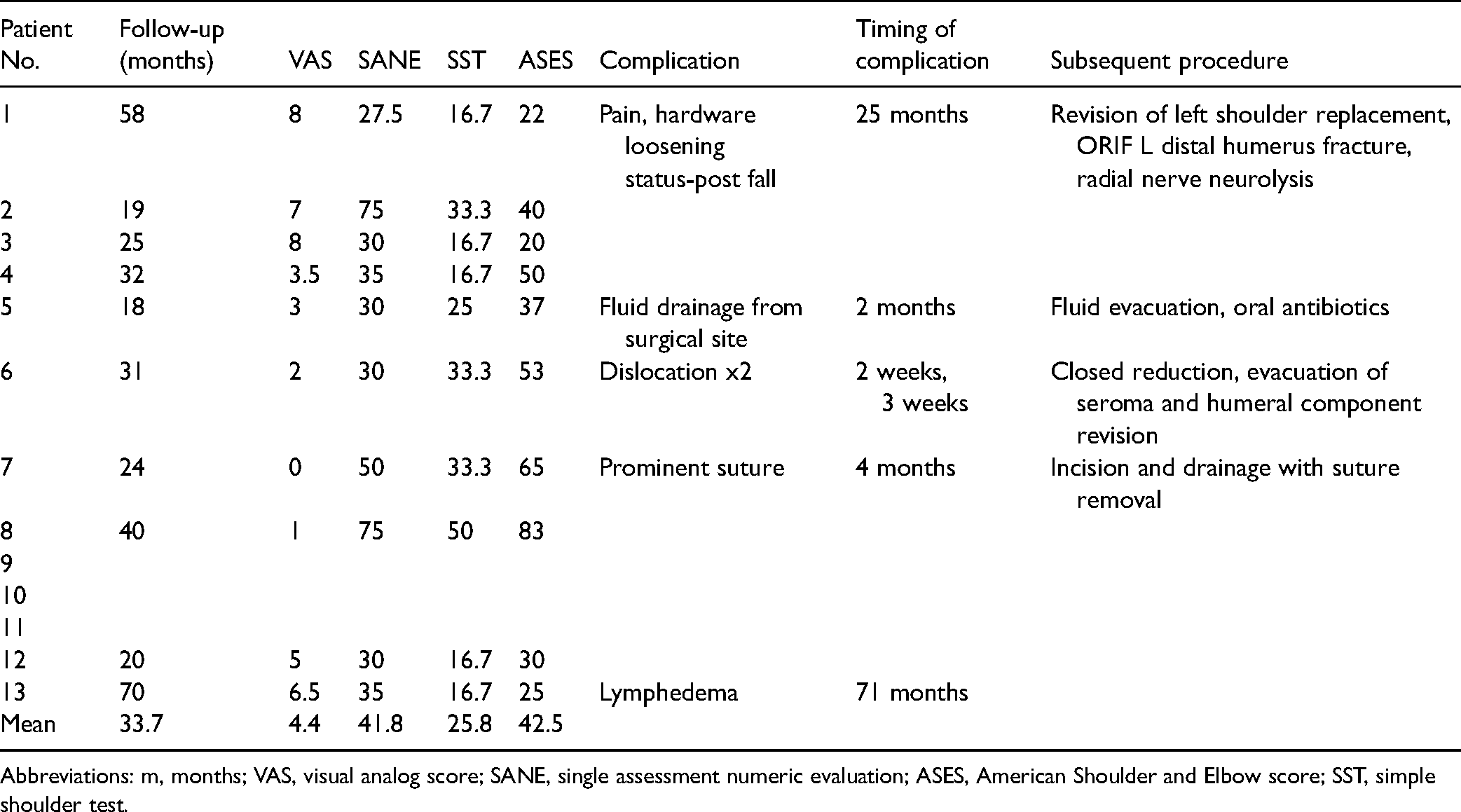
Abbreviations: m, months; VAS, visual analog score; SANE, single assessment numeric evaluation; ASES, American Shoulder and Elbow score; SST, simple shoulder test.
Radiographic Evaluation
Radiographs were obtained for all 13 patients at 2 weeks postoperatively and for 9 of 10 living patients at final follow-up at a mean of 35.3 months postoperatively (Figure 1). At 2 weeks postoperatively, 11 shoulders demonstrated satisfactory alignment with no evidence of component loosening or concerns for peri-prosthetic fracture. One shoulder demonstrated a grossly dislocated implant which was initially managed with closed reduction under anesthesia and later required revision of the humeral component with insertion of a constrained liner. A second shoulder demonstrated a small cortical fracture at the base of the humeral stem, with no evidence of component loosening. This patient was treated with continued observation and demonstrated no evidence of fracture extension at latest follow-up of 32 months postoperatively. At final follow up, all 9 shoulders demonstrated satisfactory alignment with no evidence of component loosening.

Postoperative anterior-posterior (AP) radiographs of right shoulder. (A) AP radiograph of right shoulder immediately postoperatively. (B) AP radiograph of the right shoulder 2 weeks postoperatively. (C) AP radiograph of the right shoulder 6 months postoperatively.
Complications and Reoperations
A total of 38% (n = 5 of 13) experienced a postoperative complication, with 31% (n = 4 of 13) of patients requiring a subsequent procedure. One patient suffered a fall at 25 months postoperatively, sustaining a peri-prosthetic fracture which required implant revision, open reduction with internal fixation of the distal humerus, and neurolysis of the radial nerve (Figure 2). A second patient reported wound drainage and subjective instability at 2 months postoperatively, requiring incision and drainage followed by oral antibiotics; cultures taken at the time of surgery were negative. Implant dislocation occurred twice in 1 patient at 2 and 3 weeks postoperatively, first managed with closed reduction and later requiring evacuation of a seroma and revision of the humeral component with insertion of a constrained liner. In this patient, the deltoid insertion was compromised, and a soft tissue repair was unable to be performed during the initial operation. Following revision surgery, the patient resumed normal postoperative course with no further complications. Overall, there was no association between preservation of the deltoid insertion and implant stability (P = .19). A 4th patient complained of a painful prominent suture which required surgical removal at 4 months postoperatively. This patient had been previously placed on suppressive antibiotic therapy due to an initial infection in the shoulder prior to implantation of the tumor prosthesis and remained on antibiotics following suture removal. Another patient treated for soft tissue sarcoma was noted to have painful lymphedema at final follow-up, however no surgical intervention was indicated, and medical management was pursued with improvement.

Radiographic imaging of periprosthetic fracture. (A) Preoperative AP view of shoulder with periprosthetic fracture. (B) Preoperative lateral view of humerus with periprosthetic fracture. (C) AP view of shoulder with tumor prosthesis status-post open reduction with internal fixation. (D) Lateral view of shoulder with tumor prosthesis status-post open reduction with internal fixation.
Discussion
The results of this study demonstrate that the RSA tumor prostheses functions as both a primary procedure following tumor resection and as a salvage procedure for failed shoulder arthroplasty where bone stock is limited in the proximal humerus. While patients had significant improvements in VAS and ASES scores, active forward flexion was limited with no patient achieving forward elevation beyond 100 degrees at final follow-up. Moreover, 38% of patients experienced a postoperative complication, with 31% requiring a subsequent procedure. As such, appropriate patient selection and indications, with a full understanding of the risks and limitations inherent to implant use, are essential prior to utilization of a tumor endoprosthesis.
The use of hemiarthroplasty, or less commonly RSA, with a tumor prosthesis implant system has been previously documented for the treatment of oncologic lesions of the proximal humerus.3,9,17–20 In the setting of substantial tumor burden, wide resection is required to ensure complete removal of malignant tissue from the surgical bed to lower the risk of local recurrence. 7 At a mean follow-up of 11.5 years, Raiss et al 9 reported an overall survival rate of 45% in an original cohort of 39 patients who received a hemiarthroplasty with a tumor prosthesis, with local recurrences requiring interscapulothoracic amputation noted in 2 cases. A total of 18% (n = 7 of 39) shoulders experienced complications with 13% (5 of 39) requiring reoperation including 4 dislocations, 2 deep infections, and 1 implant loosening. Moreover, in a cohort of 13 patients requiring an RSA tumor prosthesis following local tumor resection, Guven et al 3 reported 2 deaths due to systemic oncologic disease, while no local recurrence was identified in the remaining patients at a mean follow-up of 18.2 months. Complications were reported in 23% (n = 3 of 13) of cases including 2 dislocations, both of which required reoperation and 1 patient who complained of inferior instability which was managed conservatively. In our study, 6 patients received an RSA tumor prosthesis for indications related to metastasis to the proximal humerus. Postoperatively, 2 complications were reported among oncologic patients including one implant loosening secondary to a fall which required surgical intervention and one complaint of lymphedema managed conservatively. While 3 patients died within months following the operation due to complications unrelated to their shoulder surgery, the remaining 3 patients demonstrated no evidence of local recurrence at latest follow-up. Given the morbidity and high rate of complications associated with the use of a tumor prosthesis in oncologic patients, patient prognosis, pain, and functionality must be considered when evaluating patients as potential candidates.
When comparing 2 cohorts of a similar size, the complication rate in our study was higher than that reported by Guven et al (38% vs 23%). The mean follow-up was substantially longer in our study than that reported by Guven et al 3 (35 months vs 18.2 months) allowing for greater time for longer term complications to arise. Furthermore, our investigation additionally included patients undergoing revision surgery with substantial bone and soft tissue loss. In revision cases, lack of bony and soft tissue structures at the time of the operation poses additional challenges to achieving implant stability, establishing deep wound closure, and restoring function. Among patients undergoing revision surgery, 1 patient suffered 2 postoperative dislocations, 1 required removal of a prominent suture, and 1 had wound drainage requiring incision and drainage followed by antibiotic therapy. However, given the limited cohort size in this investigation, an evaluation of the clinical outcomes in patients receiving an RSA tumor prosthesis for revision surgery compared to those requiring wide tumor resection was unable to be performed. Therefore, future studies are necessary to directly compare clinical outcomes with these 2 indications.
At latest follow-up, mean forward elevation was 81° among 9 patients, with no patient achieving active forward elevation greater than 100°. Inherent to its design, RSA introduces an altered center of rotation about the glenohumeral joint. 21 In the setting of arthroplasty with massive bone loss, disruption of soft tissue stabilizers and bony attachment sites results in changes in shoulder biomechanics necessary for appropriate shoulder abduction and elevation, leading to limitations in range of motion among patients requiring a tumor prosthesis. Our results are consistent with previously published literature with well-documented restrictions in abduction and forward elevation described in multiple studies using a tumor-type prosthesis.18,19,22–24 In a study including 28 patients who received either a traditional tumor prosthesis or a reverse tumor prosthesis, Wafa et al 23 reported a maximum active forward elevation of 60°. Similarly, Cannon et al 19 found mean active forward elevation of 42° among 63 patients who received a traditional tumor prosthesis. When specifically examining clinical outcomes of a reverse tumor-type prosthesis, Guven et al 3 and Streitbeurger et al 25 demonstrated slightly better results with a mean forward elevation of 96° in 11 patients and 84° in 18 patients, respectively. Of note, all 4 oncologic patients in this study available for ROM evaluation achieved active forward elevation of 80° or greater, compared to only 2 patients undergoing revision surgery. In cases of revision surgery, failed preservation of surrounding soft tissue structures and the bone for soft tissue attachments may contribute to this decrease in active forward flexion. Additionally, significant improvements in ROM were demonstrated between 6 weeks and 3 months postoperatively with no major changes in active forward elevation beyond 3 months. Given significant improvements during this timeframe, targeted therapy programs aimed at improving ROM beginning at 6 weeks may be beneficial to restoring functional status. Based on the data from this study, utilization of the RSA tumor prosthesis demonstrated favorable ROM outcomes when compared to studies reporting on the use of hemiarthroplasty for patients with massive proximal humeral bone loss. Namely, in a retrospective review of 8 patients who received a hemiarthroplasty following malignant tumor resection of the proximal humerus, Moran and Stalley 26 reported a mean active forward flexion of 40°. Similarly, Grosel et al 27 reported a significant decrease in mean forward flexion when comparing 38 patients treated with a hemiarthroplasty (mean, 28°) to 10 patients treated using a Biomet RSA tumor prosthesis (mean, 85°) following wide tumor excision (P < .001). No significant differences were appreciated PROs or rates of complication or dislocation between treatment groups. 11
While limited ROM is expected with the tumor prosthesis, previous studies have demonstrated surgical success measured by improvements in pain scores and PROs.3,4,18,23,28 Among our patients, mean VAS score was found to improve by 3.7 points following surgery, a statistically significant improvement when compared to preoperative VAS score. Shukla et al 4 reported similar results with statistically significant improvements in pain scores by 3.5 points on a scale of 0 to 5 from preoperative measurements (P < .001). Despite overall improvements in PROs following surgery for patients in the current study, postoperative outcomes were generally poor, with less than half of maximum points achieved for average measures of ASES (47.5/100), SANE (40.3%), and SST (30.8%). In contrast, Shukla et al 4 reported an 81% satisfaction rate in patients postoperatively, and Wafa et al 23 reported an average Musculoskeletal Tumor Society functional score of 83% at latest follow-up. Guven et al 3 also demonstrated slightly better results with a mean Constant-Murley score of 53.7%. The lower postoperative PROs reported in the current investigation may be partially explained by lower preoperative status, specifically demonstrated by higher preoperative VAS scores in our patients compared to those reported by Shukla et al 4 (7.0 vs. 4.1 respectively), decreasing the ability of patients to achieve satisfactory results. However, further investigations are warranted to better understand differences in PRO based on surgical techniques, implants and indications.
Due to high levels of mechanical stress placed on the tumor prostheses and prosthetic-bone interface in the setting of substantial bone loss and soft tissue deficits, segmental replacing prosthetic reconstruction is prone to high complication rates. Mechanical complications, including dislocation, aseptic loosening, periprosthetic fracture, and failed attachment of soft tissues to the prosthesis are of major concern, with reported incidence as high as 48% in some studies.4,22,25,28 In our investigation, 2 patients experienced clinically significant mechanical complications including 1 dislocation and 1 peri-prosthetic fracture with stem loosening. In the remaining patients, radiographs obtained at 2 weeks and 6 months postoperatively demonstrated satisfactory results with no evidence of fracture or component loosening. Overall, this represents a 23% rate of mechanical complications. Prosthesis dislocation and implant migration have been associated with inadequate deltoid tensioning and lack of rotator cuff attachment, as these structures are necessary to provide additional stabilization.4,7,18 In a study by Guven et al 3 examining clinical outcomes of RSA with a tumor prosthesis following tumor resection, 2 patients experienced postoperative instability. Both patients underwent open reduction and prosthesis augmentation with remnant rotator cuff fibrous tissue, joint capsule and the deltoid muscle with no recurrent instability reported at latest follow-up. In the current study, one case of recurrent dislocation occurred in a patient whose deltoid insertion was compromised in the initial operation and a soft tissue repair was unable to be performed. Revision of the humeral component with a constrained liner resulted in improved stability. However, deltoid tension must be evaluated at the time of surgery, and soft tissue repair should be achieved whenever possible.
In the setting of revision surgery, decisions surrounding humeral stem fixation also play a role in implant stability. 4 Specifically, Shukla et al 4 reported that in 50% of cases (n = 2 of 4) complicated by humeral stem loosening, an uncemented prosthesis was placed following the removal of a cemented stem. Based on their findings, the authors recommended cementing the humeral stem in any revision case where cement was previously used in the humeral canal. In the current study, cement was used for all Exactech prostheses, while 2 of 3 of the Biomet implants were inserted by press-fit. Nevertheless, all implants were stable at 6 months regardless of fixation technique. Despite recent innovations in shoulder arthroplasty, massive humeral bone loss continues to pose challenges to surgeons during reconstruction procedures. Lack of bone stock and destruction of surrounding soft tissue stabilizers raises concerns for reconstruction failure due to component loosening or soft tissue instability. 29 However, long-term outcomes and complications in patients undergoing tumor prosthesis implantation remain limited and warrant further investigation.
Limitations
This investigation is not without limitations. Due to the small number of cases in which a tumor prosthesis is indicated, the sample size was limited to 13 patients, with only 10 patients available for final follow-up. Furthermore, a mean follow-up time of 35 months postoperatively may be too short to appreciate some complications associated with the long-term use of a tumor prosthesis. Additionally, as a single-center case series, patient demographics and surgical outcomes may not be reflective of general characteristics or postoperative results in a broader patient population performed across multiple institutions. However, this review adds to a limited pool of data describing outcomes of an RSA tumor prosthesis for shoulder arthroplasty while evaluating both patients receiving a tumor prosthesis for the treatment of tumors and during revision arthroplasty.
Conclusion
Following the use of 1 of 2 tumor prosthetic shoulder arthroplasty systems in 13 patients, statistically significant improvement in VAS and ASES scores were appreciated at final follow up despite limited improvements in postoperative forward flexion. Complications including mechanical errors due to implant placement and nonmechanical complications such as infection are of concern and must be carefully monitored. Soft tissue repair should be attempted whenever possible to reduce risk of instability. Overall, reverse tumor prostheses represent a valuable tool for shoulder arthroplasty in the setting of failed shoulder arthroplasty with excessive bone and soft tissue loss or substantial tumor burden. Further investigations are required to better understand long-term outcomes and implant longevity.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
