Abstract
Background
While initially developed to improve function in patients with chronic rotator cuff deficiency, the success of reverse total shoulder arthroplasty (RTSA) has led to expanding indications including fracture sequelae and revision of failed hemi- or total shoulder arthroplasty. While primary RTSA carries complication and revision rates of up to 75% and 2%–10%, respectively, these rates are higher in RTSA performed in a revision setting.
Methods
The current literature on RTSA and revision RTSA was reviewed to determine the most common complications and treatment strategies to address these complications.
Results
The most common complications leading to revision of RTSA are instability, infection, humeral implant loosening, and glenoid implant loosening. Each of these presents unique technical challenges for the surgeon. Retention or replacement of a reverse prosthesis affords high patient satisfaction and better functional outcomes than resection, but may require several additional surgeries. In cases in which reverse components cannot be implanted, salvage options, including resection arthroplasty and conversion to hemiarthroplasty, provide satisfactory pain relief but limited function.
Discussion
As the number of RTSA performed increase, so will the number of revisions. In this article, current treatment strategies for addressing the challenges of revision reverse shoulder arthroplasty are discussed.
Introduction
Reverse total shoulder arthroplasty (RTSA) has dramatically altered the field of shoulder reconstruction. The success of RTSA in improving function in the absence of a functioning rotator cuff has led to a broad range of applications, including massive rotator cuff tear, failed hemi- and anatomic total shoulder arthroplasty (TSA), fractures and their sequelae, and tumor reconstruction.1,2 Enthusiasm for this technique has been tempered by the highly reported complication rate. Early reported RTSA complication rates ranged widely from 0% to 75% with revision rates varying from 2% to 10%.3–5 The complication and reoperation rates tend to be higher in the revision setting with up to 22% of RTSA performed in the revision setting requiring further revision.3–7
Revision of a failed RTSA can be technically demanding due to bone loss and soft tissue deficiency. Outcomes following revision RTSA are not well documented. The few published case series suggest that, while poorer than outcomes following primary RTSA, outcomes following revision RTSA are superior to alternative salvage strategies.7–10
Preoperative Evaluation and Planning
A thorough preoperative evaluation is crucial when considering revision RTSA. Preoperative evaluation should include diagnostic imaging (radiographs which include the entire prosthesis, CT scan, MRI, and bone scan). In cases of instability, ultrasound can be useful in assessing subscapularis tendon integrity. Laboratory studies [complete blood count (CBC), erythrocyte sedimentation rate (ESR), and C reactive protein (CRP)] and joint aspiration should be obtained to rule out infection. Electromyography and nerve conduction studies are important when there is a concern for axillary nerve injury.
It is important to determine the exact prosthesis that is being revised, which often requires obtaining the operative note from the primary surgery. All of the instruments and implants from the manufacturer for both the prosthesis to be revised and the planned new prosthesis should be obtained. Revision instruments should be available at surgery. Often bone graft is required and the patient should be consented for both autograft and allograft.
Revision Technique
The typical approach for revision RTSA is a deltopectoral approach, which allows for complete access to the glenohumeral joint, rotator cuff, and associated structures. The deltopectoral approach may be extended distally to expose the humeral shaft in case of osteotomy to remove the prosthesis followed by cerclage reconstruction. An anterosuperior approach may be used where a previous anterosuperior approach has been used and extension of the incision is unlikely. When access to the posterior rotator cuff is required, a combined approach may be used, with the deltopectoral incision curved laterally to allow for a deltoid split approach.
Antibiotics are held in all revision cases until intra-operative specimens are obtained. Intra-operative frozen sections with nucleated cell counts of greater than 5 cells per high-power field are suggestive of infection. Cultures should be held for 2–3 weeks to rule out proprionobacterium acnes infection. Intra-operative antibiotics are given after cultures have been obtained.
It is often difficult to establish tissue planes in revision surgery. The coracoid process is a reliable guide and may be used to locate the rotator interval. After entering the rotator interval, the subscapularis tendon, if present, is either peeled or divided, or the lesser tuberosity insertion is osteotomized to gain access to the glenohumeral joint. The axillary nerve should be palpated and preserved throughout the approach.
Once the glenohumeral joint is entered, the prosthesis is exposed and assessed for position and stability. A stable, appropriately positioned, not infected stem does not require removal. In cases in which stem must be removed, bone loss should be minimized. Thin osteotomes can be used to break up the implant-bone or bone-cement interface within the metaphysis. If the stem cannot be dislodged, a humeral osteotomy is performed. Multiple techniques have been described in the literature for stem removal. Our preferred technique is the creation of an anterior episiotomy along the length of the stem, stopping just short of the stem tip (Figure 1(A)–(D)). For revisions of cemented stems, osteotomes can be inserted through the episiotomy site to create a longitudinal split in the cement mantle. If the stem is well fixed after creation of the episiotomy, the osteotomy can be extended transversely at the distal aspect to create a full, retractable cortical window.
A 62-year-old man s/p humeral head replacement arthroplasty for fracture with pain and inability to raise the arm. AP (A) and lateral (B) radiographs showing tuberosity resorption in a well-fixed cemented humeral prosthesis. AP (C) and lateral (D) radiographs after staged resection and removal of the prosthesis by humeral osteotomy and cerclage wiring. After infection was ruled out and osteotomy healed, reimplantation of a reverse total shoulder arthroplasty was performed. Immediate postoperative AP radiograph shows stable prosthesis (E). Subsequent AP radiograph (F) shows dislocation of the humeral prosthesis. Revision was performed with soft tissue repair, retention of the prosthesis, the addition of a metal spacer, and prosthetic lengthening. Final AP (G) and lateral (H) radiographs 6 months postoperatively show stable revision RTSA reconstruction.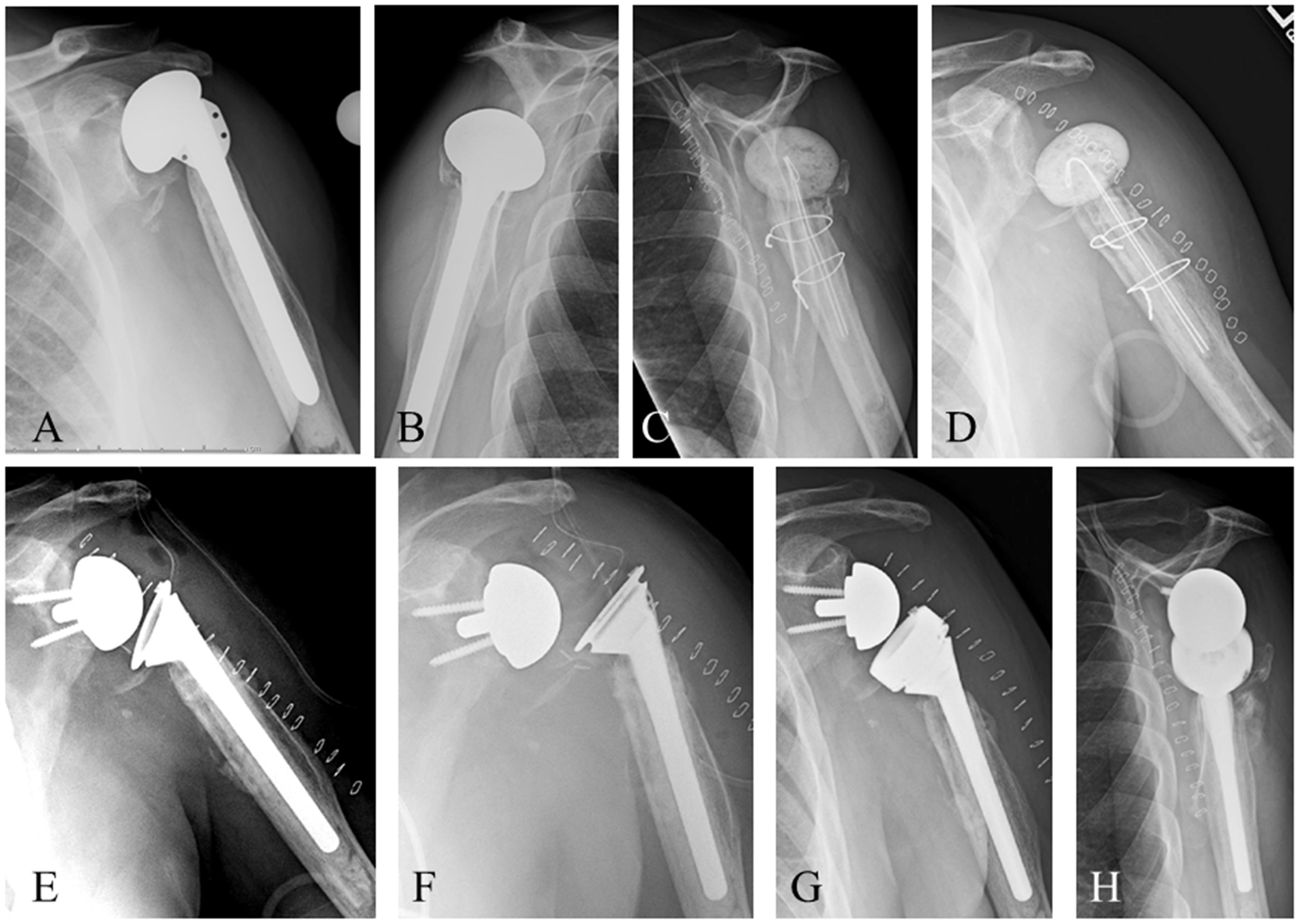
Cerclage wires or cables are carefully placed around the osteotomy site to recreate an intact cylinder prior to reaming and/or placement of the revision implant. The radial nerve should be protected during passage of cerclage wires/cables. Allograft struts may be added for thin or deficient diaphyseal humeral bone. In cases where infection is suspected, cement must be removed in its entirety. If there is no suspicion of infection, reamers can be used to penetrate the distal mantle to allow for placement of a longer stem and the revision implant may be cemented into the existing cement mantle. The revision stem should be extended 2.5 cortical diameters distal to the osteotomy site (Figure 1(E)–(H)).
Once the humeral stem is removed, the glenoid can be directly accessed. Removal of the glenoid component should be as bone preserving as possible. In cases of revision for failed TSA, the all-polyethylene glenoid component can be amputated at the implant-bone interface (Figure 2(A)–(C)). Curettes can be used to remove polyethylene plugs with minimal destruction to remaining bone. Removal of metal-backed TSA glenoid implants or glenoid baseplates can be more challenging but care should be taken to preserve glenoid bone.
A 68-year-old man s/p anatomic total shoulder arthroplasty with persistent anterior shoulder pain and subscapularis deficiency. AP (A) and axillary (B) radiographs show stable total shoulder arthroplasty with an uncemented humeral prosthesis and a cemented pegged glenoid component. Revision RTSA was performed with removal of both components and reimplantation of a long-stem humeral component. Postoperative radiograph (C) 1 year post revision show stable revision RTSA.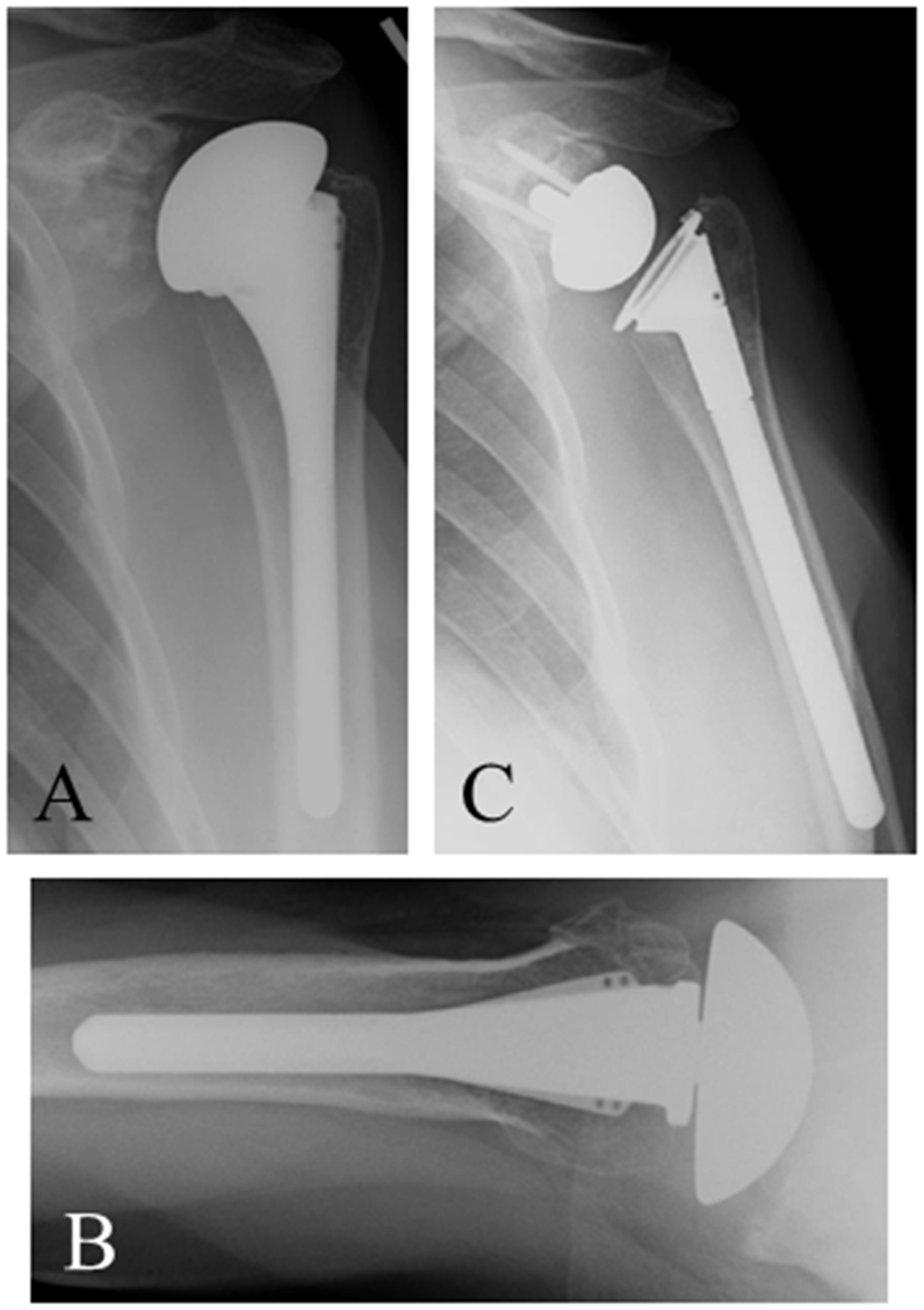
Challenges in Revision
Revision RTSA requires adequate glenoid bone stock for stable implantation of the glenoid component, sufficient proximal humeral bone to allow fixation of a humeral implant, and tension within the soft tissue envelope to enhance the stability of the implant. The surgeon must address deficiencies in any of these requirements during the revision procedure. Several strategies exist for addressing such deficiencies.
Glenoid Bone Loss
Glenoid bone loss may occur as a result of severe scapular notching, osteolysis around a loose glenoid component, or deficiency following removal of a well-fixed glenoid component. Glenoid erosion may also have been present at the primary procedure due to arthritis or prior surgery.
A preoperative CT scan of the affected shoulder is often helpful in predicting the amount and location of glenoid bone loss. Metal subtraction CT scan is useful in determining bone loss when a metal prosthesis is present. The best characterization of bone loss occurs intra-operatively once existing components have been removed, and fibrous tissue has been debrided. The most widely used glenoid bone loss classification system describes the location and severity of bone deficiency intra-operatively after component removal. 11
In cases of mild bone loss, altering the orientation of the glenoid component to take advantage of intact bone may provide sufficient stability. Klein et al. 12 have described placement of the baseplate’s central screw along the scapular spine centerline, as opposed to the line perpendicular to the face of the glenoid, with no difference in complications such as stress fractures. This strategy bypasses smaller defects and takes advantage of the intact bone deep within the glenoid vault for baseplate fixation. Most implant companies offer baseplates with longer central posts or screws to take advantage of intact native bone deeper within the vault.6,13 Some companies also offer larger diameter peripheral locking screws, which allow for improved purchase in bone where smaller diameter screws had been placed previously. 14
Larger bone defects almost always require bone grafting to recreate a stable bony platform on which to implant the baseplate (Figure 3(A)–(H)). In cases of central bone loss, the intact glenoid rim allows for impaction grafting of cancellous bone to fill the defect.15,16 With larger central defects, a structural corticocancellous graft, with the cortical portion set laterally, can be used to prevent compression of the graft and medialization of the glenoid component.
16
A 63-year-old man with posttraumatic glenohumeral arthritis. AP (A) and axillary (B) radiographs, with corresponding axillary (C) MRI image show severe posterior glenoid wear (Walch B2 glenoid). Intra-operative photographs show deficient posterior glenoid (D). Glenoid reconstruction required autograft from the humeral head (E) fashioned to match the posterior glenoid defect and is pinned in position before glenoid baseplate implantation. Intra-operative photograph (F) shows the long post glenoid baseplate implanted and securing the autograft reconstruction with 2 additional screws. Intra-operative radiograph (G) confirms correct position. Post RTSA radiograph (H) shows reconstruction of the glenoid with uncemented RTSA prosthesis.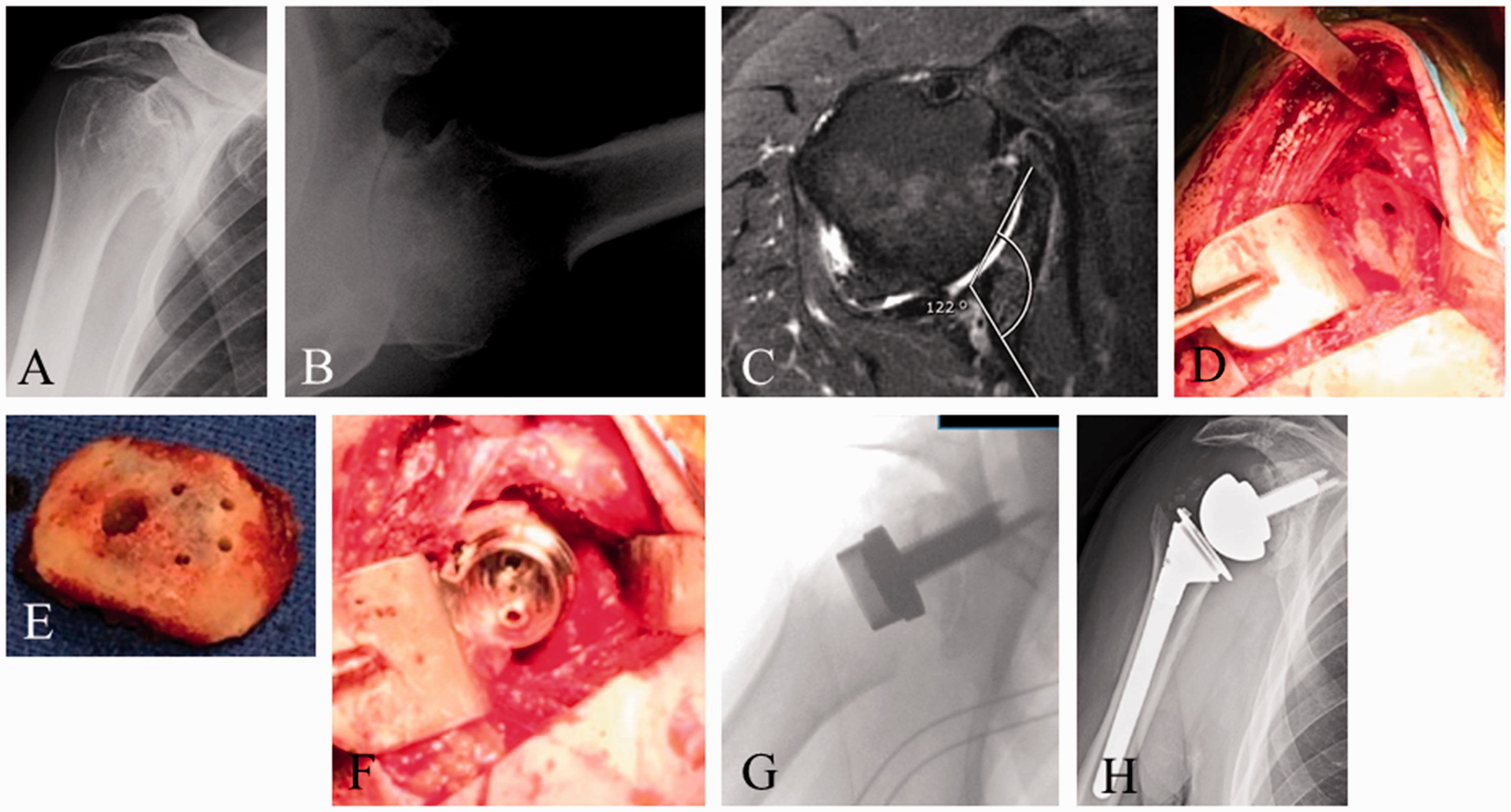
Peripheral defects are technically challenging. Cases of asymmetric superior or posterior glenoid bone loss requiring less than 15° of correction may be addressed with eccentric reaming, during which the asymmetrically elevated rim is preferentially reamed to create a flat glenoid surface onto which the baseplate is implanted. 17
Larger peripheral defects typically require reconstruction with structural graft.12,14,15,18,19 Common sources of graft material in revision cases include iliac crest autograft and fresh-frozen femoral head allograft. Shaping the graft to fit the defect can be challenging and is typically done by assessing the defect and hand-shaping the graft material to match. Iannotti et al. 20 have described creating a mold of the defect using semicured, moldable polymethylmethacrylate cement to more accurately shape the graft to fill the defect. Kelly et al. 13 have created a biocomposite graft by implanting the baseplate into a prepared section of the patient’s ipsilateral iliac crest, which was then removed en bloc, shaped, and implanted into the glenoid defect.
The graft is secured to the native glenoid by taking advantage of the baseplate’s in-growth surface, central peg or screw, and peripheral screws. The central peg or screw must engage the native glenoid bone for good stability of the construct. Some companies offer revision glenoid components that feature baseplates with attached plates and additional screw holes to improve fixation of the graft. 18 When it is not possible to secure the graft with the available baseplate fixation points, the graft may be fastened with separate wires or screws and placed strategically to avoid interference with baseplate peg and screw orientation.
When the glenoid can be reconstructed with stable implantation of the baseplate, the revision can be performed in a single stage. When this is not possible, the revision should be performed in 2 stages, with bone grafting occurring first and placement of the glenoid component during a second surgery after the graft has incorporated. Melis et al. 6 have described an alternative technique where the glenoid component is implanted to help fix the graft during the first stage and the humeral component is implanted during the second surgery, protecting the glenoid reconstruction from the shear stresses of humeral articulation as the graft incorporates.
The long-term fate of structural grafts used in glenoid reconstruction is not clear. Iannotti et al. 20 have reported a series where glenoid defects were reconstructed with fresh-frozen femoral head without implantation of a new glenoid component. Of the 5 cases that were later revised, 4 had sufficient bone graft incorporation to undergo glenoid component fixation. Scalise et al. 21 have reported on 11 patients who underwent glenoid reconstruction with allograft chips or structural allograft during revision for failed TSA. At a minimum of 2 years postoperatively, all patients demonstrated substantial graft subsidence.
Proximal Humeral Bone Loss
Proximal humeral bone loss is fairly common during revision shoulder surgery.22,23 Resorption of tuberosities following operative treatment of proximal humerus fractures may be the indication for revision. Loss of proximal humeral bone also occurs as a result of infection, tumor, and osteolysis from polyethylene debris. Proximal humeral bone may be iatrogenically destroyed in the process of removing a well-fixed humeral prosthesis.
Loss of proximal humeral bone introduces several challenges to shoulder reconstruction. Lack of bony support raises concerns about the long-term stability of an implanted humeral prosthesis. In a biomechanical study, Cuff et al.
24
have studied the torsional stability of reverse shoulder humeral components. All failures occurred in modular implants (
Proximal humeral bone loss also results in the loss of soft tissue attachment sites and altered deltoid kinematics due to changes in the line of pull and/or tension on the deltoid. These 2 factors may negatively impact the stability and function of RTSA.22,23,25,26 Use of a proximal humeral allograft-reverse shoulder prosthesis composite (APC) has been suggested as a solution to proximal humeral bone loss, restoring proximal bone to shield the implant from rotational stresses, providing soft-tissue attachment sites for subscapularis repair, and recreating the lateral contour of the shoulder to improve the trajectory and tension on the deltoid (Figure 4(A)–(F)). Use of proximal humeral allograft has the added benefit of restoring proximal humeral bone stock to improve options for possible future surgery. These benefits must be weighed against the potential downside of allograft use, including increased cost and complexity of the procedure, risk of infection, graft resorption/fragmentation,
27
allograft nonunion,28,29 and allograft failure
26
(Figure 5(A)–(H)).
A 57-year-old man with multiple myeloma of the left proximal humerus. Preoperative AP (A) and axillary (B) radiographs show proximal humeral destruction. Intra-operative photographs show resection of the tumor with rotator cuff attachments (C) and reconstruction with an alloprosthesis (D, E). Six months postoperative AP (F) and lateral (G) radiographs show RTSA reconstruction with a cemented long-stem humeral prosthesis. Graft-host bone junction is visible. A 57-year-old man with aggressive giant cell tumor of the proximal humerus. Preoperative radiographs (A, B) and coronal view MRI scan (C) demonstrate aggressive tumor eroding lateral humeral cortex. After resection of the tumor, alloprosthetic reconstruction was performed with a long stem hemiarthroplasty. Postoperative AP (D) and lateral (E) radiographs show the allograft secured to host bone with a derotation plate and screws. Radiographs 6 months (F) and 1 year postoperatively show progressive resorption of the allograft. Revision RTSA was performed with removal of the hardware and alloprosthesis. Post-RTSA radiographs (G, H) show stable revision reconstruction with a long-stem stackable revision prosthesis.

The surgical technique for reconstruction with proximal humeral bone loss begins with removal of existing hardware or implants. Judging the amount of bone loss and the appropriate height of the reverse humeral component can be difficult. Preoperative full-length radiographs of the contralateral humerus may be compared with pre- or intra-operative radiographs of the operative side to get an estimate of the amount of proximal bone loss. Alternatively, the appropriate height of the implant can be judged intra-operatively by joint stability and range of motion. Cuff et al. 24 have recommended 5 cm of bone loss as the threshold for consideration of allograft use.
To reconstruct with an APC, a transverse cut is made in the remaining native humerus to create a good interface accepting the allograft. The remaining diaphyseal bone is reamed to allow for an appropriate cement mantle. The baseplate and glenosphere are implanted, which help gauge the height of the humeral implant. A trial stem is placed into the canal. Placing a sponge down the canal before introducing the stem will often provide enough friction between the stem and the canal to hold the stem at a set height and version for trialing. After a trial liner is placed and the shoulder reduced, stability and range of motion is tested to determine whether the humeral implant should be lengthened or shortened. Once satisfied with the height of the trial stem, the distance from the shoulder of the implant to the cut surface of the diaphyseal bone is measured to determine the appropriate length of the allograft.
Proximal humeral allograft preparation includes an anatomic neck cut with the desired degree of inclination and version and diaphyseal and proximal reaming according to the technique for the particular implant being used. A transverse cut is made through the allograft diaphysis to recreate the length measured previously.
Different fixation techniques for the APC have been described. The implant can be cemented into the allograft first and the composite then cemented into the native diaphysis separately. Additional hardware fixation, such as plate and screws, can be added to supplement compression between the allograft and native bone (Figure 5(D), (E)). Alternatively, the allograft can be fixed to the native bone first. Chacon et al. 23 have described a technique involving a step-cut in the allograft to create a lateral shelf of diaphyseal bone to overlap the native humerus, allowing placement of cerclage cables to fix the allograft to native bone prior to cementing the stem in place. A revision-length stem is often needed to adequately engage the native diaphysis to ensure stability of the construct and should extend at least 2.5 humeral diameters distal to the osteotomy site for adequate stability and fixation.
Care must be taken to maintain the appropriate version on the allograft and humeral implant during implantation. The soft tissue attachments from the allograft may be used to repair tendons and to improve stability and function. During closure, the native subscapularis, if present, can be repaired to the allograft subscapularis tendon stump. Rotator cuff and deltoid attachments may also be used for repair.
Chacon et al. 23 have reported on their series of revision RTSA with use of APC for proximal humeral bone loss. Of the 90 patients, 25 underwent RTSA for failed arthroplasty required augmentation with structural proximal humeral allograft. At a minimum of 2 years postoperatively, all patients demonstrated statistically significant improvements in all clinical measures; 21/25 patients had incorporation of the allograft, 1 had graft resorption, and 2 had graft fragmentation in the metaphyseal region.
There are no studies that directly compare outcomes of revision RTSA with and without use of APC in the setting of proximal humeral bone loss. Budge et al. 22 have published their results in a series of patients with proximal humeral bone loss revised to RTSA without allograft augmentation. They noted significant humeral bone loss in 15/56 patients undergoing revision to RTSA for failed arthroplasty. The authors noted excellent clinical and functional outcomes and were satisfied with the torsional and bending stability of the humeral implants without augmentation, but recommended use of a monoblock humeral implant design.
Instability/Dislocation
Dislocation is one of the most common complications following RTSA and most dislocations occur early, within the first few months after implantation. Zumstein et al. 3 have performed a systematic review of 21 cohort studies with a minimum 2 years follow-up, collecting data on intra- and postoperative problems, complications, reoperations, and revisions. Instability was the most common postoperative complication, with a mean incidence of 4.7% and was found more frequently in the revision setting than in primary arthroplasty.
Soft tissue deficiency, inadequate deltoid function, and component malposition are important factors in postoperative instability.
Subscapularis insufficiency is a controversial cause of RTSA instability. Although some argue that the subscapularis tendon should not be repaired to allow improved motion following RTSA, others report improved shoulder stability with subscapularis repair. Trappey et al.
30
have performed a retrospective review of 212 primary and 72 revision RTSA. The rate of instability was similar between the primary and revision groups, but patients with an irreparable subscapularis tendon at surgery had a higher rate of instability (
Inadequate deltoid function, due to poor deltoid tension restoration, deltoid muscle deficiency, or axillary nerve injury, is a known cause of RTSA instability. Most of these injuries are neuropraxic and will recover with time. Revision RTSA performed in the setting of an axillary nerve injury has a high risk of postoperative instability. In rare cases, we have performed RTSA in the setting of axillary nerve injury; however, it is important to maximize other determinants of stability including humeral lengthening, center of rotation lateralization, and use of a retentive prosthesis and larger glenosphere. In general, axillary nerve injury and deltoid dysfunction is a relative contraindication to RTSA and revision RTSA.
Malposition of the prosthetic components can cause RTSA instability. Excessive retroversion of the humeral prosthesis can cause impingement of the humeral cup on the anterior glenoid (Figure 6(A)–(D)). Excessive anteversion of the humeral component can cause impingement on the posterior glenoid. Impingement may lever the humeral cup off the glenoid and dislodge the humeral cup from the glenosphere. This type of impingement may be worsened with medialization of the glenoid. Laterizalition of the glenoid baseplate and/or glenosphere may help avoid impingement. Another option is to increase the diameter of the glenosphere.
A 80-year-old man s/p RTSA for rotator cuff arthropathy. Two-week postoperative radiographs demonstrated dislocation of the prosthesis and closed reduction was performed. Subsequent 6 week postoperative AP (A) and lateral (B) radiographs show persistent anterior dislocation of the prosthesis. Revision surgery was performed and excessive retroversion (60°) of the humeral prosthesis was noted leading to anterior impingement of the humeral cup on the anterior glenoid neck. The humeral prosthesis was retained and the position of the cup revised to 10° of retroversion. Postrevision RTSA radiographs (C, D) show stable prosthesis and restoration of stability and function.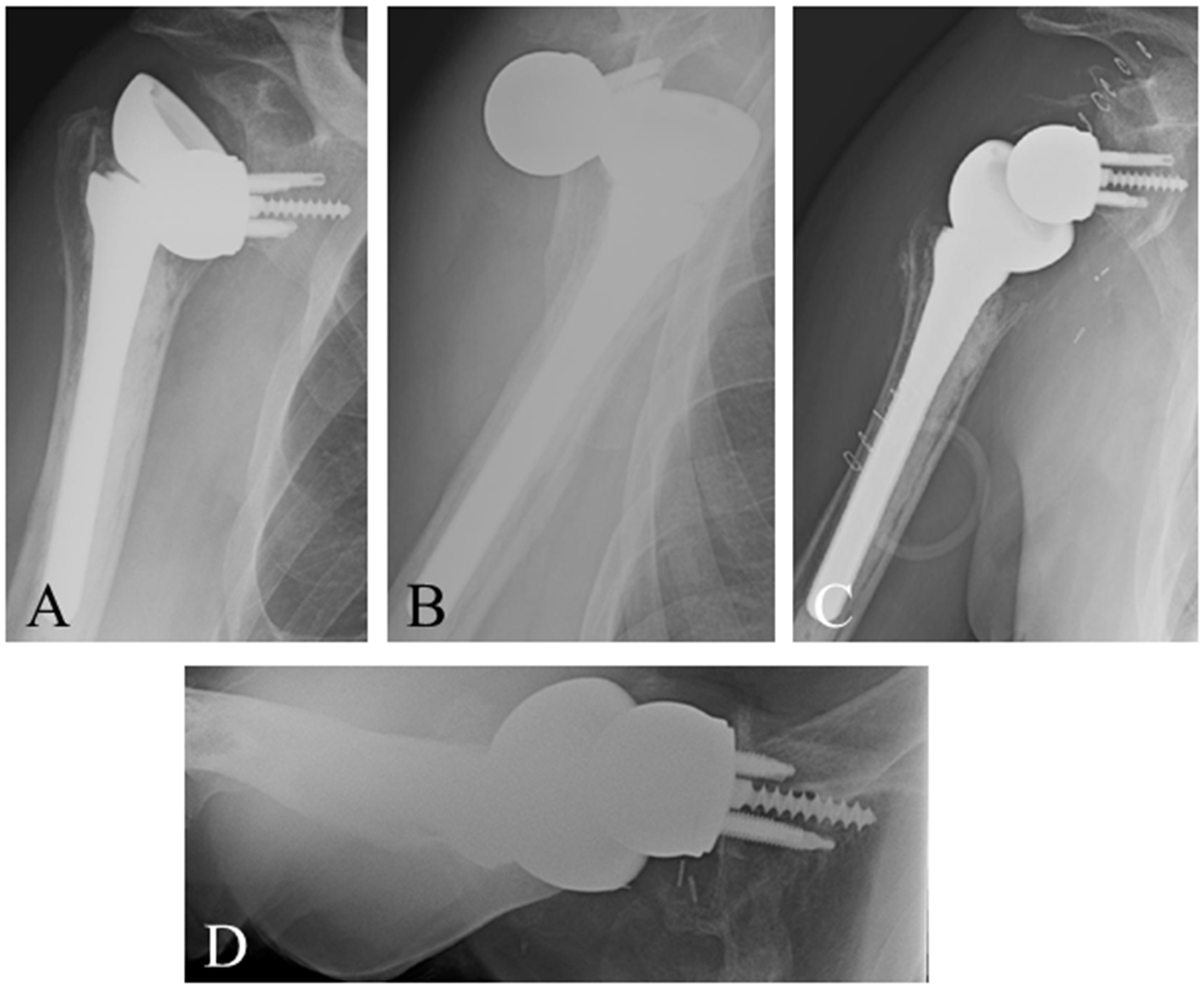
Treatment of RTSA instability can be challenging. Infection must be ruled out as instability may be accompanied by infection in ∼50% of cases. 31 If the dislocation is discovered early, the prosthesis may be reduced through closed or open reduction. This situation is rare and, in the majority of cases, there is usually an issue that needs to be addressed through revision surgery. Revision of surgery with retention of components may involve removing scar tissue and soft tissue impingement, repairing the subscapularis tendon, increasing the size of modular components, and changing the version or lateralization of modular components. Adding spacers or increasing polyethylene size improves deltoid tensioning and stability. Malpositioned components should be removed and revised to appropriate position.
Outcomes Following Revision Reverse
Outcomes following revision in which an RTSA implant remains are good. 32 Boileau et al. 1 have studied 37 patients undergoing revision in which the prosthesis was completely or partially exchanged or removed. In the 32 patients who retained an RTSA, constant scores improved significantly with acceptable active anterior elevation and 89% of patients were satisfied or very satisfied.
Salvage Options
In some cases, revision RTSA is not possible and salvage options, including hemiarthroplasty and resection arthroplasty, should be considered. Conversion to hemiarthroplasty may be facilitated by a convertible prosthesis that does not require humeral stem removal. Challenges posed by conversion with prosthesis retention include an inability to change the version of the humeral prosthesis and soft tissue tensioning. Gamradt et al. 33 have reviewed 6 patients who underwent revision to hemiarthroplasty after RTSA. All patients had good pain relief but poor function, with average forward elevation 42.5°.
When a hemiarthroplasty is not possible, resection arthroplasty is another salvage option. Muh et al. 34 have reported their series of 26 patients who underwent resection arthroplasty for failed shoulder arthroplasty and had reliable pain relief but poor function postrevision.
Conclusion
The indications and usage of reverse shoulder arthroplasty continue to expand, with revision RTSA occurring more often. The most common indications for revision RTSA are instability, infection, humeral implant loosening, and glenoid implant loosening. Surgical challenges include proximal humeral bone loss, glenoid bone loss, and soft tissue deficiency. The few published series suggest revision RTSA leads to high patient satisfaction and improved functional outcomes. In cases where reverse components cannot be implanted, salvage options, including conversion to hemiarthroplasty and resection arthroplasty, provide good pain relief but limited function.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Theodore A Blaine is a consultant for Zimmer Biomet and has equity interest in Catalyst Orthoscience. The remaining authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
