Abstract
Background
Persistent symptoms of autonomic dysregulation are common after COVID-19 infection and may result from alterations in central and/or peripheral autonomic regulatory processes. Traumatic stress can cause persistent alterations in autonomic function, potentially changing the response to future traumatic or physiologic stressors. However, the relationship between prior history of traumatic stress and autonomic symptom burden after COVID-19 infection has not been explored.
Objectives
Examine the potential for additive and/or interactive effects of traumatic stress and COVID-19 infection on autonomic symptom burden, and compare this with other common post-acute sequelae of COVID-19 (PASC) symptom domains.
Design
Observational, self-report, single time-point online assessment.
Participants
404 United States adults with (N = 289) and without (N = 112) a self-reported history of COVID-19 infection.
Main Outcomes and Measures
Autonomic symptom burden (Composite Autonomic Symptom Score [COMPASS 31]), lifetime traumatic stressors (Life Events Checklist), posttraumatic stress disorder (PTSD Checklist-5), self-reported neurocognitive functioning (Neuro-QoL), insomnia (Insomnia Severity Index), and fatigue and pain (PROMIS Fatigue and Pain Interference measures).
Results
Autonomic symptom burden was significantly and positively related to both history of COVID-19 infection and number of probable lifetime traumatic stressors, with probable lifetime traumatic stressors functioning as a positive moderator of the relationship between history of COVID-19 infection and autonomic symptom burden (Cohen's partial f2 = .11, .07 and .02 for COVID history, trauma history and interaction term respectively, all p < .05, in a model also including age and gender). The moderation effect remained significant when adjusting for both current PTSD symptoms and pre-existing multi-system PASC-like symptoms prior to COVID-19. History of traumatic stress and of COVID-19 infection each had significant and positive associations with other PASC symptom domains, but with domain-specific patterns.
Conclusions and Relevance
Prior history of traumatic stress has a positive and interactive effect on symptoms of autonomic dysregulation following COVID-19 infection, independent of PTSD symptoms. This suggests that exposure to traumatic stress may affect the response to future stressors, including physiologic stressors such as COVID-19 infection, through persistent changes in stress-threat response systems. This relationship may provide a physiologic explanation for prior observations that baseline anxiety prior to COVID-19 infection is associated with increased likelihood of PASC.
Introduction
Post-acute sequelae of COVID-19 (PASC), or ‘long COVID’, are common and can be highly functionally impairing. Symptoms are heterogeneous and include fatigue, ‘brain fog’, diffuse pain, sleep disturbances, and heart palpitations. 1 Many, but not all, implicate alterations in autonomic regulatory functions. 2
Similar constellations of complex multi-system symptoms have been described in other contexts, including posttraumatic stress disorder (PTSD), where they have been attributed to heightened activity in the body's stress-threat response systems (neuromodulatory, neuroendocrine, neuroimmune, autonomic), particularly those mediated by adrenergic signaling.3–9 While the underlying pathophysiology of PASC is almost certainly multifactorial, the similarity of symptoms suggests that changes in these interrelated threat-stress response systems could be involved. If this is the case, other experiences or diagnoses associated with alterations in these systems, such as traumatic stress, would have the potential to moderate the likelihood or severity of PASC symptoms following COVID-19 exposure.
Limited support for this possibility comes from research into Postural Orthostatic Tachycardia Syndrome (POTS), which is a common subsidiary diagnosis in PASC. 10 The role of adrenergic signaling in POTS is well established, particularly in the hyperadrenergic subtype,11,12 and illustrates the potential for changes in both central and peripheral regulatory systems to contribute in an interactive way to symptom expression. Although postural tachycardia is the defining feature of this condition, the diagnosis is associated with a range of symptoms related to altered autonomic regulation, as well as the central nervous system (CNS)-focused symptoms of brain fog and fatigue. 13 Risk factors for POTS include both prior episodes of similar symptoms and precipitating factors include acute viral illness or medical stress. 14 Although limited research exists exploring the relationship of traumatic stress to POTS, in clinical practice we and others have observed both high rates of pre-existing traumatic stress exposure and PTSD co-diagnosis in individuals meeting criteria for POTS, as well as high rates of POTS-like symptoms in people with a history of traumatic stress exposure and PTSD (unpublished observations). These findings and observations support the possibility of overlapping and at times interacting pathophysiologic mechanisms in POTS and the sequelae of traumatic stress, and provide further motivation for exploring the possibility of similar overlapping mechanisms in PASC.
Further supporting this possibility is the identification of pre-existing anxiety as a risk factor for PASC. 15 The reason for this relationship is not yet known. One possibility would be that anxiety leads to increased sensitivity to the somatic experiences of altered autonomic regulatory processes that some people experience after COVID-19 infection, increasing the likelihood that these individuals will be identified as meeting criteria for PASC. 15 Alternatively, a common risk factor, such as prior exposure to traumatic stress, could be responsible for the development of both anxiety and the increased likelihood of developing PASC following COVID-19 exposure; for example, if exposure to one or more traumatic stressors initiates persistent changes in adrenergic, immune, or other stress-regulatory processes that in turn result in an increased risk of PASC following COVID-19 infection. A prediction of this ‘common risk factor’ model is that following COVID-19 infection, the pool of individuals with PASC would be enriched in individuals with prior trauma exposure relative to those without PASC symptoms. Statistically, this would be seen as a positive moderation effect of prior experience of traumatic stress on PASC likelihood or symptom burden following COVID-19 infection At present there is limited data available addressing this prediction, although it is consistent with findings of an association between childhood trauma exposure and PASC risk.16,17
Here, we test the hypothesis that lifetime burden of probable traumatic stressors is associated with an increased autonomic symptom burden following COVID-19 infection by examining the relationship between history of traumatic stress, history of COVID-19 infection, and current burden of symptoms characteristic of autonomic dysregulation. To assess the specificity of our findings to autonomic symptoms, results are compared across other common central PASC symptom domains including subjective cognitive functioning, insomnia, pain, and fatigue.
Methods
Procedures were approved by the VA Puget Sound Healthcare System Human Subjects Committee (#1629444). Prior to enrollment, participants were provided an information statement detailing the purpose, risks, benefits, and alternatives to participation. This study uses baseline self-report data from a larger, ongoing observational study that included optional longitudinal self-report and cognitive assessments and in-person physiologic assessments for selected subsets of the study population.18,19 For the online, self-report portion of the study, participants self-enrolled by following a link provided in advertising materials or on flyers, attesting that they met the inclusion criteria, and affirming that they had read and understood the information statement and wished to participate.
Participants
Participants were recruited between January 2022 and October 2024, with the majority of data included in this analysis collected by March 2023. Recruitment occurred through targeted outreach (eg to clinicians seeing patients with symptoms of PASC, or ‘long COVID’, in contexts including Neurology, Primary Care, Psychiatry, Neuropsychology, Rehabilitation Medicine, and dedicated PASC-focused clinics) and paid advertising. Although recruitment was carried out nationally and was not restricted to Veterans, the optional in-person assessments were carried out at a VA Medical Center in Seattle, Washington, and many outreach efforts focused on this region. Advertisements were targeted at individuals interested in contributing to research into the presentation, recognition and pathobiology of ‘long COVID’ (PASC), whether or not they had personal experience of PASC or COVID-19 infection. Inclusion criteria were age 21–70, English fluency and literacy, and ability to complete online assessments; because of IRB scope, participants were required to be in the United States. Compensation was not provided for the portions of the study used in this work.
Procedures
Self-report data was collected using Qualtrics. Skipping questions was permitted; therefore, not all questions were answered by all participants.
Measures
Symptoms of autonomic dysregulation were assessed using the Composite Autonomic Symptom Score (COMPASS-31); this instrument covers 6 symptom domains (orthostatic intolerance, vasomotor, secretomotor, gastrointestinal, bladder, and pupillomotor) that can be combined into a single autonomic symptom score with range 0 to 100. 20 Symptoms of PTSD were assessed using the PTSD Checklist for DSM-521,22 (PCL-5); the total score from clusters B-D (excluding cluster E, the hyperarousal cluster) was used in multivariable regression analyses to avoid items that conceptually overlapped symptoms of autonomic dysregulation. Neurocognitive functioning was assessed using the Quality of Life in Neurological Disorders (Neuro-QoL) version 2.0 8-item cognitive function static short form,23,24 insomnia using the Insomnia Severity Index (ISI), 25 fatigue and pain using the NIH's Patient-Reported Outcome Measurement Information System (PROMIS) Fatigue 6a (6-item) 26 and Pain Intensity 3a (3-item) 27 short forms, and physical health quality of life using domain 1 of the World Health Organization (WHO) Quality of Life WHOQOL-BREF assessment (WHOQoL-D1). 28 History of exposure to probable traumatic stressors was assessed using the Life Events Checklist 29 ; a numerical index of prior trauma exposure was calculated using items selected a priori and used in previous work as representing those most likely to meet criterion A of PTSD (items 7-8, 11, and 14-16). 30
To assess history of prior COVID-19 infection despite variability in COVID-19 test availability and presentation across time and sociodemographic variables, one primary and two prespecified alternative definitions were used. First, participants were asked to self-assess if they had experienced at least one episode of known or suspected infection with COVID-19 (primary definition), and if so, 1) whether they had received a positive PCR and/or rapid antigen test result (alternate definition used in sensitivity analyses) and 2) whether they experienced any symptoms of COVID-19 during the episode. Separately, all participants were asked “How likely do you think it is that you have had COVID-19?” with a visual slider-bar response allowing answers from 0% to 100% (additional alternate definition used in sensitivity analyses).
Participants reporting at least one known or likely episode of COVID-19 were asked about new or worsening problems with their physical or mental health since recovering from their acute episode in a variety of domains (Supplemental Table 1). To address the possibility that individuals with prior trauma might report a higher burden of PASC-like, multi-system complex symptoms following COVID-19 simply because they had been experiencing these same symptoms prior to COVID-19 infection, we asked all participants to rate the frequency with which they experienced these same types of symptoms prior to either their first episode of COVID-19 infection or, if no known or suspected period of infection, prior to the COVID-19 pandemic. For the sake of clarity the phrase ‘long COVID’ or ‘long-haul COVID’ was used in place of PASC when communicating with participants and all participants were additionally asked “Do you consider yourself someone with ‘long-haul COVID’ or persistent symptoms after COVID-19?”.
Data Analysis
Data were analyzed using R/RStudio, figures created using ggplot2, 31 and tables using flextable. 32 For all analyses, p < .05 was considered statistically significant, and all comparisons were two-tailed. Participants with missing data were excluded from analyses using that data. ANOVAs were implemented using the stats package in base R. 33 Multivariable linear regression models were implemented using normalized data and the nlme, 34 package. Although multivariable linear regression model details varied across analyses (see details for each model below), all models followed the format of using either current autonomic symptom burden or another cluster of current PASC-like symptoms as the dependent variable, with independent variables including age, sex, estimated lifetime trauma burden, history of COVID-19 infection, and the interaction term of trauma history and COVID exposure; some models also include total current PTSD symptom burden (excluding hyperarousal symptoms) and/or a retrospective estimate of PASC-like symptoms experienced prior to the COVID-19 pandemic (for those without a known history of COVID-19 infection) or prior to their first episode of COVID-19. The extent to which autonomic symptom burden as measured by the COMPASS-31 served as a statistical mediator of the relationship between COVID-19 history and physical health related quality of life was assessed using the mediation package. 35 Moderation analysis testing the impact of prior lifetime trauma history on there relationship between COVID-19 infection history and autonomic symptom burden was implemented and model assumptions and fit characterized using the lmtest 36 and car 37 packages; within these packages, autocorrelation in residuals was quantified using the Durbin-Watson test, heteroscedasticity using the studentized Breusch-Pagan test, multicollinearity by calculating the variance inflation factor, unusually large or small residuals using the Bonferroni outlier test, and the influence of each observation using Cooks distance. Cohen's f2 measures of effect size were calculated using effectsize. 38
Results
Table 1 presents sample characteristics for the entire sample and stratified by subgroups. 404 participants provided complete responses to at least a portion of the measures analyzed. 72% of participants (N = 289) reported at least one episode of known or probable COVID-19 infection, of whom 69% (N = 198) reported at least one episode of symptomatic COVID-19 infection and 30% (N = 86) reported only having experienced asymptomatic infection(s) (Supplemental Table 2). Because PASC is not as commonly used outside of the scientific literature, participants were asked whether they considered themselves someone with ‘Long COVID’ or persistent symptoms after COVID-19, and 42% of participants (N = 165) reported yes, 42% (N = 164) reported no, and 15% (N = 60) were unsure. 60% of those who reported symptomatic COVID-19 infection reported experiencing ‘long COVID’ (PASC; N = 118), compared to 46% of those who reported only asymptomatic infections (N = 38). 441 episodes of known or probable COVID-19 infection were reported by participants (many participants reported more than one prior episode); of these, 62% (N = 274) were confirmed by a positive COVID test and 64% (N = 281) were symptomatic episodes.
Sample Demographics, Exposure History and Symptom Burden.
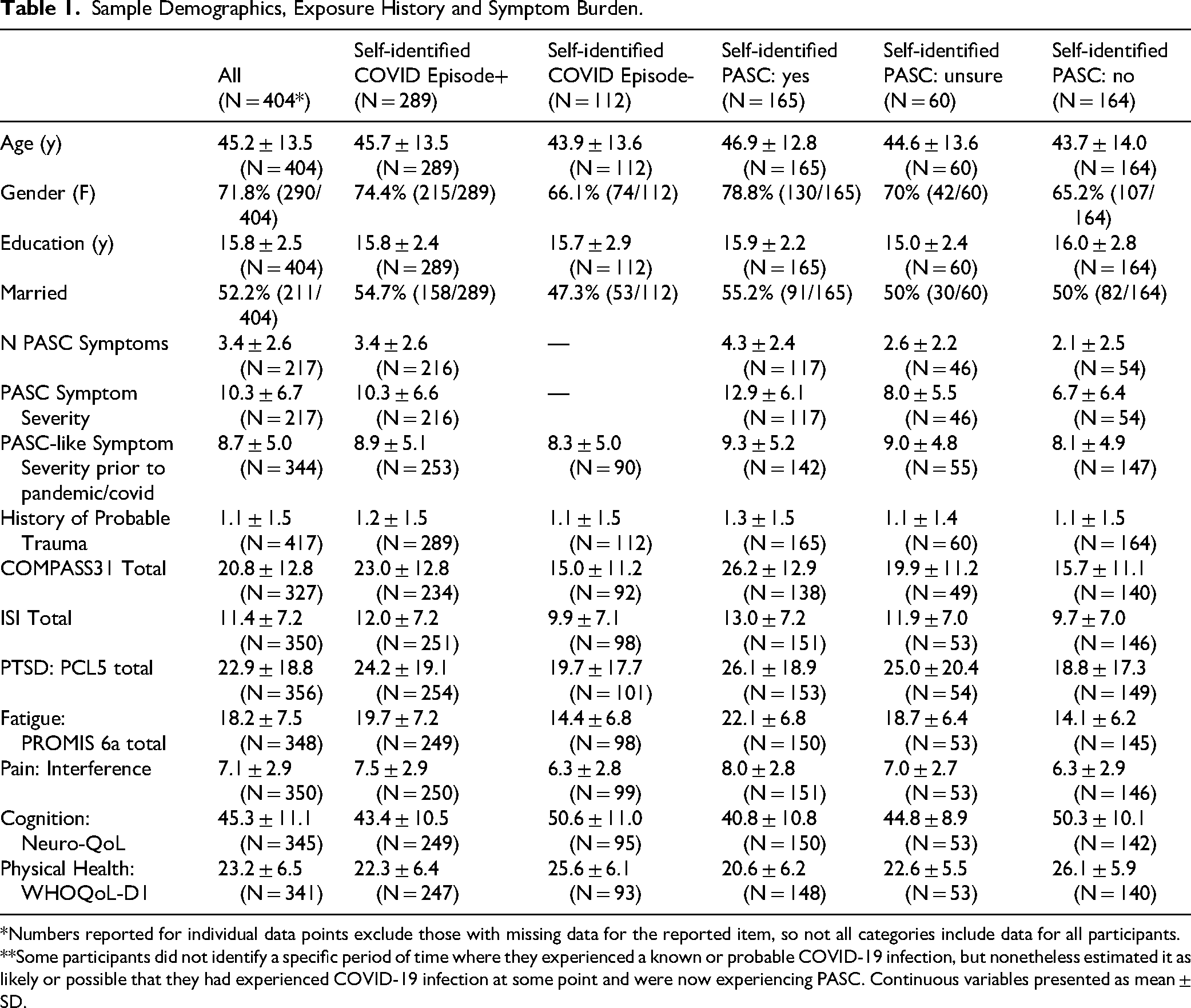
*Numbers reported for individual data points exclude those with missing data for the reported item, so not all categories include data for all participants. **Some participants did not identify a specific period of time where they experienced a known or probable COVID-19 infection, but nonetheless estimated it as likely or possible that they had experienced COVID-19 infection at some point and were now experiencing PASC. Continuous variables presented as mean ± SD.
Relationships Between Autonomic Symptom Burden, COVID-19 History, Physical Health-Related Quality of Life, and History of Traumatic Stress
Participants reporting at least one episode of known or probable COVID-19 infection (prior-COVID+) reported significantly higher total autonomic symptom burden (COMPASS-31 total score, p < 1e-7; Figure 1A) and lower physical health related quality of life (WHO-QoL BREF D1 total score, p < 1e-4; Figure 1B). Both within the prior-COVID+ sample and across all participants COMPASS-31 total score was significantly and inversely related to physical health related quality of life (prior-COVID+ only: R = −0.59, p < 1e-22, Figure 1C; all participants: R = −0.6, p < 1e-32). Autonomic symptom burden served as a statistical mediator of an estimated 78% of the relationship of COVID-19 history to physical health related quality of life (p < 2e-16, 95% CI, 52-127%).

Relationships between autonomic symptom burden, physical health-related quality of life, prior infection with COVID-19, and prior exposure to traumatic stress. Participants reporting a prior episode of known or probable COVID-19 infection reported high current autonomic symptom burden as measured with the COMPASS-31 total score (A; p<1e-7) and lower physical health-related quality of life as measured with the WHO QoL-BREF D1 total score (B; p<1e-4). Current autonomic symptom burden and physical health-related quality of life demonstrated a significant inverse relationship (C). When number of prior likely traumatic stress exposures (“Prob.Traumas”, categorized as 0, 1, or 2 or more) and history of prior COVID-19 infection are both included, both variables are significantly related to current burden of autonomic symptoms (D; two-way ANOVA: effect of COVID p<1e-7, effect of trauma: p=.0001). ***p<.001.
The potential for both prior COVID-19 infection and prior trauma to influence current autonomic symptom burden was first assessed using a two-way analysis of variance. Both infection history and history of probable traumatic stress exposure (categorized here as 0, 1, or 2 or more probable traumatic exposures) were found to be highly significant predictors (effect of COVID p < 1e-7, effect of trauma: p = .0001; Figure 1D).
To allow inclusion of age and sex as covariates and ask whether trauma exposure moderates the relationship between COVID-19 history and current autonomic symptom burden, we implemented a multivariable regression model with COMPASS-31 total score as the dependent variable and normalized independent variables of age, sex, prior-COVID+ status, estimated number of probable lifetime traumatic stress exposures, and an interaction term between COVID-19 and trauma exposure. Age but not sex was a significant predictor of current autonomic symptom burden (cohen's parial f2 = .02 with p = .03 and f2 = .01 with p = .12, respectively). COVID-19 history, trauma history, and the COVID-19 history X trauma history interaction term were all significant positive predictors of autonomic symptom burden (COVID-19 history: β = 0.22, f2 = .11, p = .0002; trauma history: β = 0.16, f2 = .07, p < 1e-6; COVID-19 X trauma interaction term: β = 0.06, f2 = .02, p = .03). There was no evidence of autocorrelation in the residuals (2.001, p = .50) or significant outliers (p = .11), residuals demonstrated equal variance (p = .74), and variance inflation factors indicated minimal multicollinearity (all <1.5). This result demonstrates a statistically significant and positive moderation effect of prior traumatic stress on the relationship between COVID-19 history and subsequent autonomic symptom burden.
Sensitivity Analyses
To assess the sensitivity of the findings to changes in model parameters and definitions, additional multivariable regression analyses were conducted (Figure 2, Supplemental Tables 3 and 4). First, we repeated the analysis varying only the definition of COVID-19 infection (Figure 2A, Supplemental Table 3). Across all three definitions, history of traumatic stress and history of COVID-19 infection remained significant independent predictors of autonomic symptom burden (p < .001) and the interaction term between trauma exposure and COVID-19 history maintained (p < .05) or increased significance (p < .01).

Independent and interactive effects of prior exposure to traumatic stress and prior history of COVID-19 infection in predicting current burden of autonomic symptoms (A) as compared to other common PASC symptom domains (B). Values plotted are the point estimates and 95% confidence intervals for the coefficients from multivariable regression models using normalized data, in order to indicate the relative contribution of each of the plotted factors to the total current symptom burden. All models include age, sex, estimated lifetime trauma burden, history of COVID-19 infection, and the interaction of trauma history and COVID exposure. Some models also include total current PTSD symptom burden (excluding hyperarousal symptoms, models shown in red and purple) and/or a retrospective estimate of PASC-like symptoms experienced prior to the COVID-19 pandemic (for those without a known history of COVID-19 infection) or prior to their first episode of COVID-19 (assessment described in supplemental Table 1, models shown in blue and purple). For models predicting autonomic symptom burden (A), each model was run in triplicate using 3 different ways of defining COVID-19 history: 1) whether the participant reported at least one specific period of time with a known or probable COVID-19 infection (solid line), 2) whether the participant reported at least one specific period of time with a known or probably COVID-19 infection confirmed by either a rapid or a PCR test (dotted line), and 3) a participant's independent estimate of the probability that they have ever been infected with COVID-19 at the time of survey completion, 0-100% (dashed line). Dependent variables are (A) the total burden of autonomic symptoms as defined by the COMPASS-31 total score, and (B) the total burden of cognitive symptoms as measured by the NeuroQoL (inverse used so that the direction of effect is constant), insomnia symptoms as measured by the Insomnia Severity Index, fatigue symptoms as measured by the PROMIS Fatigue 6a, and pain symptoms as measured by the PROMISE Pain Interference 3a. *p<.05, **p<.01, ***p<.001.
Second, we added current PTSD symptom burden (total score on the PCL-5 excluding the hyperarousal cluster) as an additional covariate (Figure 2A, red). PTSD symptom burden was a highly significant predictor of current autonomic symptom burden (p < .001). History of COVID-19 infection, history of traumatic stress, and the interaction between the two all remained significant with PTSD symptom burden included.
Third, we added participants’ retrospective estimate of the frequency of common complex multi-system symptoms prior to their first COVID-19 infection or prior to the COVID-19 pandemic (for participants without a history of known COVID-19 infection) as a covariate. When both current PTSD symptoms and prior complex multi-system symptoms were included as covariates (Figure 2A, purple), both were highly significant predictors of current autonomic symptom burden, along with COVID-19 history, and the trauma history x COVID-19 history interaction term remained statistically significant (p < .05 or p < .01, depending on criteria use for prior history of COVID-19 infection).
Specificity of Results to Autonomic Symptoms Versus Other Common PASC Symptom Domains
Although our primary hypotheses focused on the potential for an additive or interactive effect of COVID and trauma history on autonomic symptoms, we also explored whether a similar effect is seen for other common PASC symptom domains, including domains generally attributed to central nervous system dysfunction, by repeating the same analytic approach for cognitive symptoms, insomnia, fatigue, and pain (Figure 2B, Supplemental Table 4). In our simplest models, history of COVID-19 infection was a significant predictor (p < .01) for all four additional symptom domains, while prior exposure to traumatic stress was a significant predictor (p < .001) for cognitive symptoms, insomnia, and pain, but not for fatigue. When current PTSD symptom burden (red and purple) and history of prior complex multi-system symptoms (blue and purple) were added to the models, both were highly significant predictors (Figure 2B, p < .001) for all four domains. However, the details of how these additional covariates affected the influence of COVID and trauma history varied by domain; eg, the influence of both prior traumatic stress and prior COVID-19 infection on pain symptoms was robust regardless of covariates included (Figure 2B, pain: red, blue, and purple), while the influence of both prior traumatic stress and prior COVID-19 infection on insomnia symptoms was no longer statistically significant when current PTSD symptom burden was included (Figure 2B, insomnia: red and purple compared to blue). Taken together, these results indicate that both prior history of traumatic stress and prior COVID-19 infection are independent positive predictors of multiple common symptom domains reported by patients with PASC, including domains generally attributed to changes in both central nervous system and peripheral physiologic function.
Impact of Symptomatic Versus Asymptomatic Prior COVID-19 Infection
To explore whether the effects observed were different in the group that had experienced symptomatic versus asymptomatic infection, the same analyses were conducted comparing those with no known history of COVID-19 infection to those who had experienced 1) only asymptomatic infection(s) (Figure 3A, Supplemental Table 5), and 2) at least one symptomatic episode of COVID-19 infection (Figure 3B, Supplemental Table 6). History of COVID-19 infection was in general more predictive of current symptom burden in the group that had experienced symptomatic COVID-19 infections, while the interaction term between history of COVID-19 infection and traumatic stress was, where present, more consistently significant in the group of respondents who reported only asymptomatic COVID-19 infections (p < .01); this effect remained when current PTSD symptom burden was included in the model (p < .01), as well as when both current PTSD symptoms and prior PASC-like symptoms were included (p < .05).

Differences in the contributions of COVID history and history of exposure to traumatic stress for individuals reporting only asymptomatic COVID-19 infection as compared to those reporting at least one symptomatic infection with COVID-19. Models and values plotted are as described in Figure 2. For both the analysis of the impact of asymptomatic COVID-19 infections (A) and the analysis of the impact of symptomatic COVID- 19 infections (B), history of COVID-19 infection is defined as participants who reported at least one specific episode of known or probable COVID-19 infection, and participants who did not report any known history of COVID-19 infection are included. *p<.05, **p<.01, ***p<.001.
Discussion
In our sample of 404 participants who completed online self-report assessments, those with a history of known or likely COVID-19 infection reported significantly higher autonomic symptom burden and significantly worse physical health-related quality of life. Autonomic symptom burden mediated the significant majority of the statistical relationship between COVID-19 history and physical health-related quality of life. While statistical mediation does not necessarily imply causality, it does suggest that autonomic symptom burden is tightly associated with the impact of COVID-19 on physical health-related quality of life.
Current autonomic symptom burden was also significantly associated with lifetime exposure to probable traumatic stressors, consistent with existing evidence of both increased somatic symptom burden39,40 and altered autonomic regulatory functioning7,41,42 in PTSD and/or individuals with a history of traumatic stress. This finding suggests two possibilities: first, a “distal additivity” model where prior COVID-19 infection and history of traumatic stress are independent, additive factors that contribute to increased potential for autonomic symptom burden through unrelated but convergent mechanisms at one or more common distal targets (Figure 4A, top). Alternatively, these results could also support an “interactive pathophysiology” model in which there are one or more common or interacting elements in the underlying pathobiology by which COVID-19 infection and exposure to traumatic stress result in persistent altered autonomic regulatory function (Figure 4A, bottom). Recognizing interactive pathophysiology, if present, is important both to shape future specific pathophysiologic models that may lead to novel intervention options, and because it would suggest the potential for some subset of the pathophysiologic changes following traumatic stress to serve as recognizable and potentially modifiable risk factors for PASC (‘long COVID’) or other post-viral syndromes.

Conceptual models of the relationship between prior traumatic stress, COVID-19 exposure, and symptom burden. (A) Both overall burden of probable lifetime traumatic stress exposure and history of COVID-19 infection were associated with increased autonomic symptom burden at the time of assessment. This finding is consistent with two possible models: First, a distal additivity model (A, top), whereby exposure to traumatic stress (left) and COVID- 19 infection (center) lead to an increase in symptom burden that may appear similar, but occurs via different mechanisms. When an individual experiences both traumatic stress and, at another timepoint, COVID-19 infection (right), the impact is additive (as indicated by large symptom icons). In its simplest form, this model would not predict an interaction between traumatic stress exposure and COVID-19 infection history, i.e., traumatic stress would not serve as a moderator of the relationship between COVID-19 infection and symptom burden, although such an effect could be produced if there were nonlinear relationships in the reporting of symptoms, for example if only symptoms above a certain threshold tended to be reported. Second, the finding could also be explained by an interactive pathophysiology model (A, bottom), whereby traumatic stress (left) and COVID-19 infection (center) modulate common, overlapping biologic pathways. Either stressor when experienced alone may have variable impact or even no impact on symptom burden (dashed lines). Even when it does not cause immediate symptoms, however, exposure to traumatic stress may change the likelihood that a subsequent stressor, such as COVID-19 infection, may result in persistent symptoms (right panel). This model is consistent with our findings that traumatic stress is a significant moderator of the impact of COVID-19 infection history of autonomic symptom burden, and the persistent impact of prior trauma on current symptom burden across all analyses. (B) Proposed model for the relationship of traumatic stress exposure and history of COVID-19 infection to symptom burden that is consistent with the findings presented, including the variation in the relationships between traumatic stress, COVID-19 exposure, and symptom burden across symptom domains, as well as with current evidence suggesting the existence of bidirectional coregulation of central and peripheral stress regulatory systems. In this model, exposure to a traumatic stressor (left) may result in changes in both central and peripheral stress regulatory systems; these changes both reinforce each other via bidirectional coregulation, and lead to increased likelihood of symptoms relating to both altered central stress reactivity (figure representing ‘brain’ symptoms) and altered autonomic regulatory system (figure representing ‘body’ symptoms). Similarly, exposure to a physiologic stressor such as COVID-19 infection (center) may result in similar changes in both central and peripheral stress regulatory systems and downstream symptom expression, but with a differing pattern of emphasis. When an individual experiences both traumatic stressors and COVID-19 infection (right), the combination may result in increased symptom burden in both central and peripheral domains, via both additive and interactive factors.
To begin to distinguish between these two possibilities, we explored the interaction between COVID-19 history and history of traumatic stress in contributing to autonomic symptom burden using a multivariable regression model, and found a significant, positive interaction between the two; ie, prior history of traumatic stress served as a significant statistical moderator of the relationship between COVID-19 infection and autonomic symptom burden. While such a result could still occur in a distal additivity model via nonlinear translation between additive changes in autonomic symptom generation and self-reported symptom burden (eg, thresholding), it is more consistent with the expected pattern in an interactive pathophysiology model.
To further probe the relative likelihood of the distal additivity versus the interactive pathophysiology models, we added a retrospective assessment of complex, multi-system symptoms prior to any episode of COVID-19 infection (ie, PASC-like symptoms) as well as current PTSD symptoms (excluding the hyperarousal cluster to prevent conceptual overlap) as covariates in the regression analysis (Figures 2 and 3, red, blue and purple). If the distal additivity model were the primary explanation of our results the inclusion of the prior symptom assessment should account for the impact of prior traumatic stress on current autonomic symptom burden, and the moderation effect would no longer be present. However, we found that prior history of traumatic stress remained a positive moderator even when either current PTSD symptoms (excluding the hyperarousal cluster) alone or both PTSD symptoms and an estimate of prior complex multi-system symptom burden were included as covariates – consistent with an interactive pathophysiology model rather than a distal additivity model.
The strength of the interaction between COVID-19 infection history and history of traumatic stress was minimally dependent on how COVID-19 infection was determined (suspected vs confirmed infection). However, the interactive effect was stronger for participants reporting a history of only asymptomatic COVID-19 infection(s). One possible interpretation of this finding is that symptomatic COVID-19 infection was a much more potent stimulus for persistent symptoms after infection, leaving less range for history of traumatic stress or interactive factors to contribute to the overall effect.
The impact of both COVID-19 and traumatic stress on symptom burden across PASC-associated symptom domains was domain-specific, decreasing the likelihood that these effects were due to a more general influence, such as an influence of history of traumatic stress on individuals’ survey response behaviors. In general, the effect of prior traumatic stress on current symptom burden was significant but associated with concurrent PTSD symptom burden for central nervous system focused domains (ie cognitive symptoms, insomnia; Figure 2B). In contrast, fatigue was associated with current PTSD symptom burden but was not otherwise associated with prior traumatic stress, while pain was strongly associated with prior traumatic stress independent of current PTSD symptoms or prior PASC-like symptoms (Figure 2B).
Implications for the Conceptualization and Potential Underlying Pathobiology of complex, Co-Occurring Multi-System Central and Peripheral Symptoms
While these results support an interactive pathophysiology model underlying the impact of COVID-19 infection and traumatic stress on autonomic and central symptoms in PASC or ‘long COVID’, they are agnostic regarding pathophysiology. Existing evidence in similar contexts suggests a range of interactive pathophysiologic mechanisms by which traumatic stress and COVID-19 could induce persistent changes in stress-threat systems. For instance, research into the overlapping pathophysiology of PTSD and traumatic brain injury has found both actual and anticipated physical threats (ie, including traumatic stressors without direct physical harm) can activate coordinated stress-response mechanisms involving autonomic and central neuromodulatory systems (eg, central and peripheral adrenergic signaling), the neuroendocrine system including the hypothalamic-pituitary-adrenal axis, and other systems including both central and peripheral immune function. 9 These responses to acute stressors can act not only to maintain or restore homeostasis after an acute injury, but also to protect the system from future, anticipated threats; as such, they may include lasting changes in autonomic and central neuromodulatory, neuroendocrine, and/or immune regulatory systems. Furthermore, the genetic underpinnings of susceptibility to PTSD and/or changes in the immune or inflammatory systems could alter the response to Covid infection. These persistent changes, in turn, may potentiate the response to a wide variety of future stimuli. 43
When it is integrated with this existing biologic understanding, the interactive pathophysiology model provides a potential mechanistic explanation for the complex co-occurrence of central (eg cognitive, insomnia, anxiety) and peripheral (eg palpitations, gastrointestinal, pain) symptoms in PASC and other related conditions. Specifically, it suggests two mechanistically distinct reasons that central stress-related symptoms may be associated with physical or somatic stress-related symptoms (integrated in Figure 4B). First, whether a stressor is physiologic or traumatic (psychological), it can affect both central and peripheral stress-response systems in parallel. This may result in central and peripheral symptoms emerging concurrently, consistent with the frequent onset of both PTSD and somatic symptoms following a traumatic stress. Alternatively, an earlier stressor may not result in symptom expression immediately but instead increase the likelihood of symptom expression following a future stressor, consistent with the increased autonomic symptom burden following COVID-19 infection in those with a prior history of traumatic stress – even when an estimate of prior somatic symptoms was included in the model. Second, the association may occur due to the bidirectional co-regulation of central and peripheral stress systems This co-regulation means that periods of increased psychological stress may lead to increased symptom expression for peripheral somatic symptoms (or visa-versa).
This conceptualization has important clinical implications, suggesting that: 1) peripheral (somatic) symptoms could be worsened during increased stress or improved by treatment of cognitive mediators of persistent activation of the stress-threat response system (eg behavioral or pharmacologic treatments for PTSD, anxiety, or insomnia), via ongoing co-regulatory, bidirectional interactions between central and peripheral aspects of the stress-threat response systems; and 2) the effectiveness of such interventions is likely to be incomplete, with other aspects of persistent symptoms being driven by persistent alterations in peripheral regulatory systems, which may or may not respond to the same pharmacologic or behavioral interventions that are effective for centrally mediated symptoms.
Limitations
There are limitations to these results. The sample is limited in size and is not a representative population sample, limiting the interpretability of overall prevalence or symptom severity. COVID-19 history is based on self-report. To limit the impact of recall or attribution bias, our analyses focus current symptom burden with an estimate of symptoms prior to COVID-19 infection added during sensitivity analyses as a covariate, but this estimate is retrospective and likely to be affected by recall bias. Assessment of prior traumatic stress is based on the LEC5, which allows a count of the number of types of probable traumatic stressors someone has experienced but not the number of times experiencing an individual type of traumatic stress. Many of these limitations would primarily be expected to decrease detected effect size, however, rather than result in a false positive result.
As noted above, the presented work also focuses on what is expected to be only a subset of the relevant underlying mechanisms leading to symptom expression in PASC (long COVID) broadly and to the overlapping presentation and risk factors for PASC and PTSD or other persistent sequelae of traumatic stress in particular. For example, genetic factors are known to modulate the susceptibility individuals to PTSD after a trauma 44 ; if these genetic factors also contribute to the likelihood of developing PASC, it would not explain the interaction of prior traumatic stress exposure and PASC risk, but it could provide an alternative explanation for some of the relationship between pre-existing anxiety or PASC-like symptom burden prior to COVID-19 exposure and current PASC symptom burden, as well as some of the relationship between current PTSD and PASC symptoms.
Finally, as these results are based only on self-report without measures of underlying physiology, future work will be required to directly test the hypothesized models presented.
Conclusions
These findings demonstrate the relevance of prior exposure to traumatic stressors as both an independent risk factor for current PASC-like symptoms, and a factor that can modify the risk of PASC symptoms following even asymptomatic COVID-19 infection. This effect was present even when adjusting for PTSD symptoms and showed specificity for different domains of common PASC symptoms. These results suggest that behavioral and pharmacologic treatments for centrally-mediated symptoms of anxiety, PTSD, and other similar stress-related disorders may have the potential to modulate somatic PASC symptoms and reduce overall symptom burden, but that full resolution of symptoms may require interventions that target both central and peripheral stress-regulatory mechanisms.
Supplemental Material
sj-docx-1-css-10.1177_24705470251407210 - Supplemental material for Impact of Prior History of Traumatic Stress on Autonomic and Multi-System Symptoms Following COVID-19 Infection
Supplemental material, sj-docx-1-css-10.1177_24705470251407210 for Impact of Prior History of Traumatic Stress on Autonomic and Multi-System Symptoms Following COVID-19 Infection by Rebecca C. Hendrickson, Christine S. Cheah, Marlene L. Tai, Kathleen F. Pagulayan, Katharine J. Liang, Catherine A. McCall, Abigail G. Schindler, Kimberly L. Hart, Aaron F. Rosser and John C. Oakley in Chronic Stress
Footnotes
ORCID iDs
Contributions of Authors
RCH, JCO, and CSC designed the study, with input from KFP, CAM, and AGS. RCH supervised the implementation of the study. AFR and KLH provided support with study implementation. RCH, KJL and CSC carried out the analysis of all results. MLT and RCH co-wrote the initial draft of the manuscript, with contributions from JCO, CSC and KJL. All authors provided input into the interpretation of the analysis and the drafting of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Innovation Grant from the Garvey Institute for Brain Health Solutions (RCH, JCO, KFP, CAM and AGS), VA Clinical Sciences Research and Development Service Career Development Award IK2CX001774 (RCH); and the VA Northwest Network MIRECC (RCH, MAR, AGS).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Sharing
Deidentified data sets may be requested by contacting the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
