Abstract
Persistent shortness of breath is one of the most common concerns reported by patients with post-acute sequelae of SARS-CoV-2. Here, we present a case of bilateral diaphragmatic paralysis as a cause shortness of breath that developed after SARS-CoV-2 infection. A middle-aged gentleman with history of sleep apnea and body mass index 27.9 kg/m2 presented to our post-COVID clinic with 3 months of dyspnea and orthopnea after contracting SARS-CoV-2 in November 2020. During acute infection, he was hospitalized for hypoxemia, which improved with steroids and supplemental oxygen. At 3 months, he continued to report dyspnea and orthopnea. On examination, he had tachycardia and increased respiratory rate with paradoxical respiratory abdominal movement. Chest imaging showed elevated bilateral hemidiaphragms without any parenchymal lung disease. Pulmonary function test revealed severe ventilatory defect with restrictive lung disease. He was diagnosed with bilateral diaphragmatic dysfunction which was confirmed by absence of evoked potentials in diaphragm after phrenic nerve stimulation bilaterally. He was advised to use continuous positive airway pressure machine to assist with breathing at night. At his last follow-up (1-year post-infection), he was symptomatically improving without specific interventions.
Keywords
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a global pandemic with a myriad of presentation and sequelae. 1 It is not uncommon for patients to complain of shortness of breath (SOB) after recovering from acute infection.1,2 Acute pulmonary complications of SARS-CoV-2 infection include viral pneumonia, acute respiratory distress syndrome (ARDS), and pulmonary embolism whereas long-term complications include interstitial abnormalities, bronchiectasis, and pulmonary fibrosis. 2 We present an unusual cause of persistent SOB post-SARS-CoV-2 that clinicians need to consider in their differential diagnosis.
Case presentation
A 46-year-old man with history of obstructive sleep apnea (OSA), cervical spine surgery, and chronic right shoulder pain tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) in November 2020 after exposure at work. He was initially asymptomatic, but on day 7 post-positive test, he developed SOB, nausea, myalgia, fever, and loss of smell and taste. On day 18, his oxygen saturation decreased to 90% on room air and was subsequently started on oral steroids. On day 22, he was hospitalized with dyspnea at rest, tachycardia, and hypoxia (oxygen saturation 88%). His chest X-ray (Figure 1(a)) was suggestive of mild bilateral infiltrates, and the hypoxia improved on intravenous steroids and supplemental oxygen through nasal cannula. He was discharged after a 3-day hospital stay. He did not require oxygen supplementation, but continued to have dyspnea on exertion and orthopnea as reported during telemedicine appointment after hospital discharge. On day 29, he reported mild pain in neck and right arm which was non-traumatic and improved with heat and acetaminophen. Electrocardiogram and echocardiogram were unremarkable. On day 43, he was evaluated by cardiology and referred to pulmonologist. In the interim, he also followed-up with his orthopedic surgeon for his C5 C7 anterior cervical discectomy and fusion, which was performed 2 years prior. He had no concern of neck pain and arm pain at that visit and his neurological examination (including motor strength in the upper and lower extremities and reflexes) was normal. Cervical spine radiographs were normal without evidence of interval hardware complication. On day 95, he was seen by our pulmonologist in the post-COVID clinic, for persistent dyspnea on exertion and orthopnea. His Modified Medical Research Council Dyspnea Scale (mMRC) was +3. He had history of OSA, but was not using continuous positive airway pressure (CPAP) prior to COVID-19. His body mass index (BMI) was 27.9 kg/m2, and he was afebrile with regular pulse of 121/min, respiratory rate of 20/min, blood pressure of 124/85 mm Hg, and oxygen saturation of 96% on room air. He was alert and oriented. Lung exam revealed decreased breath sounds at both bases with evidence of paradoxical abdominal movement with breathing in supine position which he could not tolerate except for a few seconds.

(a) Chest X-ray taken at the time of hospitalization (day 22 since positive SARS-CoV-2 test) showing bilateral elevated diaphragms with minimal diffuse infiltrates. (b) Chest computed tomography coronal view done at 3 months after his acute infection. Full inspiratory images show bilateral elevated diaphragms (as shown by white arrows). (c) Spirogram at 3 months prebronchodilator (blue line) and post-bronchodilator (red line), and (d) spirogram at 1-year follow-up pre-bronchodilator.
Investigations
Pulmonary function test (PFT) done at 3 months post-infection in the upright position showed a forced vital capacity (FVC) of 1.76 L (37% of predicted), forced expiratory volume in the first second (FEV1) of 1.24 L (33% of predicted), and FEV1/FVC of 70%. Vital capacity (VC) was 1.89 L (42% of predicted), total lung capacity (TLC) was 3.70 L (55% of predicted), residual volume (RV) was 1.80 L (89% of predicted), and RV/TLC was 49%. Diffusion capacity of lung for carbon monoxide (DLCO) was 64% of predicted. There was no response to bronchodilators. This test highlights severe ventilatory defect with restrictive physiology with mildly reduced diffusion capacity suggesting alveolar capillary defect.
Non-contrast computed tomography (CT) of chest showed normal lung parenchyma with bilaterally elevated diaphragms during inspiration and expiration (Figure 1(b)). The diaphragmatic evoked potential by stimulation of the bilateral phrenic nerve at the posterior border of sternocleidomastoid was not elicited. Motor and sensory conduction in the right arm including ulnar, radial, and median nerves were normal (Figure 2). We did not obtain PFT in supine position given already reduced vital capacity (less than 50%) in erect position. Also, dynamic evaluation of diaphragmatic movement using fluoroscopy was not done in the setting of bilateral diaphragmatic weakness which can result in false negative test. 3
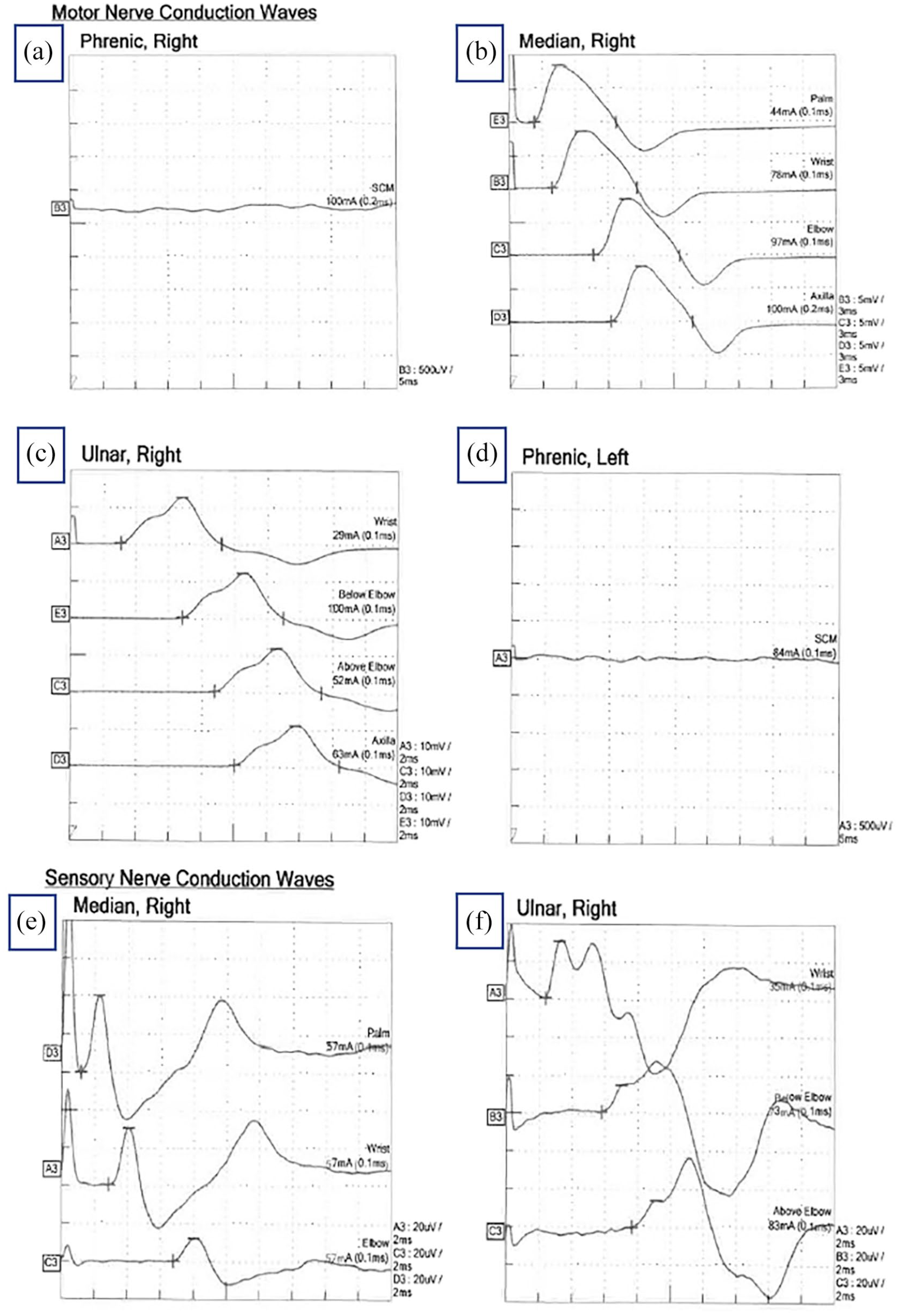
Nerve conduction study showing nerve responses after stimulation: (a) right phrenic nerve response (which is flat and abnormal), (b) normal right median nerve motor response, (c) normal right ulnar nerve motor response, (d) left phrenic nerve response (which is flat and abnormal), (e) normal right median nerve sensory response, and (f) normal right ulnar nerve sensory response.
Clinical course
He was encouraged to regularly use CPAP on previous settings (9 cm of H2O) which helped alleviate symptoms at night. We opted for a conservative management approach, and on follow-up 7 months post-infection, he reported feeling slightly better with improved SOB. His mMRC was +2 on this visit. On exam, he was able to lay flat, but still exhibited paradoxical breathing. His PFT showed improvement in FVC from 37% to 46% of percent predicted and FEV1 from 33% to 41% of percent predicted. At 1 year, FVC was 42% of percent predicted, FEV1 was 41% of percent predicted and FEV1/FVC was 73%. The DLCO improved from 64% previously to 70%. Chest X-ray showed mild elevation of bilateral hemidiaphragm. The spirogram is shown in Figure (1c) and (d).
Discussion
Our patient did not have dyspnea prior to SARS-CoV-2 infection and was diagnosed with bilateral diaphragmatic paralysis post-infection, which was confirmed with nerve conduction studies. Although chest imaging during acute infection also showed elevated diaphragms, this was not recognized until at a later follow-up. Considering other differentials, the right-sided musculoskeletal pain raised suspicion for neuralgic amyotrophy, an idiopathic inflammatory neurological condition with unilateral phrenic palsy. However, the presence of normal sensory and motor potentials in nerve conduction study goes against right brachial plexopathy. It is possible that cervical spine surgery may have predisposed him to have traumatic phrenic nerve injury. However, considering the fact that he was asymptomatic prior to his acute SARS-CoV-2 infection, and the timing of his neck surgery (2-years prior), the new findings of diaphragmatic dysfunction are unlikely a sequelae of his previous neck surgery.
We reviewed several other cases of diaphragmatic dysfunction reported till date following SARS-CoV-2 infection4–8 (Supplementary Table 1). We want to highlight that, during acute infection, our patient was hospitalized briefly and did not require invasive mechanical and non-invasive positive airway pressure ventilation (NPPV), yet developed significant complication whereas others report a much-severe SARS-CoV-2 disease preceding its development. In a small cohort of patients with severe COVID-19 admitted for inpatient rehabilitation, evidence of diaphragmatic dysfunction was identified by ultrasound modality, the role of which is emerging during COVID-19 pandemic.9,10 Besides SARS-CoV-2, there are other viral illness such as influenza, herpes, and HIV which can also lead to diaphragmatic dysfunction.11,12 Also, critical illness itself has been associated with diaphragm weakness which can exist either prior to admission to critical care or after prolonged stay. 13 It is possible that these neurological manifestations of SARS-CoV-2 may be due to direct neuro-invasion or/and post-infectious immune-mediated neuronal injury; however, further research is needed to elucidate the underlying pathophysiological mechanisms.2,6,14
Treatment of bilateral phrenic nerve palsy is largely targeted at identifying and treating underlying causes and concomitant conditions (obesity, deconditioning). 3 In cases of para or post-infectious immune-mediated neurological conditions, gradual recovery is expected with time. 3 However, the role of immune therapy in acute phase needs to be further investigated. In addition, there is a role of inspiratory muscle training and NPPV in setting of underlying sleep disordered breathing, severe diaphragm weakness and underlying progressive neurological or muscular conditions.3,15 Nonetheless, diaphragmatic paralysis is an underrecognized condition, but early diagnosis and treatment can help minimize morbidity and mortality. 15
Conclusion
Clinicians should be aware of diaphragmatic dysfunction as a possible complication of SARS-CoV-2 infection. There is a role of systematic work up (including tests like PFT) and following patients with ongoing concerns after SARS-CoV-2 infection. In this case, the patient reported gradual symptomatic improvement suggesting that spontaneous resolution may occur in these cases, but careful monitoring is required.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X221105990 – Supplemental material for Bilateral diaphragmatic dysfunction: A cause of persistent dyspnea in patients with post-acute sequelae of SARS-CoV-2
Supplemental material, sj-docx-1-sco-10.1177_2050313X221105990 for Bilateral diaphragmatic dysfunction: A cause of persistent dyspnea in patients with post-acute sequelae of SARS-CoV-2 by Christine Gill, Alpana Garg, Rami Fakih and Nabeel Y Hamzeh in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
The authors thank Heather Healy for assistance with literature search.
Author contributions
Data collection and writing the paper: C.G. and A.G. contributed equally. RF and NYH reviewed and editted the manucript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
