Abstract
Objective
We aimed to describe the prevalence and factors associated with the need for supplemental oxygen and persistent symptoms 1 year after severe SARS-CoV-2 infection.
Methods
In this historical cohort and nested case–control study, we included adults with severe COVID-19 (requiring admission to the intensive care unit or invasive mechanical ventilation). We evaluated factors associated with a need for supplemental oxygen and persistent symptoms 1 year after severe infection.
Results
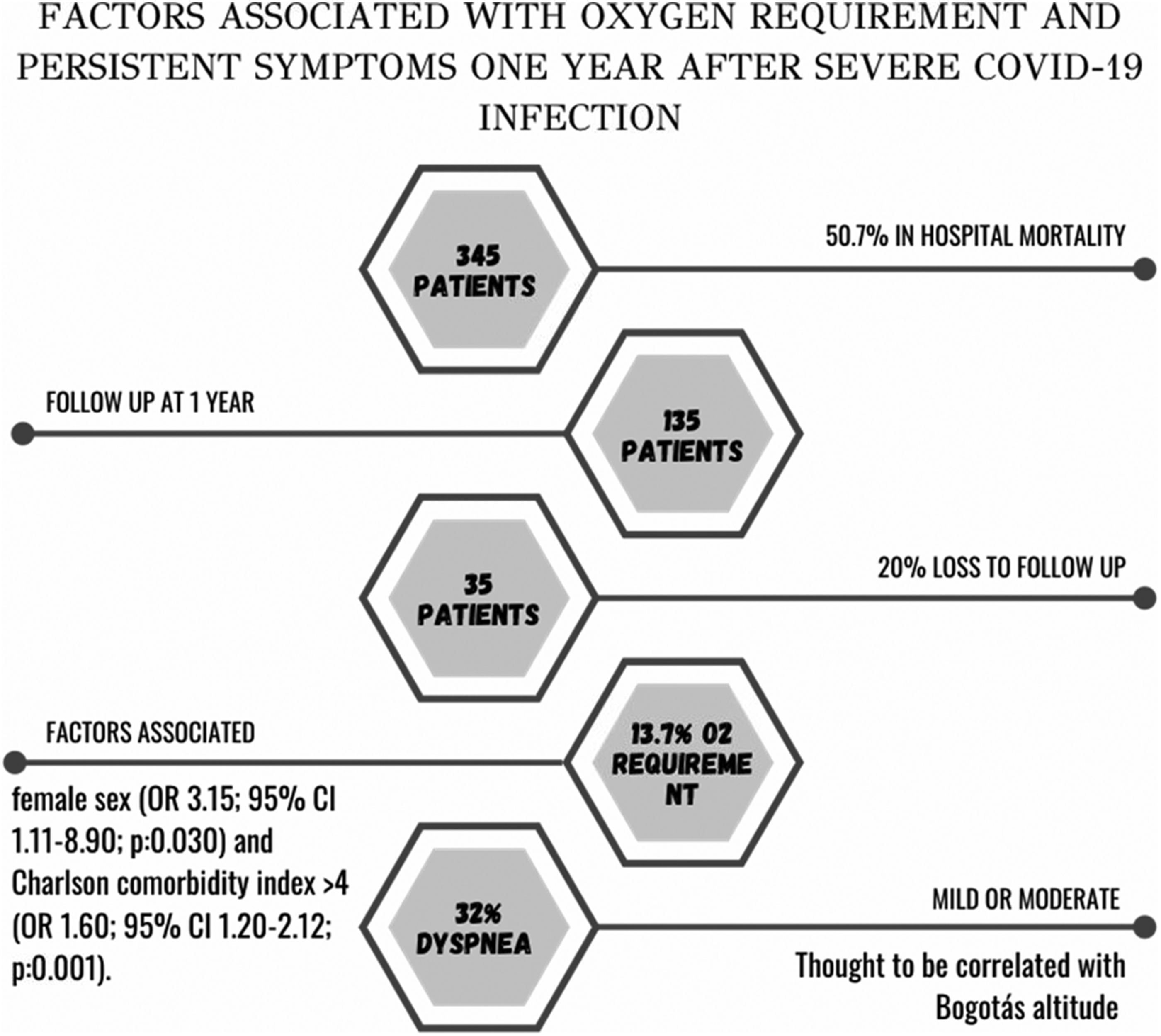
We included 135 patients (median age 62 years, 30% women). At 1-year follow-up, the main symptoms were dyspnea (32%), myalgia (9%), cough (7%), anxiety (4%), and depression (5%); 12.59% of patients had prolonged requirement for supplemental oxygen. Factors associated with a persistent requirement for supplemental oxygen were female sex (odds ratio 3.15, 95% confidence interval 1.11–8.90) and Charlson Comorbidity Index > 4 (odds ratio 1.60, 95% confidence interval 1.20–2.12).
Conclusions
We found that a high prevalence of supplemental oxygen requirement 1 year after severe COVID infection was associated with female sex and a baseline high rate of comorbidities. It is unknown whether this prevalence was related to other factors, such as the altitude at which patients lived. More than half of patients had prolonged post-COVID syndrome.
Keywords
Introduction
The COVID-19 pandemic has had a profound impact worldwide owing to its risk of high contagion and associated morbidity and mortality. 1 According to global statistics of the World Health Organization, by March 2023, there were more than 760 million confirmed COVID-19 cases with more than 6.8 million deaths. 2 The proportion of severe cases and deaths varies depending on the rate of vaccination in the population, availability and access to health services, population density, income per capita, and many other factors.
Some data are now available regarding the persistence of symptoms in the medium and long term, an phenomenon termed prolonged COVID or long COVID. Pathophysiologically, it has been proposed that long COVID is secondary to persistence of an inflammatory state, with dysfunction of different effectors of the immune system such as CD8+ T lymphocytes and B lymphocytes, with autoantibody production and hyperactivity of neutrophils that generate persistent damage to target organs such as the endothelium, heart, and lungs. 3
The symptoms most frequently experienced by patients with long COVID at 1 year are fatigue (61.2%), dyspnea (23.3%), chest pain (6.5%), and cough (2.5%). 4 According to a meta-analysis, the prevalence of fatigue is 28% and that of dyspnea is 18%. 5 However, clinical factors associated with long-term symptoms or a persistent requirement for supplemental oxygen have not been fully described. Additionally, whether these clinical manifestations are more frequent in patients living at higher altitudes is unclear.
The aim of the study was to describe the frequency of symptoms associated with prolonged COVID and to identify factors associated with oxygen requirements and persistent symptoms at 1 year of follow-up among patients treated for severe COVID-19 infection in a reference hospital in Bogota, Colombia.
Methods
This was a historical cohort and nested case–control study. We included consecutive adult patients with a diagnosis of COVID-19 confirmed in polymerase chain reaction who had severe manifestations, defined as requiring admission to the intensive care unit (ICU) or invasive mechanical ventilation, who were treated at the Hospital Universitario San Ignacio (HUSI) in Bogotá, Colombia between October 2020 and April 2021. We excluded those patients who, despite having COVID-19, were admitted to the ICU for another condition, those referred to another institution, and those who withdrew their consent to participate in the study when contacted for follow-up. The authors de-identified all patient details. All patients provided verbal informed consent solicited at the start of telephone follow-up. This study was approved by the institutional ethics committee of Pontificia Universidad Javeriana (approval number 217/2021).
Patient data were collected from the institutional COVID-19 registry, where information on all patients treated at HUSI has been systematically recorded since the start of the pandemic, using the REDCap platform. 6 The information collected included sociodemographic, clinical (including maximum oxygen requirement during hospitalization), and paraclinical variables from patients taken at admission to the hospital, including markers of poor prognosis such as lactate dehydrogenase, C-reactive protein, D-dimer, and lymphopenia. We also recorded the treatments used and patient outcomes during hospitalization (mortality, ICU stay, and hospital stay). To guarantee the quality of the data, the rate of missing data was recorded, and the information was verified when extreme data points were found; the rate of missing data was less than 1%. If there were any missing data, we searched for the missing information in the electronic medical records. To determine the persistence of symptoms and the need for supplemental oxygen, telephone follow-up was conducted exactly 1 year after the date of hospital admission. At 1-year follow-up, we used a standardized questionnaire that included closed-ended questions such as the presence of dyspnea and requirement for supplemental oxygen. We determined scores according to the Modified Medical Research Council (mMRC) Dyspnea Scale. We also asked open-ended questions such as the presence of other symptoms. All data were recorded in a data extraction format using REDCap for secure and confidential recording of all patient information.
The cutoff points for the predictors of poor prognosis of COVID-19 were selected according to published reports. 7
Statistical analysis
Patients who had the outcomes of interest (persistent symptoms or oxygen requirement at 1 year of follow-up) were considered cases, and all patients who did not have those outcomes were considered controls. In the descriptive analysis, categorical variables are presented using absolute and relative frequency and percentage. Continuous variables are presented as mean (standard deviation) or median (interquartile range, IQR) according to whether the assumption of normality was met; the Shapiro–Wilk test was used to test normality. Comparisons between groups were made using the t-test, Mann–Whitney U test, or chi-square test, as appropriate.
We conducted logistic regression, taking a persistent oxygen requirement or persistent symptoms at 1 year of follow-up as the outcome. We conducted univariate analysis, followed by multivariate analysis. All variables that were significant in the univariate analysis were included in the multivariate analysis, as well as those that have been described as being associated with chronic symptoms in previous studies, such as age, smoking, and days requiring mechanical ventilation. Associated variables were selected using a stepwise-backward method, and we included only those with p value <0.05. Stata version 16.0 software was used for the statistical analysis (StataCorp LLC, College Station, TX, USA). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
Results
During the observation period, 345 patients who met the inclusion criteria were identified. Of these, 175 died during hospitalization. Among the remaining 170 patients, follow-up was completed for 135 patients (21% loss to follow-up). Supplementary Table 1 presents a comparison between the included patients and those lost to follow-up, with no significant differences found between groups.
Table 1 shows the general characteristics of the 135 patients. Among included patients, the median age was 62 years (IQR 53–70), and 30% were women. The main comorbidity was arterial hypertension (36.3%) and 17% of patients had multiple comorbidities, scoring > 4 on the Charlson Comorbidity Index. Only one patient had an oxygen requirement prior to COVID-19 infection. Regarding the paraclinical predictive factors of poor prognosis for COVID-19 infection, most patients met the cutoff points established in the literature. A small proportion of patients had suspected organizing pneumonia (12.59%) or suspected pulmonary thromboembolism (8.15%). Most patients (99%) received steroid as an immunomodulator. The median hospital stay was 23 days (IQR 15–32) and 94% of patients required orotracheal intubation.
General characteristics of patients surviving severe COVID-19 at 1-year follow-up, according to oxygen requirement.

LDH, lactate dehydrogenase; CRP, C-reactive protein; ICU, intensive care unit, BMI, body mass index; HFNC, high-flow nasal canula; OTI, orotracheal intubation; PE, pulmonary embolism; O2, oxygen; CCI, Charlson Comorbidity Index; IQR, interquartile range.
Among patients who completed telephone follow-up at 1 year, 17 (12.59%) continued to require supplemental oxygen. Table 1 shows differences between patients with and those without an oxygen requirement at 1-year follow-up. Patients with a persistent oxygen requirement had higher proportion of women (52.9% vs. 26.3%, p = 0.024), older patients (68 vs. 62 years, p = 0.039), patients with comorbidities (47.06% vs. 11.86%, p < 0.001), and patients with prior oxygen use (5.88% vs. 0%, p = 0.008), in comparison with those who did not have a persistent oxygen requirement.
Table 2 shows those factors associated with a persistent oxygen requirement. In both the univariate and multivariate analysis, an association was found with female sex (OR 3.15, 95% CI 1.11–8.90; p = 0.030) and Charlson Comorbidity Index > 4 (OR 1.60, 95% CI 1.20–2.12; p = 0.001). The variables age, smoking, and days of hospitalization were not statistically significant. Figure 1 summarizes the main results of the study.
Univariate and multivariate analysis of factors associated with oxygen requirement at 1 year.

LDH, lactate dehydrogenase; CRP, C-reactive protein; PE, pulmonary embolism; OR, odds ratio; CI, confidence interval; CCI, Charlson Comorbidity Index.
Summary of key findings.
Persistent symptoms at 1 year of follow-up are listed in Table 3. The main symptoms reported by patients were dyspnea (32%), followed by myalgia (9%), cough (7%), and neuropsychiatric symptoms such as anxiety (4%) and depression (5%). In 27% of patients with persistent dyspnea, this was classified as mild to moderate (mMRC scale score 1–2).
Main symptoms at 1-year follow-up after severe COVID-19 infection, N = 135.

mMRC, Modified Medical Research Council Dyspnea Scale.
When comparing patients with persistent dyspnea at 1 year with those who were asymptomatic (Table 4), there were no differences between the two groups regarding clinical factors related to COVID-19 severity or the burden of comorbidities.
Differences between patients with and without persistent dyspnea at 1-year follow-up.

LDH, lactate dehydrogenase; CRP, C-reactive protein; ICU, intensive care unit, IQR, interquartile range; BMI, body mass index; HFNC, high-flow nasal canula; OTI, orotracheal intubation; PE, pulmonary embolism; O2, oxygen; CCI, Charlson Comorbidity Index.
Discussion
In our study, we found that the prevalence of an oxygen requirement at 1 year after severe COVID-19 infection was 13%. This was the first report on a persistent oxygen requirement with a follow-up period of 1 year after severe COVID-19. In a comparison with data from a cohort of patients referred to COVID recovery clinics in Texas in the United States, the reported persistence of a supplemental oxygen requirement (13%) was similar to our finding. However, that study had a median follow-up of only 37 days and included fewer patients with severe COVID-19 infection. 9 Another retrospective study performed in Atlanta, Georgia found that 13.2% of patients required supplemental oxygen upon discharge. Although that series included 13.5% with a requirement for orotracheal intubation and 21.6% required ICU admission, the median patient follow-up time was not specified. 10 A study in a veterans hospital in Chicago, Illinois reported that 23% of patients hospitalized with COVID-19 were discharged with a supplemental oxygen requirement. Of those, only 3% required orotracheal intubation. At 8 weeks after hospital discharge, 52% of patients no longer required supplemental oxygen. 11 According to follow-up reports at 60 days after hospital discharge in another study in the United States, 2.6% of patients required supplemental oxygen. In that study, 13.2% of the included patients had been admitted to the ICU. 12 It is difficult to make comparisons between our data and those of previous reports given the heterogeneity of disease severity and shorter follow-up times in past studies. However, our estimates regarding supplemental oxygen requirements at 1 year of follow-up were greater than those reported in other study populations, which may be explained our inclusion of only patients with severe COVID-19 infection. Another possible explanation is that altitude is a factor associated with a greater need for supplemental oxygen after severe COVID-19 infection; the study city, Bogotá, Colombia, is at 2600 meters above sea level (m.a.s.l.). However, there are no published data in this regard. Future studies are needed to confirm this hypothesis.
The prevalence of dyspnea at 1 year in our population was 32%; among these patients, 27% were classified as having mild to moderate dyspnea. Global studies show conflicting data regarding the presence of dyspnea, making comparisons with our results difficult. A multicenter study in Madrid, Spain with a 7-month follow-up and 7% of patients who had severe COVID-19 reported rates of 55% dyspnea with some degree of physical activity and 23.5% at rest. 13 Another Spanish study that followed hospitalized patients with COVID-19 infection for 1 year found persistent dyspnea in 23.3%, but a low proportion of the study population had experienced severe COVID-19 infection (6.6%). 4 Two European cohorts with 1-year follow-up reported the presence of dyspnea in approximately 70% of patients, although severe COVID-19 cases in those studies only comprised 10.4% and 27.8% of patients.14,15 A meta-analysis with 3-month follow-up found a prevalence of dyspnea of 0.37 (95% CI 0.28–0.48, I2 = 93%). 16 More recent data from an Italian cohort found that 45% of patients discharged after severe COVID-19 infection reported dyspnea, most with mild severity (mMRC 1–2). The authors of that past study also carried out pulmonary function tests and found greater regional homogeneity among patients with dyspnea both when breathing at rest and during forced expiration. 17 Differences between the prevalence of dyspnea in our study and those reported in the above studies may be mainly associated with COVID-19 severity and the duration of follow-up.
The prevalence of symptoms at 1 year in our population was more than 50%, mainly owing to dyspnea, followed by myalgia in 9% of patients, cough in 7%, asthenia in 6%, and muscle weakness in 2%. The percentages of persistent symptoms were similar to those of previous reports. A multicenter study in Madrid with a 1-year follow-up found that more than 80% of patients had persistent symptoms. Among patients, the most frequently reported symptoms were fatigue (61.2%), chest pain (6.5%), and cough (2.5%). 4 Another multicenter study in Madrid with a 7-month follow-up showed that 61% of patients had fatigue. 13 A series in Italy found a high prevalence of persistent symptoms after COVID-19 infection, in 91.7% of patients. 14 As mentioned, our data could differ owing to disease severity among the included patients and the longer follow-up time in this study.
Regarding neuropsychiatric symptoms, we found a 1-year prevalence of anxiety in 4% and depression in 5% of patients. One month after COVID-19 infection, up to 56% of patients present alterations in at least one clinical dimension. 18 In China, after 6 months of follow-up in hospitalized patients, a 76% prevalence of neuropsychiatric symptoms is reported. 19 Another study reported that 17.39% of patients were diagnosed with anxiety disorder after COVID-19 infection. 20 Follow-up of 4 to 7 months in critically ill patients has revealed a 20% prevalence of anxiety and depression. 21 As above, heterogeneity is evident with respect to our present findings and past reports. In addition to the specific characteristics of our study population, we cannot rule out that the definitions of neuropsychiatric symptoms did not adhere to classifications of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.
A recent publication focusing on quality of life assessment 1 year after severe COVID-19 infection according to ICU discharge reports found lower scores in physical and mental components. The factors associated with lower health-related quality of life were female sex, a history of cardiovascular and liver disease, and length of ICU stay. That study also identified a 30% prevalence of post-traumatic stress disorder in the patient population. 22
We found that factors associated with the need for supplemental oxygen 1 year after severe infection with COVID-19 corresponded to sex, with female patients having a three times greater risk than male patients, as well as Charlson Comorbidity Index > 4. No previous analysis has been done regarding this association. We examined reported factors associated with a lower probability of recovery from severe COVID-19, which include female sex (OR 0.68, 95% CI 0.46–0.99), obesity (OR 0.50, 95% CI 0.34–0.74), and the requirement for invasive ventilation (OR 0.42, 95% CI 0.23–0.76). 15 Another study also found a higher proportion of women among patients with persistent symptoms (OR 1.75, 95% CI 1.37–2.24, p < 0.001). 13 Until now, it remains unknown whether these rates correspond to the pathophysiological basis of the disease or where there is a direct association with sex. However, deconditioning as a cause of exercise limitation has been consistently documented in this population. 23
One of the main limitations of our study is the high rate of loss to follow-up (21% of patients). However, a sensitivity analysis (Supplementary Table 1) showed no significant difference between patients lost to follow-up and those included in the study.
A second limitation is that we could not clearly differentiate whether all reported symptoms were exclusively associated with long COVID or whether symptoms were a manifestation of post-intensive care syndrome, which is related to chronic cognitive and psychological symptoms. 24 However, this syndrome has not been found to be related to a chronic oxygen requirement.
Another study limitation is the relatively small number of outcomes, which limits our findings regarding the importance of associated factors. Multicenter studies with a larger sample size are needed to assess these associations with greater precision. Finally, pulmonary outcomes were only assessed according to symptomatic evaluation and the persistence of a supplemental oxygen requirement. Further studies are needed that include complimentary evaluations, such as echocardiogram to evaluate pulmonary hypertension, thorax tomography to evaluate pulmonary fibrosis, and pulmonary function tests to more thoroughly evaluate long-term pulmonary function.
Conclusions
We found a high prevalence of supplemental oxygen requirement at 1 year after severe COVID-19 infection. This percentage was higher than expected and may be associated with the altitude of the location where our patients lived and where the present study was carried out (Bogotá, at 2600 m.a.s.l.). Our data suggest that female sex and a Charlson Comorbidity Index score >4 are factors associated with a persistent requirement for supplemental oxygen after severe infection with COVID-19.
There is a high burden of symptoms 1 year after severe COVID-19, mainly owing to dyspnea, musculoskeletal, and neuropsychiatric symptoms. No factors have been identified to predict which patients will present long-term symptoms. The present findings will be useful to generate follow-up programs in which complete functional evaluation can be carried out and effective rehabilitation strategies can be established.
Research data
Research Data for Factors associated with oxygen requirement and persistent symptoms 1 year after severe COVID-19 infection
Research Data for Factors associated with oxygen requirement and persistent symptoms 1 year after severe COVID-19 infection by María Natalia Serrano, Oscar Mauricio Muñoz, Camilo Rueda, Alejandra Cañas- Arboleda, Juan David Botero, Marlon Mauricio Bustos in Journal of International Medical Research
Footnotes
Acknowledgements
We gratefully acknowledge the role of all our colleagues, nurses, and others involved in the care of patients.
Author contributions
Conception and design: NS, OM; collection of study materials or patients: collection and processing of data: MS, CR, JDB; data analysis and interpretation: NSS, OMM; manuscript writing: AC, MB; final approval of the manuscript: all authors.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors received financial support for this research from Hospital Universitario San Ignacio.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
