Abstract
Background
Understanding distinct neurobiological mechanisms underlying bipolar disorder (BD) and major depressive disorder (MDD) is crucial for accurate diagnosis and the discovery of novel and more effective targeted treatments. Previous diffusion-weighted MRI studies have suggested some common frontotemporal corticolimbic system white matter (WM) abnormalities across the disorders. However, critical to the development of more precise diagnosis and treatment is identifying distinguishing abnormalities. Promising candidates include more prominent frontotemporal WM abnormalities observed in BD in the uncinate fasciculus (UF) that have been associated with frontal-amygdala functional dysconnectivity, and with suicide that is especially high in BD. Prior work also showed differentiation in metabotropic glutamate receptor 5 (mGlu5) abnormalities in BD versus MDD, which could be a mechanism affected in the frontotemporal system. However, associations between WM and mGlu5 have not been examined previously as a differentiator of BD. Using a multimodal neuroimaging approach, we examined WM integrity alterations in the disorders and their associations with mGluR5 levels.
Methods
Individuals with BD (N = 21), MDD (N = 10), and HC (N = 25) participated in structural and diffusion-weighted MRI scanning, and imaging with [18F]FPEB PET for quantification of mGlu5 availability. Whole-brain analyses were used to assess corticolimbic WM matter fractional anisotropy (FA) across BD and MDD relative to HC; abnormalities were tested for associations with mGlu5 availability.
Results
FA corticolimbic reductions were observed in both disorders and altered UF WM integrity was observed only in BD. In BD, lower UF FA was associated with lower amygdala mGlu5 availability (p < .05).
Conclusions
These novel preliminary findings suggest important associations between lower UF FA and lower amygdala mGlu5 levels that could represent a disorder-specific neural mechanism in which mGluR5 is associated with the frontotemporal dysconnectivity of the disorder.
Keywords
Introduction
Bipolar disorder (BD) and major depressive disorder (MDD) are debilitating and unfortunately common mood disorders with a lifetime prevalence of 2.4% and 13.2%, respectively.1,2 It is often challenging to differentiate between BD and MDD during depressive episodes.3,4 In fact, about 60% of BD individuals are initially misdiagnosed with MDD possibly due to the earlier onset of depression than hypomania and the higher prevalence of depressive over hypomanic symptoms.4,5 This can significantly delay appropriate treatment and worsen their symptoms. 6 Therefore, it is important to understand the overlaps and differences in neuropathology between BD and MDD. One potential differentiator is white matter structural alterations, which have been consistently reported across BD and MDD7,8; however, the biological mechanisms underlying such deficits in these disorders remain poorly understood. Identification of common and distinct white matter abnormalities between BD and MDD may not only yield critical information for understanding neuropathology but also help provide precise neural targets for accurate diagnosis and effective treatment for both disorders.
Neural models of BD and MDD have proposed that abnormalities in frontal-limbic connectivity interfere with emotional regulation processes and thus may contribute to the development and expression of mood symptoms.9,10 While these models rely on evidence that has stemmed primarily from functional magnetic resonance imaging (fMRI) studies, results from studies examining white matter integrity have shown far more extensive alterations in BD and MDD.7,8 The most recent meta and mega-analytic findings revealed that both BD and MDD (separately compared to health controls (HC)), showed lower values of fractional anisotropy (FA)—a metric derived from diffusion tensor imaging (DTI) that can be used as a measure of structural integrity within wide-spread white matter tracts. 11 The largest effect sizes were observed from both BD and MDD studies in the corpus callosum (CC).7,8 From the BD meta-analysis findings, the additional biggest effect size was reported in the cingulum, whereas in the MDD study, the corona radiata was found to show the largest differences in addition to the CC.7,8 Importantly, observed white matter deficits in both BD and MDD are frequently associated with the presence and severity of clinical symptoms, cognition and suicidality.12-16 Nonetheless, direct comparisons focused on studying the nature of white matter FA and its underlying biological mechanism between BD and MDD relative to HC are scarce,5,17,18 thus limiting our understanding of how white matter deficits distinctively affect these disorders.
Of note, evidence has linked abnormal cortical neuron activity with reduced white matter integrity in various pathological states, providing one possible explanation for observed white matter reduction in MDD and BD.19-21 For example, an association between the availability of metabotropic glutamate receptor 5 (mGlu5)—a g-protein coupled receptor which plays a key role in neuromodulation—and white matter integrity has been observed in healthy subjects. 22 More specifically, mGlu5 modulates synaptic transmission and plasticity of glutamatergic synapses with glutamate being the primary excitatory neurotransmitter governing the frontal-limbic circuitry that subserves emotion regulation.23-25 A dysfunctional glutamatergic system is thought to play a critical role in mood disturbances in both BD and MDD. 26 MGlu5 is thought to be a main contributor to synaptic plasticity and has important implications for both cognitive function and mood regulation.3,23,27 Postmortem work shows lower mGlu5 receptor levels in individuals with BD,28-30 a finding recently confirmed by the first in vivo work from our group. 3 Specifically, we observed significantly lower prefrontal cortex (PFC) mGlu5 availability in BD versus MDD and HC. Notably, a larger body of work regarding mGlu5 in MDD shows mixed findings,3,31-34 with some studies showing lower receptor availability while others not showing alterations in vivo or postmortem. In our recent study, no difference was found between MDD and HC, but lower dorsolateral PFC (dlPFC) mGlu5 was associated with worse depressive symptoms in MDD. 3
MGlu5-mediated glutamate signaling in the gray matter has been suggested to play a potential role in maintaining or promoting white matter properties in healthy human brains.22,35 Despite the growing evidence showing white matter deficits and altered mGlu5 availability in BD and MDD, no published studies have trans-diagnostically examined the potential relationship between white matter deficits and mGlu5 abnormalities. This knowledge could lead to more efficacious and precise treatments for individuals with these disorders. Here, by conducting a preliminary study, we aimed to explore (1) identify white matter alterations common and distinct in BD and MDD groups and (2) examine potential relationships between the identified white matter alterations and the neighboring gray matter mGlu5 availability. We tested the hypothesis that BD and MDD would show common decreases in white matter FA in the CC and frontotemporal regions of the frontal-limbic circuitry and explored other potential differences within each diagnosis. Secondarily, we explored whether the identified lower white matter FA in BD and MDD would be associated with neighboring gray matter mGlu5 availability and differ in patterns from HC. Exploratory correlational analyses were also conducted to examine the associations between symptom measures, white matter FA and mGlu5 availability.
Methods
Participants
A total of 65 participants were included in the current investigation. Twenty-one subjects with BD (Mage ± SD = 31.2 ± 10.6; 13 males and 8 females), 10 individuals with MDD (Mage ± SD = 27.9 ± 5.4; 3 males and 7 females) and 25 HC (Mage ± SD = 36 ± 15.1; 13 males and 12 females) completed the study. All participants were recruited from New Haven and the greater New Haven area by flyers, community outreach, referrals from local clinics/hospitals and word of mouth. Written informed consent was obtained from all participants in accordance with Yale University’s institutional review board. Screening procedures included electrocardiography, complete blood counts, serum chemistries, thyroid function test, liver function test, urinalysis and urine toxicology screening, and plasma pregnancy tests (for female participants). The Structured Clinical Interview for DSM-V was used to confirm subjects’ diagnoses. 36 Depressive symptoms were assessed using the Montgomery–Åsberg Depression Rating Scale (MADRS) 37 and anxiety symptoms were assessed by the Hamilton Anxiety Rating Scale (HAM-A). 38 No subjects had major medical or neurological disorders including a history of loss of consciousness for more than 5 min. Demographic, clinical, and radiotracer characteristics are shown in Table 1.
Demographic and clinical characteristics for BD, MDD, and HC.

Data are presented as M ± SD or percentage.
Abbreviations: BD, bipolar disorder; MDD, major depressive disorder; HC, health controls; MADRS, Montgomery–Åsberg Depression Rating Scale; HAM-A, Hamilton Anxiety Rating Scale.
p < .01 HC versus MDD.
p < .01 HC versus BD.
Main effect of three groups.
MRI Acquisition
T1-weighted MR and diffusion MR scans were acquired for all participants on a single 3-Tesla Siemens Trio MR system (Siemens, Erlangen, Germany). The T1-weighted MR scans with magnetization-prepared rapid gradient-echo (MPRAGE) sequence (TR = 1500 ms, TE = 2.83 ms, matrix = 256 × 256, field of view = 256 × 256 mm2, 160 1-mm slices without gap and two averages) were obtained to exclude structural abnormality and for coregistration with positron emission tomography (PET) scans. DTI data were obtained with 32 noncolinear directions (b = 1000 s/mm2) and acquisition without diffusion weighting b = 0 was acquired with the alignment of the anterior commissure-posterior commissure plane (TR = 7400 ms, TE = 115 ms, matrix = 128 × 128, field of view = 256 × 256 mm2, 40 3-mm slices without gap).
MR Data Processing
The diffusion MRI data were preprocessed for motion and eddy-current correction by using the FMRIB Software Library (FSL5; http://fsl.fmrib.ox.ac.uk/fsl). By fitting a tensor model to the raw diffusion data, voxel-wise values of FA were obtained. All subjects’ FA data were aligned to MNI standard space by using the nonlinear image registration tool (FNIRT). Next, the mean FA image was created and thinned to create a mean FA skeleton, which represents the centers of all tracts common to the group. Each subject's aligned FA data were then projected onto this skeleton and the resulting data were fed into voxel-wise cross-subject statistics.
Brain [18F]FPEB PET Imaging Acquisition
The radiotracer [18F]FPEB was synthesized onsite at the Yale PET Center (as previously). 39 [18F]FPEB was injected i.v. using a bolus plus infusion (B/I) paradigm over 120 min.39,40 Equilibrium was reached at 60 min, and emission data were acquired 90-120 min after the start of injection on the high-resolution research tomograph (HRRT; Siemens/CTI). The HRRT has an intrinsic spatial resolution of approximately 2.5 mm full width at half the maximum. Blood samples were drawn at 15, 20, 25, 30, 40, 50, 60, 75, 90, 100, and 120 min postinjection to permit calculation of a metabolite-corrected venous input function. This procedure has been validated by our research team in previous studies.32,39 A 6-min transmission scan was obtained for attenuation correction. Head motion was tracked using the Polaris Vicra optical tracking system (Vicra, NDI System Waterloo). There were no significant differences in the injected dose or mass between BD, MDD, or HC groups (overall injected M = 168 MBq, SD = 0.85). Dynamic scan data were reconstructed with corrections for attenuation, normalization, randoms, scatter, dead time, and motion using the ordered-subset expectation maximization-based MOLAR algorithm. 41
PET Data Analysis
All PET images were first coregistered to the participant's T1-weighted MRI images using a six-parameter mutual information algorithm (FLIRT, FSL 3.2, Analysis Group, FMRIB). Images were then coregistered to the MR template via nonlinear transformation using the Bioimagesuite software (Version 2.5; www.bioimagesuite.com). Regions of interest were identified using the Anatomical Automatic Labeling for Statistical Parametric Mapping (SPM) 2 template (http://www.fil.ion.ucl.ac.uk/spm). PET time-activity curves were generated for our regions of interest (ROIs) covering the frontal-limbic circuitry: the orbital frontal cortex (OFC), anterior cingulate cortex (ACC), ventral medial prefrontal cortex (vmPFC), dorsal lateral PFC (dlPFC), insula cortex, amygdala, hippocampus as well as CC. Gray matter segmentation was conducted using the computational anatomy toolbox for SPM2 (CAT). No appropriate reference region completely devoid of mGlu5 is available in the human brain. 41 As such, our outcome measure was calculated as volume of distribution (VT: ratio of radioligand concentration in region of interest to the metabolite-corrected radioligand concentration in plasma, calculated at equilibrium). VT was estimated using the equilibrium analysis method with venous input function.32,39,42 No differences were observed in free fraction in plasma. Therefore, as previously, [18F]FPEB VT represents mGlu5 availability.
Diffusion MR Data Statistical Analysis
We compared the skeletonized diffusion parameters between BD, MDD and HC subjects. Permutation-based testing (5000 permutations) was carried out with Randomise, using Threshold-Free Cluster Enhancement (TFCE), 43 and p-values were controlled for family wise error rate (FWE) taking into account multiple spatial comparisons. Randomise was used to compare FA values between BD, MDD and HC. TFCE controlled uncorrected p < .05 was considered significant. Age and sex were added as covariates. Of voxels that showed main effect of three groups, the mean FA values were extracted for each subject. By using SPSS Statistics v22 (IBM), one-way ANOVA and post-hoc 2-sample-T tests were subsequently performed to investigate the overall three-group effect and differences between each diagnostic group relative to HC of FA (p < .05).
To explore potential FA differences in BD and MDD relative to HC, respectively, permutation-based testing (5000 permutations) was carried out with Randomise, using TFCE 43 on two-group comparisons (BD vs HC and MDD vs HC). TFCE controlled uncorrected p < .05 was considered significant. Age and sex were added as covariates.
Diffusion and PET Data Statistical Analysis
Potential associations between white matter FA deficits and regional mGlu5 levels within each diagnostic group were assessed using an ROI approach, including white matter regions showing between-group differences of FA in the frontal-limbic circuitry. The Johns Hopkins University (JHU) ICBM-DTI-81 white-matter labels atlas provided by FSL was chosen to define the ROIs. Regression analyses within each diagnostic group were conducted by applying permutation-based testing (5000 permutations) with Randomise, using TFCE. 43 TFCE, family-wise error controlled uncorrected p < .05 was considered significant. Age and sex were added as covariates.
Associations Between Altered White Matter and Clinical Characteristics
To investigate the relationship between FA and subjects’ clinical measures, mean FA values were first extracted per individual on voxels showing significant group differences (three-group as well as two-group comparisons). Pearson's correlation analysis was then performed to study the association between these measures by using SPSS v22 (IBM).
Results
Demographic and Clinical Characteristics
Age and sex did not differ significantly between BD, MDD and HC. For mood states in BD subjects, one individual was in manic state, 8 individuals were in depressed state and 12 individuals were in a euthymic state at the time of scans. Moreover, 15 out of 21 BD subjects and 4 out of 10 MDD subjects had taken antipsychotic/SSRI medications. Between subject groups, there was a significant main effect of the group on the MADRS and HAM-A ratings with both BD and MDD showing significantly higher scores than HC (p < .001) (see Table 1).
Differences in Whole Brain White Matter Integrity Across BD, MDD, and HC
We observed a significant main effect of three groups in the CC and the thalamic radiation (TR) but not in other frontal-limbic regions (see Figure 1). Mean FA values per individual were extracted from the bilateral thalamic radiation and the corpus callosum at TFCE controlled p < .05. The post hoc t-tests revealed that the mean FA values in the MDD subjects were significantly lower than the HCs in the TR, while the CC mean FA values were significantly lower in both MDD and BD individuals relative to HCs (see Figure 1). Since distinct functional neural deficits have been reported between manic and depressive mood sates in BD, 44 subanalysis was conducted excluding one BD individual with a manic mood state and we found these findings remained significant after the exclusion (data not shown). Additional subanslysis was performed by adding medication as a covariate and findings also remained significant (data not shown).

(A) Whole-brain tract-based spatial statistics analysis shows main effect of groups between bipolar disorder (BD), major depressive disorder (MDD), and healthy control (HC). Panel 1A shows the main effect of groups between BD, MDD, and HC in the corpus callosum (circled in blue) and the bilateral thalamic radiation (circled in yellow). The whole brain skeleton of mean fractional anisotropy is displayed in green and the significant main effect of the group is displayed in red. (1B–1D). The extracted mean fractional anisotropy per subject of the (1B) corpus callosum and (1C) left and (1D) right thalamic radiation from the main effect of groups. Data are graphed as the M ± SD with significant post hoc independent samples t-tests revealed that lower fractional anisotropy (FA) in the BD group (p = .003) and MDD group (p = .01) relative to HC. (C) showed a significant main effect of groups in the left thalamic radiation. (D) The significant main effect of groups in the right thalamic radiation with post hoc independent samples t-tests revealed lower FA in the BD relative HC (p < .001).
Differences in Whole Brain White Matter Integrity Between BD Versus HC and MDD Versus HC
When BD individuals were compared to HCs, significantly lower FA was observed in the CC and the bilateral TR, the bilateral uncinate fasciculus (UF), the bilateral superior longitudinal fasciculus (SLF), the bilateral inferior frontal-occipital fasciculus (IFOF) and the cerebellum (see Figure 2). These findings remained significant when subanalysis was conducted excluding one BD individual with a manic mood state. Sensitive analysis was also performed by only including BD subjects with euthymic mood states (N = 5), no significant findings were observed. Additional subanslysis was performed by adding medication as a covariate and findings also remained significant (data not shown).

Whole-brain tract-based spatial statistics analysis shows lower fractional anisotropy in bipolar disorder subjects relative to health control. Lower fractional anisotropy (FA) was seen in BD as compared to HC in the corpus callosum (circled in blue) (A); bilateral uncinate fasciculus (circled in blue) and the bilateral inferior frontal-occipital fasciculus (B); left SLF (C); right SLF (D); bilateral cerebellum white matter (E). The whole brain skeleton of mean fractional anisotropy is displayed in green and significantly lower fractional anisotropy in BD relative to HC is displayed in red.
When compared MDD relative to HC groups, lower FA was observed in the CC, bilateral TR, the fornix, the acoustic radiation (AR), and the bilateral IFOF (see Figure 3). In BD, the lower FA in the bilateral SLF was identified near the middle frontal gyrus and motor brain regions, while in MDD the FA alterations were concentrated in the temporal part of the brain. Additional subanslysis was performed by adding medication as a covariate and findings remained significant (data not shown).

Whole-brain tract-based spatial statistics analysis shows lower fractional anisotropy in major depressive disorder subjects relative to health control. (A) shows lower fractional anisotropy (FA) in the corpus callosum (circled in blue) in individuals with major depressive disorder (MDD) relative to healthy control (HC). (B) Shows lower FA in the fornix (circled in blue) in the MDD relative to HC. (C) Shows lower FA in the bilateral thalamic radiation (circled in yellow) and the bilateral acoustic radiation (circled in blue) in the MDD relative to HC. (D) Shows lower FA in the left superior longitudinal fasciculus in the MDD relative to HC. (E) Shows lower FA in the right superior longitudinal fasciculus in the MDD relative to HC. (F) Shows lower FA in the bilateral inferior frontal-occipital fasciculus (circled in blue) in the MDD relative to HC. Whole brain skeleton of mean fractional anisotropy is displayed in green and significantly lower fractional anisotropy in MDD relative to HC is displayed in red.
Associations Between White Matter Integrity and mGlu5
In the BD group, the UF FA was positively associated with mGlu5 availability in the amygdala. These findings remained significant when subanalysis was conducted excluding one BD individual with a manic mood state. Sensitive analysis was also performed by only including BD subjects with euthymic mood states (N = 5), no association between FA and mGlu5 were observed (data not shown). We did not observe any significant associations between white matter FA and mGlu5 availability in MDD or HC groups (see Figure 4).
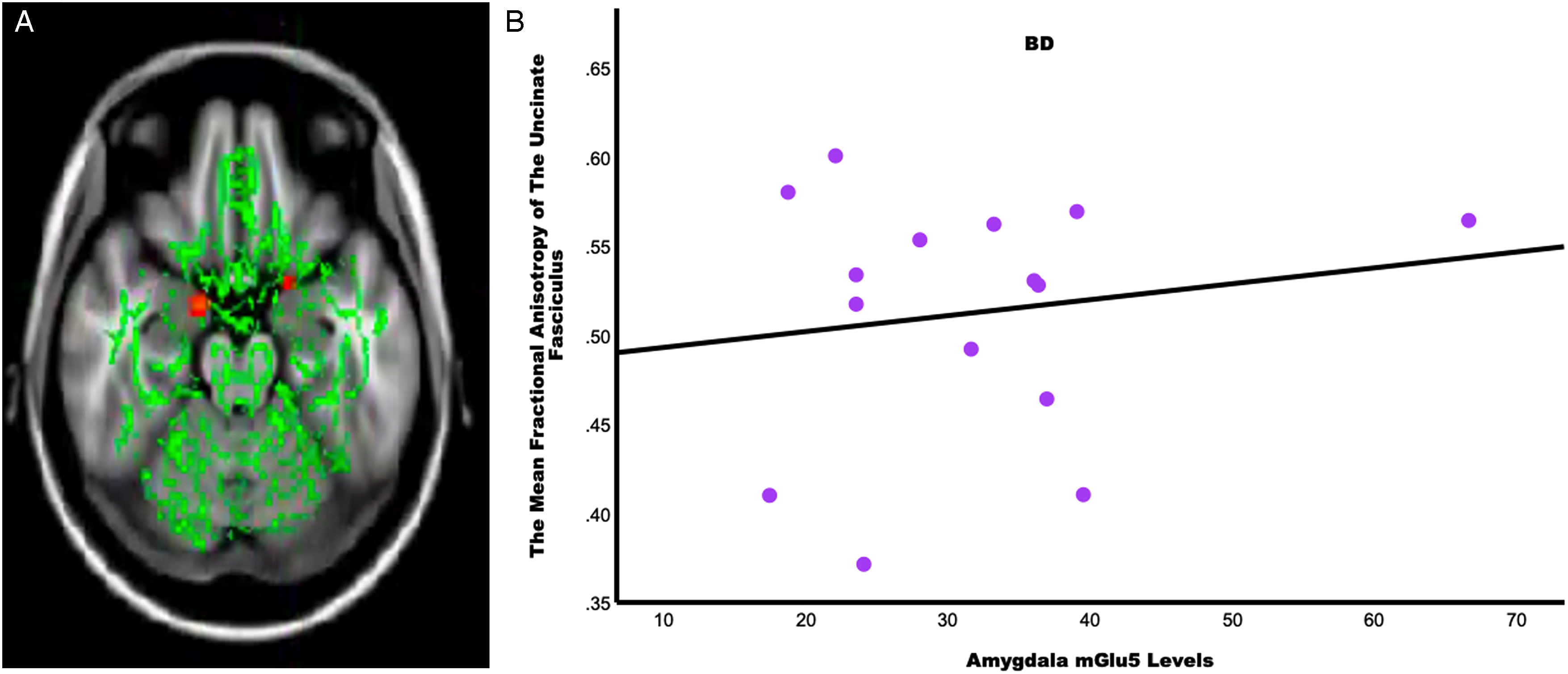
In subjects with bipolar disorder (BD), region of interest tract-based statistics analysis shows lower fractional anisotropy in the bilateral uncinate fasciculus is associated with lower metabotropic glutamate receptor 5 (mGlu5) in the amygdala. (A) Shows lower fractional anisotropy in the bilateral uncinate fasciculus is associated with lower mGlu5 levels in the bilateral amygdala. The whole brain skeleton of mean fractional anisotropy is displayed in green and a significant positive association between fractional anisotropy and amygdala mGlu5 levels in BD is displayed in red. (B) shows lower fractional anisotropy in the bilateral uncinate fasciculus (in Figure 4A) was associated with higher levels of mGlu5 in the amygdala (r = .39, p = .039).
Relationship Between White Matter Deficits and Clinical Measures
Within BD, the UF FA was found negatively correlated with anxiety levels (left: r = −.55, p = .034; right: r = −.61, p = .015) (see Figure 5). No significant correlation was found between white matter alterations and clinical measures in MDD. Additional explorative analysis was conducted to examine whether/how UF FA and amygdala mGlu5 might be together driving the anxiety symptoms in BD. Nevertheless, no significant interaction effect between altered FA in the UF and amygdala mGlu5 levels were observed on BD subjects’ anxiety levels.

In subjects with bipolar disorder, lower fractional anisotropy in the bilateral uncinate fasciculus was associated with worse anxiety symptoms. (A) Shows lower fractional anisotropy (FA) in the left uncinate fasciculus was associated with higher scores from the Hamilton Anxiety Rating Scale (HAM-A) (r = −.55, p = .034). (B) Shows lower FA in the right uncinate fasciculus was associated with higher HAM-A scores (r = −.61, p = .015).
Discussion
To our knowledge, this is the first study providing preliminary evidence of associations between white matter alterations and mGlu5 receptor availability across BD and MDD. Partially consistent with the study hypotheses, lower white matter FA was observed in the CC and the TR across BD and MDD. In BD, additional widespread altered FA was seen in the UF, SLF, IFOF, and cerebellum. In MDD, abnormal FA was also found in the fornix, AR, SLF, and IFOF. Within BD subjects, amygdalar mGlu5 availability was associated with UF FA, and lower FA in the UF was associated with greater anxiety symptoms.
Lower FA observed in the CC across BD and MDD provided robust and promising findings in line with the most recent published meta and mega studies7,8 suggesting its important role in mood disorders. The CC is the largest interhemispheric commissure in the brain8,45 and provides vital connections for the ACC and the OFC in both hemispheres. The ACC and the OFC are key brain regions in the frontal-limbic circuitry for emotion regulation, and abnormalities in these two regions have been often reported in both BD and MDD.46-48 It has been suggested by other preliminary data that abnormal interhemispheric connectivity could be a disease marker rather than a vulnerability marker for affective disorders. 49 Perhaps with a moderate effect size relative to the CC, FA alterations in the TR were also reported by several studies in both BD and MDD.50,51 Our post hoc t-test revealed significantly lower FA was only present in MDD relative to HC. The TR are bilateral fibers connecting the thalamus and cerebral cortex. 51 The thalamus is a key structure in the limbic system with vast connections to many important brain areas.52,53 It supports emotional, cognitive and motor processes, and deficits in the thalamus have been often implicated in the neuropathology of MDD.51,52 The current findings suggest BD and MDD might be both vulnerable to white matter deficits in the TR; however, in comparison to MDD, reduced TR FA seemed to be more pathologically prone to BD.
Comparing BD to HC, additional reduced FA was observed in the bilateral UF, bilateral SLF, bilateral IFOF and the bilateral cerebellum. The UF carries a major connection from the ventral PFC to the amygdala and decreased FA in the UF has been associated with anxiety, impulsivity and suicidality in BD.13,54-56 The observed reduced FA in the UF in BD subjects are consistent with previous DTI studies.57,58
Consistent with previous studies, decreases in FA in the SLF and IFOF were reported in BD and MDD, emphasizing their important involvement in the neuropathology of mood disorders.5,59,60 The SLF is an association tract passing through the frontal, parietal, temporal, and occipital lobes. 61 In this BD sample, SLF deficits were mainly seen in the PFC and motor brain regions. The main component of SLF terminates in the PFC and may provide support to working memory function 62 ; whereas the dorsal part of the SLF passes through the motor areas and is involved in the regulation of motor behavior. 61 Therefore, it is reasonable to speculate that such SLF abnormalities might intensify cognitive and motor impairments that have been reported in BD.3,63 In the MDD individuals, decreases in FA were more prominent in the temporal brain regions. The temporal lobe is involved in a variety of important cognitive functions, such as sensory and language processing, and impairments in these functions have been reported in MDD.64,65 The IFOF also bridges between the frontal, parietal, temporal and occipital lobes. 66 Evidence has shown that IFOF is associated with attention and affective behavior.66,67 Here, BD subjects showed widespread FA reductions in the IFOF, while in the MDD individuals altered FA was more localized to the occipital area. Since no main effects of group were observed in the IFOF, we could only tentatively speculate that this region might be more affected in BD pathology relative to MDD and could be contributing to the altered affective processing.
More specific to MDD, decreased FA was found in the fornix and the AR. The fornix is part of the limbic system and the major output tract of the hippocampus. 68 It is thought to be involved in memory functioning. 68 This finding is again consistent with previous studies69,70 highlighting the vital role underlying the neuropathology of MDD. The AR is part of the auditory system, and the tract terminates in the primary auditory cortex. 71 The functions of AR are less understood, and its deficits seem to be underreported in psychiatric disorders relative to other white matter tracts. Nonetheless, a study showed altered mean diffusivity (a measure of white matter integrity) in MDD subjects and its association with worse depressive symptoms. 72
When examining associations between FA and mGlu5 availability, we observed significant associations in the BD group only. Specifically, we showed for the first time that lower FA in the UF was significantly associated with lower amygdala mGlu5 availability. As mentioned, the UF is known for its important connection to the amygdala involving emotional processing, impulsive control, and suicidality, and its abnormalities have been consistently reported in BD.13,54-58 A recent study showed evidence of lower mGlu5 levels in the PFC regions in BD relative to HC. 3 Although the study did not examine the amygdala region, the current finding provides a suggestion that suboptimal levels of mGlu5 in the amygdala seem to relate to the reductions in UF FA. At this stage, it remains challenging to unfold the underlying neural mechanism of the association between lower FA in the UF and lower amygdala mGlu5 levels, however, their relationships with clinical measures may add more insight. We found that worse anxiety in BD was associated with lower FA in the UF but not with amygdala mGlu5 levels. This evidence suggests that white matter deficits between the PFC and amygdala seem to contribute to BD subjects’ anxiety levels while such symptoms were not affected by suboptimal levels of mGlu5 in the amygdala. Hence, we may speculate that abnormalities in the amygdala mGlu5 availability might disrupt the normal glutamatergic signaling in the frontal-limbic circuitry and in some way amplify the decreases in the UF FA and subsequently encourage the anxiety symptoms in BD individuals.
In MDD, we did not observe significant associations between altered FA and mGlu5 availability in the frontal-limbic circuitry. This may not be surprising as there has not been clear evidence indicating abnormalities in mGlu5 levels in MDD. 3 Thus, it might be that other neurotransmitter systems are associated with (or subserve) this particular alteration in MDD. For example, GABAergic deficits have been strongly implicated in MDD neuropathology. Neurotransmitter γ-aminobutyric acid (GABA) is the main inhibitory neurotransmitter in the brain and there has been abundant evidence suggesting GABA contributes prominently as a biological risk factor to MDD.73,74
Study limitations include a relatively small sample size for MRI-PET investigation that might limit the observations of significant findings especially in the MDD group as well as enhancing the likelihood of type 2 error for all the analyses we have conducted to study the relationships between white matter FA, mGlu5 and clinical characteristics. However, given the novelty of the work and the need to better differentiate and treat BD and MDD, we believe these data are critical. Second, even though the study used prior-selected ROIs based on the previous literature, a rather lenient thresholding was used in this suboptimal sample size to avoid false negatives. Third, our study only included one BD subject in a manic mood state which restricts our ability to generalize these findings across the BD mood states. Four, although subanalysis was conducted by adding medication as a covariate, we cannot completely rule out medication use as a potential confound in our findings. Five, the BD group had more males relative to the MDD group while the MDD group had more females relative to the BD group. Although we controlled for sex in all the main analyses, potential sex differences might be underdetected due to the small sample size. Six, in order to rule out mood state-related changes in mGlu5 relationship with FA, subanalyses were conducted including only the 5 euthymic BD subjects and no significant associations were observed. Nonetheless, it is possible that the low sample size restricted the statistical power for detecting the potential effects of euthymia. Therefore, future studies need to replicate these findings using larger transdiagnostic sample size and with individuals across BD mood states to examine associations between white matter deficits and mGlu5 levels (Table 2).
Means and standard deviations of FA values for BD, MDD, and HC in the corpus callosum and bilateral thalamic radiation.

Abbreviations: BD, bipolar disorder; MDD, major depressive disorder; HC, health controls.
p = .01 HC versus MDD.
p = .003 HC versus BD.
cp < .001 HC versus BD.
d Main effect of three groups.
In conclusion, the results of the current study are consistent with some of the previous findings of white matter alterations in BD and MDD and importantly provided the first preliminary evidence showing an association between white matter FA alterations in the UF and amygdala mGlu5 levels in BD individuals. Moreover, lower FA in the UF was significantly correlated with greater anxiety symptoms in the BD. These findings suggest white matter deficits in the CC and the TR might be a common disease marker for these affective disorders. However, the association with mGlu5 levels in the BD group only suggests the molecular underpinnings of white matter alterations may differ between these disorders and may point to precision medicine to treat symptoms of BD specifically.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the common fund (grant number R01MH116657).
