Abstract
Background
Females are twice as likely to experience post-traumatic stress disorder (PTSD) than males, yet specific factors contributing to this greater risk are not fully understood. Our clinical and recent preclinical findings suggest a role for the metabotropic glutamate receptor 5 (mGlu5) in PTSD and differential involvement between males and females.
Methods
Here, we further investigate whether mGlu5 receptor availability may contribute to individual and sex differences in PTSD susceptibility by quantifying receptor availability using the mGlu5 receptor-specific radiotracer, [18F]FPEB, and positron emission tomography in male (n = 16) and female (n = 16) rats before and after traumatic footshock exposure (FE) and assessment of stress-enhanced fear learning (SEFL) susceptibility, as compared with no-shock controls (CON; n = 7 male; n = 8 female).
Results
Overall, FE rats displayed greater fear generalization as compared with CON (p < .001). Further, greater mGlu5 receptor availability at baseline (p = .003) and post-test (p = .005) was significantly associated with expression of the SEFL phenotype. Notably, FE female rats displayed a shift to more passive coping (ie, freezing), and displayed greater SEFL susceptibility (p = .01), and had lower baseline mGlu5 availability (p = .03) relative to their FE male rat counterparts.
Conclusion
Results are consistent with clinical findings of higher mGlu5 receptor availability in PTSD, and add to growing evidence implicating these receptors in the pathophysiology of PTSD and sex-differences in susceptibility for this disorder.
Keywords
Introduction
Post-traumatic stress disorder (PTSD) is classified as a Trauma- and Stressor-Related Disorder in the 5th edition of the Diagnostic and Statistical Manual of Mental Disorders. 1 Its diagnosis requires that an individual experiences one or more traumatic event(s) with subsequent development of symptoms, such as new or worsening anxiety and depression, re-experiencing symptoms (such as flashbacks or nightmares), hypervigilance and arousal, and increased startle response. While it is estimated that most individuals (∼90%) experience at least one event that could be classified as a trauma, lifetime prevalence of PTSD is only 8.3%. 2 While the nature of the trauma itself, including severity, chronicity, and trauma type (eg, sexual violence), all significantly contribute to post-traumatic outcomes,3,4 these factors alone cannot account for the vast discrepancy between rates of trauma exposure and prevalence of PTSD in the general population and the extreme heterogeneity of post-traumatic outcomes. 5 Similarly, the well-documented sex differences in PTSD prevalence and presentation2,6 cannot be fully explained by differences in the rates of trauma exposure or the nature of traumatic experiences of men and women. 7 Collectively these data suggest additional factors contribute to individual and sex-differences in risk for developing PTSD following a traumatic event. Identification of predictive factors and elucidation of mechanisms which they mediate PTSD risk or promote resilience is paramount to the development of novel targeted treatments for what can otherwise be a debilitating, and oftentimes chronic, disorder.
In recent years, the metabotropic glutamate receptor 5 (mGlu5) has gained attention as a potential biomarker and molecular mediator of PTSD susceptibility.8,9 This Gq/11 protein-coupled receptor mediates forms of synaptic plasticity underpinning hippocampal-dependent memory, and particularly, affective learning processes.9‐11 Further, there is mounting evidence from human postmortem investigations and clinical neuroimaging studies that suggest dysregulation of the mGlu5 receptor in PTSD. Specifically, using positron emission tomography (PET) to measure mGlu5 receptor availability in vivo, our group has previously demonstrated higher mGlu5 receptor availability in PTSD that significantly correlates with symptom severity.12,13 However, it is important to recognize the limitations of these clinical studies due to their prospective and cross-sectional nature. Thus, it remains unclear if the higher mGlu5 receptor availability observed represents a difference that predates the trauma (that is, a PTSD risk biomarker), or rather a change which occurs in response to the trauma itself and/or develops over the course of PTSD pathology progression.
The ability to assess mGlu5 receptor availability in animals, before and after trauma exposure, provides a unique opportunity to model PTSD risk by conducting prospective studies that can also address mechanistic questions that are otherwise not feasible in human populations. Therefore, the primary aim of the present study was to investigate mGlu5 as a predictive biomarker of PTSD susceptibility by combining longitudinal PET imaging with a well-established rodent model of PTSD-like behavior. Specifically, we utilized stress-enhanced fear learning (SEFL), a behavioral paradigm developed to recapitulate critical aspects of PTSD, including enhanced startle response and sensitization of fear learning following exposure to an acute stressor.14‐16 We recently reported that behavioral responses to footshock stress and expression of contextual fear memory positively correlate with and increases in mGlu5 receptor availability following exposure to footshock stress relative to baseline. 17 However, in that study footshock exposure (FE) rats did not display enhanced fear learning and memory in a novel context as typically demonstrated with the SEFL protocol. Therefore, in the present study, we adopted a modified version of the SEFL paradigm based on the 2019 report of Hersman et al, 14 which allows for the assessment of enhanced fear learning with a conditioned stimulus in addition to contextual fear learning sensitization. Further, we wanted to examine behavioral differences between male and female rats exposed to the SEFL paradigm in order to assess the potential role of mGlu5 in mediating any observed sex-differences.
Methods
Animals and Experimental Timeline
Male (n = 24) and female (n = 24) Long Evans rats (Charles River Laboratories), age 55 and 60 days (∼8 weeks), were single housed in a climate-controlled room and maintained on a 12h light/dark cycle (lights on at 7:00) with ad libitum access to food and water. Prior to the start of the experiments, rats were given 5 to 6 days to acclimate to the facility and were handled daily by the experimenter. An overview of the experimental timeline is provided in Figure 1A. Briefly, PET with [18F]FPEB was conducted for baseline quantification of mGlu5 receptor availability. One male rat died during the baseline PET session, thus n = 23 was the final number of male rats included for these studies. Following baseline PET imaging, rats were randomized to either the FE (male: n = 16; female: n = 16) or no-shock control (CON; male: n = 7; female: n = 8) group prior to conducting a 4-day SEFL paradigm, and as detailed below. Twenty-four hours after day 4 of the SEFL paradigm, rats underwent scanning with [18F]FPEB for a second (ie, post-test) time. All experimental procedures were performed as approved by the Institutional Animal Care and Use Committee at Yale University and according to NIH and institutional guidelines and the Public Health Service Policy on Humane Care and Use of Laboratory Animals.

PET evaluation of mGlu5 receptor availability during SEFL. (
Behavioral Testing
The SEFL paradigm performed here was modified from that of Hersman et al. 14 Behavioral testing on each day of the 4-day protocol (Figure 1B) was conducted in chambers individually housed within a sound-attenuating enclosures and equipped with an infrared camera (Med Associates, Inc, Fairfax, VT). On day 1, rats were transported to the behavioral suite adjacent to the housing facility and placed into context A. For context A, the chamber square walls were left exposed and corncob bedding was placed in the catch-tray below the metal grid floor. FE rats received 15 footshocks (1.0 mA, 1 s, pseudorandom ITI) over a 90min session, while CON rats exposed to context A for 90 min in the absence of footshocks. Context A fear memory was assessed 24h later (day 2) during a 5min re-exposure to context A. The following day (day 3), all rats received a single tone presentation (80 dB, 20 s) co-terminating with a footshock (1 mA, 1 s) after 3 min in a novel context (context B) and were removed from the context 1 min after the tone/shock. For context B, a white curved wall and floor (beneath the grid) insert were used, along with 3 drops of lemon scent. The test of SEFL and fear generalization (the ‘SEFL-test’) was performed 24h later (day 4): rats were returned to context B and re-exposed to the tone 3 min into the session, then removed 1 min after presentation of the tone. The chambers were cleaned before and after each trial. For context A, the corncob bedding in the catch-tray was replaced and 70% ethanol was used as the cleansing/deodorant agent. For context B, 5% bleach was used as the cleansing/deodorant agent.
Freezing behavior during each session was quantified using VideoFreeze software (Med Associates, Inc, Fairfax, VT). The use of this software has undergone extensive validation to ensure good agreement freezing as calculated by the software and manual scoring by an unbiased, trained human observer.18,19 Percent freezing (%freezing) was calculated as:

Footshock exposure in females is associated with a shift to a more passive stress response. (
Positron Emission Tomography With [18F]FPEB
All rats underwent 2 scanning sessions with the mGlu5 receptor-specific radiotracer, [18F]FPEB. Scans with excessive motion (n = 2) or failed radiotracer injection (n = 2) were not included in the final analyses.The baseline scan took place 3 to 10 days prior to behavioral testing. The second scan was acquired 24h after the day 4 SEFL test (ie, ‘post-test’). Scans were performed as previously described.17,20 To summarize briefly, high molar activity [18F]FPEB (5.24 ± 2.70 MBq/nmol) was synthesized in-house as previously detailed. 21 Rats were transported to the Yale PET center at 08:00, with [18F]FPEB injections, scans, and data processing proceding as dedailed in the Supplemental material. The primary outcome measure was [18F]FPEB nondisplaceable binding potential (BPND) as estimated by the simplified reference tissue model with cerebellum as the reference region. 22 For this study, we used a regions of interest (ROIs) approach, which included amygdala (AMY), hippocampal formation (HIP, including dorsal and ventral aspects and subiculum), prefrontal cortex (PFC, including orbitofrontal and medial prefrontal regions), and striatum (STR) as ROIs. These ROIs were selected based on previous studies implicating them in fear learning and stress-related psychopathology that overlap with regions of mGlu5 receptor expression, both in preclinical and human work.19,23‐27
Statistical Analyses
We tested for the homogeneity of variance and normal distribution of data before proceeding with further analyses. Freezing behavior for each day or specified timeframe was assessed using 2 (sex: male vs female) by 2 (treatment group: CON vs FE) way analysis of variance. Comparison of freezing across time was performed with a mixed-effects model, with time/context (context A vs context B) within-subject measure and sex a between-group effect, followed by post hoc tests with Bonferroni's correction for multiple comparisons. An independent samples t-test was used to assess differences in the values of context B – context A %freezing between male and female FE rats. For PET data, multivariate analysis of variance (MANOVA) models were performed with sex and treatment group (CON vs FE) as fixed effects, and BPND values for the 4 ROIs (AMY, HIP, PFC, and STR) at baseline or post-test as the dependent variables. For comparisons between behavioral phenotype [SEFL(−) vs SEFL(+)], additional MANOVAs were performed with phenotype and sex entered as fixed factors, and the 4 ROI BPND values as dependent variables. We used nonparametric tests (Kruskal-Wallis) to further explore the interaction between phenotype and sex (see the Supplemental material). IBM SPSS Statistics, version 29.0.1.0 (IBM Corp., Armonk, NY) was used for all statistical analyses and Prism GraphPad, version 10.0.2 (GraphPad Software, Boston, Massachusetts) was used for data visualization. All statistical tests were 2-tailed, and results were considered statistically significant when p < .05.
Results
Footshock Exposure Enhances Generalization of Fear Learning
On day 1 of the SEFL paradigm, greater freezing was observed in the FE rats who received 15 footshocks over 90 min in context A relative to CON rats (F1,43 = 91.8, p < .001), with no significant main effect of sex or sex*treatment interaction (Figure 3A). Similarly, during the day 2 test of context A fear memory (Figure 3B), FE rats displayed greater freezing behavior as compared with CON (F1,43 = 28.9, p < .001), with the main effect of sex and sex*treatment interactions not achieving statistical significance (p = .086, p = .102, respectively). On day 3, all rats were introduced to context B and exposed to a single tone that co-terminated with a footshock. Freezing in context B during the 3 min prior to the tone/shock pairing (Figure 3C) and during the 20 s tone (Figure 3D) was similar for all rats, regardless of prior FE or sex. However, for the 1 min following the tone/footshock (Figure 3E), FE rats displayed significantly greater freezing behavior relative to CON (F1,43 = 21.5, p < .001) with no significant main effect of sex (p = .782) or sex*treatment interaction (p = .278).

Prior footshock exposure is associated with sensitization and generalization of fear learning in male and female rats. Freezing behavior of FE (n = 16 male, n = 16 female) and no-shock control (CON, n = 7 male, n = 8 female) rats during the (
On day 4—the SEFL test—FE rats displayed enhanced fear memory for context B, as evidenced by greater freezing in the 3 min prior to the tone as compared with CON (Figure 3F: F1,43 = 19.4, p < .001). Cue fear memory, that is, freezing during the 20 s tone (Figure 3G), was elevated in FE rats relative to CON, but this difference was not statistically significant (p = .054) due to high variability across all groups. Yet, FE rats tended to display freezing behavior for the 1 min after the tone terminated at a rate that was significantly greater than CON rats (Figure 3H: F1,43 = 22.5, p < .001).
Footshock Exposure in Female Rats is Associated With a Shift to a More Passive Stress Response
To assess the degree to which FE in context A enhanced fear learning and memory for context B, we conducted repeated measures analysis to compare freezing during the first 3 min of the day 2 context A fear memory test to freezing during the first 3 min of the day 4 SEFL test in male and female FE rats (Figure 2A). While there was neither an overall effect of the repeated measure, context (p = .159), nor a significant effect of sex (p = .127), there was a significant context*sex interaction (F = 8.11,30, p = .008). Post hoc tests revealed that FE males displayed similar freezing behavior in both contexts (padj = .331), whereas freezing behavior increased in context B relative to context A among the FE females (padj = .005). Then, the difference in contextual freezing between context B and context A was calculated for each FE rat and classified as SEFL(+) or SEFL(−) (see Methods section and Figure 2B). This context B – context A value was greater in female FE rats relative to FE male rats (t30 = 2.8, p = .008), however, there was not a significant difference in the proportion of female (n = 12, 75%) versus male (n = 8, 50%) rats classified as SEFL(+) (
Footshock Exposure is Not Associated With Differences in mGlu5 Receptor Availability
In vivo receptor availability, as estimated by [18F]FPEB nondisplaceable binding potential (BPND), in 4 regions of interest (AMY, HIP, PFC, and STR) was measured at baseline and post-test using [18F]FPEB and PET. At baseline, there were no differences in mGlu5 receptor availability between rats that would go on to be randomized to the CON group versus the FE group (Supplemental Figure S1A; F4,36 = 1.1, p = .362). There were also no differences between CON and FE rats at the post-test timepoint (Supplemental Figure S1B; F4,38 = 0.894, p = .477). Notably, there was a significant effect of sex at both baseline (F4,36 = 5.8, p < .001) and post-test (F4,38 = 6.5, p < .001), with female rats showing lower receptor availability. This difference was especially evident in AMY, where receptor availability in females was 17.2% lower at baseline (F1,39 = 4.8, p = .034) and 12.0% lower post-test (F1,41 = 5.9, p = .019) as compared with males. Sex-by-group interactions were not significant at baseline (F4,36 = 1.2, p = .321) or post-test (F4,38 = 0.9, p = .362), and no within-sex comparisons of CON versus FE, with or without correcting for multiple comparisons.
Differences in mGlu5 Receptor Availability Associated With the SEFL Phenotype
We additionally tested for differences in mGlu5 receptor availability among the FE rats, and more specifically, differences between rats classified as SEFL(+) versus SEFL(−) (Figure 4). A multivariate analysis of baseline regional BPND values (Figure 4A) revealed overall differences in mGlu5 receptor availability associated with rats that would go on to develop the SEFL(+) phenotype versus those that would not, regardless of sex (F4,24 = 3.4, p = .026). This included, on average, 10.6% lower in AMY and 6.9%, 6.4%, and 8.4% higher receptor availability in HIP, PFC, and STR, respectively, in SEFL(+) relative to SEFL(−) rats. However, these group differences were not statistically significant for any single ROI. The main effect of sex was significant at baseline (F4,24 = 3.8; p = .005), with significantly lower AMY BPND in female rats overall as compared with males (F1,27 = 6.6; p = .016). Supplemental Kruskal-Wallis tests to examine behavioral phenotype*sex interactions at baseline were all nonsignificant (all H3 < 5.68, all p's > .10).
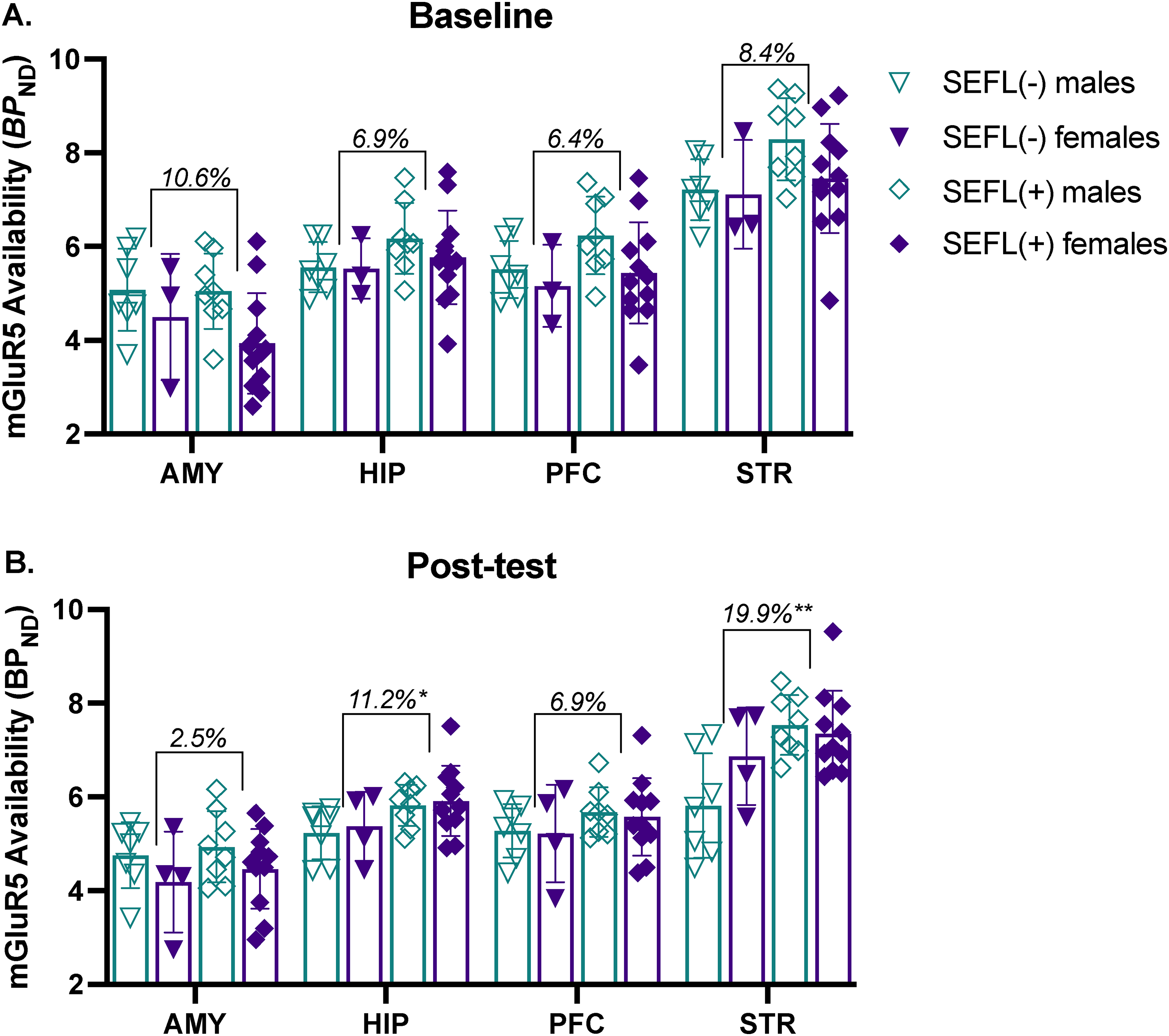
MANOVA, assessing regional difference in mGlu5 receptor availability as estimated by binding potential (BPND) using [18F]FPEB and PET. (
At the post-test timepoint (Figure 4B), BPND values were higher in the SEFL(+) group across all 4 ROIs (F4,24 = 3.6, p = .019), including 2.5% and 6.9% higher in AMY and PFC. Further, group differences were statistically significant in HIP (11.2%, F1,27 = 5.5, p = .027) and STR (19.9%, F1,27 = 10.2, p = .003). While the post-test main effect of sex was not significant (F4,24 = 2.6; p = .060), a significant Kruskal-Wallis test was observed for post-test STR (H3 = 8.7, p = .034), with pairwise comparisons indicating that SEFL(−) males had lower post-test mGlu5 receptor availability relative to both SEFL(+) males (punadjusted = .005, pBonferroni corrected = .027) and SEFL(+) females (punadjusted = .024, pBonferroni corrected = .144).
Discussion
We showed that mGlu5 receptor availability at baseline was predictive of rats who would go on to express the SEFL(+) behavioral phenotype (ie, SEFL susceptibility). Additionally, at the post-test timepoint (ie, after completing the SEFL procedure), mGlu5 receptor availability was higher in SEFL(+) rats relative to SEFL(−) rats for all 4 brain regions of interest. Together these data argue that mGlu5 receptor signaling may be a key biobehavioral marker and risk factor for the pathoetiology of PTSD-like behavior in rodent models of PTSD, and suggest novel sex-specific factors, as detailed below.
Critically, the with regard to the behavioral responses with the SEFL paradigm in male and female rats, we found footshock stress-enhanced future responding to a single footshock presented in a novel context. This was evidenced by greater freezing among FE rats following the single tone-shock pairing in context B on day 3 as compared with CON rats. We previously observed this behavioral sensitization and enhanced fear learning in FE rats, 17 as have others using the SEFL paradigm.14,28‐30 Additionally, on the day 4 SEFL test, FE rats displayed greater freezing behavior in the 3 min before the tone onset and during the 1 min after termination of the tone. This pattern of behavior is consistent with enhanced fear learning and fear generalization typically observed with the SEFL paradigm and argued to model PTSD-like behavior in rodents.14,28‐30 Discreate fear memory for the cue, as measured by freezing during the tone on the day 4 SEFL test, was greater in the FE rats relative to CON rats, a difference approaching statistical significance (p = .054). It is noteworthy that for the above analyses of freezing behavior, we observed no significant main effects of sex or interactions between sex and treatment group. However, when looking at changes in contextual freezing for individual FE rats (ie, the measure used to determine SEFL susceptibility), we found FE female rats tended to display increased freezing in context B relative to context A; this is in contrast to FE male rats, among whom freezing tended to not change between the 2 tests of contextual fear memory. A potential interpretation of this phenomenon is that exposure to traumatic footshock stress induced a shift from the more “active” stress coping strategies (eg, darting or climbing) typically observed in female rodents in response to classical Pavlovian fear conditioning, to freezing, a more “passive” coping strategy more commonly observed in males.31‐33 Previously, studies relying solely on freezing as a measure of susceptibility to acute stress and fear learning classified female rats as more “resilient,” although there is now a greater appreciation that less freezing does not necessarily indicate reduced sensitivity or greater resilience in female animals, but more likely a reflection of differences in fear expression and selection of coping strategy between male and female rats.31‐33 To our knowledge, the findings presented here would be a unique demonstration of traumatic footshock stress inducing such a shift in behavioral stress coping in female rats. Further, the difference between male and female rats reported here as they relate to SEFL expression and SEFL susceptibility are consistent with marked sex-differences in PTSD prevalence and presentation observed in human clinical populations.2,6
We observed significant effect of sex, both at baseline and post-test, on mGlu5 availability. Notably, this difference was especially evident in AMY, where receptor availability in female rats was on average 15.5% lower than males, irrespective of treatment group. This finding is consistent with our previous study, 17 and it is possible that differences in mGlu5 receptor availability contributed to the sex difference in SEFL expression. Furthermore, our work is consistent with previous findings in an evaluation of a large sample of healthy human participants by Smart et al who found significantly higher mGlu5 availability in men (+17% overall) as compared with women. 34 To our knowledge, there is no published evidence of interactions between a PTSD diagnosis and sex as related to mGlu5 expression or its relationship with psychiatric symptoms. Given the present findings, it will be important to assess and characterize potential molecular mechanism of mGlu5 expression that we hypothesize may contribute to differences in PTSD between women and men.
Additionally, at baseline, and more critically, at post-test, there were no differences between the CON and FE groups, suggesting FE itself did not result in changes in mGlu5 availability within the timeframe examined in this study. However, when limiting the analysis to FE rats, baseline measures of mGlu5 availability were predictive of SEFL susceptibility. Specifically, regardless of sex, lower receptor availability in AMY and higher receptor availability in HIP, PFC, and STR was observed in rats that would go on to be SEFL(+) relative to those that would be SEFL(−). These findings add to the growing preclinical evidence implicating differences in mGlu5 receptor expression and activity contributing to susceptibility or resilience in rodent models of stress and fear learning,8,17,35‐37 and likewise are consistent with clinical neuroimaging studies from our lab12,13 where corticolimbic mGlu5 receptor availability was higher in individuals with PTSD relative to individuals with major depressive disorder and healthy controls. Collectively, these data support the hypothesis that higher mGlu5 availability could indeed represent a molecular “biomarker” for PTSD vulnerability and novel treatment target.
The primary limitation of the present study is the modest sample size, which restricted statistical power to examine mGlu5 availability at individual ROIs. Nevertheless, the effect sizes reported here can help inform the design and sample size requirements for future studies. Further, given the small number of female rats who ended up expressing the SEFL(−) phenotype, we were not able to fully investigate phenotype*sex interactions. There is mounting evidence implicating a modulatory role of sex hormone signaling in response to stress, 38 PTSD and mood symptoms,39‐41 pain processing, 42 and interactions between estrogen and mGlu5 receptor signaling. 43 Therefore, future investigations into whether interactions between estrogen and mGlu5 could influence resilience and vulnerability in the SEFL paradigm would be of merit. Second, given mGlu5's involvement in nociception and chronic pain,44‐46 it is possible that the painful nature of footshocks is a necessary mediator for a relationship between higher mGlu5 availability and SEFL susceptibility, and that is related to differences in pain processing. This potential confound would fit in with clinical observations suggesting overlapping molecular mechanisms mediating chronic pain syndromes and stress-related psychiatric disorders, including PTSD.47‐49 Third, we do not have postmortem analyses to correlate with our in vivo findings, as the cohort of rats included in this analysis were used in subsequent behavioral studies beyond the scope of this report. Fourth, to be consistent with previous studies,14,17‐19 all rats were single-housed for the duration of the study. However, single housing can be considered a mild stressor, and a systematic evaluation of single versus pair-housing during the SEFL procedure was found to not influence behavior of either control or FE animals. 50 Therefore, group housing is an option that could be considered in future work and could additionally be used to examine possible sex-differences. Despite these caveats the strengths of this study include the prospective design, which allowed us to make statements on the predictive nature of mGlu5 availability and the directionality of relationships with PTSD susceptibility; the use of in vivo PET measurements which mirror the methodology used in human populations, making the findings here more immediately translatable to clinical findings; inclusion of both male and female rats and consideration of the main and interaction effects of sex for all analyses.
In the future, we hope to further this line of investigation. Specifically, we would like to examine potential relationship and shared mechanisms (including mGlu5 receptor) between the SEFL phenotype and other behavioral phenotypes associated with PTSD, such as deficits in fear extinction and retention of extinction learning. 51 Additionally, taking a pharmacological approach, we would like use mGlu5 receptor modulators as a method for testing the necessity and sufficiency of mGlu5 signaling in development of the SEFL and how this may differ between male and female rats. In these studies, it will be important to consider that mGlu5 receptor engagement facilitates fear extinction and extinction retention,52,53 as well as enhancing performance in tests of hippocampal-dependent learning. 54 Therefore, if mGlu5 modulation is pursued as a strategy for reducing risk or enhancing resilience following trauma exposure, it will be critical to ensure desirable forms of mGlu5-mediated plasticity, learning, and memory are not compromised.
Conclusion
In summary, here we combine longitudinal PET imaging with the SEFL rodent model of PTSD. Our results provide novel evidence regarding sex-differences in behavioral susceptibility, as well as insight regarding the potential utility of mGlu5 receptor availability as a biomarker of elevated risk for developing a PTSD-like phenotype following trauma exposure. Our data also highlight the potential contribution of mGlu5 availability in individual and sex-differences responses to stress and trauma. We hope further research efforts aimed at understanding mechanisms by which mGlu5 may contribute to individual and sex-differences will ultimately translate into more personalized treatment approaches and the development of novel preventative or therapeutic strategies that can only be born out of a greater mechanistic understanding of the pathophysiological underpinnings of PTSD and related trauma-related and stressor-related disorders.
Supplemental Material
sj-docx-1-css-10.1177_24705470231215001 - Supplemental material for Examining mGlu5 Receptor Availability as a Predictor of Vulnerability to PTSD: An [18F]FPEB and PET Study in Male and Female Rats
Supplemental material, sj-docx-1-css-10.1177_24705470231215001 for Examining mGlu5 Receptor Availability as a Predictor of Vulnerability to PTSD: An [18F]FPEB and PET Study in Male and Female Rats by Ruth H. Asch, Krista Fowles, Robert H. Pietrzak, Jane R. Taylor and Irina Esterlis in Chronic Stress
Footnotes
Data availability statement
The datasets generated during the current study and presented here are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by the United States Department of Veterans Affairs National Center for PTSD. Additional support was provided by the Biological Sciences Training Program (BSTP) in Psychiatry (T32 MH014276) and the State of Connecticut, Department of Mental Health and Addiction Services. This publication does not express the views of the United States Department of Veterans Affairs, Department of Mental Health and Addiction Services, or the State of Connecticut.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

