Abstract
Background
Fibromyalgia is a chronic pain syndrome which occurs in the absence of an organic damage, whom causes is still unclear. Aims of this pilot study were to investigate the neural correlates of fibromyalgia in response to pain-related visual stimuli and explore the psychological differences among fibromyalgia, chronic low back pain (CLBP) and healthy conditions.
Methods
After a clinical assessment, electrophysiological responses to pain-related visual stimuli were recorded using a 256-Hydrocel Geodesic-Sensor-Net. Event-related potentials (ERPs), standardised low-resolution electromagnetic tomography (sLORETA), and psychological (Symptom Checklist-90-Revised) data were analysed for a total sample of 23 women (5 healthy volunteers, 12 fibromyalgia patients, 6 CLBP patients).
Results
The main finding was that fibromyalgia women reported a different brain response to pain-related visual stimuli on the frontal montage compared to women with CLBP (p = .028). Moreover, fibromyalgia women showed an increased activity mainly on the hippocampus (p = .003) and the posterior cingulate cortex (p ≤ .001) in response to algic stimuli compared to not algic ones. Lastly, these women presented higher scores on the somatization (p = .002), obsession-compulsion (p = .045), depression (p = .043) and positive symptom distress (p = .023) dimensions compared to the healthy women.
Conclusions
These preliminary results suggest that although the painful symptoms are similar, the central elaboration of pain could be different between women with fibromyalgia and those with CLBP. Moreover, these findings provide preliminary evidences about the great alert and the central sensitivity to pain-related information regarding fibromyalgia patients.
Introduction
Fibromyalgia (FM) is a syndrome characterized by widespread chronic pain, which occurs without a clear organic cause.1–3 As reported by previous studies, these patients presented a reduced pain threshold, higher sensitivity to sensory stimuli and hypervigilance towards pain.4–9 However, it has been pointed out that an interweaving of genetic, biological, and psychological factors plays a role in the development and maintenance of this condition.10,11
From a neurobiological perspective, FM seems to be associated with an increased activation of afferent pathways in response to pain-related stimuli.12–14 In particular, these patients showed an enhanced activity on the somatosensory primary and secondary cortices, insula, and association brain areas, which are structures involved in processing the sensory characteristics of painful stimulations. 15 A recent event-related potential (ERP) study reported that pain facial expressions elicited a greater N100 amplitude on the frontal montage in FM patients but not in the control groups. 16 Moreover, different amplitudes were found on the occipital montage at P100, and on the central-parietal montage on late positive potential components. 13 Although recent literature seems to agree about the different activity of central structures involved in pain processing, the aetiology of the disorder and its clinical specificity in relation to others chronic pain conditions are still unclear. 17 Interestingly, psychological and affective disorders seem to be involved in the pathology of FM.18–21 Recent studies highlighted the differences between the FM participants and the healthy volunteers in terms of psychopathology and personality.19–23 Moreover, women with FM seem to have fewer psychological resources, showing an enhanced vulnerability to the negative effects of stress.19,23,24 Currently, the treatment prescribed to these patients includes tricyclic anti-depressants, selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.25,26 The psychological characteristics of the FM require further studies in the light of the pharmacotherapy used and the presence of co-comorbidities with mental disorders.
Another common form of chronic pain condition is the chronic low back pain disorder (CLBP) resulting from injury or overuse, which is an important cause of disability.27–30 Also in this pain condition, according to several evidence, psychological factors significantly contribute to disability.31,32 To the best of our knowledge, there are no studies comparing the electrophysiological activity of patients with FM and CLBP in response to painful images.
The aim of this pilot investigation was to explore the electrophysiological responses to pain-related visual stimuli of FM patients and evaluate the psychopathological characteristics of this syndrome. The hypotheses were that the FM patients would present different amplitude and latency of the early and late ERP components on the occipito-temporal, parietal, and frontal montages, as well as a wider involvement of the cortical and subcortical areas in response to pain-related visual stimuli compared to both the CLBP patients and the healthy participants. Lastly, it was hypothesized that patients with FM would show higher levels of psychopathological symptoms compared to the CLBP and healthy participants.
Methods
Participants
The pilot study was carried out at the Department of Dynamic and Clinical Psychology, and Health Studies of Sapienza University. After local Ethical Committee approval, 30 volunteers (9 healthy women, 12 women with a FM diagnosis, and 9 women with a CLBP diagnosis) signed the informed consent, obtained for experimentation with human subjects in accordance with the 1964 Declaration of Helsinki. Only women between 25 to 65 years of age were included in the three groups and without pregnancy, pacemaker, other metal implants, systemic infectious diseases, epilepsy, neoplastic diseases, neurological injury, severe heart disease, previous spinal and lumbar hernias surgery, and other rheumatic diseases. The inclusion criteria for the FM and CLBP groups were: a FM and CLBP diagnoses (following the 1990 American College of Rheumatology criteria), a stable pharmacological treatment for at least 3 months, and a score higher than 3 on the Visual Analogue Scales (VAS).3,33 The exclusion criteria for the FM group was the presence of overlapping syndrome and for the CLBP group was the presence of radiating pain to the lower limbs.
Stimuli
The pain-related visual stimuli included 36 black and white images chosen from the Back school manual. 34 The manual aims at teaching to use correct movements and postures in everyday life in order to avoid painful perceptions. It also provides illustrations related to the perception of the body and the correct and incorrect movements control. 34 Eighteen images depicting a character doing an activity in a painful position were selected for the algic condition, while 18 images depicting a character in a painless position were collected for the not algic condition.
Clinical Assessment
The clinical assessment regarding psychological symptoms, pain, and disability were conducted using the following instruments. The Symptom Checklist-90-Revised (SCL-90-R), which is a self-report instrument designed to evaluate nine psychological problems and symptoms (somatization, obsession-compulsion, interpersonal sensitivity, depression, anxiety, hostility, phobic anxiety, paranoid ideation, psychoticism) and three global indexes (the global severity index, the positive symptom distress index, the positive symptom total). 35
The VAS, which is a widely used instrument to assess the unidimensional measure of pain intensity (0-10). 36
The McGill Pain Questionnaire (MPQ), a self-report instrument that allows patients to give a description about the quality and intensity of their painful experience through 78 adjectives. 37 The outcomes of the test consist in the following scores: somatosensory score; affective score; emotional score; mixed score; number of words chosen; present pain intensity; and a ratio between somatosensory and affective scores. 37
The Oswestry Disability Index (ODI) is a self-report questionnaire, which contains ten topics concerning daily life activities. 38 Each topic is followed by 6 statements describing potential scenarios. The patient checks the statement which most closely resembles their situation. 38
The Health Assessment Questionnaire-Disability Index (HAQ-DI) is an instrument, usually self-administered, which measures the functional disability in patients with a variety of rheumatic diseases. 39
The Fibromyalgia Assessment Status (FAS) is a self-administered index that combines the patient's assessment of fatigue, sleep disturbances, and pain evaluated on the basis of 16 non-articular sites listed. 40
The Fibromyalgia Impact Questionnaire (FIQ), a self-administered instrument which assesses the health status of patients with FM and the impact of the syndrome on their functions. 41
Experimental Procedure
After signing the informed consent, each participant provided information about age and body mass index (BMI). Moreover, the clinical assessments were conducted as follows: the SCL-90-R was administered to each of the three groups; the VAS, MPQ, and ODI were administered to both the FM and CLBP groups; the HAQ-DI, FAS, and FIQ were administered only to the FM group. Subsequently, the participants were seated at a viewing distance of 80 cm from a PC monitor (27 cm, 75-Hz, 1024 × 768), in order to perform the visual task. The pain-related visual stimuli were presented using E-Prime (v.2.0.8.90; Psychology Software Tools, Inc.; Pittsburgh, PA, USA). The visual task procedure was reported in Figure 1. The electroencephalographic (EEG) signal of each participant was recorded during the visual task. After the visual task, the participants were asked to evaluate each of the 36 pictures (18 algic and 18 not algic) indicating how much pain they would feel doing the activities showed by the character in the images, using a Likert scale (0-9).

Visual task procedure: the visual task started with the instructions for the participants, each trial started with a fixation cross displayed for 1000 ms, followed by the pain-related visual stimulus (algic or not algic condition) presented for 2000 ms, the trial ended with inter-stimulus interval (ISI) of 400 ms. A total of 108 trials [18 trials per condition (algic and not algic), repeated for three times] were presented in a random order for about 7 min. Note. Instructions: “Now we will show you some images. We invite you to pay attention to the pictures and to image yourself doing the activities carried out by the character. When you are ready to begin, please press the spacebar.”.
EEG Registration and Analysis
The EEG signal was recorded continuously at 250 Hz using Net Station4.4.2 and a 256-Hydrocel Geodesic-Sensor-Net, with an impedance kept below 50kΩ and the reference to the vertex (Cz). The acquired data was digitally filtered (30 Hz low-pass) offline. The EEG data of each participant were segmented in epochs of 100 ms before the presentation of the stimulus to 600 ms after stimulus onset. The artefacts detention was set at 200μV for bad channels (noisy electrodes), at 100μV for eye's blinks, and at 140μV for electrodes revealing eye movements (Electrical Geodesic, Inc., Eugene, OR, USA).42–44 The segments with an eye blink, an eye movement or more than 30 bad channels were excluded. Baseline correction −100 ms before the onset of the stimulus was applied. Through the visual inspection of the grand-averaged waveforms, the following intervals were set for the ERP components: 60 to 160 ms for the P100, 160 to 260 ms for the N200, 260 to 360 ms for the P300, 360 to 460 ms for the LC1, and 460 to 600 ms for the LC2. After the EEG signal cleaning from artefacts, the following electrode locations were chosen for each montage: occipito-temporal (left electrodes 106-107-114-116; right electrodes 150-159-160-169), parietal (left electrodes 75-76-84-87-97-99; right electrodes 140-141-152-153-161-172), and frontal (left electrodes 28-40-41-48-49; right electrodes 5-13-206-212-222) montages. The analyses on the mean amplitude and latency of the early components (P100;N200;P300) were conducted on each montage. The analysis on the mean amplitude of the late components (LC1;LC2) were conducted on the parietal and frontal montages.
Source Analysis (sLORETA)
The default standardized low-resolution electromagnetic tomography (sLORETA) 45 inverse model of the GeoSource software (version_2.0; EGI, Eugene, OR), with the Sun-Stok4-Shell Spere head model and Tikhonv 1 × 10 to 4 regularization was used to identify the locations of the neural generators of the ERP components. sLORETA is based on the assumption of the standardization of the current density which implies that not only the variance of noise in the EEG measurements but also the biological variance in the actual signal is taken into account. 45 This biological variance is taken as independent and uniformly distributed across the brain resulting in a linear imaging localization technique having an exact, zero-localization error. 46 Source locations were derived from the probabilistic map of the Montreal Neurological Institute 305 average. Based on the probabilistic map, gray matter volume was parcelled into 7-mm voxels. Each voxel served as a source location with three orthogonal orientation vectors, resulting in a total of 2447 source triplets whose anatomical labels were estimated through the use of a Talairach daemon.42,43,47–53
The Magnetic Resonance Imaging normalization and extraction of the data of each subject were performed and mean intensity of each Brodmann area (BA) was extracted for each ERP component. Referring to the recent literature on the neurobiological correlates of the pain perception in FM and CLBP syndromes, eight regions of interest (ROIs), corresponding to specific combinations of BAs, were defined.8,13,30,54–57 Specifically, the motor ROI included BA04-BA06, the occipital ROI included BA17-BA18-BA19; the somatosensory ROI included BA01-BA02-BA03-BA05-BA07, the limbic ROI included amygdala-insula-BA27-BA28-amygdala hippocampus junction-BA35-BA36-hippocampus; the temporal ROI included BA20-BA21-BA22-BA37-BA38-BA41-BA42-BA43; the anterior cingulate cortex ROI included BA24-BA32-BA33; the posterior cingulate cortex ROI included BA23-BA29-BA30-BA31; and the prefrontal cortex ROI included BA09-BA10-BA11-BA25-BA46-BA47. The mean intensity (nA) of each BA in response to pain-related visual stimuli was extracted and analysed for each ERP component. The nA of the occipital and temporal BAs were extracted and analysed only for early components (P100;N200;P300).
Statistical Analyses
For what concerns the psychological data, the analyses of variance (ANOVAs) with group (controlvs.FMvs.CLBP) as the between-subjects factor were performed on the SCL-90-R scores, and the planned comparisons among the three groups (controlvs.FMvs.CLBP) were carried out. For the behavioural data, the 3 × 2 repeated-measures ANOVAs with group (controlvs.FMvs.CLBP) as the between-subjects factor and condition (algicvs.not algic) as the within-subjects factors were conducted on the pain valence assignment of the visual stimuli. For the ERP data, the 3 × 2 × 2 repeated-measures ANOVAs with group (controlvs.FMvs.CLBP) as the between-subjects factor, condition (algicvs.not algic) and hemisphere (leftvs.right) as the within-subjects factors were conducted on amplitude and latency of each ERP component (P100;N200;P300;LC1;LC2) on the occipito-temporal, parietal, and frontal montages. Partial eta-squared was calculated.
For the sLORETA data analyses, the 3 × 2 × 2 repeated-measures ANOVAs with group (controlvs.FMvs.CLBP) as the between-subjects factor, condition (algicvs.not algic) and hemisphere (leftvs.right) as the within-subjects factors were performed on the mean intensity of each BA for each component (P100;N200;P300;LC1;LC2). Basic single comparisons on each BA intensity were carried out and Bonferroni corrections were applied for each ROI. The significant threshold for .05 p-value was set at (Bonferroni correction): .0125 for the motor ROI, where 4 comparisons (2BAs × 2hemispheres: .05/4 = .0125) were identified; .008 for the occipital and anterior cingulate ROIs, where 6 comparisons (3BAs × 2hemispheres: .05/6 = .008) were identified; .005 for the somatosensory cortex ROI, where 10 comparisons (5BAs × 2hemispheres: .05/10 = .005) were identified; .003 for the limbic and temporal ROIs, where 16 comparisons (8BAs × 2hemispheres: .05/16 = .003) were identified; .006 for the posterior cingulate ROI, where 8 comparisons (4BAs × 2hemisphere: .05/8 = .006) were identified; and .004 for the prefrontal ROI, where 12 comparisons (6BAs × 2hemispheres: .05/12 = .004) were identified.
All statistical analyses were performed using Statistica8 (StatSoft, Inc. 2007).
Results
Seven participants from the initial thirty were excluded due to artefacts in the EEG data. The final sample was composed by twenty-three participants: 5 women in the control group, 12 women with FM diagnosis, and 6 women with CLBP diagnosis. The descriptive statistics of the three groups were reported in Table 1.
Descriptive statistics of the clinical cssessment [age, body mass index (BMI), visual analogue scale (VAS), mcgill pain questionnaire (MPQ), oswestry disability index (ODI), health assessment questionnaire-disability index (HAQ-DI), fibromyalgia assessment status (FAS), and fibromyalgia impact questionnaire (FIQ)] of the control, fibromyalgia, and chronic low back pain groups (n = 23).
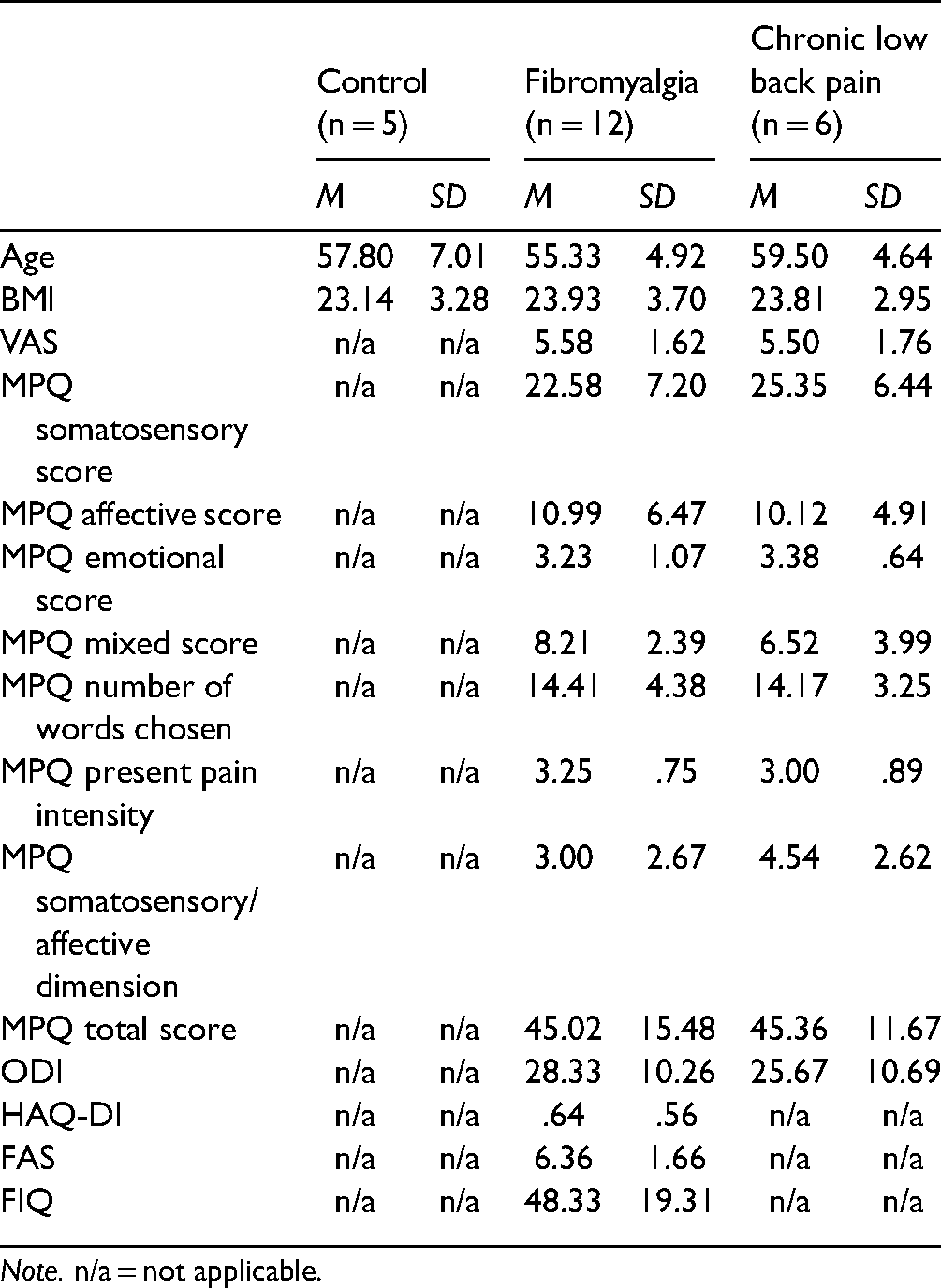
Note. n/a = not applicable.
Psychological Data
The planned comparisons among the three groups (controlvs.FMvs.CLBP) on the SCL-90-R scores showed a significantly lower score of the control group compared to the FM one for what concerns somatization, obsession-compulsion, depression, and positive symptom distress index (Table 2).
Planned comparisons of the symptom checklist-90-revised (SCL-90-R) among the three groups (Control vs Fibromyalgia vs Chronic low Back Pain) (n = 23).

Note. SOM = somatization scale; O-C = obsessive-compulsive scale; I-S = interpersonal sensitivity scale; DEP = depression scale; ANX = anxiety scale; HOS = hostility scale; PHOB = phobic anxiety; PAR = paranoid ideation; PSY = psychoticism; GSI = global severity index; PSDI = positive symptom distress index; PST = positive symptom total; n.s. = not significant.
Behavioural Data
The 3 × 2 repeated-measures ANOVAs group (controlvs.FMvs.CLBP) per condition (algicvs.not algic) performed on the pain valence assignment of the visual stimuli showed a significant main effect of the group [F(2,20) = 14.7;p < .001;ηp2 = .59], in which the control group rated the stimuli as less painful compared to the FM (p < .001) and CLBP (p = .002) groups. Moreover, a significant main effect of the condition was found [F(1,20) = 26.0;p < .001;ηp2 = .57], in which the algic stimuli versus the not algic stimuli were rated as more painful (p < .001).
ERPs Data
To analyse the ERPs data, the 3 × 2 × 2 repeated-measures ANOVAs group (controlvs.FMvs.CLBP) per condition (algicvs.not algic) per hemisphere (leftvs.right) were performed on the amplitude showing the following results (Table 3, Figure 2). A significant main effect of the group was found on the frontal montage at P300 in which the control group showed a greater amplitude compared to the other two groups. A significant main effect of the condition was found on the frontal montage at N200, LC1, and LC2, in which the algic stimuli versus the not algic stimuli evoked a greater amplitude. A significant main effect of the hemisphere was found on the parietal montage in all components in which the left hemisphere showed a lower amplitude compared to the right one. A significant interaction group × condition was found on the occipito-temporal and frontal montages, starting from the N200 component, in which in response to the algic stimuli the control group showed a greater amplitude compared to the other two groups. On the contrary, the control group reported a lower amplitude on the occipito-temporal montage at N200, and on the parietal montage at N200 and LC1 in response to the not algic stimuli compared to the FM and the CLBP groups. Moreover, the algic stimuli in the control group evoked a greater amplitude compared to the not algic stimuli on each montage in all components. A significant interaction group × hemisphere was found on the parietal montage at N200, in which in the CLBP group left hemisphere showed a lower amplitude compared to the right one, and on the frontal montage at P300 where the control group showed a greater amplitude mainly on the left hemisphere compared to the other two groups. Moreover, the left hemisphere in the FM group showed a lower amplitude compared to the right one.

ERP grand averages of the left and right occipito-temporal, parietal, and frontal montages of the three groups (control vs fibromyalgia vs chronic low back pain) in response to pain-related visual stimuli (algic stimuli vs not algic stimuli).
The ANOVAs group [control (C) versus fibromyalgia (F) versus chronic low back pain (L)] per condition [Algic stimuli (Algi) versus not algic stimuli (NoAlgi)] per hemisphere [Left (L) versus right (R)] on the amplitude and latency of each component (P100, N200, P300, LC1, and LC2) on the occipito-temporal, parietal, and frontal montages (n = 23).

Note. n.s. = not significant.
The ANOVAs on latency showed the following results (Table 3, Figure 2). A significant main effect of the group was found on the frontal montage at N200 where the control group showed a longer latency compared to the other two groups. A significant main effect of the condition was found on the occipito-temporal montage at P300 where the algic condition elicited a shorter latency compared to the not algic ones. A significant main effect of the hemisphere was found on the parietal montage at P300 where the left hemisphere showed a shorter latency compared to the right one. A significant interaction group × hemisphere was found on the frontal montage at N200 where the control group showed a longer latency compared to the other two groups mainly in the left hemisphere. In the CLBP group, the left hemisphere showed a shorter latency compared to the right one. A significant interaction condition × hemisphere was found on the frontal montage at P300 where the algic stimuli elicited a longer latency in the right hemisphere compared to the not algic ones, and where the algic stimuli evoked a longer latency in the right hemisphere compared to the left one. A significant interaction group × condition × hemisphere was found on the parietal and frontal montages at P300. In the control group the algic stimuli elicited a longer latency compared to the not algic stimuli in both hemispheres, while in the CLBP group this trend was inverted. Moreover, on the frontal montage, the CLBP group showed a shorter latency in the left hemisphere in response to algic stimuli compared to the other two groups. The not significant effects were reported the Supplemental material (Table 1S).
sLORETA Data
The sLORETA analyses did not show significant differences among the three groups neither in response to the algic stimuli nor in response to the not algic ones as reported in the Supplemental material (Table 2S-7S).
Algic Stimuli versus Not Algic Stimuli
In the control group (Table 4), the algic stimuli elicited a lower intensity compared to the not algic ones on the occipital ROI (BA17;left-BA18) at N200, on the somatosensory cortex ROI (right-BA05) at P100, and on the posterior cingulate cortex ROI (BA23;left-BA30;BA31) between N200 and LC1. The response of the control group to the algic stimuli compared to the not algic ones passed from lower to higher on the limbic ROI (right-amygdala hippocampus junction) between P300 and LC1.
Significant basic single comparisons on mean intensity of each brodmann area (BA) for each region of interest (ROI), including motor ROI (BA04 and BA06), occipital ROI (BA17, BA18, and BA19), somatosensory ROI (BA01, BA02, BA03, BA05, BA07), limbic ROI [amygdala (AMG), insula, BA27, BA28, amygdala hippocampus junction (AHj), BA35, BA36, and hippocampus (HPC)], temporal ROI (BA20, BA21, BA22, BA37, BA38, BA41, BA42, and BA43), anterior cingulate cortex ROI (BA24, BA32, and BA33), posterior cingulate cortex ROI (BA23, BA29, BA30, and BA31), and prefrontal cortex ROI (BA09, BA10, BA11, BA25, BA46, and BA47), for each components (P100, N200, P300, LC1, and LC2) in both hemispheres left (l) and right (r) in response to the algic stimuli compared with the not algic ones in each group (Control, Fibromyalgia, and Chronic low Back Pain) (Bonferroni Correction was Applied with Accepted

Note. ↑ = BAs intensity in response to the algic stimuli was higher compared with the not algic stimuli; ↓ = BAs intensity in response to the not algic stimuli was higher compared with the algic stimuli; n.s. = not significant; n/a = not applicable.
In the FM group (Table 4), the algic stimuli elicited a greater intensity compared to the not algic stimuli on the occipital ROI (right-BA18;right-BA19) at N200, on the limbic ROI (right-BA28;right-amygdala hippocampus junction;right-hippocampus) at N200 and LC2, on the posterior cingulate cortex ROI (BA23;right-BA30;BA31) at N200, LC1, and LC2, and on the prefrontal ROI (left-BA25) at LC2. This trend was inverted on the motor ROI (left-BA06) at LC1 and LC2, on the somatosensory cortex ROI (left-BA02;BA05) at P300 and LC2, on the anterior cingulate cortex ROI (right-BA32;right-BA33) at P100 and LC1, and on the posterior cingulate cortex ROI (left-BA23;left-BA29;left-BA30) at P300.
In the CLBP group (Table 4), the algic stimuli elicited a lower intensity compared to the not algic stimuli on the anterior cingulate cortex ROI (BA24;left-BA32;BA33) at P300 and LC2, on the posterior cingulate cortex ROI (BA23;right-BA30;right-BA31) starting from the P100 component, and on the prefrontal cortex ROI (left-BA10;left-BA11;BA25) at N200, P300, and LC2. The not significant basic single comparisons have been reported in the Supplemental material (Table 8S).
Discussion
The main finding of this pilot study was that FM patients showed a different brain response to pain-related visual stimuli compared to CLBP patients. In particular, the FM group showed a longer latency of the early component (260-360 ms) on the left frontal montage in response to the algic stimuli compared to the CLBP group. This preliminary finding does not seem to confirm the evidences of a previous study, which reported a similar brain response in the two clinical groups during tactile stimulations. 58 These contrasting results could be due to the different tasks performed in the two experiments. In fact, in the Giesecke and colleagues’ experiment, the pressure task allowed to obtain evidence regarding the processing of the sensory inputs of the peripheral tissue.29,58 On the other hand, the pain-related images used in the present study, allowed to highlight differences regarding complex affective processes, such as mentalization skills and identification with the observed pain.13,59,60 Despite the small number of participants, in the light of the differences found in the brain response between the FM and the CLBP groups, it is possible to speculate that even if the manifestation of the painful symptoms is quite similar, the psychological and affective processing related to the pain elaboration could be different between the two clinical groups. Moreover, these results seem to confirm the involvement of the frontal structures in the pathophysiology of the CLBP condition, and support the presence of a facilitated pain response, as previously reported.30,57
Another interesting result was that the FM and the CLBP groups showed a different brain response to pain-related visual stimuli compared to the healthy volunteers. In particular, starting from the N200 component, the two clinical groups reported a different amplitude on the occipito-temporal, parietal, and frontal montages in response to algic stimuli compared to the control group. This neurophysiological response to the painful images extends the recent clinical insights, which reported an altered pain processing mechanism in patients with FM and CLBP.4,56,59–61 Since there are no studies comparing directly the ERP data of the FM and the CLBP patients in response to pain-related visual stimuli, this preliminary finding seems to confirm the evidences of a previous research, which reported a different involvement in brain activity related to pain processing in the two clinical conditions compared to the healthy volunteers during a pressure-pain threshold task. 58 Moreover, it is interesting to note that both clinical groups reported a greater amplitude compared to the control one mainly on the parietal montage, in response to the not algic stimuli. According to the current literature, the parietal circuits should be involved in the detection of the visuo-spatial and motor information and in processing the sensory discriminative characteristics of a painful stimulation.15,62,63 The parietal pathways responsivity of the two clinical groups even to not algic stimuli seems to support the evidences of the generalized hyper-vigilance to visual stimuli with motor and somatic connotations, already found in FM patients. 13 These evidences were confirmed also by the behavioural finding of this study, in which the FM and CLBP groups rated the stimuli as more painful compared to control group.
From a psychological perspective, the results showed that patients with FM reported worse scores compared to the healthy volunteers on the SCL-90-R. In particular, FM patients seemed to present a psychological profile characterized by somatization, obsessive-compulsive attitude, depression, and the tendency of overestimate the intensity of the symptoms. These results seem to confirm the enhanced susceptibility to negative experiences19,23,59,60 and could support the evidences about the tendency of patients with FM to show pain catastrophizing symptoms.15,64,65
The source analyses did not show significant differences among the three groups; it is possible to suppose that this lack of effect could be due to the involvement of central networks more complex than a single BA in the development of brain sensitization mechanisms. 66 Nonetheless, the source analyses performed on each group showed significant differences on BA intensity between the algic and the not algic conditions. In particular, it was interesting that while the CLBP group and the control one presented a lower brain intensity in response to the algic stimuli compared to the not algic ones, the FM group reported an inverse intensity pattern, showing a more widespread brain activation in response to the algic stimuli compared to the not algic ones. The latter result seems to confirm the presence of an altered activation of several brain areas, and seems agree with previously findings about a different pain-related information perception of patients with FM.6,13,15,17,67 Interestingly, the FM group showed increased activity in response to algic stimuli in the limbic brain areas and in the posterior cingulate cortex. As previously reported, the enhanced activity of these neural circuits in response to painful images could be related to the greater sensitivity to the pain-related experience than what actually shown by these patients. 58
Furthermore, the FM group showed a greater intensity of the motor, the somatosensory and the anterior cingulate cortex brain areas in response to the not algic stimuli compared to the algic ones. A similar pattern was found in patients with generalized anxiety disorders, who showed a heightened brain response to both aversive and neutral images.68,69 These results seem to confirm that patients with FM show an increased sensitivity also to not painful images with somatic and motor connotations, supporting the presence of an altered affective evaluation of their pain experiences.13,15 Previous studies reported that the anterior cingulate cortex activity was associated with pain expectation mechanism and with paying attention to pain information, while the motor brain areas activity was associated to expressive responses to pain.15,70,71 Moreover, the involvement of these brain areas seems to be associated with coding the affective dimension and the empathic responses related to painful experiences. 72
The present study had some limitations. The low number of participants does not allow one to consider the results as representative of the two clinical groups. Nonetheless, the conservative criterion used during the statistical analyses (Bonferroni corrections with accepted p-value from .0031 to .0125) allows us to assume the present findings as a preliminary investigation into the study of neural correlates of FM and CLBP clinical conditions, providing indications to future research to extend and eventually confirm these results. Moreover, from a clinical perspective, the findings could provide useful preliminary information for the psychological assessment and treatment of FM and CLBP.
In conclusion, this study underlines brain activity differences among FM, CLBP patients, and healthy persons, supporting the presence of a greater sensitivity to pain-related stimuli in FM patients and a facilitated pain-related response in patients with CLBP.
Supplemental Material
sj-docx-1-css-10.1177_24705470211046881 - Supplemental material for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study
Supplemental material, sj-docx-1-css-10.1177_24705470211046881 for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study by Carlo Lai, Chiara Ciacchella, Gaia Romana Pellicano, Daniela Altavilla, Daniela Sambucini, Teresa Paolucci, Maria Laura Sorgi, Manuela Di Franco, Raoul Saggini and Paola Aceto in Chronic Stress
Supplemental Material
sj-docx-2-css-10.1177_24705470211046881 - Supplemental material for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study
Supplemental material, sj-docx-2-css-10.1177_24705470211046881 for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study by Carlo Lai, Chiara Ciacchella, Gaia Romana Pellicano, Daniela Altavilla, Daniela Sambucini, Teresa Paolucci, Maria Laura Sorgi, Manuela Di Franco, Raoul Saggini and Paola Aceto in Chronic Stress
Supplemental Material
sj-doc-3-css-10.1177_24705470211046881 - Supplemental material for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study
Supplemental material, sj-doc-3-css-10.1177_24705470211046881 for Different Electrophysiological Responses to Pain-Related Visual Stimuli Between Fibromyalgia and Chronic low Back Pain Women: A Pilot Case-Control Study by Carlo Lai, Chiara Ciacchella, Gaia Romana Pellicano, Daniela Altavilla, Daniela Sambucini, Teresa Paolucci, Maria Laura Sorgi, Manuela Di Franco, Raoul Saggini and Paola Aceto in Chronic Stress
Footnotes
Acknowledgments
The authors would like to thank all participants of the present study and Denis Mariano, a native English speaking teacher for the careful language revision of the manuscript.
Data Accessibility Statement
The data will be available with a reasonable request.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
The work has been approved by the Ethical Committee of the Department of Dynamic and Clinical Psychology, and Health Studies, of “Sapienza” University of Rome (approval number 24/2017) and followed ethical guidelines for experimentation with human subjects in accordance with the 1964 Declaration of Helsinki.
Informed Consent
The Informed consent was signed by each participant.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
