Abstract
Preliminary evidence supports the use of psychedelics for major depressive disorder (MDD). However, less attention has been given to the neural mechanisms behind their effects. We conducted a systematic review examining the neuroimaging correlates of antidepressant response following psychedelic interventions for MDD. Through MEDLINE, Embase, and APA PsycINFO, 187 records were identified and 42 articles were screened. Six published studies and one conference abstract were included. Five ongoing trials were included from ClinicalTrials.gov. Our search covered several psychedelics, though included studies were specific to psilocybin, ayahuasca, and lysergic acid diethylamide. Three psilocybin studies noted amygdala activity and functional connectivity (FC) alterations that correlated with treatment response. Two psilocybin studies reported that FC changes in the medial and ventromedial prefrontal cortices correlated with treatment response. Two trials from a single study reported global decreases in brain network modularity which correlated with antidepressant response. One ayahuasca study reported increased activity in the limbic regions following treatment. Preliminary evidence suggests that the default mode and limbic networks may be a target for future research on the neural mechanisms of psychedelics. More data is required to corroborate these initial findings as the evidence summarized in this review is based on four datasets.
Keywords
Introduction
Depressive disorders are among the leading causes of disability worldwide. 1 From 1990 to 2017, the number of global cases of depression reportedly increased by 49.86%. 2 With the global incidence of depression on the rise, the social, economic, and health-related burdens of this psychiatric disorder are increasingly evident at both the individual and societal levels.
Monoaminergic antidepressants have been used extensively as a first-line treatment for depressive disorders. 3 However, it is estimated that only 42–53% of depressed patients respond to antidepressant medications. 4 Psychotherapies, such as cognitive behavioural therapy and interpersonal psychotherapy, have also been used as a first-line treatment for depression, though as many as 40% of patients with Major Depressive Disorder (MDD) do not benefit from these therapies.5,6 Due to the high rates of non-response to first-line interventions, there is a growing need for novel, more effective treatment options.
In recent years, psychedelic compounds have gained interest in the field of psychiatry as viable pharmacological treatments for psychiatric disorders. 7 Such compounds include psilocybin, ayahuasca, lysergic acid diethylamide (LSD), 3,4-methylenedioxymethamphetamine (MDMA), and N,N-Dimethyltryptamine (DMT). Their antidepressant effects appear to be notably more rapid and long-lasting compared to traditional antidepressant medications. 8 Additionally, psychedelics appear to effectively alleviate depressive symptoms while also providing depressed patients with unique and adaptive psychological benefits, including increased cognitive flexibility, 9 mindfulness, 9 psychological flexibility, 10 acceptance, 11 connectedness, 11 and catharsis. 12 One clinical trial 13 implied that these unique effects may be exclusive to psychedelics, in that they are not commonly found with classical antidepressant medications. Moreover, psychedelic interventions have shown preliminary clinical efficacy in treating depressive disorders, including MDD and treatment-resistant depression (TRD). 14 For instance, psilocybin with psychological support has been shown to reduce anhedonia 15 and pessimism bias 16 in patients with TRD. Psilocybin has also been shown to rapidly alleviate symptoms of anxiety and depression in MDD patients17–19 and patients with life-threatening diseases, 20 as well as effectively treat secondary depression in cancer patients. 19 Moreover, single-dose ayahuasca administrations have shown a rapid reduction in depressive symptoms in patients with recurrent depression. 21
Recent evidence has pointed to disrupted functional connectivity (FC) within and between higher-order intrinsic connectivity networks (ICNs) in depression. 22 Neuroimaging research has suggested that MDD is characterized by functionally deviant coupling within the central executive network (CEN), default mode network (DMN), and salience network (SN).22–24 The CEN and SN have been connected to attentional processes, cognitive control, task-switching, and decision-making,25–27 all of which are often impaired in depression.28–30 The DMN has been associated with internalized thought and self-reflection. 31 Individuals with depression have been found to excessively engage in these cognitive processes.32,33 Hyperactivity of the DMN is linked to depressive symptomatology, including rumination 34 and disturbed self-referential processing. 35 In patients with TRD, studies have shown reduced FC between the CEN and DMN and between the anterior and posterior DMN. 36 Depressed patients have also exhibited decreased flexibility in the SN compared to healthy controls. 37
Depressed patients have also been found to have aberrant FC within the brain's limbic regions, including the amygdala, hippocampus, medial prefrontal cortex (mPFC), and anterior cingulate cortex (ACC). 38 Enhanced FC between the amygdala and prefrontal regions has been associated with rumination and worry in depressed patients. 39 Given that the limbic regions are central to emotional regulation, it is unsurprising that disturbances in emotional processing are a prominent feature of depressive symptomatology.40,41
In MDD and TRD patients, select ICNs and brain regions are implicated in mediating antidepressant response. For MDD, a meta-analysis 42 found that following antidepressant drug treatment, the amygdala, hippocampus, parahippocampal region, ventral ACC, orbitofrontal cortex, and insula showed decreased activation, while the dorsolateral, dorsomedial, and ventrolateral prefrontal cortices (PFC) showed increased activation. In TRD, cerebellar–cerebral FC in the PFC and DMN were significantly decreased. At the same time, increased FC was shown in the parahippocampal gyrus and visual recognition network (lingual gyrus, middle occipital gyrus, and fusiform) following antidepressant treatment. 43 Identifying neuroimaging correlates of antidepressant treatment response has allowed researchers to better understand the mechanisms behind the action of monoamine-based psychopharmacology – specifically, how antidepressant medications impact select brain regions and ICNs, leading to clinical improvement of MDD symptoms.
Previous research has highlighted efforts taken toward understanding the therapeutic effects of psychedelics by studying brain activity and FC. 44 However, the neural mechanisms behind the antidepressant effects and unique psychological benefits of psychedelics remain less understood compared to conventional antidepressant medications. This gap may be due to the lack of neuroimaging outcomes in MDD clinical trials exploring the efficacy and feasibility of psychedelic treatments, given the novelty of the field. Existing evidence concerning the neural underpinnings behind the effects of psychedelic treatments primarily emerges from studies conducted among healthy participants. Psychedelics appear to target the frontomedial, medial temporal, and occipital cortices, as well as exert an inhibitory effect on the DMN. 45 One model proposes that psychedelics dysregulate neuronal activity among various brain regions by agonistically acting on 5-HT2A serotonin receptors, which gives rise to transient breakdowns of ICNs. 46 The 5HT2A receptor is a main proteomic binding site of serotonergic psychedelics 47 (i.e., ayahuasca, DMT, LSD, psilocybin, etc.) and is abundantly expressed throughout the CEN, DMN, and SN. 48 Psychedelics may, therefore, bring about their therapeutic effects by targeting particular ICNs in which 5HT2A receptors are heavily populated. In line with this serotonin receptor model, a recent meta-analysis 49 on psychedelic interventions in healthy individuals found altered FC and activation patterns in regions with high densities of 5HT2A receptors – notably, the peristriate area 19 of the occipital cortex, dorsal anterior and posterior cingulate cortices (PCC), mPFC, dorsolateral PFC (DLPFC), supramarginal gyrus, temporal cortex, and right amygdala. 48 Several of these brain regions comprise the DMN.
Given the lack of research investigating the neural mechanisms of psychedelic treatment response in MDD, there is a growing need to examine neuroimaging studies in this domain. Specifically, neuroimaging research may demonstrate how the therapeutic effects of psychedelics are achieved via functional alterations within ICNs and implicated brain regions. Furthermore, the identification of these neural correlates may give rise to valuable prognostic tools that can be used to predict successful responses to psychedelic interventions among patients with depression. This knowledge could lead to an appropriate selection of MDD patients to undergo psychedelic treatments as opposed to conventional antidepressant treatments, which are often accompanied by adverse effects,50,51 delayed onset of action, 52 and lack of therapeutic response. 53
A previous systematic review 45 provided a comprehensive outline of clinical studies that studied neuroimaging correlates of psychedelic interventions in healthy individuals. However, to our knowledge, there is no systematic review that examines such correlates in patients with depression. We review both published and ongoing clinical trials to provide a more detailed and practical overview of the current research landscape, avoid positive results bias, and highlight the advancements and future directions in this field.
Methods
We conducted a comprehensive search of both published studies and registered clinical trials encompassing various neuroimaging modalities and psychedelic interventions in MDD patients. This article was prepared in accordance with the PRISMA guidelines for systematic reviews. 54 Refer to Supplementary 1 to view a detailed description of the Methods for registered clinical trials.
Search Strategy
Published studies
A neuroimaging-focused search of three databases (MEDLINE, Embase, and APA PsycINFO) was conducted through the OVID and PubMed search engines. The following search terms were included: psilocybin, ayahuasca, LSD, DMT, MDMA, mescaline, depress*, neuroimag*. To view the complete search strategy, refer to Supplementary 2. We searched grey literature using the European database OpenGrey (http://www.opengrey.eu/), which included an advanced search of the term psychedelic* in conjunction with the terms pertaining to various neuroimaging modalities (ie, positron emission tomography [PET], single-photon emission computed tomography [SPECT], functional magnetic resonance imaging [fMRI], magnetic resonance imaging [MRI], electroencephalography [EEG], magnetoencephalography [MEG]). The end date of the searches was April 18, 2022. Two authors (A.D. and S.K.) were responsible for independently conducting the primary screening and assessment of retrieved articles for inclusion.
Inclusion Criteria
The present systematic review included published studies, registered/ongoing clinical trials, and conference abstracts involving psychedelic interventions in patients with depression. Studies had to contain neuroimaging measures as primary or secondary outcomes. The enrolled participants had to have a valid depression diagnosis according to a recognized depression scale or the MDD diagnostic criteria as per the Diagnostic and Statistical Manual of Mental Disorders (i.e., DSM-IV or DSM-5) or the International Classification of Diseases (i.e., ICD-9 or ICD-10). Studies that enrolled patients with comorbid psychiatric disorders were excluded, with the exception of comorbid anxiety disorders due to the high rate of their co-occurrence with MDD. Moreover, non-primary articles (i.e., reviews, systematic reviews, meta-analyses, etc), non-interventional studies (ie, observational studies), and non-human trials were excluded from the review. Non-English articles were also excluded. Studies were only included if subjects were 18 years or older. Finally, no date limit was applied.
Screening Process
The PRISMA flow diagram outlines the screening process for both published and registered clinical studies (Figure 1). The screening was conducted independently by the two first authors (A.D and S.K). Results were compared to detect any potential discrepancies, which were resolved by a tertiary arbitrator (I.D).

PRISMA flow diagram of published studies and registered trials screened and evaluated for eligibility.
Variable Extraction
Published studies
The following variables of interest were extracted: the mean age, participant sex, as well as the intention-to-treat (ITT) and per-protocol (PP) sample sizes. The psychiatric diagnosis of the participants was recorded, as well as the inclusion criteria required to meet a valid depression diagnosis at screening (i.e., Hamilton Rating Scale for Depression [HAM-D], Montgomery-Åsberg Depression Rating Scale [MADRS], Beck Depression Inventory [BDI], etc) or the MDD diagnostic criteria as per the Diagnostic and Statistical Manual of Mental Disorders (ie, DSM-IV or DSM-5), including any reported minimal scale thresholds set up by researchers for inclusion. Furthermore, several study characteristics were extracted: study duration, drug administration route, dosage, number of treatments, comparison type (ie, placebo-controlled or open-label), allocation (ie, randomized or nonrandomized), masking procedure (ie, single-masked or double-blind), and interventional model (ie, single group assignment, parallel assignment, or crossover assignment).
Furthermore, information about the treatment protocol of the psychedelic intervention was extracted (i.e., the class of psychedelic, dosage, and duration of the treatment period). The neuroimaging modalities used in the studies (i.e., EEG, fMRI, MEG, MRI, MRS, PET, and SPECT) and analyzed metrics (i.e., the amplitude of low-frequency fluctuation [ALFF], blood-oxygen-level-dependent [BOLD] response, cerebral blood flow [CBF], FC, neurometabolic concentrations) were reported. Moreover, the neural correlates of assessor-rated clinical outcomes, subjective clinical outcomes (i.e., self-report measures), and adverse effects within each study were also extracted. Notably, the p-values and correlation coefficient (Pearson's r) values were recorded when presented to highlight the statistical significance of the given findings, as well as the direction and strength reported in the correlation analyses.
Quality of Assessment
All published studies included in this systematic review were assessed for quality of evidence using the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) checklist. 55 The corresponding table summarizing the included studies in terms of the GRADE criteria can be found in Supplementary 3. All but one trial included in this review was open-label, creating a high potential for performance bias. In addition, all included studies reported data consistent with their outcomes of interest, suggesting a low risk of selective reporting bias.
Results
Search Results
A total of 187 relevant articles were retrieved through the OVID databases and assessed for eligibility. Overall, 42 articles remained after applying automation tools to limit the search results to only English language articles, clinical trials, and studies containing adult participants. Of the 42 articles, 16 were excluded due to focusing on psychedelic interventions in healthy individuals, 14 due to being unrelated to psychedelic interventions or depression, 5 due to being non-primary articles (i.e., book chapters, reviews), and 1 due to having no accessible results. After assessing the full text of the 6 remaining articles for eligibility, 5 published studies56–60 and 1 conference abstract61,62 were included. An additional article by Daws et al 63 (2022) was retrieved from other sources (i.e., PubMed). The conference abstract included in this review by Wall et al61,62 (2021) has also been recently made available as a pre-print. 62 Detailed results of the published studies are provided in Table 1. It is important to note that 4 out of 7 published studies56–58,61,62 included in this review reported their neuroimaging and clinical findings from the same cohort of subjects. Therefore, the main characteristics of the studies listed in Table 1 were similar to each other. However, we have displayed these characteristics to resemble how they were reported in the included studies, to highlight pertinent neuroimaging aspects in this review. No relevant clinical studies were obtained from the OpenGrey database. Additionally, detailed results of ongoing clinical trials are provided in Supplementary 1.
Summary of Published Psychedelic Interventional Studies.

Psychedelic Type
Five published studies56–58,60,63 and one conference abstract61,62 used psilocybin as their primary intervention. Of these studies, Daws et al 63 (2022) conducted both an open-label trial and a randomized controlled trial (RCT); these two trials will be referred to separately throughout the review. One published study 59 examined ayahuasca (Figure 2).
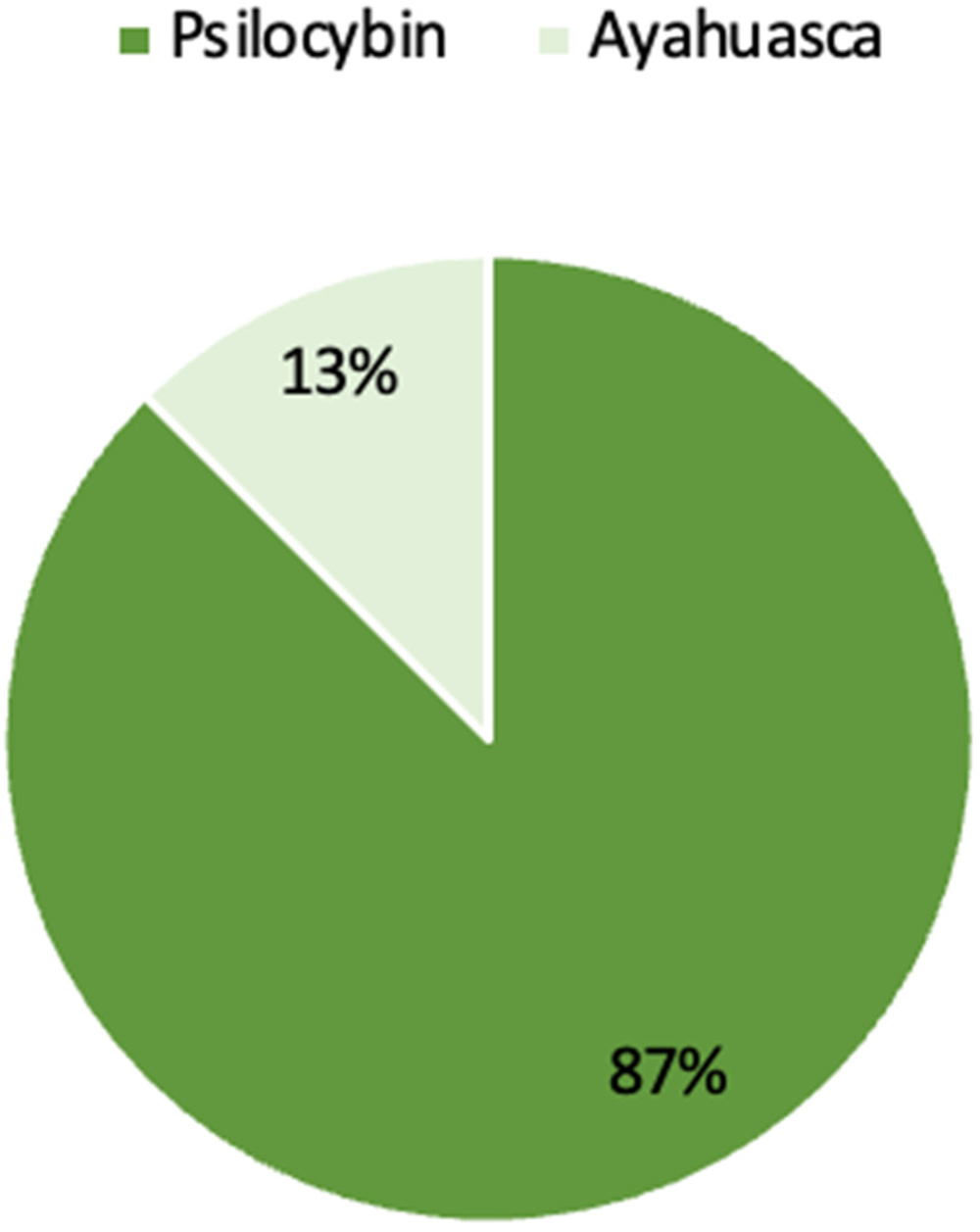
Type of psychedelic intervention examined in the published studies and conference abstract.
Research Design
Four studies56–59 followed an open-label, non-randomized, single-group interventional model. One clinical trial from Daws et al 63 (2020) also used an open-label, non-randomized, single-arm design, whereas the second clinical trial from Daws et al 63 (2020) was a double-blind RCT. Doss and colleagues 60 (2021) used an open-label interventional design that was randomized and not placebo-controlled. Roseman et al 58 (2018) was categorized as a feasibility trial but did not include a placebo group. Lastly, the conference abstract61,62 followed an interventional, open-label and single-group assignment design.
Sample Size and Clinical Diagnosis
All six published studies56–60,63 had ITT and PP sample sizes of less than 30 participants. Wall et al61,62 (2021) did not report their ITT sample size but had a PP sample size of fewer than 30 participants. Carhart-Harris et al 57 (2017) had PP sample sizes of 16 participants for their arterial spin labeling (ASL) analysis and 15 for their resting-state FC (RSFC) analysis. Doss et al 60 (2021) had PP sample sizes of 20 for resting-state analysis and ACC scans, 19 for left hippocampus scans, and 18 for right hippocampus scans. Daws et al 63 (2022) had PP sample sizes of 16 for their open-label trial and 22 for the RCT. Three studies56–58 and one conference abstract61,62 included patients diagnosed with TRD in the trials, while two studies59,60 included patients with MDD. Additionally, Daws et al 63 (2022) recruited TRD patients for their open-trial study but MDD patients for their RCT (Figure 3).

Published studies by the per-protocol sample size for each study. Note: Carhart–Harris et al (2017) has two different sample sizes for their ASL and RSFC sample sizes. Doss et al (2021) has four different sample sizes for their RSFC, ACC, left hippocampus and right hippocampus analyses. Daws et al (2022) has two different sample sizes for their psilocybin open-label trial and double-blind RCT.
Treatment Parameters
Four studies56–58,63 and one conference abstract61,62 investigating psilocybin used the same dosage and number of treatments. These studies each had a first session consisting of a 10 mg per os (p.o.) test dose followed a week later by a 25 mg p.o. therapeutic dose. Doss et al 60 (2021) used a moderately high dose of 20 mg/70 kg followed by a second high dose of 30 mg/70 kg administered 1.6 weeks apart. The RCT in Daws et al 63 (2022) used 25 mg psilocybin sessions on two separate dosing days with 3 weeks of daily placebo capsules following each dosing day. Mertens et al 56 (2020), Roseman et al 58 (2018), Doss et al 60 (2021) and Daws et al 63 (2022) specified that psilocybin was administered via a capsule. Carhart-Harris et al 57 (2017) and Wall et al61,62 (2021) did not specify the route of administration. Each subject in Sanches et al 59 (2016) drank a single dose of 120 to 200 mL of ayahuasca (See Table 2).
Treatment Dose and Duration of Treatment Period for Published Studies.

Abbreviations: LSD = lysergic acid diethylamide, RCT = Randomized Control Trial.
Clinical, Safety, and Tolerability Outcomes
All six published studies56–60,63 analyzed changes in depression scores using either the 21-item HAM-D, BDI, GRID-HAMD or MADRS. Only one study 59 reported vomiting as an adverse effect of ayahuasca, present in 47% of their patients. the RCT by Daws et al 63 (2022) did not observe adverse effects following psilocybin treatment. The remaining studies did not report or mention collecting the data on adverse effects (See Table 2).
Neuroimaging Modality & Psychedelic Type
Six studies56–58,60 and one conference abstract61,62 collected neuroimaging measurements at baseline and post-treatment. Roseman et al 58 (2018) and Mertens et al 56 (2020) collected functional neuroimaging data the morning after psilocybin administration, while Carhart-Harris et al 57 (2017) and the open-label trial by Daws et al 63 (2022) collected functional neuroimaging data 1 day after treatment. Roseman et al 58 (2018) and Mertens et al 56 (2020) collected fMRI data during an emotional face paradigm pre- and post-psilocybin. Carhart-Harris et al 57 (2017) collected fMRI data at resting-state pre- and post-psilocybin. These researchers also used perfusion-weighted imaging (PWI) to measure changes in CBF at baseline and 1 day after a psilocybin treatment. Doss et al 60 (2021) collected fMRI and magnetic resonance spectroscopy (MRS) data pre-and post-psilocybin. The RCT by Daws et al 63 (2022) collected data at baseline and at 1 day and 3 weeks post-treatment. During the psilocybin therapy session, patients were sitting in a comfortable environment and listening to music. Additionally, follow-up sessions took place after the psilocybin therapy session to discuss participants’ experiences. Wall et al61,62 (2021) collected fMRI data 1 week prior to the first psilocybin session and 1 day after the second psilocybin session. fMRI scans were acquired at rest and also while the participants were listening to music. Sanches et al 59 (2016) collected SPECT data before ayahuasca intake and 8 h following treatment (See Table 3).
Neuroimaging Technique by Psychedelic Intervention in Published Studies.
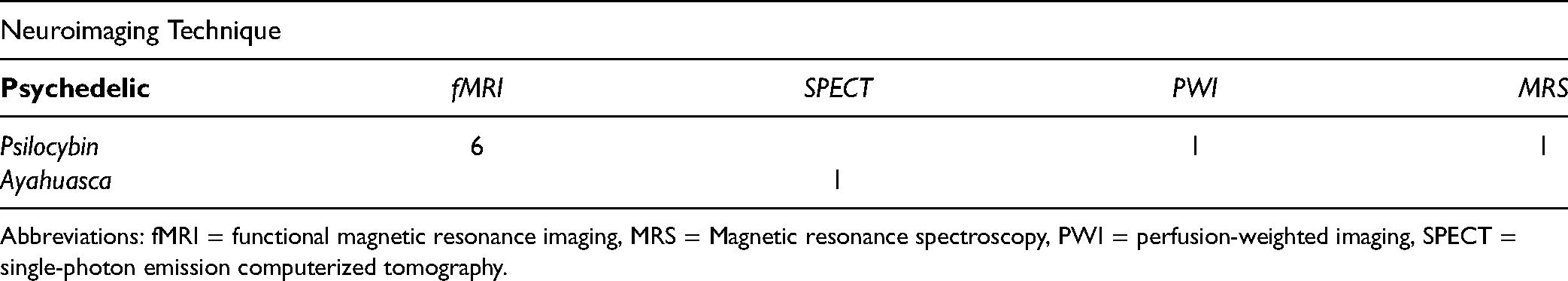
Abbreviations: fMRI = functional magnetic resonance imaging, MRS = Magnetic resonance spectroscopy, PWI = perfusion-weighted imaging, SPECT = single-photon emission computerized tomography.
Neuroimaging Outcomes
fMRI BOLD response/activity
Roseman et al 58 (2018) was the only published study that used an emotional faces task with BOLD fMRI to measure changes in neural activity following psilocybin administration. Following the second dose, increased activity was found in the right amygdala, temporal lobe and visual areas. The conference abstract by Wall et al61,62 (2021) used ALFF to measure changes in brain activity following psilocybin. Following psilocybin intake, greater ALFF was observed in the bilateral superior temporal cortex during music listening and in a small cluster of the right ventral occipital lobe during the resting state. Additionally, increased responsiveness was located in the superior and bilateral superior temporal lobes and supramarginal gyrus during the music listening scans.
fMRI functional connectivity
Mertens et al 56 (2020) used a task-based emotional faces paradigm while Carhart-Harris et al 57 (2017) and Daws et al 63 (2022) measured FC at resting state. Mertens et al 56 (2020) found decreases in ventromedial PFC (vmPFC) to right amygdala FC during face processing post-treatment. They also found increases in FC between the amygdala and vmPFC to occipital-parietal cortices during face processing. In addition, increases in FC were found between the amygdala and visual areas, as well as between the vmPFC and right angular gyrus, right lateral occipital cortex, and other visual areas in the right hemisphere following psilocybin treatment. Carhart-Harris et al 57 (2017) reported decreases in FC between the DMN and right frontoparietal network as well as increased DMN within-network FC post-treatment. Carhart-Harris et al 57 (2017) also found increases in FC within the anterior and posterior cingulate cortices and decreases in FC between the medial/lateral PFC and parahippocampus following treatment. Finally, they reported increases in FC between the vmPFC and bilateral inferior-lateral parietal cortex (ilPC) post-treatment. Doss et al 60 (2021) found no significant changes in ACC-PCC static FC. The researchers did find increased dynamic functional connectivity (dFC) between the ACC and PCC 1 week after psilocybin therapy. However, this was no longer present at 4 weeks post-psilocybin administration. Daws et al's 63 (2022) open-label trial found increased between-network FC between the DMN and CEN and the DMN and SN as well as reductions in within-DMN FC one day after psilocybin treatment.
fMRI brain modularity
Daws et al 63 (2022) used brain network modularity as a metric following psilocybin treatment. In both their open-label trial and RCT, they found brain network modularity was significantly reduced at 1 day and 3 weeks post-psilocybin therapy.
PWI cerebral blood flow
Carhart-Harris et al 57 (2017) used whole-brain analysis to measure CBF and found decreases in the resting-state CBF within the amygdala and temporal lobe following a second psilocybin dose.
SPECT
Sanches et al 59 (2016) used SPECT imaging to measure brain functioning following ayahuasca administration. Researchers reported increases in activation within the left nucleus accumbens (NAcc), right insula, and left subgenual area.
MRS
Doss et al 60 (2021) used MRS to investigate biochemical changes following psilocybin administration. The researchers found that glutamate and N-acetylaspartate (NAA) were decreased in the ACC 1-week post-psilocybin (See Table 4).
Main Imaging Findings from Published Studies.

Neuroimaging Correlates of Treatment Response
In three psilocybin studies,56–58 changes in amygdala activity and FC were correlated with treatment response. Mertens et al 56 (2020) found that reduced right amygdala to vmPFC FC was significantly associated with reduced depressive rumination scores at 1-week post-treatment. This study 56 also reported increased FC between the amygdala and visual areas under psilocybin, though these changes were not significantly associated with depressive outcomes. Carhart-Harris et al 57 (2017) reported that decreased CBF in the amygdala was predictive of lower depressive scores. Moreover, Roseman et al 58 (2018) found that increased right amygdala activity in response to fearful versus neutral faces was strongly predictive of favorable clinical outcomes.
Changes in the ventromedial and medial PFC FC were significantly correlated with depressive outcomes in two studies. Mertens et al 56 (2020) found that 1 week following psilocybin treatment, decreased right amygdala to vmPFC FC was associated with depressive rumination scores. Additionally, they found vmPFC-occipital lobe-parietal lobe FC was negatively affected which was correlated with BDI scores. 56 Carhart-Harris et al 57 (2017) found that increased vmPFC to bilateral ilPC FC and decreased parahippocampal to PFC FC were predictive of psilocybin treatment response at 5 weeks post-treatment. In the latter study, reduced FC between the parahippocampus and the medial and lateral portions of the PFC also significantly predicted treatment response at 5 weeks post-psilocybin. 57
Decreased brain modularity was significantly correlated with improvements in BDI scores at 1 week, 3 months and 6 months post-psilocybin in the open-label trial by Daws et al 63 (2022). The RCT by Daws et al 63 (2022) also found that decreases in brain network modularity were significantly associated with clinical improvements in depressive symptoms at 3 weeks post-psilocybin. In the same RCT, 63 increased CEN dynamic flexibility also strongly correlated with greater improvement in depressive symptoms at 6 weeks post-treatment.
Ongoing Clinical Trials: Overview of Results
Detailed information on ongoing clinical trials can be found in Supplementary 1. Four of the five ongoing clinical trials64–67 are using psilocybin as their primary intervention, and one trial 68 is investigating LSD. When examining changes in brain activity, all clinical trials will assess changes in depressive symptomatology using various standardized questionnaires following psilocybin and LSD administration (See Table S1 in the supplementary for more detailed findings). fMRI is the most common neuroimaging modality being used to measure psilocybin effects in clinical trials, with three trials65,66,68 including it as their outcome measure. One clinical trial 64 is using PET to measure the effects of psilocybin on brain activity, while another study 67 is using EEG. Another fMRI-based clinical trial 68 is also using both DTI and PWI (ie, ASL) to measure brain perfusion in LSD administration.
Discussion
Summary of the Findings from Published Studies
The present systematic review investigated six interventional studies and one conference abstract on neuroimaging correlates of psychedelic response in MDD patients. Due to the high variability in neuroimaging metrics, sample sizes, and study designs, a meta-analysis was considered unsuitable for examining the current state of the literature. 69
Preliminary evidence suggests that FC and brain activity changes in the DMN—particularly, the amygdala and mPFC/vmPFC— may be associated with psilocybin treatment response in MDD patients. Select psilocybin studies reported alterations in the activity and FC of the temporal gyri, ACC and PCC/precuneus, and visual areas following psilocybin intake. However, alterations in these regions were not associated with treatment response. The single study on ayahuasca reported increased activation in the limbic regions, including the left NAcc, right insula, and left subgenual areas, though these changes did not correlate with treatment response.
Neuroimaging Correlates of Psychedelic Treatment Response in MDD Patients
Psilocybin
Previous research has illustrated that select ICNs and brain regions are implicated in the neural mechanisms behind antidepressant response. In particular, the CEN, DMN and SN, which integrate critical brain areas including the dorsal ACC (dACC), anterior insula, DLPFC, and amygdala, all have demonstrated specific alterations in MDD patients who underwent treatment with typical antidepressants. 70 In the open-label trial by Daws et al 63 (2022), psilocybin led to reductions in within-DMN FC and increases in DMN to SN and DMN to CEN FC. Though these FC changes did not correlate with clinical outcomes in said trial, 63 changes in within-DMN and between-DMN FC may explain the observed improvements in depressive symptoms following psilocybin treatment given that changes to this network have previously been associated with antidepressant response.71,72 Furthermore, it is plausible that FC reductions to this network may alleviate depressive symptoms, considering that greater DMN dominance and heightened FC have been associated with depressive symptomatology, particularly rumination, among MDD patients.34,73
The RCT by Daws et al 63 (2022) observed that psilocybin-induced increases in CEN dynamic flexibility strongly correlated with greater improvement in depressive symptoms, even when combining several CEN regions with the SN and dorsal attention network. Diminished dynamic flexibility has been closely linked to depressive symptoms. 74 Thus, psilocybin may lead to clinical improvements in depressive symptoms by altering functional dynamics of specific neural networks.
A key finding was that psilocybin-induced changes in the amygdala activity and FC correlated with treatment response in select studies.56–58 Recent studies have highlighted the amygdala's role in antidepressant treatment response in MDD patients. For example, changes in intrinsic connectivity between the amygdala and subgenual cingulate cortex have been associated with antidepressant treatment response to ketamine in patients with TRD. 75 Moreover, changes in amygdala BOLD response to sad facial stimuli have been predictive of antidepressant treatment response to scopolamine in MDD patients. 76 Changes in the amygdala activity and FC may, therefore, broadly characterize antidepressant response and extend to psilocybin treatment. However, it remains unknown whether the reported activity and FC changes result from the specific effect of the psychedelic. Future studies should focus on differentiating the amygdala functional properties specific to psilocybin from those characterizing responses to other antidepressant interventions that target neurotransmission.
It is of note that clinical trials enrolling healthy volunteers have also reported changes in the amygdala activity and mood following psilocybin treatment. Barrett et al 77 (2020) reported that a single dose of psilocybin in healthy volunteers reduced negative affect and the amygdala responsiveness to facial stimuli 1-week post-treatment. In another study 78 involving a single-dose administration of psilocybin to healthy subjects, psilocybin-induced decreases in the right amygdala responsiveness to negative stimuli were correlated with enhanced positive mood following intake. Therefore, psilocybin-induced changes in the amygdala activity may in fact explain improvements in depressive symptoms in MDD patients, given that associations between the amygdala activity and mood are well established in healthy individuals.
Another preliminary finding emerging from two reviewed studies56,57 was that FC changes in the mPFC/vmPFC correlated with improvements in depressive symptoms. Recent studies have highlighted the role of the vmPFC and mPFC, which are integral components of the DMN, in mediating antidepressant response among MDD patients. For instance, vmPFC to cerebellar vermis FC has been implicated in selective serotonin reuptake inhibitor (SSRI) treatment response in MDD patients, with responders showing decreased FC between these regions compared to non-responders. 79 Moreover, mPFC intrinsic connectivity prior to antidepressant treatment has been predictive of remission outcomes in MDD patients. 80 Despite existing evidence supporting the amygdala and mPFC/vmPFC as neural correlates of treatment response following conventional antidepressants, more data are necessary to determine how robust and replicable these neural correlates are and whether they are equally pertinent in the context of psilocybin.
As summarized in the review by Dos Santos et al 45 (2016), Carhart-Harris et al 81 (2012) reported that the magnitude of decreased mPFC activity was related to the subjective effects of psilocybin in healthy volunteers. Additionally, Vollenweider et al 82 (1997) found that increased activity in the PFC, as revealed by increased CMRglu, was positively associated with psilocybin's dose and subjective effects in healthy subjects. Thus, changes in depressive symptoms associated with altered FC in the mPFC/vmPFC among MDD patients may similarly resemble subjective state changes associated with altered PFC activity among healthy volunteers, so this neural correlate is well worthy of more detailed investigation.
With regard to large-scale neural effects of psilocybin, two trials by Daws et al 63 (2022) reported global decreases in brain modularity which significantly correlated with treatment response. Interestingly, enhanced resting-state network modularity has been linked to depression severity.83,84 Thus, it is plausible that psilocybin mediates its therapeutic effects by diminishing network modularity among certain neural networks that are commonly overactive in depressed patients.
Other brain regions that showed altered activity and/or FC under psilocybin were the right middle 58 and left superior temporal gyri, 57 bilateral temporal cortex,61,62 visual areas,56,58,61,62 as well as regions of the DMN, including the ACC57,60 and PCC/precuneus.56,57,60 However, these reported changes did not correlate with improvements in depressive symptoms. Increased PCC/precuneus and middle temporal gyrus activation has been predictive of SSRI treatment response in MDD patients.85,86 Taken together, activity changes in these brain regions warrant further investigation into their associations with depressive symptoms and treatment response.
In contrast, reported changes in brain activity may be confounded by experimental tasks that participants were required to complete. For instance, Roseman et al 58 (2018) found increased activation in the right amygdala and right middle temporal gyrus for the fearful > neutral contrast during an emotional faces image task. In a clinical trial by Pourtois et al 87 (2005), perception of emotional faces was associated with activation in the medial temporal gyrus and enhanced activity in the right amygdala during exposure to fearful faces. Moreover, in a meta-analysis 88 exploring emotional face processing in healthy subjects, the amygdala and middle temporal gyrus were among the key regions found to be significantly activated while processing emotional stimuli. Therefore, it remains unclear whether the observed alterations in brain activity in Roseman et al 58 (2018) result from the unique effects of psilocybin or due to the processing of emotional stimuli.
Furthermore, the conference abstract61,62 noted greater ALFF in the bilateral superior temporal cortex during music listening. However, a recent meta-analysis 89 reported activation in the bilateral superior temporal gyrus during music perception in healthy subjects. Consequently, it remains unclear whether the increased ALFF in the bilateral superior temporal cortex in Wall et al61,62 (2021) results from psilocybin intake or is due to the neural mechanisms of music perception. Future research should take task-based paradigms into account and introduce control groups to determine the isolated effects of psilocybin on neural activity.
Ayahuasca
A single SPECT study 59 investigated the effects of ayahuasca in patients with recurrent depression. Ayahuasca was associated with increased activity in the limbic regions, including the left NAcc, right insula, and left subgenual area. Though no specific neural correlates of psychedelic effects were reported, the authors did note that ayahuasca intake led to increases in psychoactivity and reductions in depressive symptoms.
Previous research has highlighted the involvement of the limbic system in the neural mechanisms behind antidepressant response. For instance, increased insula activity in response to emotional stimuli following antidepressant treatment has been correlated with clinical improvements in MDD patients. 90 Moreover, clinical improvements in depressive symptoms following fluoxetine treatment have been associated with decreased glucose metabolism in the limbic areas, particularly in the subgenual area, hippocampus, insula, and pallidum. 91 Therefore, changes in the limbic system activity may offer valuable insight into the neural correlates of treatment response following ayahuasca.
Furthermore, clinical trials in healthy controls have highlighted ayahuasca's activity in the limbic region. Specifically, the amygdala 92 and parahippocampus92,93 have also shown increased activity under ayahuasca. Another clinical trial 94 involving healthy participants reported increased ACC to SN connectivity, decreased PCC to DMN connectivity, and increased SN to DMN connectivity following a single session of ayahuasca. In the same trial, 94 increases in FC between the SN and DMN were significantly associated with negative affect scores. Considering the role of the limbic system in emotion regulation, ayahuasca-induced activity changes within this region may, therefore, explain clinical improvements in depressive symptoms among MDD patients.
Registered Clinical Trials
Five registered clinical trials provide insight into the changing landscape of psychedelic neuroimaging studies in MDD. While four clinical trials64–67 are investigating the neural effects of psilocybin (80%), one trial 68 (20%) is exploring the neural effects of LSD-assisted psychotherapy. Given the shortage of research investigating LSD for the treatment of MDD, this trial is especially important as it may increase our understanding on the unique effects of certain psychedelics on brain activity. Neuroimaging research in healthy volunteers has shown that LSD reduces FC of the anterior mPFC and globally alters dynamic FC. 95 Moreover, LSD-induced decreases in FC between the parahippocampus and retrosplenial cortex have been associated with self-reports of mystical experiences, such as ‘ego-dissolution’ and ‘altered meaning.’ 96
However, neuroimaging research exploring LSD for depression is lacking. For this reason, the single, upcoming LSD trial 68 can provide valuable insight into the neural mechanisms of LSD in the context of depression. Moreover, this is the only trial to use psychedelic-assisted psychotherapy. Published studies have shown variable methods when using psychedelic-assisted psychotherapy, however, it is important to examine this aspect as well. Comparing interventional methods while investigating neuroimaging correlates and psychedelic efficacy can contribute to the evidence supporting one treatment regimen over another.
Furthermore, all but one 67 clinical trials investigating psilocybin have estimated sample sizes of 30 or more. This provides researchers with more accurate and reliable data that may be used to refine sample size estimates for future large-scale psychedelics trials for MDD. Overall, unpublished clinical trials offer insight into the neural interactions of psychedelics in MDD patients and how their clinical effects are achieved.
Strengths, Limitations, and Future Directions
A strength of this systematic review is the inclusion of both published and registered clinical trials to provide a more realistic representation of the existing state of research. Moreover, three databases (MEDLINE, Embase, APA PsycINFO), one clinical registry (Clinicaltrials.gov), and one grey literature database (OpenGrey) were searched to gather as much relevant literature as possible.
The main limitation of the current research landscape is the low number of published studies exploring the neuroimaging correlates of psychedelic treatment response in MDD patients, given the relatively recent emerging interest in this area of research. A new systematic review 97 reported the use of oral psilocybin as a well-tolerated and efficacious treatment for psychiatric conditions such as depression, anxiety, and substance use disorders. Therefore, though the current scope of psychedelic efficacy trials supports their use, mechanistic neural studies exploring psychedelic activity remain scarce. In a rigorous methodological review of psychedelic clinical trials, Aday et al 98 (2022) emphasize the importance of including objective and reliable biomarker measures, including predictive biomarkers and surrogate endpoints, in the design of future trials to reduce bias associated with self-report and clinician-administered assessments of clinical outcomes. Including an fMRI component in the design of future psychedelics trials represents one of the avenues for this emerging field of research to delineate plausible neural mechanisms that could predict or explain the action of psychedelics in MDD. Efforts should be made to standardize this fMRI component as more trials enter the psychedelic neuroimaging space – acquisition sequences and data analysis scripts should be shared across labs, new data analysis models should be tested against previously reported models, and hypotheses should be based on previously reported neuroimaging findings, including the neural signatures identified in this systematic review, in attempts to replicate them with independent data. 44
A secondary key limitation of this review, related to the main limitation, is the small sample sizes used in all the included studies and the identical cohort of subjects employed in Carhart Harris et al 57 (2017), Roseman et al 58 (2018), Mertens et al 56 (2020) and Wall et al61,62 (2021). In addition, several studies did not include control groups. Future research should aim to increase study sample sizes and allocate sizeable participants to both control and experimental study groups to increase the validity and strength of the findings and minimize confounding effects. A review by Aday et al 98 (2022) emphasized the importance of having control groups in psychedelic research. Particularly, there is a need for active control groups in psychedelic research, such as a placebo that mimics some of the effects of psychedelics, to exclude treatment-nonspecific factors as one of the sources of treatment outcome differences. This also applies to neuroimaging studies of psychedelics for MDD - for example, most of the studies included in our review were open-label, resulting in a heightened risk of treatment-nonspecific effects on corresponding clinical outcomes, such as the Hawthorne effect, 99 whereby MDD patients may have unconsciously changed their behaviour in response to being observed by research personnel.
In the field of neuroimaging, treatment-nonspecific effects associated with the administration of psychedelics remain unknown in relation to how they affect fMRI metrics in participants with MDD. Therefore, in line with the general recommendations to improve the ability to discern the treatment-specific effects of psychedelics, 98 fMRI studies should aim to identify neural characteristics associated with treatment-nonspecific effects, such as placebo and nocebo response, as well as with a single versus repeated administration of psychedelics. From the study design perspective, future trials will also benefit from introducing control groups, including active comparator, to identify the impact of treatment-nonspecific effects on clinical and neuroimaging outcomes.
Another limitation of our review concerns the fact that all included studies conducted neuroimaging scans at baseline and shortly after treatment, thereby concealing the long-term impact of psychedelics on neural activity. Future research should aim to corroborate the literature's current findings by conducting clinical trials investigating the neuroimaging correlates of antidepressant response following psychedelic treatments at multiple time points post-treatment and during follow-up to assess the sustainability of treatment effects at the neural level. Moreover, as evidenced by this review, psilocybin and ayahuasca are currently the only psychedelics that have been explored in the context of MDD. Therefore, studies are yet to investigate other psychedelic treatments, such as DMT and LSD, in MDD patients.
Another significant limitation of the included psilocybin studies is the variation in study design concerning the use of either task-based, task-free, or music listening neuroimaging scans, as well as the presence or absence of psychedelic-assisted psychotherapy. While resting-state studies may naturalistically provide insight into the brain functions associated with the experience of being treated with psychedelics, potential for the use of task-based fMRI has also been demonstrated. 44 It is, therefore, difficult to equally weigh and compare the findings across the studies bearing that methodological variability in scan and treatment protocols may potentially confound neuroimaging results. A review by McCulloch et al 44 (2022) highlighted this methodological heterogeneity within psychedelic literature, as the field is still in its nascent stages. For instance, eyes-closed and eyes-open resting-state fMRI metrics have both been employed across psychedelic neuroimaging studies. 44 While it is suggested that scans are acquired with the eyes closed due to visual alterations and associated changes in brain dynamics produced by psychedelics, this underscores the need for more research to determine which procedures work best 44 by replicating the designs of the included psilocybin studies. This will help determine if these conflicting study methodologies do, in fact, significantly impact neuroimaging outcomes.
Conclusion
Psychedelic interventions appear to have potential clinical benefits for patients with MDD and TRD as fast-acting alternatives to first-line antidepressants. The findings of the present review indicate that the DMN— particularly, the amygdala and mid-prefrontal regions (i.e., mPFC, vmPFC) —may offer a preliminary foundation for hypothesis generation and theory interrogation for future research aiming to investigate the underlying neural mechanisms of antidepressant response following psilocybin treatment. Although these mechanisms are not clearly understood, the evidence suggests that these brain regions and associated ICNs may be involved in mediating antidepressant response, although this evidence is based only on four datasets with extremely different methods. Psilocybin also appears to target multiple other brain regions within the temporal lobe, DMN (ie, ACC and PCC) and visual areas, though there is no evidence to date to suggest that these correlates can be used to predict antidepressant response following psilocybin treatment. Moreover, ayahuasca may mediate its therapeutic effects by increasing activity in the limbic regions, though this assumption is constrained to the findings of a single clinical trial. 59 Several ongoing, registered clinical trials plan to further investigate the neuroimaging correlates of psychedelics, such as psilocybin and LSD, in MDD patients specifically. Importantly, incorporating neuroimaging modalities in psychedelic interventions could enhance this developing area of research by providing insight into the neural mechanisms underlying psychedelic treatment efficacy in MDD, although standardization across neuroimaging acquisition parameters and data analysis pipelines is critically needed. Importantly, the neural signatures identified in this review represent early observations. Further research and integration among datasets are required to replicate these observations and draw more definitive conclusions regarding the effect of psychedelics on the brain in patients with MDD.
Supplemental Material
sj-docx-1-css-10.1177_24705470221115342 - Supplemental material for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review
Supplemental material, sj-docx-1-css-10.1177_24705470221115342 for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review by Sarah Kuburi, Anne-Marie Di Passa, Vanessa K. Tassone, Raesham Mahmood, Aleksandra Lalovic, Karim S. Ladha, Katharine Dunlop, Sakina Rizvi, Ilya Demchenko and Venkat Bhat in Chronic Stress
Supplemental Material
sj-docx-2-css-10.1177_24705470221115342 - Supplemental material for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review
Supplemental material, sj-docx-2-css-10.1177_24705470221115342 for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review by Sarah Kuburi, Anne-Marie Di Passa, Vanessa K. Tassone, Raesham Mahmood, Aleksandra Lalovic, Karim S. Ladha, Katharine Dunlop, Sakina Rizvi, Ilya Demchenko and Venkat Bhat in Chronic Stress
Supplemental Material
sj-docx-3-css-10.1177_24705470221115342 - Supplemental material for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review
Supplemental material, sj-docx-3-css-10.1177_24705470221115342 for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review by Sarah Kuburi, Anne-Marie Di Passa, Vanessa K. Tassone, Raesham Mahmood, Aleksandra Lalovic, Karim S. Ladha, Katharine Dunlop, Sakina Rizvi, Ilya Demchenko and Venkat Bhat in Chronic Stress
Supplemental Material
sj-docx-4-css-10.1177_24705470221115342 - Supplemental material for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review
Supplemental material, sj-docx-4-css-10.1177_24705470221115342 for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review by Sarah Kuburi, Anne-Marie Di Passa, Vanessa K. Tassone, Raesham Mahmood, Aleksandra Lalovic, Karim S. Ladha, Katharine Dunlop, Sakina Rizvi, Ilya Demchenko and Venkat Bhat in Chronic Stress
Supplemental Material
sj-docx-5-css-10.1177_24705470221115342 - Supplemental material for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review
Supplemental material, sj-docx-5-css-10.1177_24705470221115342 for Neuroimaging Correlates of Treatment Response with Psychedelics in Major Depressive Disorder: A Systematic Review by Sarah Kuburi, Anne-Marie Di Passa, Vanessa K. Tassone, Raesham Mahmood, Aleksandra Lalovic, Karim S. Ladha, Katharine Dunlop, Sakina Rizvi, Ilya Demchenko and Venkat Bhat in Chronic Stress
Footnotes
Declaration of Conflicting Interests
SK, AD, VKT, RM, AL, SR, and ID declared no potential conflicts of interest with respect to the research, authorship, and publication of this article. KSL is supported in part by Merit Awards from the Department of Anesthesiology and Pain Medicine at the University of Toronto (Toronto, Canada). KD is supported by an Academic Scholar Award from the University of Toronto, Department of Psychiatry. VB is supported by an Academic Scholar Award from the University of Toronto Department of Psychiatry and has received research support from the Canadian Institutes of Health Research, Brain & Behavior Foundation, Ministry of Health Innovation Funds, Royal College of Physicians and Surgeons of Canada, Department of National Defence (Canada), and an investigator-initiated trial from Roche Canada.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data Availability
The data that support the findings of this study are available from the corresponding author, VB, upon reasonable request.
Author Contributions
S.K, A.D and V.B conceptualized the review. S.K and A.D conducted a systematic search of the available literature and performed the data extraction and analysis. S.K and A.D also produced the first version of the manuscript. All aspects of the manuscript were overseen by I.D and V.B. The remaining authors provided critical revision of the article. All authors read and approved the final version of this manuscript.
Supplemental Materials
Supplementary 1 is a text document with further data on the ongoing clinical trials included in this review
Supplementary 2 is a text document on the search strategies that were used to retrieve the records for this systematic review (including both OVID search and ClinicalTrials.gov search)
Supplementary 3 is a text document with the results of the quality assessment as per the GRADE checklist for use in systematic reviews
Supplementary 4 is a text document with the PRISMA Checklist to support this systematic review
Supplementary 5 is a text document with the PRISMA Abstract Checklist to support this systematic review
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
