Abstract
Cystic fibrosis (CF) is an autosomal recessive genetic disease caused by mutations in the cystic fibrosis transmembrane conductance regulation (CFTR) anion channel. Loss of CFTR protein and/or function disrupts chloride, bicarbonate, and fluid transport and also impacts epithelial sodium transport. Such altered ion and fluid transport produces mucus obstruction, inflammation, pulmonary infection, and damage to multiple organs. Although an autosomal disease, it is apparent that gender differences in life expectancy and quality of life do exist. Conventionally established therapies have treated the downstream sequelae of CFTR dysfunction and have led to a steady increase in life expectancy. Physicians now have access to medications that treat the basic defect in CF, in the form of CFTR modulators. These drugs target the trafficking and/or function of CFTR to improve clinical outcomes for patients. This review summarizes the science behind CFTR modulators and shows how these drugs have dramatically changed how patients with CF are treated. Surprisingly, although the drug target(s) are identical in males and females, CF females seem to display a greater improvement than their male counterparts.
Introduction
Cystic fibrosis (CF) is the most common lethal genetic disease in Caucasian populations, occurring in approximately 1:3500 births in the United States, with a slightly higher level in Northern European countries. 1,2 The discovery of the CF gene, cftr, some 30 years ago was a major milestone in unraveling the mechanistic defect causing CF, 3 -5 and researchers and clinicians assumed a therapy based on knowledge of the genetic defect was only a matter of time. In fact, it took another quarter of a century before the first drug to treat the basic defect in CF received Federal Drug Administration (FDA) approval. Cystic fibrosis is caused by mutations in the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR) protein, an anion channel capable of transporting chloride and bicarbonate ions. 6,7 The cftr gene is located on the middle of the long arm of chromosome 7 and inherited as an autosomal recessive disease according to classical Mendelian genetics. Disease-causing variants alter ion and fluid transport across epithelia leading to altered viscoelastic properties in airway mucus. 5,8 In normal airways, the height of the airway surface liquid (ASL) is maintained by the opposing actions of chloride secretion and sodium absorption, causing the appropriate amount of osmotic water transport (Figure 1). Fully hydrated mucus released from submucosal glands sits on top of the ASL to attract dust particles, viruses, bacteria, and other debris, that is moved to the back of the throat by the mucociliary escalator. In CF, the loss of chloride secretion, coupled to enhanced sodium absorption markedly reduces ASL height. Together with thick unhydrated mucus from submucosal glands, the cilia are compressed, and mucociliary clearance fails to occur. Increased secretion of dehydrated mucus in the lungs predisposes the tissue to chronic infection, inflammation, and airway damage that can progress to respiratory failure. Although CF is often thought of as a pulmonary disease, and indeed this contributes the most to current morbidity and mortality, it nonetheless impacts multiple organ systems, including the gastrointestinal (GI) tract, reproductive tract, sweat glands, endocrine systems, and bone. In the pancreas, failure to properly hydrate and flush out digestive enzymes from the exocrine pancreas into the duodenum not only leads to poor nutrition and failure to thrive but also leads to pancreatic autodigestion and the appearance of pancreatic enzymes in the blood. There are more than 2000 variants of the cftr gene that have been identified, though only a subset of these have been well described, and are clearly disease-related (www.genet.sickkids.on.ca/cftr and www.cftr2.org). The vast majority of patients with CF display a relatively small number of variants that can be broken down into 2 broad categories, a lack of CFTR protein at the cell surface or production of a non (or poorly) functioning protein at the cell surface. 9,10
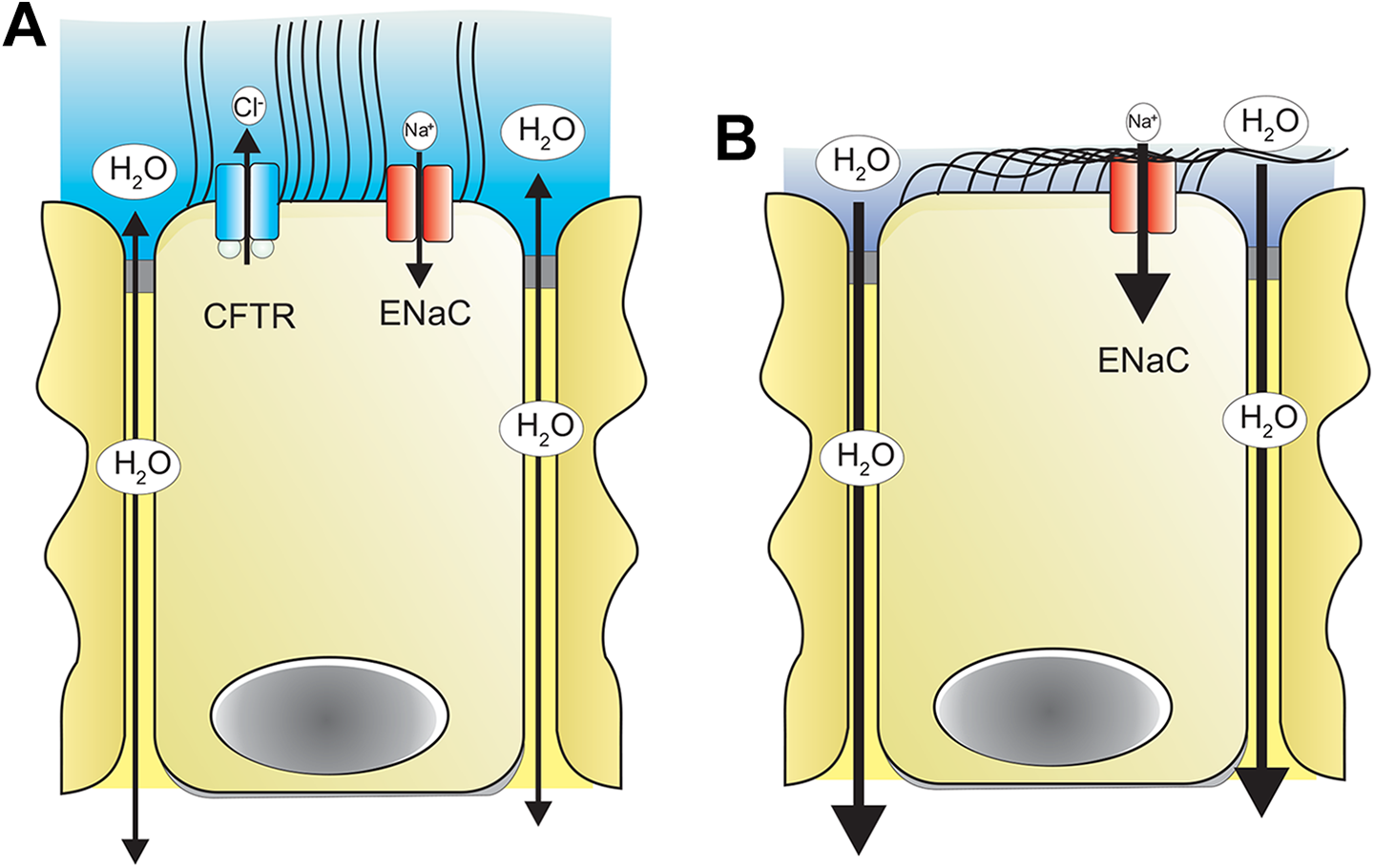
Role of CFTR in airway ion and fluid movement. In healthy airways (A), CFTR is present in the apical membrane of bronchial epithelial cells. The combination of CFTR-mediated chloride secretion and ENaC-mediated sodium absorption controls proper airway surface hydration and proper ASL height, leading to effective mucociliary clearance. In CF (B), a variety of mutations lead to either absent or nonfunctioning CFTR in the apical membrane. This leads to a lack of chloride secretion. For reasons not fully understood, the absence of CFTR protein and/or function enhances Epithelail sodium channel activity, unbalancing ionic levels and leading to osmotic water absorption from the airway surface. With little or no ASL, the cilia are unable to beat effectively and mucociliary clearance is severely compromised. ASL indicates airway surface liquid; CF, cystic fibrosis; CFTR, cystic fibrosis transmembrane conductance regulation.
Symptom-Based Therapies
Until recently, the mainstay of CF therapies has been the treatment of symptoms. Although not treating the basic defect, they nonetheless have been highly successful in bringing about a steady increase in patient life span and life quality outcomes (Figure 2). While symptom-based, these established therapies did address critical aspects of disease pathology, including airway bacterial infections (with inhaled and systemic antibiotics such as tobramycin and azithromycin), dehydrated mucus (with inhaled osmotic agents such as hypertonic saline), mucus plugging (recombinant human DNAse), inflammation (high-dose non-steroidal antiinflammatory drugs, azithromycin), pancreatic insufficiency (with pancreatic replacement enzymes), and nutritional deficits (with fat-soluble vitamins and high caloric supplements). 10 -16 Even with the remarkable strides seen in the predicted survival of patients with CF, the median life expectancy of a patient with CF is still well below than that of a non-CF newborn. 17 Thus, there is a clear need for the development of more effective therapies targeting the manifold aspects of CF, if the disease is to change from the most common lethal genetic disease of Caucasians into nothing more than a genetic trait. 18 Among the non-CFTR, drug targets are mucoactive compounds, anti-inflammatories, antibiotics, and treatments to address the nutritional deficiencies, 18 -21 as well as other ion channels (sodium channels, eg, ENaC inhibitors, or alternative chloride channels, eg, CaCC). This review will focus on compounds directed toward modulating mutant CFTR.
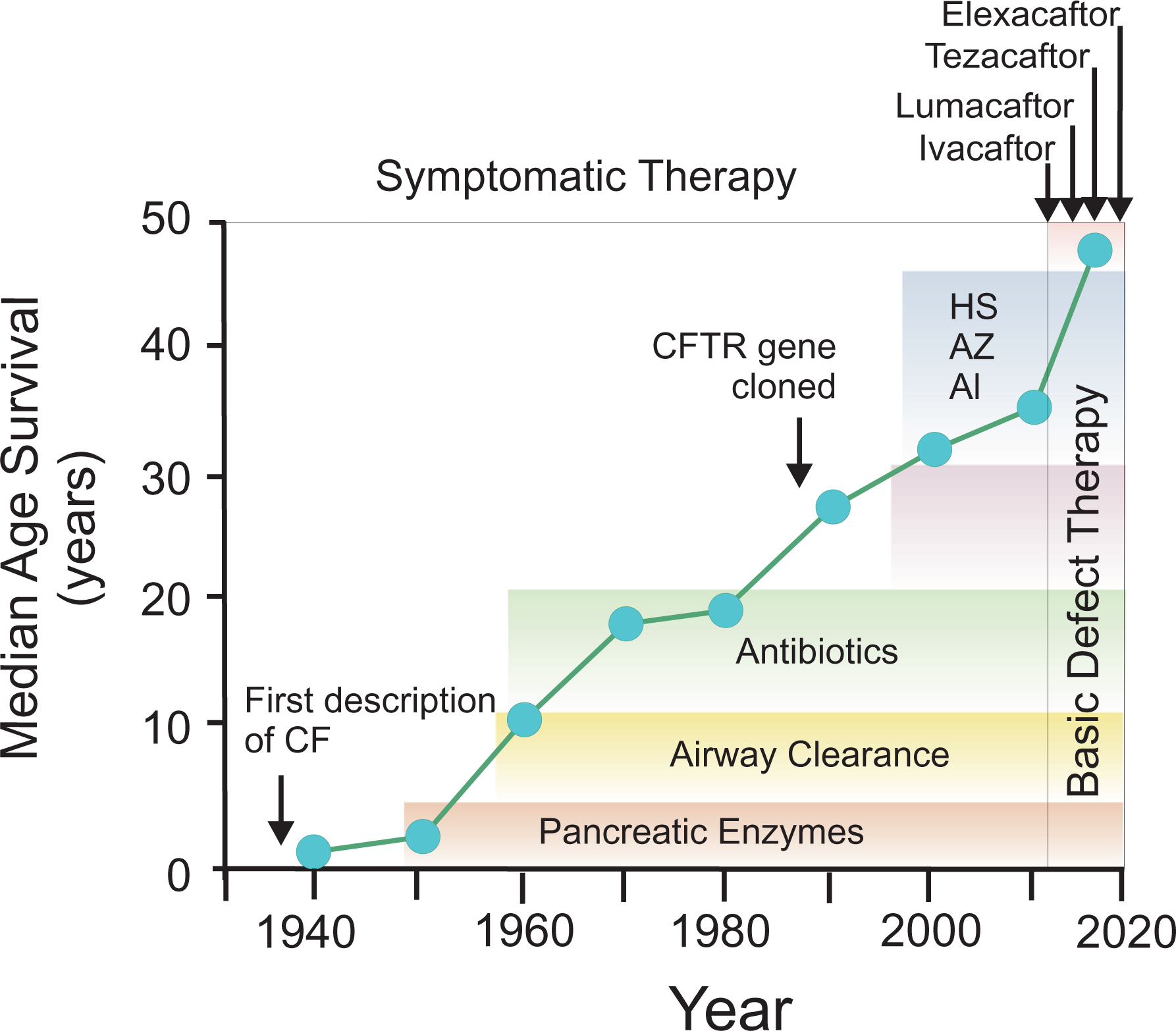
Median survival in CF. Cystic fibrosis survival over time, associated with mile-stones (arrows) and CF therapies (boxes). Steady increases in median survival have followed the introduction of new therapies and changes in care delivery. AI indicates inhaled aztreonam; AZ, azithromycin*; CF, cystic fibrosis; CFTR, cystic fibrosis transmembrane conductance regulator; HS, hypertonic saline. Shown is the distinction between symptomatic therapies and therapies directed against the molecular defect. *Although azithromycin is a macrolide antibiotic, it also has well-described anti-inflammatory properties, with actions at least partly ascribed to macrophages145.
Precision Medicine
In contrast to established symptom-based therapies for CF, which have no regard to a patient’s genotype, recent therapies termed CFTR modulators have generated a lot of excitement in the CF research and care community, by directly targeting mutant CFTR in a mutation-specific manner. 10,18,21 -24 Indeed, accumulating clinical trial data argues that such modulators have tremendous benefit for patients with CF outcomes, including lung function (measured by forced expiratory volume in one second [FEV1]), pulmonary exacerbation rates (worsening of respiratory symptoms and lung function), weight gain, growth, and in some cases, sweat chloride. 13,21,23,25 -29 Unlike some genetic conditions where mutations are found on the sex chromosomes, the gene for CFTR is found on chromosome 7 and has equal penetrance in both males and females. However, the severity of symptoms in patients with CF does appear to vary between men and women (discussed in detail later), yet to date, no consideration of gender has routinely been applied to drug development. There were several reasons for initially not considering gender. Cystic fibrosis is an autosomal disease, and so there was no a priori reason to assume that meaningful differences between genders would exist. Gender had not been considered with symptom-based therapies, and the application of such therapies had greatly improved life quality and survival in both males and females. With the advent of technologies enabling high-throughput screening (HTS) of drugs against relevant airway epithelia, there was a growing demand for human airway tissue. Given the limited availability of lung transplant tissues at the time, with which to perform HTS and drug development assays, investigators were happy just to get any airway samples regardless of gender. In fact, the consideration was given exclusively to genotype since the focus of drug companies was on developing drugs effective against specific CFTR mutations. The increased use of nasal epithelial cells, which do not rely on cadaveric of transplant tissues, and are easy to obtain, will in future afford more gender selection in tissues for drug screening. Perhaps, the most important reason that gender was not considered in drug development was simply because the FDA did not require them, and so teasing out gender-specific information was not warranted. As pharmaceutical companies move forward with drug development for patients with CF, this may be the information the FDA requires in the future.
Although CFTR modulators display reasonable efficacy, they suffer from 2 important problems. Firstly, not all mutations are covered by existing modulators. This is especially true for nonsense mutations. Secondly, even though the CFTR protein can be somewhat “corrected,” it is still not a wild-type protein. Full restoration of wild-type CFTR activity will obviously rely on some form of nucleotide therapy, whether this is an insertion of a wild-type complementary DNA (cDNA) or repair of genomic DNA through CRISPR gene editing approaches. Although such approaches have clear advantages, their implementation has proven difficult, with the problem of nucleotide entry still an issue. 30
When considering how CFTR modulators improve mutant CFTR function, it is perhaps beneficial to have insight into the nature of the problem. Cystic fibrosis transmembrane conductance regulator is a member of the traffic ATPase or ABC (adenosine triphosphate [ATP]–binding cassette) family. Many other members of this family are also associated with clinical disease. For example, P-glycoprotein (MDR1) confers resistance to chemotherapeutic drugs by pumping the drugs out of the cell using energy from ATP hydrolysis. 31 Similarly, the ABC transport adrenoleukodystrophy protein (ABCD1) is present in peroxisomal membranes and works to transport fatty acids into peroxisomes for degradation. Mutations in ALDP are associated with X-linked adrenoleukodystrophy, 32,33 a condition brought to the attention of many through the Hollywood movie “Lorenzo’s Oil.” Cystic fibrosis transmembrane conductance regulator is a 1480 amino acid integral membrane protein that normally resides in the apical membrane of polarized epithelial cells lining the airways, GI tract, vas deferens, pancreatic duct, biliary tree, and sweat gland ducts. 5,34 -38 Like all members of the ABC family, CFTR is composed of 2 membrane-spanning domains (TMD-1 and -2), which anchor the protein in the plasma membrane, and 2 nucleotide-binding domains (NBD-1 and -2), which form a heterodimer to bind and hydrolyze ATP, presumably to control channel gating (CFTR opening and closing). 5,39,40 Cystic fibrosis transmembrane conductance regulator is unique, however, in having an unstructured central linking regulatory, or R, domain, which imparts phosphorylation-sensitive activation (predominantly protein kinase A), and thus controls the transport of chloride and bicarbonate 41 -43 (Figure 3). Recently, the structure of human CFTR has been determined by cryo-EM at a resolution of 3.9 Å. 39 This certainly allows for in silico molecular docking of pharmacologic compounds into mutant CFTR, though translating such docking into therapeutic reagents is an enormous task. 44
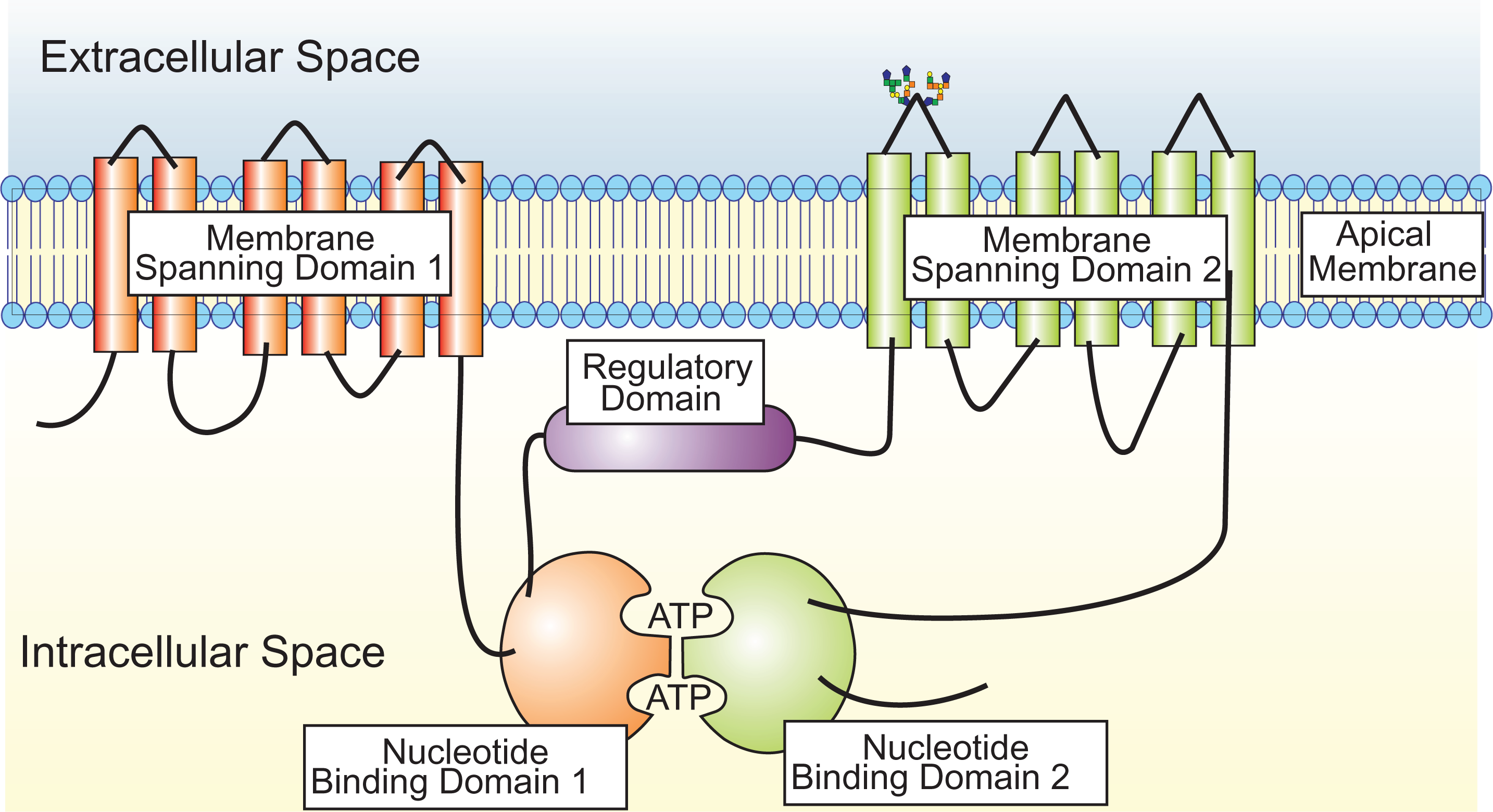
Model for CFTR model showing the domain structure of CFTR. The protein has 1480 amino acids, with both the amino and carboxy termini within the cell cytoplasm. The 2 membrane spanning domains each containing 6 transmembrane segments, forming the ion channel. There are 6 extracellular loops, with glycosylation occurring on asparagine residues at 2 sites on extracellular loop 4. Also shown are 2 nucleotide-binding domains (the F508del mutation is located in NBD1). Phosphorylation of the regulatory (R) domain by cAMP-dependent protein kinase (PKA) is necessary for channel function. cAMP indicates cyclic adenosine monophosphate; CFTR, cystic fibrosis transmembrane conductance regulation.
What goes wrong?
When thinking about what can be altered to improve CFTR activity (or indeed any ion channel), there are only 3 things that can be manipulated. Movement of an ion through a channel, in this case, chloride or bicarbonate through CFTR, is directed according to the formula I = i * N * Po , where I is the overall current passing through all CFTR molecules in the plasma membrane, i is the conductance or current passing through an individual CFTR channel, N is the number of channels in the membrane, and Po is the open probability for each channel (ie, what percentage of the time the channel is open and passing ions). So far, we know of no way to alter i, and so currently available modulators include Potentiators which increase open channel probability, or Po , and correctors which improve N, the number of CFTR channels in the membrane.
Gene sequencing has revealed over 2000 mutations in CFTR. Despite this large number, there are 2 general principles that apply. Firstly, all mutations can be broadly categorized into mutations that affect the number of channels in the apical membrane and those that reach the membrane but have altered function (Figure 4). For example, the premature stop mutation G542X results in an absence of protein from the cell surface. In contrast, the G551D mutation generates a protein that reaches the cell surface but has altered channel kinetics. Secondly, out of this number of mutations, only 16 mutations account for 85% of CF alleles in the population (http://www.genet.sickkids.on.ca/CFTR). As the research community has learnt more about the most common clinically important CFTR variants, it has become clear that they often have more than 1 defect affecting Po , N, or i. This is perhaps no better illustrated than with the most common CFTR disease-causing mutation F508del, which is caused by a 3 base pair deletion in exon 10, and results in the omission of a phenylalanine residue at position 508 in the full-length protein. 5
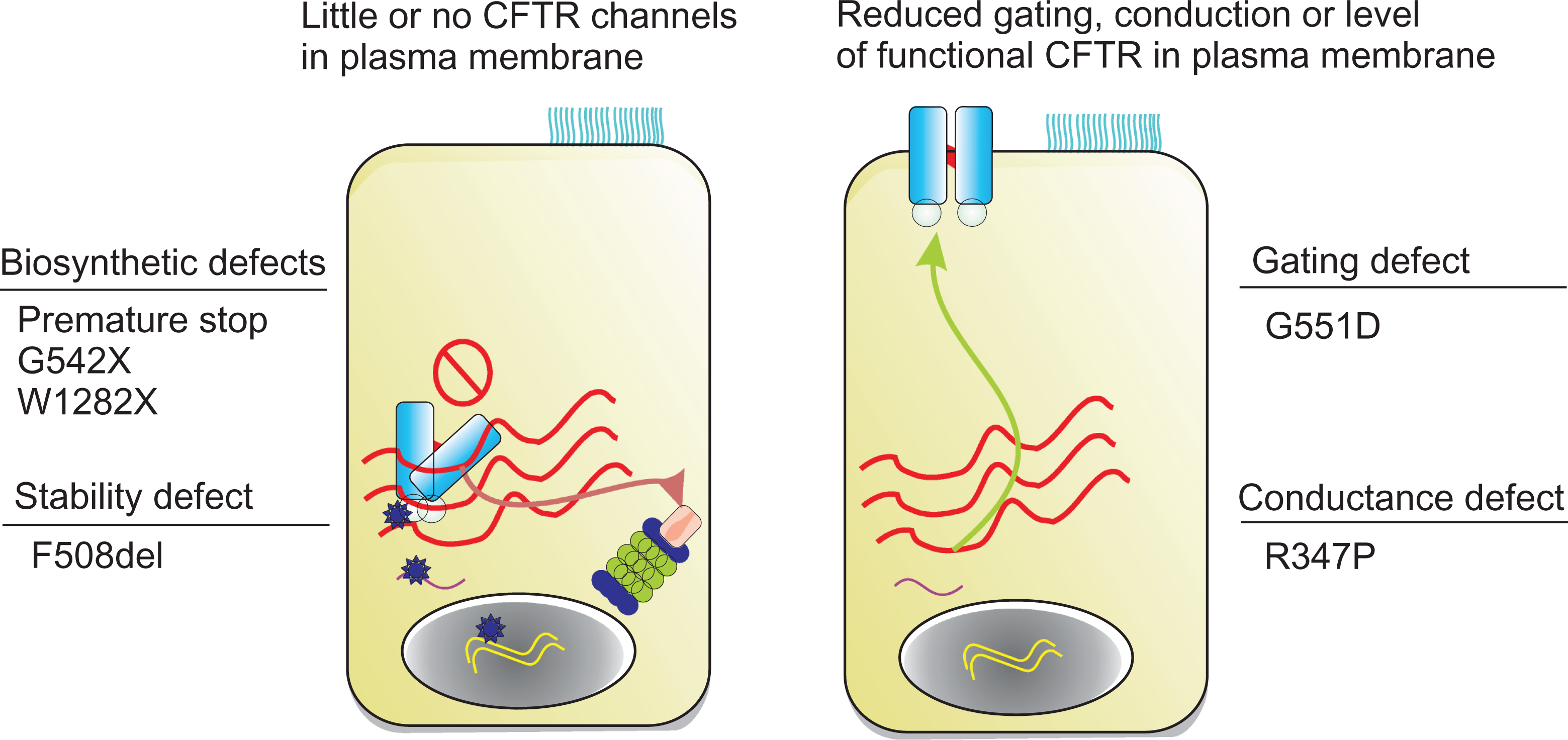
CFTR mutation classes. Disease-causing CFTR variants are determined by the presence or absence of CFTR at the plasma membrane 10 . Left: CFTR variants that result in little or no CFTR at the plasma membrane. CFTR is ER export incompetent and subject to proteasomal degradation. Right: CFTR variants with residual function are localized to the plasma membrane. Examples within each group are given. CFTR indicates cystic fibrosis transmembrane conductance regulation; ER, endoplasmic reticulum.
The primary defect with F508del CFTR is a problem with protein stability. The F508del protein folds to an approximation of the wild-type structure but is very unstable and denatures easily. The mutant protein is recognized as impaired by the endoplasmic reticulum (ER) quality control machinery, failing to mature and exit the ER to become a mature fully glycosylated protein. F508del CFTR is rapidly degraded by the 26S proteasome, and little, if any, CFTR reaches the plasma membrane (ie, reduced N). 34,45 -47 Shortly after the cloning of CFTR, it was realized that F508del CFTR could be encouraged to reach the plasma membrane through a variety of experimental manipulations, including low temperature or glycerol 48,49 ; however, even when at the cell surface, F508del CFTR shows defects in channel gating (ie, reduced Po ) and defects in stability, which reduces N. 50 -52 F508del is not the only mutation to sit in both categories of mutations. The P67L mutation also exhibits defects in both plasma membrane expression and gating. 53
Mutations that reduce the number of apical membrane CFTR channels to a very low or zero level, including premature termination of mutations, frameshifts, or splice variants, are typically associated with a severe phenotype and have little if any residual exocrine pancreatic function. Such mutations give rise to the so-called pancreatic insufficient conditions and are associated with a poorer prognosis. Indeed, pancreatic insufficient patients account for 85% of all patients with CF and is associated with maldigestion as evidenced by steatorrhea. Such patients require pancreatic enzyme replacement therapy with each meal. However, just getting to the cell surface is not sufficient, if mutant CFTR also harbors severe gating mutations. Thus, G551D CFTR, which has appreciable N, has a very low Po and is considered a severe pancreatic insufficient mutation. Mutations that permit CFTR to reach the cell surface and have some residual function have a milder phenotype and are often associated with pancreatic sufficiency. 2,8,54 It should be borne in mind, however, that an individual with ostensibly 2 severe mutations may, in fact, present with mild clinical disease, whereas patients with an allele predicted to be a milder mutation may have typical severe CF manifestations. While the exact reasons for this apparent discrepancy are still unclear, it is known that allelic variations on other non-CFTR genes (modifier genes) can impact disease severity. 55 -57
CFTR Modulators
Since mutant CFTR can lead to disease through different general mechanisms, which ultimately is a reflection of a patient’s individual genotype, it is clear that a single pharmacophore is unlikely to be a panacea for all mutations. Drugs that are currently available for prescription fall into 2 categories, correctors and potentiators. Correctors are designed to allow mutant CFTR to be ER export competent and reach the cell surface. Potentiators work on those mutations that reach the plasma membrane but have deficits in function. Of course, mutations like F508del and P67L will need drugs from both categories to be treated efficiently. Since 90% of patients with CF carry at least F508del allele, and this is obviously a critical mutation to target. Despite the prominence of the F508del mutation, it was however not the first mutation to receive an FDA-approved drug. That honor went to the G551D mutation since this variant reaches the cell surface reasonably well but displays a gating defect. Fixing only 1 thing seemed easier than trying to fix 2 different things at the same time.
Cystic Fibrosis Transmembrane Conductance Regulation Potentiators
Cystic fibrosis transmembrane conductance regulation potentiators are small molecule drugs that increase the gating of CFTR, increasing open probability (P o), causing the channel to spend more time in the open configuration and facilitating the movement of anions across the cell membrane. 10,58,59 Potentiators were identified using heterologous gene expression, coupled with HTS approaches. 58 The HTS screening target used was the G551D mutation. Although the G551D variant represents only 5% of all patients with CF globally, it is still the third most prevalent disease-causing mutation (www.genet.sickkids.on.ca). Moreover, G551D represented an important proof of concept and seemed an ideal initial target. G551D has no difficulty being exported from the ER and reaching the plasma membrane but does display a very low P o, a characteristic that could possibly be increased. Following initial HTS, Vertex Pharmaceuticals discovered VX-770, also known as ivacaftor, which was subsequently evaluated against primary human bronchial epithelial (HBE) cells grown in planar culture. 58 Using this model system, ivacaftor was found to increase total G551D CFTR activity to almost 50% of that observed in HBE cultures from non-CF individuals (ie, wt-CFTR). 23,25,58,60 -62 Such improvement, or potentiation, of gating activity resulted in several key downstream benefits, including improvement of ASL volumes (ASL height), improved ciliary beat frequency, and improved mucociliary clearance. In contrast to the animal testing pathway that many FDA-approved drugs undergo, ivacaftor was developed independently of animal models since the only available model (genetically engineered mice) displayed minimal airway disease. As a result, the HBE assay has become the gold standard for which all CFTR modulators are tested against.
Although ivacaftor (sold as Kalydeco) identified by screening G551D expressing tissues and approved for use in patients with CF who have at least 1 G551D mutation, other gating mutations such as R117H, G551S, S549N, G970R, and G1349D have also shown responses to the drug. 25,26,28,29,61,63 -70 Improvements on FEV1 percent predicted of greater than 10%, and a reduction in pulmonary exacerbations of greater than 50%, as well as weight increases, have all been noted in patients receiving ivacaftor relative to placebo control. Moreover, reductions in sweat electrolytes of around 50 mM have been observed with CFTR-gating mutations exposed to ivacaftor, with most improvement seen in younger patients. 61,69 Encouragingly, toddlers treated with ivacaftor have shown increased fecal elastase levels, suggesting an improvement in exocrine pancreatic function. 61,69 Prolonged treatment of homozygous G551D individuals with ivacaftor appears to reduce the rate of loss in lung function relative to untreated controls and possibly reduces mortality in patients with advanced pulmonary disease. 71,72 Based on accumulating evidence of potentiator efficacy in other CFTR-gating mutants, the FDA has expanded the number of mutations for which ivacaftor can be prescribed on the label. 72 This is an exciting development, as the expansion now covers several rare mutations, which would have difficulty recruiting enough patients for a rigorous clinical trial.
Mechanistic studies suggest that ivacaftor directly binds to CFTR. 73 -75 Whether knowing the precise binding site for potentiators on CFTR will allow for in silico predictions of improved modulators is still not known. Moreover, formal determination that binding of ivacaftor to CFTR is directly related to its potentiator activity remains to be done. Obviously for potentiators to be effective, they must work on CFTR mutants that are present in the plasma membrane. Thus, ivacaftor has failed to show any demonstrable clinical improvement in patients homozygous for the F508del mutation. 76
CFTR Correctors
Although F508del CFTR is missing an entire amino acid, the crystal structure of mutant and wild-type CFTR is remarkably similar. 77 Indeed, the problem with F508del CFTR appears to be more of a stability issue, with F508del unfolding more easily than its wild-type counterpart. The loss of phenylalanine at position 508 results in a protein whose primary defect is an inability to be exported from the ER and fails to reach the plasma membrane. 34,78,79 Since 90% of all patients with CF harbor at least 1 F508del allele, it is clear that drugs that target F508del CFTR will impact most patients.
Certain experimental manipulations such as low temperature 49 can get CFTR to the cell surface, but even then it is not as functional as wt-CFTR. 51,52 The first identified correctors were Corr-4a (bisamionomethylbithiazole C4) and VRT-325 (quinazolinone C3); however, they were not selective for CFTR and displayed low efficiency and high toxicity. The next-generation correctors included lumacaftor (VX-809), developed by Vertex Pharmaceuticals. VX-809 showed promise in preclinical studies, restoring the trafficking surface expression of F508del CFTR to around 15% wild-type CFTR activity, 80,81 at least it showed modest efficacy in patients with CF with the F508del mutation in phase 2 clinical trials. Indeed, although lumacaftor in combination with a potentiator was later approved for clinical use, lumacaftor alone is not approved by the FDA. In contrast to the effect of the potentiator ivacaftor on multiple gating mutations, so far lumacaftor appears to be selective for the F508del protein. 81 Disappointingly, lumacaftor appears to have little efficacy toward N1303K CFTR, the second most common “folding” defect. Clinical trials with lumacaftor showed limited bioavailability and was not effective in improving lung function decline, 82 although small dose-dependent decreases in sweat chloride were observed. 83 From such studies, it appears that for individual patients, sweat chloride correction is not necessarily correlated with improved lung function. It is also apparent, though in retrospect obvious, that a corrector alone is unlikely to be effective for F508del CFTR. Tezacaftor (VX-661) is the latest generation CFTR corrector, but like lumacaftor is not approved on its own but is approved when used in combination with a potentiator. Cystic fibrosis transmembrane conductance regulation correctors are thought to bind directly to CFTR, and there is some evidence in support of this, though whether that is the mechanism of action is still unresolved.
Drug Combos
Although Kalydeco (ivacaftor) had some efficacy toward G551D CFTR, it was ineffective on its own against F508del CFTR since there was nothing in the plasma membrane to potentiate. Conversely, lumacaftor could get some F508del CFTR to the cell surface, but CFTR’s activity was very small. A combination of both drugs therefore seemed a good idea, and a combination of lumacaftor (corrector) plus ivacaftor (potentiator) received FDA approval and was marketed as Orkambi. An observational study using Orkambi showed that it does improve F508del function, at least as assessed by nasal potential difference, and short-circuit current measurements, to levels around 10% to 20% of wild-type. 67 The rate of pulmonary decline in patients, though, did seem to lessen in response to a combination of corrector and potentiator, 84 though side effects of chest tightness were observed. 85
Given the poor efficacy of the lumacaftor/ivacaftor combination, it became apparent that a second-generation corrector would be needed to treat F508del CFTR patients. Based on HTS, the corrector tezacaftor was developed that appears to have a similar mechanism of action on CFTR as lumacaftor. Importantly though, tezacaftor does not appear to activate the cytochrome P450 system (which was a problem with the lumacaftor/ivacaftor combination) and also does not have the side effects of chest tightness. 60 Six-month testing of tezacaftor/ivacaftor (marketed as Symdeko) in patients homozygous for the F508del allele showed improvements in FEV1 compared to control (∼4%), 86 -89 similar to that seen with the lumacaftor/ivacaftor combination, and a reduction in pulmonary exacerbations of ∼35%. 90 More recently, a triple combination therapy comprised of the potentiator ivacaftor, along with 2 correctors, tezacaftor and elexacaftor, has been approved for patients with CF by a fast-tracked FDA approval process. The efficacy of the triple combination, going by the name Trikafta, showed an increase in FEV1 of 10% to 14% compared to placebo in 2 independent clinical trials. Moreover, Trikafta appeared to improve sweat chloride, reduce the number of pulmonary exacerbations, and increased body mass index. 89,91
With recent advances in FDA-approved drugs, and the likelihood of new therapies from other pharmaceutical companies, the impact on CF pathogenesis will only increase. The transformation of CF from a lethal condition to a disease with significantly less morbidity and mortality is clearly within reach. However, with new-generation drugs to treat patients with CF, it is perhaps timely to start looking at the long known but little investigated gender gap in CF.
The Gender Gap in CF
A major difference between males and females with CF has been known for some time, but until recently, with many patients moving into adulthood, has not been an issue much raised. The difference concerns reproductive tissues, where males with CF are infertile due to the absence of vas deferens, but females are capable of conceiving and carrying to full term. 1 While this is likely to become an increasing issue among patients with CF, this review is focused on addressing possible fundamental differences in disease severity and life expectancy. Whether sex is a risk factor in CF morbidity and mortality remains somewhat contentious. The cftr gene is located on an autosome and therefore, at least theoretically, should have equal penetrance among males and females. Indeed, patients with CF are equally represented by both males and females, yet suggestions of gender disparities in disease severity and survival have been brought forward. Unfortunately, a clear picture of gender-dependent morbidity and mortality in CF has been complicated by the fact that different studies have employed widely different age cohorts, different criteria, and a mix of patient data from single- and multiple-combined CF centers. As an example of how large a difference is made just by patients being in 1 center versus another, a comparison of lung function outcomes in patients with CF showed that in 2018, a 10-year gap in the median age of survival existed between US and Canadian patients. 92 However, for patients born after 1990, that gap has diminished considerably. Accounting for the mix in methodologies and treatments, O’Connor and colleagues concluded that for patients with CF, being female was indeed associated with greater risk of death, 93 strengthening the argument for a gender gap in CF. Despite the recent successes in pharmacological therapies targeted to the basic defect, females with CF still have worse outcomes than males. Females with CF still score worse than males with CF in a health-related quality-of-life studies, 94 and females have greater exacerbations of the disease. 95,96 Although both males and females with CF show a greater age-related decrease in lung function compared to non-CF individuals, females have a greater decline in percent-predicted FEV1, compared to males in the 18 to 28 years of age range −1.76% (95% CI: −2.06 to −1.46) and −1.61% (95% CI: −1.91 to −1.31) per year in females and males, respectively. 97 Even accounting for key CF-related comorbidities, females with CF have a shortened life expectancy than their male counterparts (Figure 5). 98 -106 The most recent analysis shows a median life expectancy of 38.7 years for male patients with CF, compared to 36.0 years for females. 99 Remarkably, even since the publication of this report in 2014, the 2018 patients with CF registry predicts that for those patients born in 2018, the average survival age will increase by a decade to 47.4 years (https://www.cff.org/Research/Researcher-Resources/Patient-Registry/2018-Patient-Registry-Annual-Data-Report.pdf), an increase for both genders.
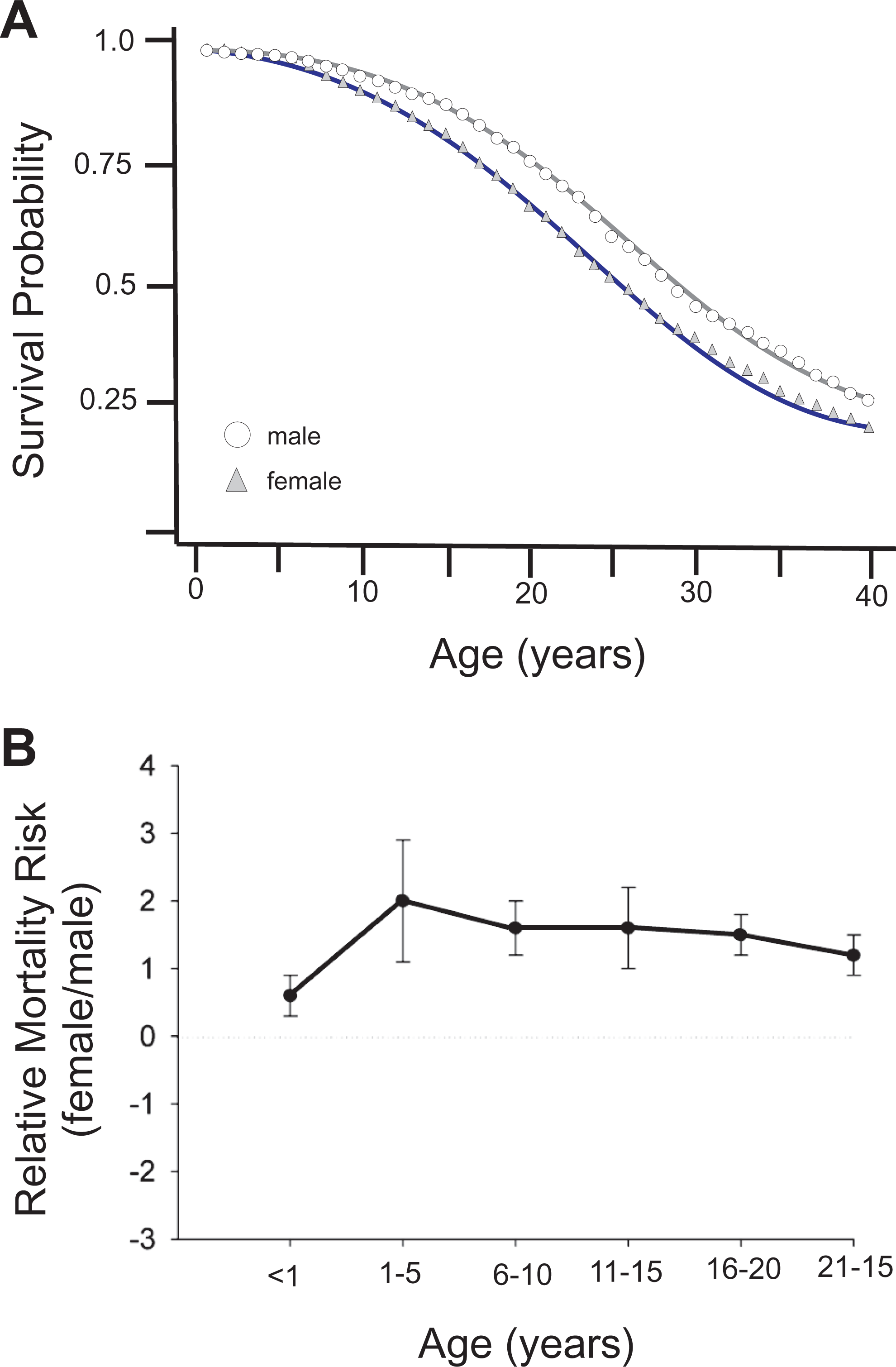
Gender gap in CF. A, Survival of 21 047 CF patients seen at CF foundation accredited CF centers between 1988 and 1992. Curves are derived using the life-table method, with 1-year age interval. Subjects were left-entered according to their age at the entry to the cohort. From Rosenfeld et al, 98 (B) relative risk of death for females compared to males, in 5-year increment. 98 CF indicates cystic fibrosis.
Although a greater mortality is seen for older females with CF compared to males, it is known that there is a higher mortality rate in males compared to females for patients with CF under 1 year of age. 107 Whether this reflects a greater male infant mortality rate also seen in the general population, 108 or a poorer survival rate from lower respiratory viral infections, an important cause of infant mortality in CF, which is also seen in non-CF males, 109 is not known.
Chronic respiratory infections remain the largest contributor to morbidity and mortality in patients with CF, which, in the context of this review, raises the question of whether being female imposes a disadvantage and a predilection toward bronchiectasis and airway destruction. Indeed, it is interesting to note that a wide variety of respiratory diseases also show a gender disparity in terms of prevalence and/or outcomes. For example, idiopathic pulmonary hypertension is more prevalent in women, whereas idiopathic pulmonary fibrosis is more common in males. 110 -112 A gender gap in outcomes is found in asthma and chronic obstructive pulmonary disease, where women generally have poorer outcomes than men. 98,113 -116 Interestingly, postpubertal girls with asthma have more frequent and severe exacerbations than boys, 117 whereas prior to puberty, girls have a lower incidence. 118 Lymphangioleiomyomatosis is a rare cystic lung disease of unknown origin, predominantly affecting females, 119 with very few males ever having been diagnosed with the condition. Even though studies that have failed to identify any gender differences in key clinical outcomes in patients with CF, including lung function, body mass index, and frequency of CF-related diabetes, females were found to have a higher frequency of Burkholderia infection compared to males. 120 Moreover, female patients seemed to require more intensified treatments regarding antibiotics and increased hospitalization days compared to males. Given the wide gender gap, it was hypothesized that the disease may be hormone-mediated, but hormone-based treatments have not yet established a clear benefit. 119
As with lymphangioleiomyomatosis, the gender gap in CF has been hypothesized to be related to hormones, specifically estrogen. 17β-estradiol, the major circulating estrogen in premenopausal women, causes a rapid and reversible inhibition of forskolin-stimulated (cAMP-activated), CFTR-dependent chloride secretion in T84 intestinal epithelial monolayers. 121 Synthetic estrogens and the selective estrogen receptor modulator, tamoxifen, also inhibited chloride secretion in this mode. 17α-estradiol, a stereoisomer that fails to bind and active nuclear receptors, is equally potent, arguing against genomic effects. Indeed, studies have shown a direct binding of estrogens to CFTR 121, although whether this causes channel inhibition through direct pore blocking or allosteric effects is unknown.
Efficient mucociliary clearance in the airways relies on a thin film of liquid (airway surface liquid or ASL) within which the cilia of airway epithelia cells beat, to remove viruses, bacteria, and debris from the lungs. In normal airways, CFTR-mediated chloride secretion, and epithelial sodium channel, ENaC-mediated sodium absorption, work in concert to provide a healthy ion composition and depth of ASL, within which the cilia of airway epithelial cells beat to remove debris and bacteria from the lungs. In CF airways, the physical or functional absence of CFTR (depending on the specific mutation), along with enhanced ENaC activity alters the balance of ion transport in favor of ion and fluid absorption. The airway becomes dehydrated, and mucociliary clearance is impaired. Studies by Fanelli and colleagues revealed that incubation of airway cells with 10 nM estradiol increased the expression of F508del CFTR at the apical membrane, potentially correcting in transport defects. 122 Effects peaked at 6 to 7 hours suggesting a transcriptional effect. Marginal increases in function were seen, although this was evaluated by fluorescence ratios rather than the gold standard of electrophysiological measurements. Moreover, although CFBE41o cells are nominally F508del, they have been stably transfected with a cDNA plasmid expressing F508del, although the transgene contains the TTT trinucleotide deletion rather than the naturally occurring CTT deletion. Whether the endogenous or trans (or both) proteins were affected by estrogen is not known. One could imagine that if estrogens affected the native protein, then females would be better off than males, particularly after puberty, but this appears not to be the case.
The absence of CFTR from the airway surface in patients with CF has led to the suggestion that activation of alternative chloride channels in the airways, such as the calcium-activated chloride channel (CaCC), could be of therapeutic benefit in restoring ion and fluid balance to the airway surface. But CaCC is not without its own issues since its activity is influenced by estrogens and therefore shows differential activity in males and females, especially cycling females. 17β-estradiol decreases purinergic signaling of CaCC, such that calcium-activated chloride secretion is diminished. 123 In this case, the effects of 17β-estradiol are mediated by modifying the signaling cascade, rather than directly on the channel itself. In the context of a female CF airway, this could potentially worsen the degree of airway surface dehydration and exacerbate any pulmonary infection and inflammation. Physiologic elevations in preovulatory estrogen levels may further exacerbate diminished airways surface liquid by preventing calcium-dependent chloride and water transport and leading to reduced mucociliary clearance and enhanced susceptibility to infection. While ENaC- and CFTR-mediated currents were unaffected by the menstrual cycle, CaCC currents (measured by uridine-5'-triphosphate, a pruingergic receptor agonist-mediated purinergic stimulation) varied by 50% in both CF and non-CF patients during the menstrual cycle. It may be that in CF, even a modest decrease in CaCC activity, in an already hyperinflammatory environment, may predispose females to exacerbations in pulmonary problems, but this has not been evaluated.
Estrogen may also impact the ability of the immune system to respond to Pseudomonas infections. Estrogen is shown not only to aggravate inflammatory responses following Pseudomonas exposure but also to suppress innate antibacterial defenses. 124,125 Thus, estrogen inhibits the production of the pro-inflammatory cytokine IL-8 in bronchial epithelial cells, which would hinder neutrophil recruitment and inflammatory responses in women. Moreover, estrogens appear to promote the conversion of Pseudomonas aeruginosa from a drug-sensitive non-mucoid phenotype to a drug-resistant mucoid form. 125,126 However, other publications have shown that it is colonization by Burkholderia cepacia rather than P aeruginosa that seems to affect survival among females more negatively than males, although both colonizers impact overall survival independent of gender. 127
Despite the various effects of sex hormones on ion transport and inflammation, whether such effects translate to the gender gap in CF mortality is still uncertain. Indeed some reports suggest that gender differences in CF may start prior to puberty and that earlier bacterial infections are more common in girls than boys (Table 1). 98 Since this would relate to a time of minimal estrogen expression in females, it is unlikely that estrogens are playing a major role in disease disparity before puberty. Thus, while there is fairly strong evidence of a gender gap in CF outcomes, it is unlikely that it comes down to something as simple as estrogen. The increasing understanding that sex chromosomes express regulators that can modulate expression from autosomes, 128 suggests that it is likely a complex interplay of differential gene expression and signaling that impacts gender differences in CF. How easily such interplay can be unraveled to identify key pharmacological targets is not certain.
Comparison Between Genders of Age-Dependent Pseudomonas Infection.a
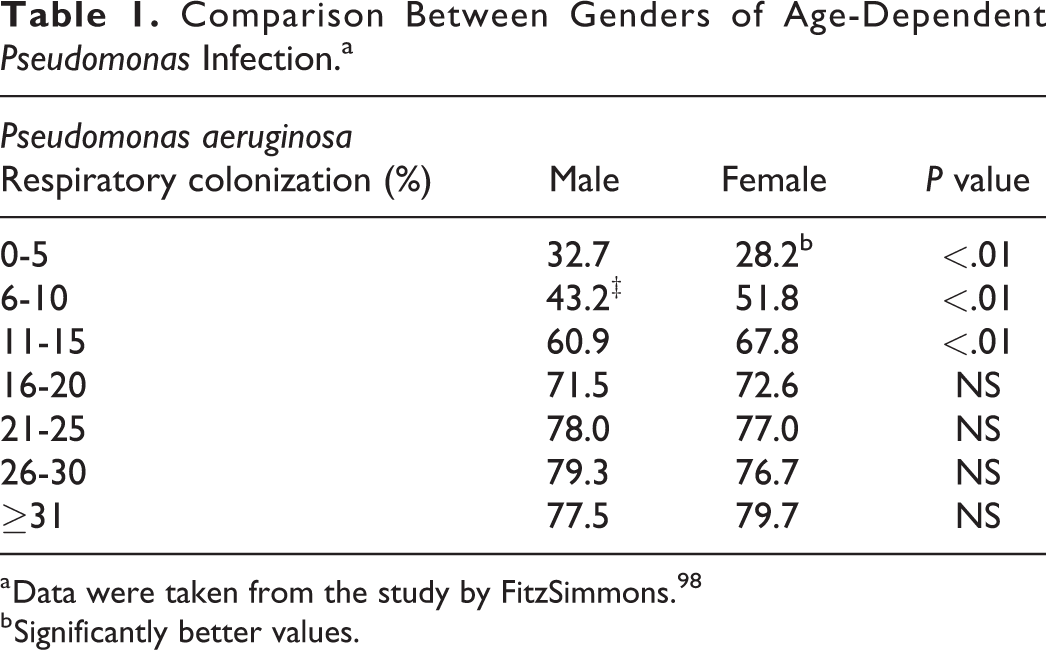
a Data were taken from the study by FitzSimmons. 98
b Significantly better values.
Cystic fibrosis-related bone disease is also a common complication in patients with CF and is characterized by a low bone mineral density (BMD) and increased rates of fracture compared to non-CF individuals. 129 Steady-state BMD is maintained by the relative activities of osteoclasts and osteoblasts. In recent studies using cftr knock-out mice, no differences were observed between male and female bone-derived osteoclasts. 130 However, a significant impairment in osteoblast differentiation and function was observed for female mice compared to males. Although a mechanistic basis for such observations is not fully elucidated, it nonetheless is consistent with the observations that low BMD is more prevalent in females with CF compared to males. 131
The effect of gender on CF pharmacophores
The GOAL (G551D Observational) study enrolled patients of greater than 6 years of age with at least 1 G551D allele and who were beginning treatment with ivacaftor. 66 Participants were 46% female, 54% male, with mean age of 21.1. In a retrospective analysis of the data, Secunda and colleagues made 2 interesting observations. 130 Firstly, ivacaftor-treated females had a greater reduction in pulmonary exacerbations compared to their male counterparts. This may in part be explained by the higher number of baseline exacerbations in females compared to males. Secondly, females older than 18 years had a greater reduction in sweat chloride in response to ivacaftor than males (55.2 mEq/L for females vs 44.1 mEq/L for males). In contrast, females younger than 18 years had a similar decrease in sweat chloride relative to males (55.8 mEq/L for females vs 53.9 mEq/L for males).
Future Perspective
Estrogen has been shown to modulate drug pharmacokinetics, 132 with differences in drug metabolism likely contributing to drug efficacy. 128 For example, both ivacaftor (VX-770) and lumacaftor (VX-809) are potent inducers of the cytochrome P450 system, 133,134 and since there are well-characterized gender differences in the expression of P450 enzymes, 135 it is not unreasonable to suppose that the half-lives of drugs in patients may vary with gender. For example, Ambien, zolpidem, is metabolized by the p450 system much slower in females than males and therefore has a longer half-life. 136 However, such differences have not yet been reported for CFTR pharmacotherapy. Whether this is because no difference exists or the data have not been evaluated with respect to gender is not known. Certainly, 1 direction for manipulating endogenous estrogen concentrations, to reduce their potentially detrimental effects, is through the use of the antiestrogen agent tamoxifen or the oral contraceptive pill; each approach achieving its goal through different mechanisms. Tamoxifen in vitro reverses the impact of estrogens on diminishing ASL levels, returning them to pre-estrogen levels. 123 As more females with CF reach child-bearing age, more patients are taking oral contraceptives. 137,138 Five-year follow-up studies have shown a decline in lung function of around 1% to 2% predicted FEV1 per year, consistent with other investigations. Moreover, the decline in lung function in females taking oral contraceptives was not different from those not taking oral contraceptives, 139 indicating that such medication did not affect disease severity.
A potential alternative non-CFTR therapeutic in CF are microRNAs. 140,141 MicroRNAs are small noncoding, 21 to 23 nucleotide RNAs capable of suppressing the expression of target genes. Numerous microRNAs are differentially expressed CF females compared to males. 142 For example, miR-885-5p is significantly increased in female compared to males with CF, and miR-885-5p has a number of validated targets, many of which are associated with the Rho GTPase, RAC1. Rho GTPases impact CFTR trafficking and expression 141,143 and hepatocyte growth factor along with RAC1 enhances the rescue of F508del CFTR by pharmacological correctors. 142 Indeed, preclinical studies have shown that 15-day treatment of airway monolayers significantly improves the functional rescue of F508del CFTR by and ivacaftor/lumacaftor combination. 142 The observation that miR-885-5p is elevated in females may therefore have implications for not only a gender gap in patients with CF but also for the sensitivity to current and future therapeutic compounds between males and females.
Despite huge strides in the treatment of patients with CF leading to greater life spans, the question of gender disparity is still not clearly delineated. Certainly below 1 year of age for males, and early postpuberty for females, appear to be important time points in overall survival. The GOAL study suggests that there are differences in response to CFTR drugs in females compared to males. This was a retrospective study and not one a priori designed to evaluate gender in drug responses. While the FDA does not require gender data, it is not likely that drug companies will go out of their way to perform such evaluations. Moreover, it is only recently that drugs to treat the basic defect in CF have been available, and their long-term effectiveness and gender disparity (if any) will have to wait for several years. Data on any gender disparities with respect to patients taking the triple corrector/potentiator Trikafta will not be available for a while. Whether different dosing parameters should be considered for males and females is an issue that may be worth investigating. What is clear is that both symptomatic- and gene-specific therapies have dramatically increased the life span and quality of life for patients with CF. Getting to the point where most patients with CF are seen in adult clinics rather than by pediatricians is a remarkable achievement itself. Targeting those few mutations not currently covered by pharmacophores (ie, premature stop, splice variants) is currently a major area of intensive investigation. Certainly, further studies are required to determine whether CFTR modulation will lead to long-term equivalency in females compared to males, so that both males and female patients with cystic fibrosis can lead long healthy, happy and productive lives.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
