Abstract
Background:
Data on sex differences in heart failure (HF) with reference to left ventricular ejection fraction (LVEF) are limited.
Methods and Materials:
We examined 4683 consecutive patients (mean 69 years) with HF in the CHART-2 study.
Results:
Compared to men (N = 3188), women with HF (N = 1495) were older and had a lower prevalence of ischemic heart disease and cancer, received less implementation of evidence-based treatment, and were characterized by more severe HF in terms of higher New York Heart Association (NYHA) functional class and increased brain natriuretic peptide (BNP) levels, despite greater preservation of LVEF. During the median 6.3-year follow-up, all-cause mortality was comparable between women and men (32.8% vs 33.2%, P = .816), while women had higher cardiovascular mortality, particularly among those with LVEF ≥50%. Although no sex differences existed in cause of death among patients with LVEF ≤ 40% and 41% to 49%, women had a higher proportion of cardiovascular death and lower proportion of noncardiovascular death than men among those with LVEF ≥ 50%. Multivariable Cox regression models showed that women with HF had reduced risk of both cardiovascular and noncardiovascular death, regardless of LVEF category. Beta-blockers were associated with improved mortality in women but not men with LVEF ≤ 40%, while renin–angiotensin system inhibitors were not associated with improved mortality in women with LVEF ≥ 50% but were in men.
Conclusion:
In addition to sex-specific differences in the age of onset, etiology and response to treatment, women with heart failure and preserved left ventricular ejection fraction (LVEF ≥ 50%) have higher cardiovascular mortality than men. Sex-related management of congestive heart failure should include a consideration of LVEF.
Introduction
Substantial sex differences in the clinical features of heart failure (HF) have been reported, specifically with regard to clinical characteristics, etiology, treatment, and outcome. 1 –3 However, these sex differences in HF should be reevaluated in the current era, since the overall picture of HF has recently changed, particularly regarding an increase of HF in women 4 –8 and HF with preserved left ventricular ejection fraction, LVEF (HFpEF). 7,9 –12 Roger reported a greater increase of HF incidence in women than men from 1979 to 2000 among 4537 cases of new-onset HF in Olmsted County, Minnesota (8% vs 3%), 2 while Huffman et al reported that women have recently shown similar or in fact increased lifetime risk of HF compared to men (30%-42% in white males vs 32%-39% in white females, and 20%-29% in black males vs 24%-46% in black females). 8
Another emerging change in HF structure affecting sex differences in this condition is an increase in HFpEF. 7,11 –14 This is now recognized as a new entity of HF. 13,14 Since women at least have a generally higher prevalence of HFpEF than men, 15 –17 a proper addressing of HF in the present era warrants investigation of potential interaction in women having HF with LVEF. To date, however, data on women with HF or on sex differences in HF are limited, particularly with reference to LVEF.
In this study, we aimed to elucidate the characteristics of HF in women in an aged society, with special reference to LVEF.
Methods and Materials
The Chronic Heart Failure Analysis and Registry in the Tohoku District-2 Study
The Chronic Heart Failure Analysis and Registry in the Tohoku District-2 (CHART-2) study is a multicenter, prospective observational study that enrolled 10 219 patients older than 20 years with significant coronary artery disease (stage A) or in stages B to D between October 2006 and March 2010. 11,17 –22 Heart failure in the CHART-2 study was diagnosed by attending physicians based on the Framingham criteria. 23 Staging was conducted at the time of registration according to the American college of Cardiology (ACC)/American Heart Association (AHA) guidelines classification, namely, stage A at high risk of HF but without structural heart disease or symptoms of HF; stage B with asymptomatic cardiac structural and/or functional abnormalities; stage C with HF symptoms; and stage D with severe HF. 24 All information, including clinical characteristics, medical history, laboratory data, and echocardiography data were recorded at the time of enrollment and thereafter annually by trained physicians and clinical research coordinators. The CHART-2 study was approved by the local ethics committee in each participating hospital, and informed consent was obtained from all patients.
Study Design
The diagram for the present study is shown in Figure 1. Among the 10 219 patients registered in the CHART-2 study, we enrolled 4683 consecutive stage C/D patients with HF having baseline echocardiographic data. In accordance with the ACC/AHA guideline classification, 24 we divided them into 3 groups, namely, patients with HF with preserved LVEF (HFpEF, LVEF ≥ 50%, n = 3193), borderline HFpEF (LVEF 41% to 50%, n = 709), and HF with reduced LVEF (HFrEF, LVEF ≤ 40%, n = 781). When prior myocardial infarction or coronary artery disease was present, the main etiology of HF was determined to be ischemic heart disease (IHD). Those without IHD but with a previous diagnosis of dilated cardiomyopathy (DCM), hypertrophic cardiomyopathy (HCM), and valvular heart disease (VHD) were then classified as having DCM, HCM, or VHD, respectively. Valvular heart disease was specifically defined as severe aortic or mitral valvular disease by echocardiography. Hypertensive heart disease (HHD) was diagnosed when a patient did not have IHD, DCM, HCM, or VHD but did have a history of hypertension. If a patient was classified as not having IHD, DCM, HCM, VHD, or HHD, the HF etiology was classified as “others.” Using the registry data of these patients, we examined sex differences in clinical characteristics, management, prognosis, prognostic factors, and cause of death in patients with stage C/D HF, with special reference to sex and HF categories. Primary endpoint was all-cause death, and the secondary endpoints were cardiovascular death, noncardiovascular death, and admission for HF.
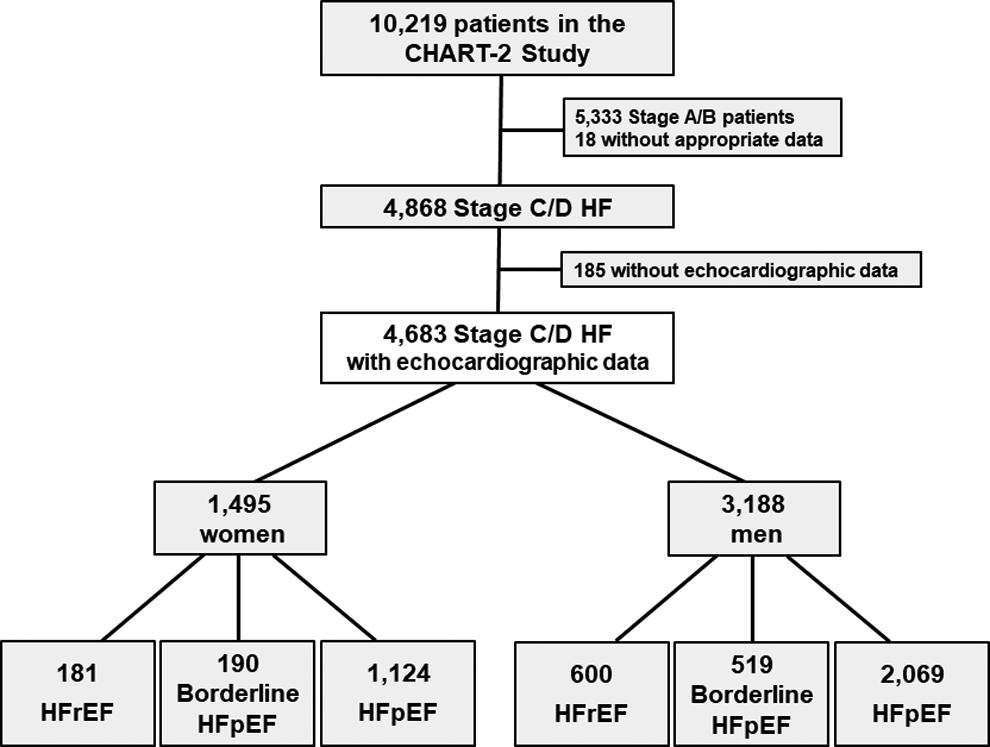
The consort diagram of the present study.
Statistical Analysis
All continuous variables are shown as mean (standard deviation [SD]). Continuous clinical characteristics were compared by Welch’s t test or Wilcoxon rank-sum test for 2 groups, and analysis of variance or the Kruskal-Wallis rank sum test for 3 groups. All discrete variables are shown as frequency (percentage). Discrete characteristics were compared by Fisher’s exact test. Kaplan-Meier curves and log-rank tests were used to estimate and compare sex differences in the occurrence of primary endpoint. Incidence rates per 1000 person-year for primary and secondary endpoints were compared with the Mid-p exact test. Determinants of each endpoint were examined by univariable Cox proportional hazard models and multivariable Cox proportional hazard models with stepwise variable selection. All potential confounding factors were included in the univariable Cox regression; factors for the multivariable model were then selected from the set of covariates with P < .2 in the univariable analysis using stepwise backward elimination procedure. For the overall cohort analysis, the optimal set of covariates was selected from all samples, and the same set of the variables were applied to temporal change and/or LVEF category analysis except to the drug response analysis, in which the optimal set of covariates was selected each time for every LVEF category. The covariates included in the multivariable analysis were age, sex, body mass index, systolic blood pressure, diastolic blood pressure, heart rate, left ventricular diastolic dimension, LVEF, levels of albumin, blood urea nitrogen, BNP, creatinine, hemoglobin, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol and triglyceride, previous or current smoking, HF etiologies (IHD, HT, DCM, HCM, and VHD) and comorbidities (amyloidosis, adult congenital heart disease, atrial fibrillation, cancer, diabetes mellitus, hypertension, hyperuricemia, myocardial infarction, sarcoidosis, and stroke), history of prior admission for HF, coronary artery bypass graft, cardiac resynchronization therapy, implantable cardioverter defibrillator, pacemaker implantation, and percutaneous coronary intervention and medications at baseline (antiplatelet, aldosterone antagonist, beta-blocker, calcium channel blocker, diuretic, renin–angiotensin system [RAS] inhibitor, statin, and warfarin). All statistical analyses were performed using the open-source statistics computing software R version 3.4.2., 25 with a P value of <.05 and a P value for interaction <.1 considered to indicate statistical significance in the present study.
Results
Baseline Characteristics
Baseline characteristics of overall women and men are shown in Table 1. Among the 4683 stage C/D patients with HF, 1495 (32%) were women, aged 3.9 years older than men. Compared to men, women with HF were characterized by a lower body mass index, and lower prevalence of diabetes, smoking history, myocardial infarction, and cancer as well as by a higher prevalence of atrial fibrillation and history of HF hospitalization. Although women with HF had more preserved LVEF, they had relatively severe HF manifestations compared to men, characterized by a higher heart rate, higher New York Heart Association (NYHA) class, and increased BNP levels. Women with HF were less likely to undergo percutaneous coronary intervention or coronary artery bypass graft surgery. Furthermore, women were less frequently treated with an implantable cardioverter–defibrillator and/or cardiac resynchronization therapy, although they were more frequently treated with other cardiac pacemakers. Regarding HF treatment at baseline, women were less frequently treated with beta-blockers, RAS inhibitors consisting of angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARB), and statins but were more frequently treated with aldosterone antagonists and diuretics in the overall population.
Baseline Characteristics.a
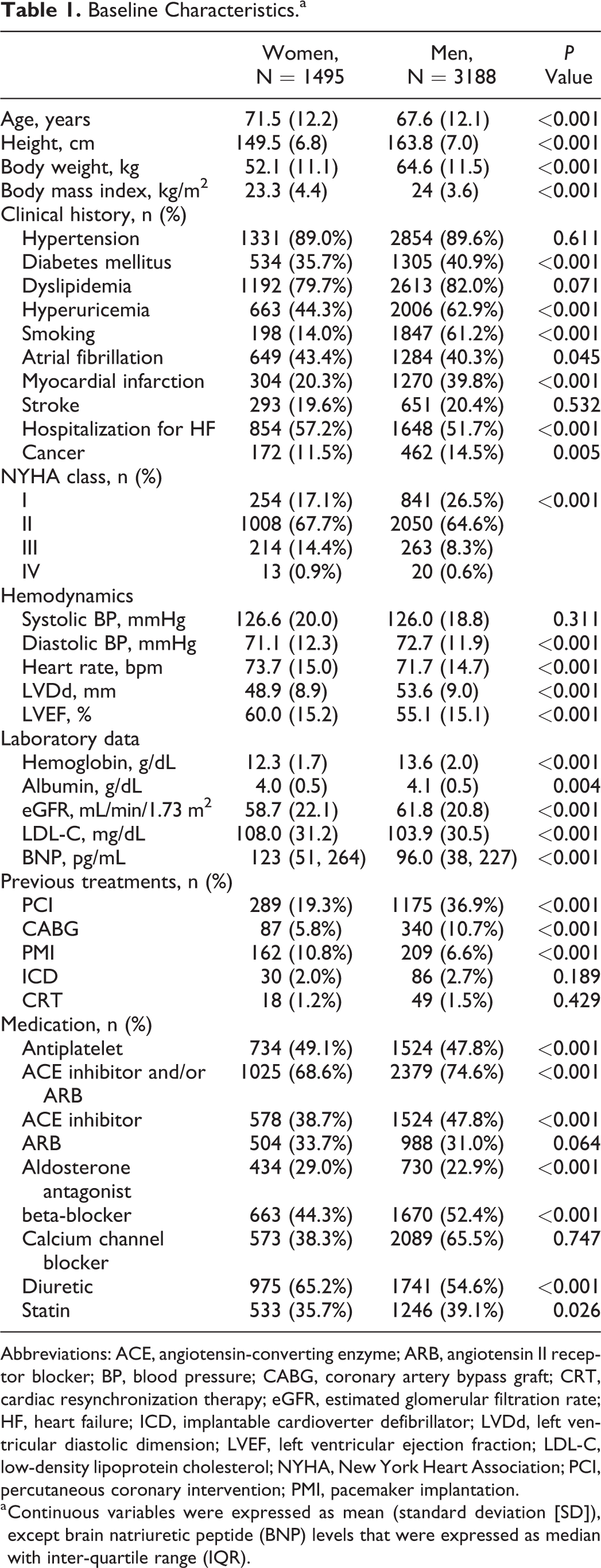
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor blocker; BP, blood pressure; CABG, coronary artery bypass graft; CRT, cardiac resynchronization therapy; eGFR, estimated glomerular filtration rate; HF, heart failure; ICD, implantable cardioverter defibrillator; LVDd, left ventricular diastolic dimension; LVEF, left ventricular ejection fraction; LDL-C, low-density lipoprotein cholesterol; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; PMI, pacemaker implantation.
a Continuous variables were expressed as mean (standard deviation [SD]), except brain natriuretic peptide (BNP) levels that were expressed as median with inter-quartile range (IQR).
Sex differences in baseline characteristics of patients with HF were also found when the groups were divided by LVEF category (Table 2). From HFrEF to borderline HFpEF, and then to HFpEF, mean age and the prevalence of hypertension and cancer significantly increased in men but remained unchanged in women, whereas the prevalence of atrial fibrillation increased in women but not in men. As for treatment, it was noted that the use of ARB was increased from HFrEF to borderline HFpEF and then to HFpEF in men but did not differ in women. Comparison of clinical background in each LVEF category indicated that sex differences were particularly noted in the HFpEF population but were not particularly evident in the HFrEF population. Notably, sex differences in NYHA classes and BNP levels were noted in the HFpEF and the borderline HFpEF groups but were no longer observed in the HFrEF population. In contrast, with regard to medication, sex differences were noted in the HFrEF group as well as in the HFpEF and borderline HFpEF groups. For example, even in the HFrEF group, women still had a tendency to be treated less frequently with β-blockers and had a significantly decreased prescription rate of ACEI compared to men.
Baseline Characteristics By Sex And HF Categories.a
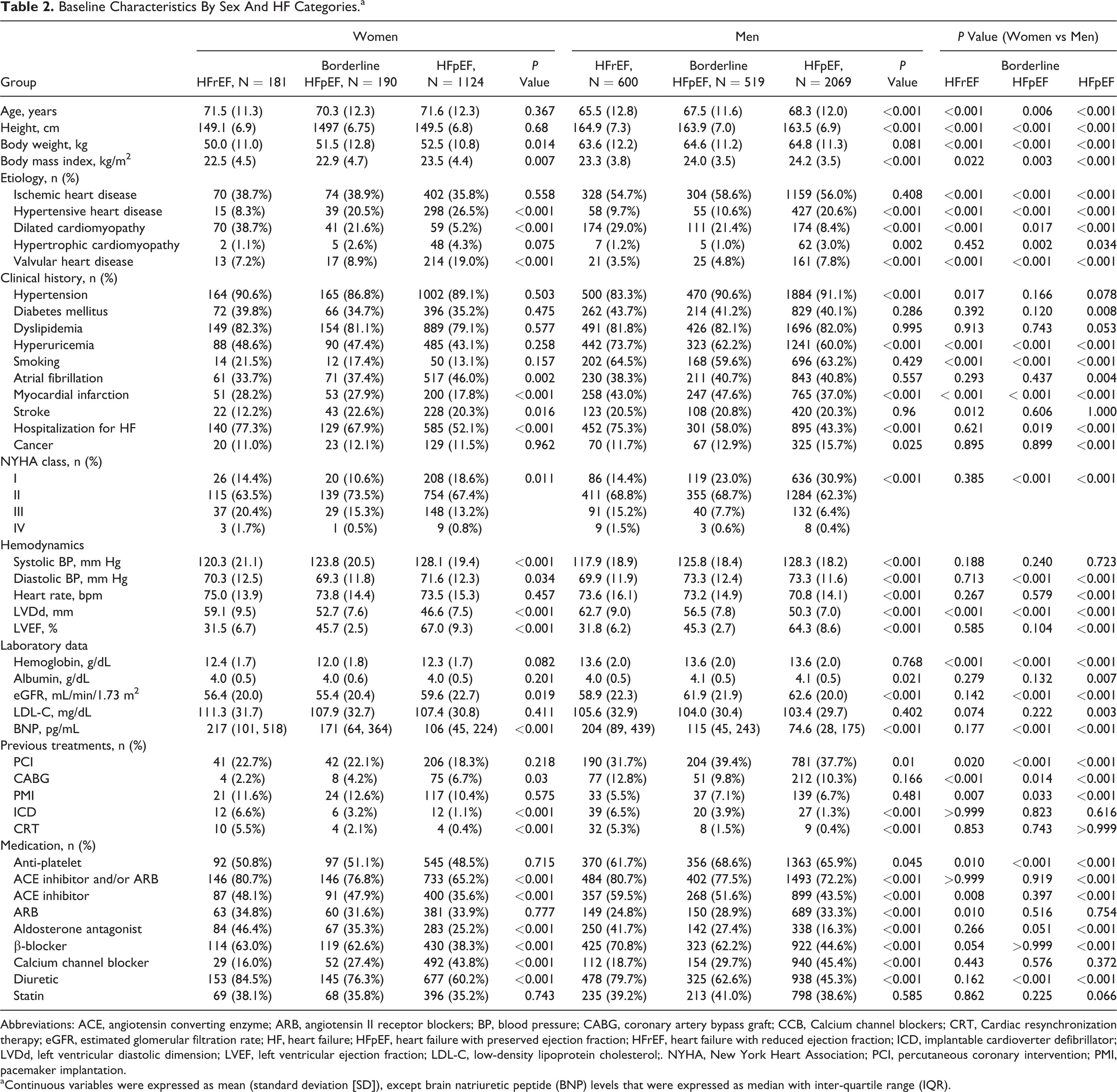
Abbreviations: ACE, angiotensin converting enzyme; ARB, angiotensin II receptor blockers; BP, blood pressure; CABG, coronary artery bypass graft; CCB, Calcium channel blockers; CRT, Cardiac resynchronization therapy; eGFR, estimated glomerular filtration rate; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; ICD, implantable cardioverter defibrillator; LVDd, left ventricular diastolic dimension; LVEF, left ventricular ejection fraction; LDL-C, low-density lipoprotein cholesterol;. NYHA, New York Heart Association; PCI, percutaneous coronary intervention; PMI, pacemaker implantation.
a Continuous variables were expressed as mean (standard deviation [SD]), except brain natriuretic peptide (BNP) levels that were expressed as median with inter-quartile range (IQR).
Heart Failure Etiologies
The most frequently observed HF etiology was IHD (49.9%), followed by HHD (19.0%) in both sexes (Table 1). However, the proportion of IHD was lower in women than in men (36.5% vs 56.2%, P < .001) whereas that of HHD was higher in women than in men (23.5% vs 16.9%, P < .001). Following IHD and HHD, VHD and DCM were the third and fourth etiologies, respectively, in women, while DCM was the third and VHD was the fourth in men. While the proportion of IHD was the most frequent and comparable among the HFpEF, borderline HFpEF, and HFrEF groups regardless of sex, the proportion of DCM was as high as that of IHD in women with HFrEF while not so high in men with HFrEF (Table 2). In both sexes, the proportions of HHD and VHD were most increased in the HFpEF group whereas that of DCM was most frequently increased in the HFrEF group.
Long-Term Outcomes
There were 1550 deaths (491 in women (32.8%) vs 1059 in men (33.2%), P = .816) during the median follow-up of 6.3 years. Kaplan-Meier curves indicated that women with HF had comparable all-cause mortality to men but had an increased incidence of cardiovascular death and admission for HF and a tendency toward decreased noncardiovascular mortality (Figure 2). When divided by LVEF category, the increased incidence of cardiovascular death and HF admission in women was statistically significant in the HFpEF group but not in the HFrEF or borderline HFpEF groups (Figure 3). The incidence of cardiovascular death and HF admission was decreased in the HFrEF and borderline HFpEF groups and then to the HFpEF in both sexes, with the exception of a lack of difference in cardiovascular death between the borderline HFpEF and the HFpEF in women. In contrast, the incidence of noncardiovascular death did not differ by LVEF category in either sex (Figure 4).
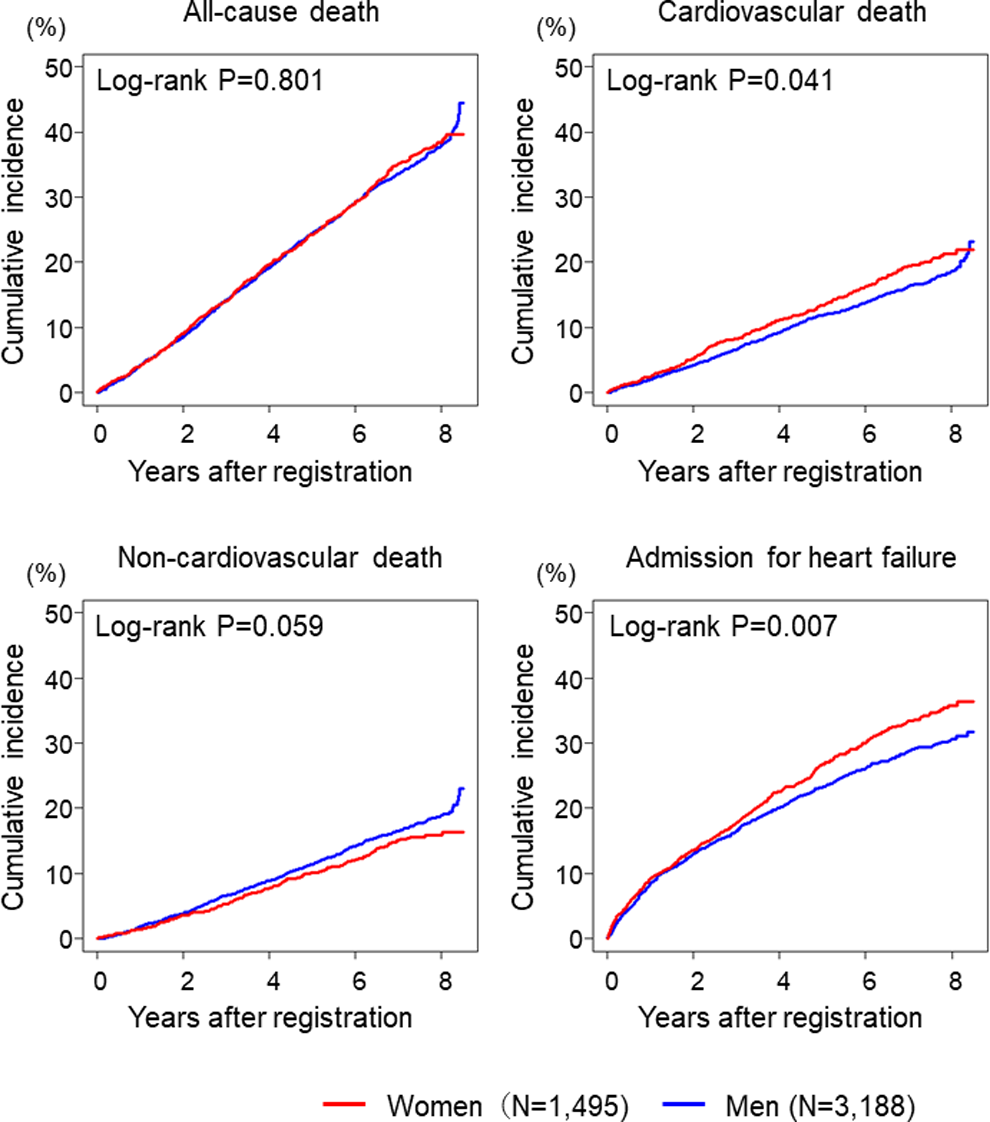
Kaplan-Meier estimates for clinical outcomes.
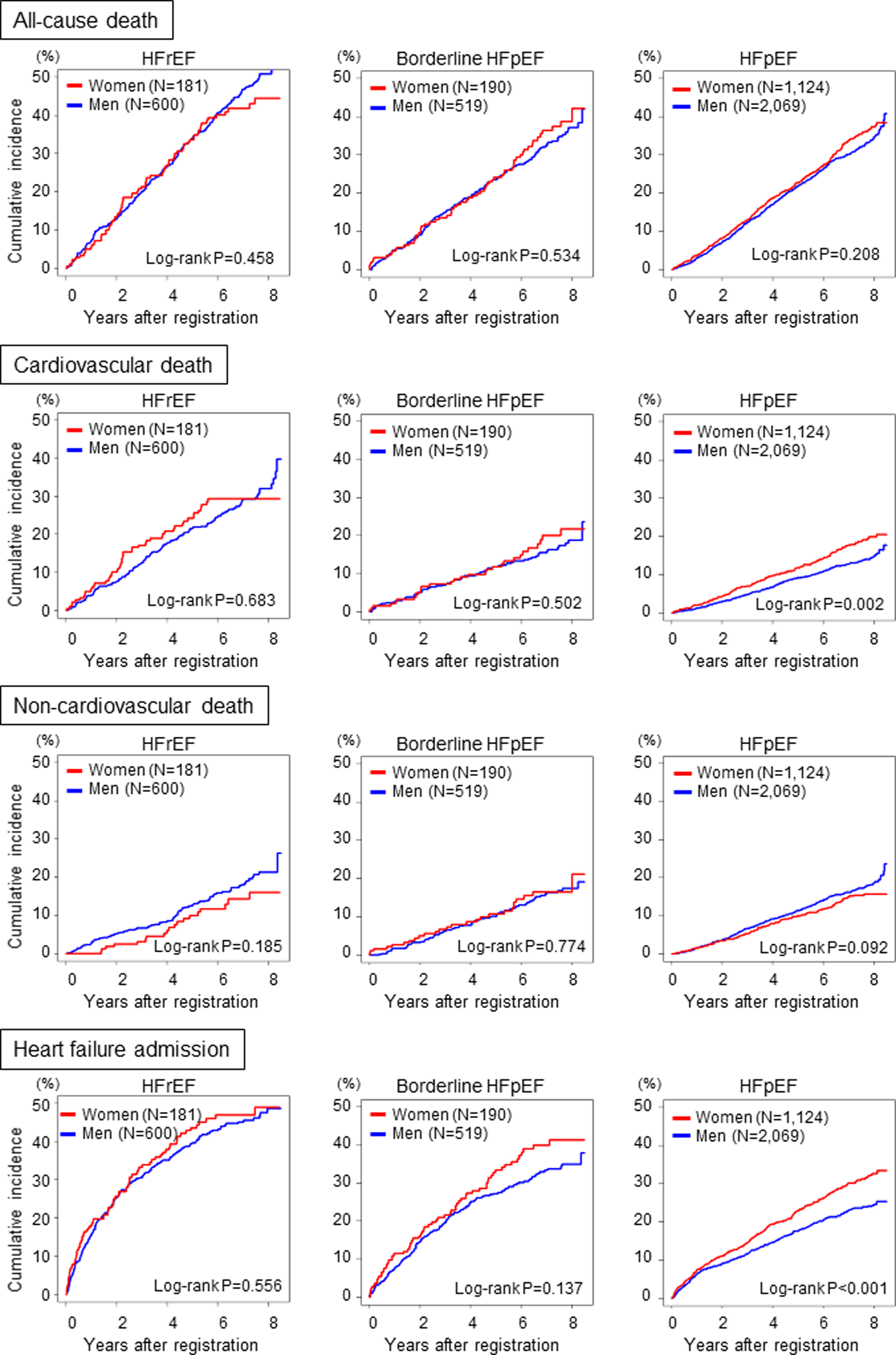
Kaplan-Meier estimates for clinical outcomes by LVEF category. LVEF denotes left ventricular ejection fraction.
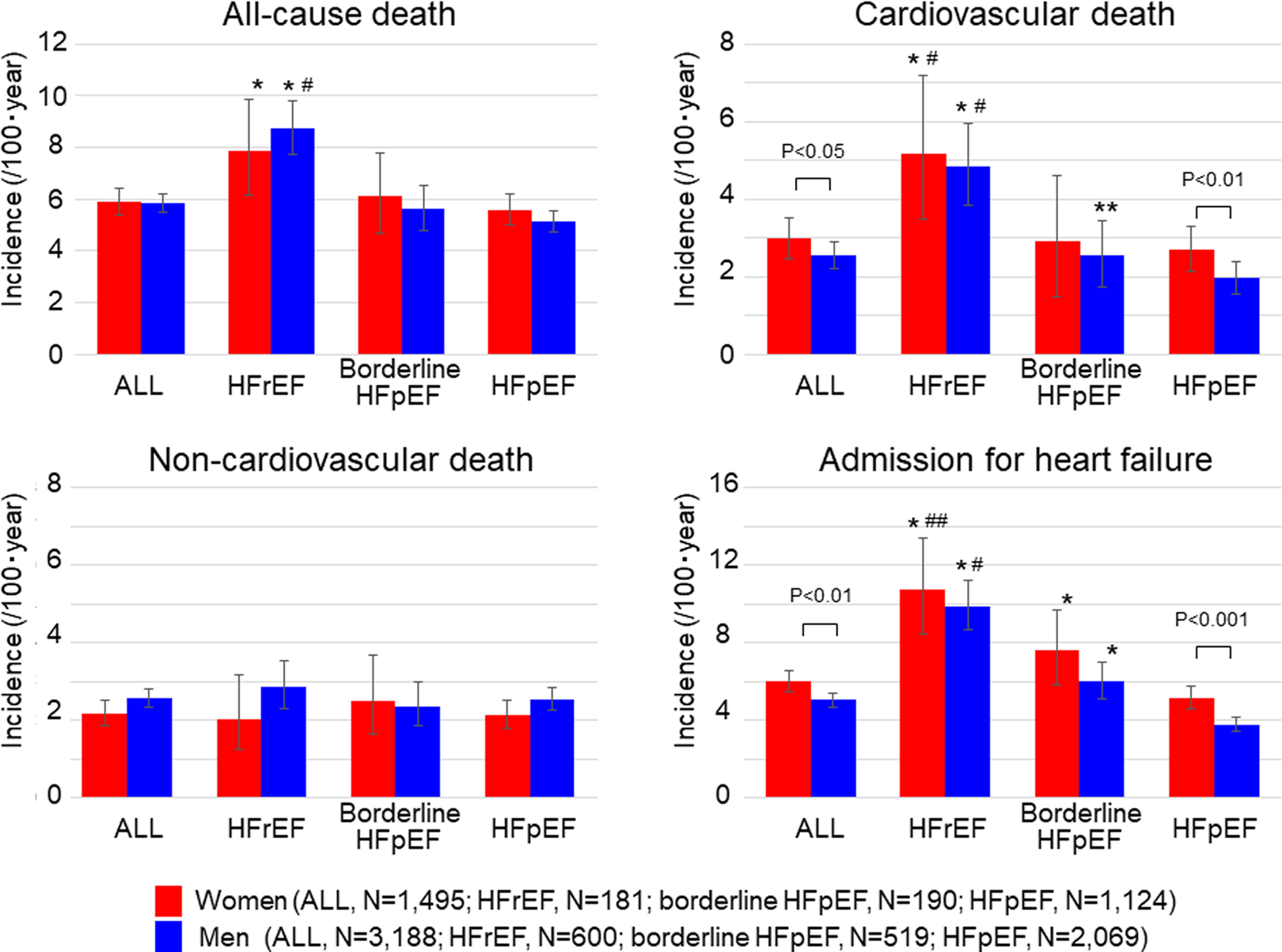
Incidence of clinical outcomes by HF categories. *P < .01 versus HFpEF; **P < .05 versus HFpEF; # P < .01 versus borderline HFpEF; ## P < .05 versus borderline HFpEF. HF denotes heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.
Cause of Death
Of 1550 deaths during the median follow-up of 6.3 years, 713 (46.0%), 647 (41.7%), and 190 (12.3%) were due to cardiovascular, noncardiovascular, and unknown causes, respectively. Table 3 shows sex differences in the cause of death. In the overall population, when compared to men, women had a higher incidence of deaths due to HF (9.4% vs 7.0%, P = .004) and a lower incidence of deaths due to noncardiovascular origins (12.1% vs 14.6%, P = .021), which was likely attributable to deaths due to cancer (3.3% vs 5.9%, P < .001) and infectious pneumonia (1.5% vs 2.3%, P = .075). Subgroup analysis by LVEF category indicated that these sex differences in the cause of death were statistically evident only in the HFpEF population (death due to HF, 8.6% vs 5.2%, P < .001; death due to cancer, 3.6% vs 5.9%, P = .005; and death due to infectious pneumonia, 1.2% vs 2.3%, P = .021).
Cause of Death by Sex and HF Categories.
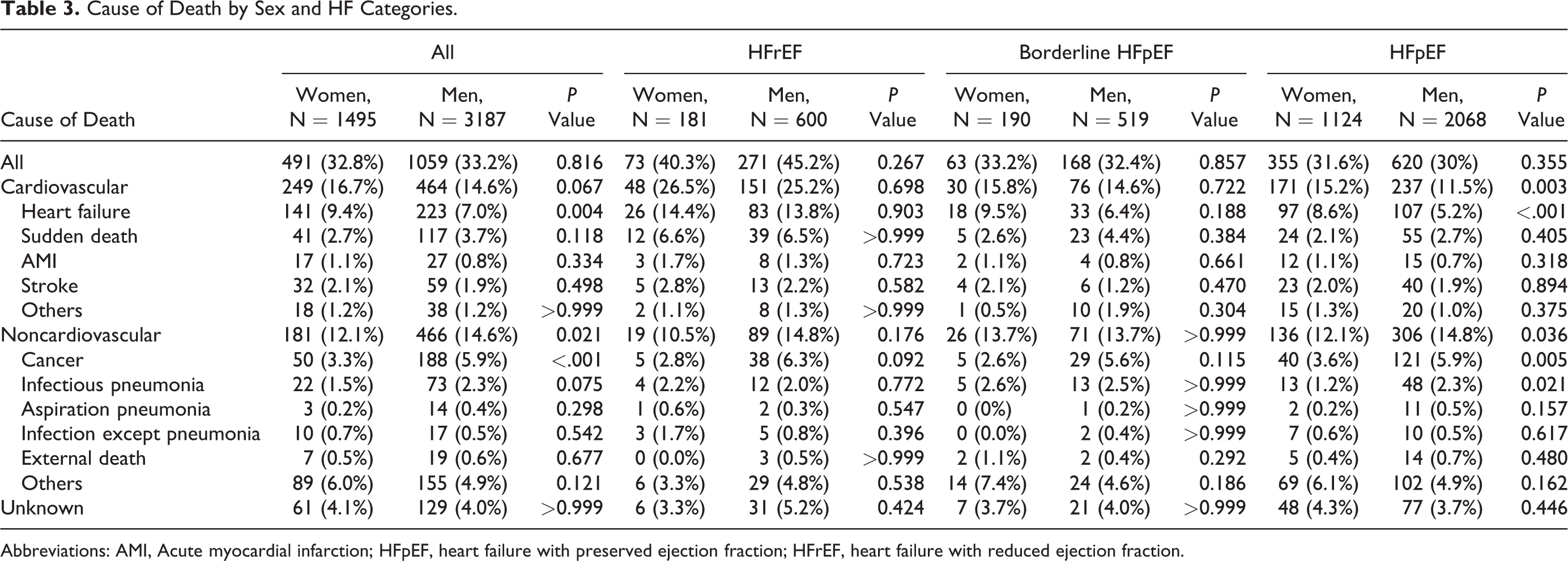
Abbreviations: AMI, Acute myocardial infarction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.
Prognostic Impact of Female Sex
Multivariable Cox proportional hazard models indicated that female sex was associated with decreased all-cause mortality (adjusted hazard ratio (HR) 0.743; 95% confidence interval (95% CI): 0.650-0.849, P < .001), which was most attributable to a significant decrease in noncardiovascular mortality (adjusted HR 0.582; 95% CI: 0.482-0.703, P < .001) and modestly attributable to cardiovascular mortality (adjusted HR 0.817; 95% CI: 0.688-0.970, P = .021; Table 4). Women had comparable adjusted risk for HF admission (adjusted HR 0.917; 95% CI: 0.806 -1.044, P = .191). Furthermore, no statistically significant interaction was seen between female sex and LVEF category in all-cause death. There were also no significant interactions between female sex and LVEF category in cardiovascular death, noncardiovascular death, or HF admission. Figure 5A shows temporal changes in the prognostic impact of female sex on primary and secondary end points during the observational period. When compared to men, women tended to have reduced risk of all-cause mortality at 1-year and had statistically significant reduced risk after 1-year follow-up. It was noted that women did not have a reduced risk of cardiovascular death at 1- and 3-year follow-up but then had a significantly reduced risk at the 5-year and overall follow-up. Reduced risk for noncardiovascular death was observed in women compared to men throughout the period, while risk of HF admission was comparable between the sexes throughout the follow-up. Figure 5A shows temporal changes in sex differences in prognostic impacts of BNP level during the follow-up period. In each end point, the prognostic impact of BNP level was decreased across the follow-up in women but not in men. As a result, interaction between female sex and prognostic impact of BNP level decreased across the time. These observations in Figure 5 did not differ by LVEF category.
Prognostic Impact of Female Sex by HF Categories.a
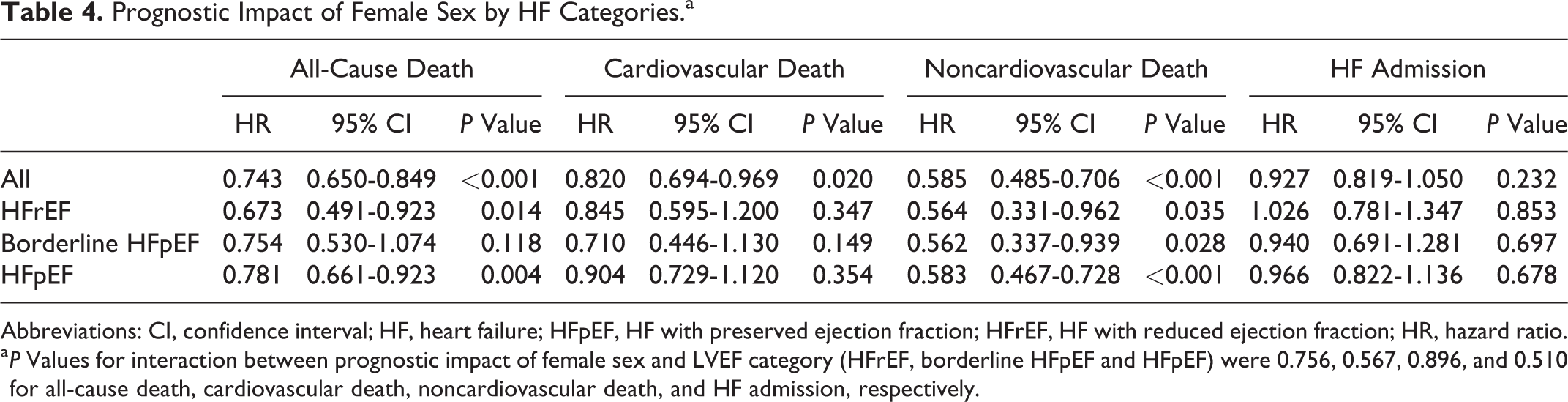
Abbreviations: CI, confidence interval; HF, heart failure; HFpEF, HF with preserved ejection fraction; HFrEF, HF with reduced ejection fraction; HR, hazard ratio.
a P Values for interaction between prognostic impact of female sex and LVEF category (HFrEF, borderline HFpEF and HFpEF) were 0.756, 0.567, 0.896, and 0.510 for all-cause death, cardiovascular death, noncardiovascular death, and HF admission, respectively.
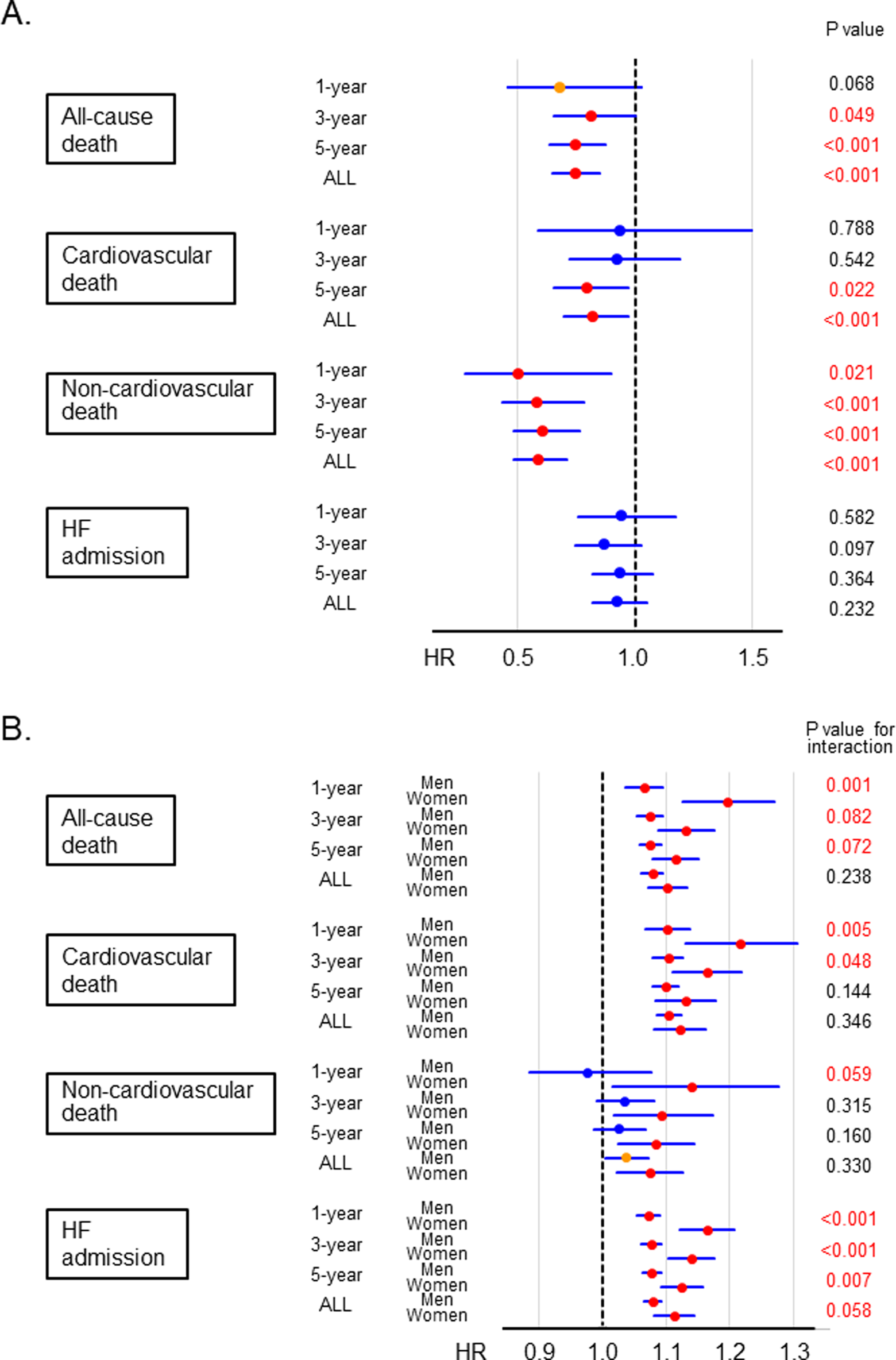
Temporal changes in (A) prognostic impact of female sex and (B) sex differences in prognostic impact of BNP level during the observational period. BNP denotes brain natriuretic peptide; HR, hazard ratio.
Sex Differences in Prognostic Factors and Drug Responses
Table 5 shows sex differences in the prognostic impact of each factor indicated in the stepwise selection of multivariable Cox proportional hazard models for all-cause death. No significant interaction between sex and each factor was indicated for all-cause death in the overall cohort. Subgroup analysis also indicated that impact of each factor on all-cause death was not influenced by sex in any LVEF category. Figure 6 compares the prognostic impact of medications between the sexes. In the overall population, β-blockers and RAS inhibitors were associated with improved all-cause mortality. Notably, although there was no significant interaction with sex on the prognostic impact of β-blockers in the overall population, sex difference was noted for the use of beta-blockers in the HFrEF group (adjusted HR 0.457; 95% CI: 0.235-0.888, P = .021 in women, vs adjusted HR 0.874; 95% CI: 0.639-1.196, P = .400 in men, P for interaction = .042) while not in the borderline HFpEF or HFpEF groups. In contrast, with the use of RAS inhibitors, sex difference was observed in the overall patients (P for interaction = .045), which was most evident in the HFpEF group (adjusted HR 0.962; 95% CI: 0.721-1.284, P = .794 in women, vs adjusted HR 0.701; 95% CI: 0.566-0.869, P = .001 in men, P for interaction = .053).
Prognostic Factors by Sex.
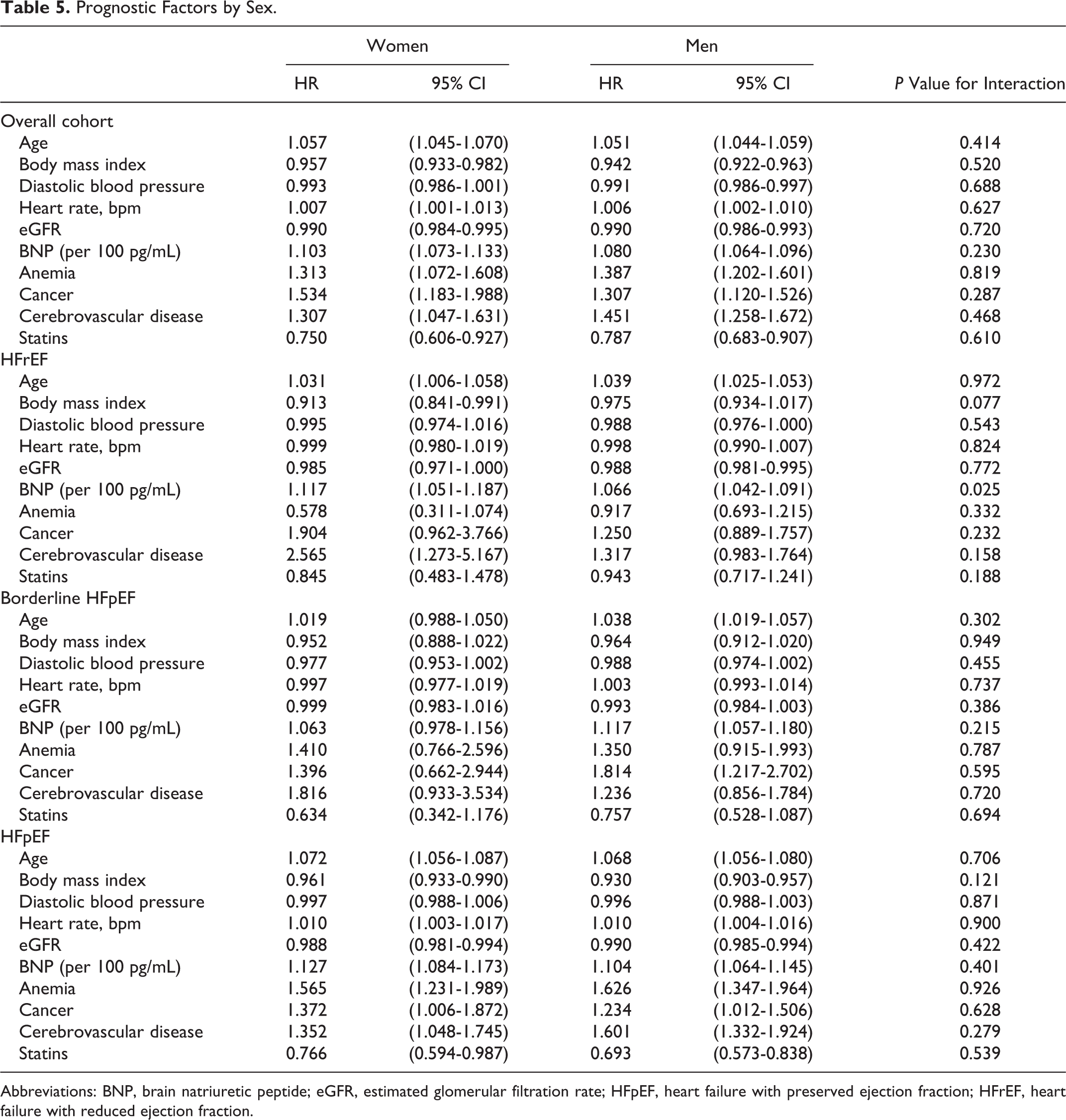
Abbreviations: BNP, brain natriuretic peptide; eGFR, estimated glomerular filtration rate; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.
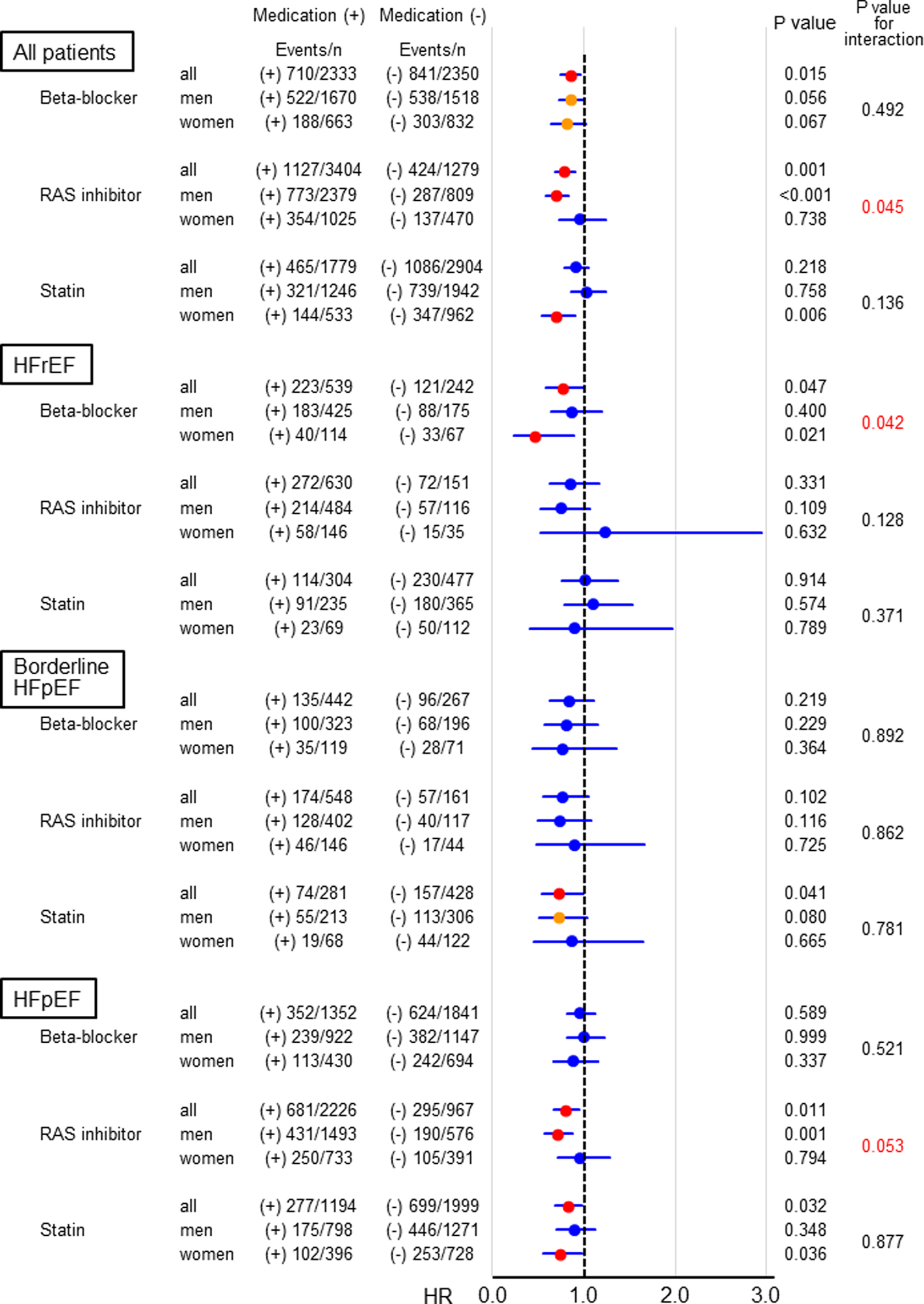
Prognostic impact of medication and its interaction with sex. HFpEF denotes heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; HR, hazard ratio; RAS, renin–angiotensin system.
Discussion
Using the database of the CHART-2 study, a large-scale observational study for HF in Japan, the present study demonstrated substantial sex differences in HF in the current era. When compared to men, women with HF had distinct clinical characteristics in terms of age, etiology, comorbidity, management, drug response, cause of death, and outcomes, which differed by LVEF category. In particular, we noted that women with HF had more severe HF and more increased cardiovascular mortality than men, despite their reduced cardiovascular risk after adjustment by clinical background compared to men. To our knowledge, this is the first study to comprehensively elucidate sex differences in HF with reference to LVEF and provides insights into future directions for sex-specific HF management in the contemporary era.
Sex Differences in Clinical Characteristics and Adherence to Clinical Practice Guidelines
First, the present study demonstrated sex differences in the clinical characteristics of 4683 consecutive patients with stage C/D HF enrolled from the CHART-2 study, one of the largest scale prospective observational studies for HF in the world (N = 10 219). Women in the present study were characterized by older age, more preserved LVEF, lower prevalence of IHD, and higher prevalence of VHD. These findings are consistent with previous reports from Western countries 26 –28 and our preliminary report. 18 The present study revealed that the clinical manifestations of HF appeared to be more severe in women than in men, since women had more advanced NYHA functional class and elevated serum BNP level despite more preserved LVEF. The insufficient implementation of evidence-based medication (beta-blockers, RAS inhibitors, and statins) in women compared to men could be explained, at least in part, by these sex differences in the present study, particularly by more preserved LVEF and lower prevalence of IHD in women, since the current clinical guidelines recommend treatment with β-blockers, RAS inhibitors, and statins in patients with HFrEF and/or IHD. 24,29,30 However, subgroup analysis by LVEF category revealed decreased prescription rates of β-blockers and ACEI in women even in the HFrEF population. In addition, women with HF having IHD underwent PCI less frequently than men, even in patients with IHD (data not shown). Thus, women with HF are less adequately treated and consequently manifest more severe HF conditions than men.
Sex Difference in Prognosis and Cause of Death
Although women had significantly reduced risk for all the mortality outcomes after adjustment with clinical background, women had comparable all-cause mortality to men and an increased incidence of cardiovascular death and HF admission in the present study. This observation is not consistent with those from several previous landmark studies, which showed a reduced crude incidence of death in women with HF. 1 –3,5,6,26 –28 Considering that an increased incidence of cardiovascular death and HF admission in women was statistically evident only in the HFpEF population, the lack of prognostic advantage in women with HF could be explained by the higher proportion of HFpEF in women in the present study. Indeed, a lack of sex difference in crude mortality was also reported in patients with HFpEF enrolled in the ancillary arm of the Digitalis Investigation Group trial. 28 Since both the number and the proportion of patients with HFpEF are increasing in aged societies, 7,9 –11 these lines of observation are quite important in considering the future direction of HF management in the aged and super-aged societies.
In the present study, reduced risk of noncardiovascular death was more evident than that for cardiovascular risk: Overall hazard risk of noncardiovascular death was smaller than that for cardiovascular death. Notably, reduced risk of cardiovascular death in women was not significant during early follow-up but became significant later whereas that for noncardiovascular death was constantly evident in women throughout the observational period. Considering a decline in prognostic impact of BNP level across the time in women but not in men, this discrepancy in temporal changes of prognostic impact for women between cardiovascular and noncardiovascular death could be explained by a more severe HF status in women: More women with severe HF, for example, those with increased BNP levels, could have died due to cardiovascular causes in the early period, abolishing the reduced risk of female sex in nature at least in the early follow-up period. Therefore, we should pay more attention to women with HF for further implementation of evidence-based medicine and better adherence to the treatment to improve mortality of women with HF.
We also emphasize that the prognostic impact of female sex did not differ among HFrEF, borderline HFpEF, and HFpEF in the multivariable Cox hazard models in the present study, indicating that gender-specific cardiovascular risk may exist regardless of LVEF in the current era. Importantly, this gender-specific risk could be social rather than biological, since women likely visit hospitals later with a more advanced stage of HF than men, a common observation in daily practice. From this viewpoint, education for both primary and secondary prevention of HF to improve adherence to HF prevention and management is more important in women than in men.
Factors Influencing Sex Difference in Prognosis
There has been little consensus on the explanation for the sex difference in prognosis. Although previous studies suggested that LVEF has been suggested to explain sex differences in HF prognosis, the reduced risk of each end point in women did not differ among the HFrEF, borderline HFpEF, and HFpEF groups in the present study. This supports the finding that the lower risk in women with HF was not explained by LVEF in a recent post hoc analysis of the Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM) program. 26
Recently, Lam et al assessed sex differences in baseline characteristics and outcomes among 4128 patients with HFpEF in the Irbesartan in Heart Failure with Preserved Ejection Fraction trial and found that the sex-related difference in risk of all-cause events was modified by 4 factors, namely, renal dysfunction, advanced NYHA class symptoms, the presence or absence of atrial fibrillation, and stable angina pectoris. 27 In the present study, however, no clinical background, including these 4 factors, showed significant interactions with sex, indicating that these factors were not strong enough to affect sex differences. Kajimoto et al reported marked differences between men and women with respect to the association of anemia and LVEF with survival in Japanese acute decompensated patients registered in the Acute Decompensated Heart Failure Syndromes registry: After adjustment for multiple co-morbidities, anemia was an independent predictor of all-cause death for HFrEF but nor for HFpEF in men, while it was for HFpEF but not for HFrEF in women. 31 In the present study, however, sex differences in prognostic impact of anemia were not observed regardless of LVEF category. Although these discrepancies could be explained by several factors, including differences in ethnicity or nature of the studies (post-hoc analysis of the randomized study vs observational study, or acute decompensated HF vs stable chronic HF), further studies are warranted to elucidate factors modifying sex differences in patients with HFpEF to improve prognosis of patients with HF in the contemporary or future aged societies.
Sex Differences in Prognostic Impacts of Cardiovascular Medications
In the management of HF, the use of cardioprotective drugs, particularly β-blockers and RAS inhibitors, plays important roles. The current clinical guidelines for HF, however, do not provide sex-specific recommendations due to the underrepresentation of women and lack of sex-specific evaluation in previous trials. Thus, one of the most important messages from the present study is the presence of sex differences in the prognostic impacts of cardiovascular medications: Compared to men, use of a β-blocker was associated with better mortality in women, whereas RAS inhibitors produced better mortality in men. This finding of a greater benefit with beta-blockers and an attenuated benefit of RAS inhibitors (particularly ACE inhibitors) in women is also of clinical significance given that it had been suggested in the post hoc analysis of the previous landmark trials of beta-blockers and RAS inhibitors but remained to be confirmed. For example, in the US Carvedilol Heart Failure Study, it was shown that carvedilol improved survival to a greater degree in the 256 women with moderate HF symptoms and LVEF ≤ 35% (HR: 0.23, 95% CI: 0.07-0.69) than in the men (HR: 0.41, 95% CI: 0.22-0.80). 32 In the Cardiac Insufficiency Bisoprolol Study (CIBIS II), a significant reduction in all-cause mortality among women treated with bisoprolol compared to men was observed, although this was not significant in multivariate analysis. 33 The Studies of Left Ventricular Dysfunction (SOLVD) investigators also reported that treatment with enalapril was associated with a reduction in mortality and hospitalizations, albeit less so for women. 34 Furthermore, in a meta-analysis of ACE inhibitors, attenuation of benefit from ACE inhibition in women when compared to men was indicated recently. 35 Since these landmark studies, however, no subsequent randomized clinical studies have been conducted in recent years, as evidence on the benefits of β-blockers and RAS inhibitors was so robust. Thus, findings from the large-scale observational cohorts are significant in providing important insights to confirm sex differences in the benefits of cardiovascular medications. Nevertheless, prospective trials to examine sex differences in the effects of cardiovascular medications in the near future are strongly warranted, particularly considering factors that may affect sex differences including age, LVEF, HF severity, renal function, atrial fibrillation, and IHD.
Study Limitations
Several limitations of this study should be mentioned. First, the CHART-2 study is a prospective observational study for HF in Japan with a larger proportion of patients with HFpEF compared to previous studies in Western countries. Second, we used the clinical data at enrollment in the CHART-2 study and did not consider drug adherence or initiation and/or discontinuation during follow-up. Third, because the CHART-2 study is an observational study, we cannot rule out the possibility of significant confounding factors associated with management and prognosis. Thus, caution should be taken when generalizing the present findings to other populations, and validation studies in other countries should be performed.
Conclusion
In this study, we demonstrated substantial sex differences in patients with HF in our CHART-2 study. In the current era, the reduced risk of female sex in nature might be abolished by the more severe status of HF in women, particularly in those with preserved LVEF. Establishing sex-related HF management with consideration to LVEF is an emerging agenda item in the aged society.
Footnotes
Appendix
Acknowledgments
We thank all the members of the Tohoku Heart Failure Society and the staff of the Departments of Cardiovascular Medicine and Evidence-based Cardiovascular Medicine, Tohoku University Graduate School of Medicine, for their contributions (Appendix).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Grants-in Aid from the Ministry of Health, Labour, and Welfare, the Ministry of Education, Culture, Sports, Science, and Technology (H. S. and Y. S.) and the Agency for Medical Research and Development (H. S.; No 15ek0210043h0001), Tokyo, Japan. The Department of Evidence-based Cardiovascular Medicine, Tohoku University Graduate School of Medicine, is supported in part by unrestricted research grants from Daiichi Sankyo (Tokyo, Japan), Bayer Yakuhin (Osaka, Japan), Kyowa Hakko Kirin (Tokyo, Japan), Novartis Pharma (Tokyo, Japan), Dainippon Sumitomo Pharma (Osaka, Japan), Astellas Pharma, AstraZeneca, Chugai Pharmaceutical, GlaxoSmithKline, Kowa Pharmaceutical, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, MSD, Nippon Boehringer Ingelheim, Otsuka Pharmaceutical, Shionogi, and Takeda Pharmaceutical. H. S. has received lecture fees from Bayer Yakuhin (Osaka, Japan), Daiichi Sankyo (Tokyo, Japan), and Novartis Pharma (Tokyo, Japan).
