Abstract
This meta-analysis evaluated the efficacy of mesenchymal stem cell (MSC) treatment on cardiovascular function and major adverse cardiac events (MACE) in patients with acute myocardial infarction (AMI) at various follow-up intervals. Clinical studies comparing MSC therapy with control treatments for AMI were identified from databases including Cochrane, Web of Science, PubMed, Embase, CNKI, and Wanfang, covering publications up to August 2024. Data analysis was conducted using Review Manager 5.4 software. MSC treatment significantly improved left ventricular ejection fraction (LVEF) compared to controls at follow-up intervals <6 months (MD = 3.42; P < 0.0001), 6 months (MD = 4.15; P = 0.006), and 12 months (MD = 2.77; P = 0.006). However, no significant effect on LVEF was observed after 12 months (MD = 3.50; P = 0.17). MSC therapy did not significantly affect left ventricular end-diastolic volume (LVEDV) at any interval. Left ventricular end-systolic volume (LVESV) significantly decreased only within the first 6 months (MD = −11.35; P = 0.11) but not at subsequent follow-ups. Wall motion score index (WMSI) significantly improved at <6 months (MD = −0.06; P < 0.0001), 6 months (MD = −0.04; P = 0.006), and >12 months (MD = −0.03; P = 0.02). However, the improvement at 12 months was borderline significant (MD = −0.06; P = 0.06). MSC therapy showed no significant reduction in MACE (odds ratio [OR] = 1.61; P = 0.10). Subgroup analyses indicated intracoronary MSC administration notably improved LVEF (MD = 4.27; P < 0.0001), while intravenous MSC administration showed no significant effect. Neither administration route significantly affected MACE outcomes. No publication bias was detected. In conclusion, MSC therapy significantly enhances LVEF and WMSI within the first 12 months post-AMI, with intracoronary administration showing greater efficacy than intravenous delivery. However, MSC treatment did not significantly reduce MACE incidence. Further rigorous clinical trials are needed to confirm these findings.

Keywords
Coronary artery disease is a major cause of mortality, with acute myocardial infarction (AMI) being a major contributor to the increasing prevalence of heart failure and mortality 1 . AMI, a frequent cardiovascular emergency, is characterized by prolonged myocardial ischemia, hypoxia, and necrosis due to the narrowing or blockage of coronary arteries2,3. Clinically, it manifests as severe, persistent chest pain behind the sternum lasting over 30 minutes, often radiating to the back, jaw, and upper limb4,5. Certain people might encounter lightheadedness, queasiness, vomiting, and difficulty breathing. Patients typically show dynamic electrocardiogram (ECG) changes and elevated cardiac enzymes, with complications like arrhythmias, heart failure, and shock posing life-threatening risks4,5. Risk factors for AMI include genetic predisposition, smoking, high cholesterol, hypertension, diabetes, older age, and male gender6–8. Diagnosis is generally based on clinical symptoms, ECG results, cardiac biomarkers, and coronary angiography6–8. Advances in drug therapy and interventional techniques have significantly reduced the acute mortality rate for myocardial infarction patients6–8. However, the incidence of heart failure postmyocardial infarction remains high. Therefore, improving cardiac function after percutaneous coronary intervention (PCI) and enhancing long-term survival rates for AMI patients is a pressing issue 9 .
Mesenchymal stem cells (MSCs) are a variety of stromal cells that can be isolated from various adult tissues, including bone marrow, umbilical cord, and adipose tissue 10 . Recently, they have attracted attention in various medical fields due to their potential therapeutic benefits. MSCs are mature stem cells capable of differentiating into various cell types and demonstrating considerable plasticity11–13. Under specific conditions, they can transform into mesodermal and ectodermal tissue cells, including adipocytes, osteocytes, chondrocytes, skeletal myocytes, cardiomyocytes, smooth muscle cells, hepatocytes, and neuron-like cells11–13. MSCs can be conveniently and safely harvested with minimal harm and are easy to expand in vitro, making them suitable for autologous transplantation11–13. They have the capacity to repair and regenerate various functional cells, making them important “seed cells” for tissue engineering11–13. Several prior studies have demonstrated that after MSC transplantation in patients with cardiovascular diseases, these stem cells can colonize the infarcted myocardial tissue, directly proliferating, and differentiating into cardiomyocyte-like cells and vascular endothelial cells14–16. They can also secrete various vascular endothelial growth factors through paracrine mechanisms to promote blood vessels formation, enhance myocardial contractility, and reduce myocardial fibrosis while preventing left ventricular remodeling14–16. Currently, MSCs are used in treating various heart conditions, including heart failure and myocardial infarction17,18.
MSC transplantation in patients with AMI is typically performed using three methods: intracoronary injection, intramyocardial injection, and intravenous injection19,20. Intravenous infusion often results in low cell survival rates within the myocardium, which can greatly impact the effectiveness of the treatment19,20. Research indicates that direct intramyocardial injection of stem cells is less effective than intracoronary injection and may cause uneven distribution of stem cells, leading to dys-synchrony in ventricular wall motion 21 . In addition, intramyocardial injection carries a higher risk of triggering malignant arrhythmias. Consequently, intracoronary injection of stem cells is considered the most optimal transplantation method 21 . To our best knowledge, most studies reported small sample sizes or conflicting results22–35, making it difficult to draw a reliable and authoritative conclusion for clinical guidance. Hereby, we carried out a meta-analysis to summarize efficacy and safety of MSC therapy in patients with AMI and to investigate the immediate and prolonged impacts of MSC on patients with AMI.
Methods
Literature retrieval strategy
According to the principles and relevant guidelines proposed by the Cochrane Handbook, we conducted a computer search in databases including “PubMed,” “The Cochrane Library,” “Embase,” “Web of Science,” “CNKI,” and Wanfang. The search aimed to identify clinical studies on MSCs therapy for treating AMI patients, covering period from inception of databases up to August 2024. Search terms or keywords in English were: “mesenchymal stem cells,” “mesenchymal stromal cells,” “stem cell(s),” “cell transplantation” “acute myocardial infarction,” “AMI,” “myocardial infarction,” “MI,” and “coronary artery disease.”
Inclusion and exclusion criteria
Inclusion criteria were (1) clinical studies with a minimum follow-up of 2 months. (2) Both experimental and controls received standard treatment, with the control group not receiving stem cell transplantation. (3) Patients clinically diagnosed with AMI who underwent PCI. (4) Studies in which the experimental group received cell therapy within 3 months post-PCI. (5) MSCs sourced from human bone marrow, umbilical cord, or adipose tissue. (6) Primary endpoints included left ventricular ejection fraction (LVEF), with secondary endpoints being left ventricular end-diastolic volume (LVEDV) and left ventricular end-systolic volume (LVESV). (7) Only MSCs were used for transplantation, with no restrictions on cell dose or administration timing.
The exclusion criteria were: (1) Studies involving cytokine interventions. (2) Trials lacking a control group. (3) Research involving specific populations, such as pregnant women. (4) Studies that did not assess any of the primary or secondary endpoints. (5) Studies with incomplete data or duplicate publications.
Literature selection and data extraction
Two reviewers independently performed the literature search based on the defined criteria. All selected literature was evaluated according to the inclusion and exclusion criteria, removing irrelevant trials, reviews, or nonclinical studies. Any disagreements or differing viewpoints were addressed through discussion and consultation with a third reviewer.
Two researchers independently extracted data from the included studies, ensuring accuracy through cross-checking. Discrepancies were resolved via discussion or by consulting a third reviewer. The extracted data included: general study details (author, publication year, number of cases, demographic information, follow-up duration, and baseline LVEF), the exact number of MSCs transplanted, and the time elapsed from PCI to transplantation. In addition, measurement methods including echocardiography, magnetic resonance imaging (MRI), SPECT, positive emission tomography (PET) and angiography, and outcome measures, such as, LVEF, LVEDV, LVESV, wall motion score index (WMSI), and major adverse cardiac events (MACE) were also recorded.
Quality assessment
Two researchers assessed the quality of the included studies across seven dimensions. Any disagreements or differing views were resolved through discussion and consultation with a third reviewer. The evaluation criteria were based on the quality assessment standards for RCTs outlined in the Cochrane Handbook. This assessment covered aspects such as random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, completeness of outcome data, selective reporting, and other potential biases. The results were classified as low risk of bias, high risk of bias, or unclear risk of bias.
Statistical analysis
Data from the included studies were organized and analyzed using Review Manager 5.3. For continuous variables, the mean difference (MD) was used as the effect size in studies employing the same statistical methods, helping to adjust for variations in absolute values across studies. For categorical variables, the odds ratio (OR) was used. Statistical significance was defined as P < 0.05. Heterogeneity among studies was evaluated using chi-square tests to compute P and I² values. If P > 0.1 and I² < 50%, a fixed-effect model was applied for the meta-analysis; otherwise, a random-effect model was used after investigating sources of heterogeneity. Funnel plots were employed to evaluate potential publication bias.
Results
Study flow
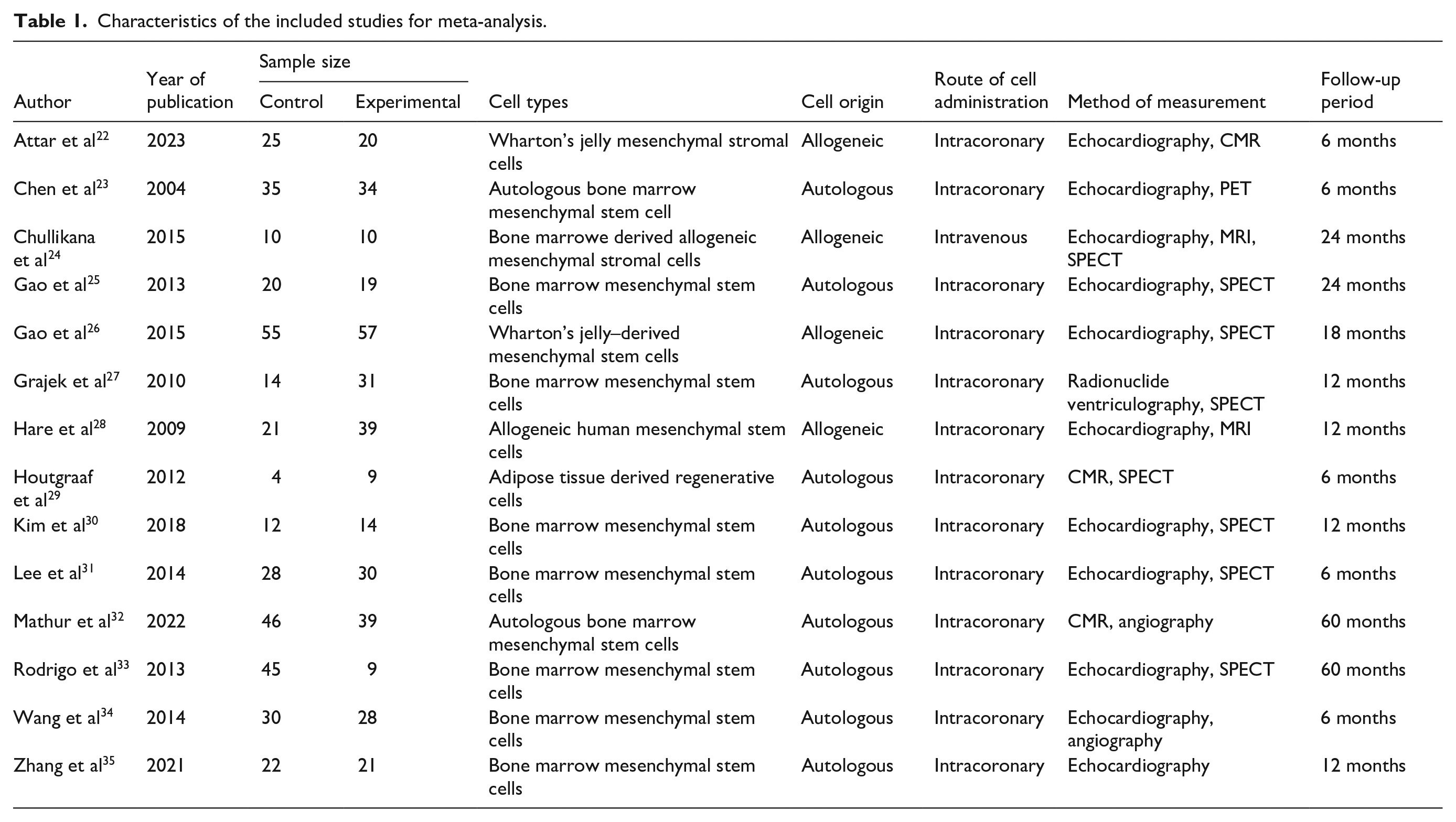
Meta-analysis flow is illustrated in Figure 1. Initially, 1,475 articles were retrieved, with an extra 27 obtained from other sources. Of these, 983 records were excluded. The remaining 62 studies were reviewed in detail, resulting in the inclusion of 13 studies involving 727 patients (360 in the experimental group and 367 in the control group). Table 1 provides a summary of the characteristics of these included studies, which were published between 2004 and 2023.

Flow diagram of the meta-analysis.
Characteristics of the included studies for meta-analysis.
Quality assessment of included literature
Risk bias assessment indicated varying degrees of uncertainty and potential bias among the eligible studies. Specifically: Four studies were categorized as having an “unclear risk” of selection bias. One study was classified as having a “high risk” of selection bias due to insufficient descriptions of random sequence generation. Five studies had an “unclear risk” of selection bias due to a lack of clarity regarding allocation concealment. Three studies were labeled as having an “unclear risk” and one as having a “high risk” due to insufficient details concerning the blinding of participants and personnel. Three studies were deemed to have an “unclear risk” of detection bias because of incomplete information on the blinding of outcomes assessment. Seven studies were categorized as having an “unclear risk” of other forms of bias. A visual representation of the quality assessment of the eligible clinical studies is shown in Figure 2.

Quality assessment analysis: (a) risk of bias graph and (b) risk of bias summary.
LVEF assessment at different time points
For LVEF assessed at <6 months, eight studies compared LVEF between experimental (234 cases) and control groups (229 cases). The results indicated heterogeneity in studies (I² = 81%, P < 0.00001); thus, a random effect was used. Meta-analysis showed a notable difference in LVEF < 6 months between these groups [MD = 3.42, P < 0.0001, 95% CI (1.86, 4.98)] (Figure 3a). This indicates a substantial elevation in LVEF in experimental group within 6 months following treatment compared to controls.

Forest plot of meta-analysis of LVEF: (a) Forest plot of LEVF <6 months; (b) Forest plot of LVEF at 6 months; (c) Forest plot of LVEF at 12 months; (d) Forest plot of LVEF >12 months.
For LVEF assessed at 6 months, 10 studies compared LVEF between experimental (227 cases) and control groups (223 cases). The results showed heterogeneity among studies (I² = 94%, P < 0.00001); thus, a random-effects model was used. The meta-analysis revealed a notable difference in LVEF at 6 months between these groups [MD = 4.15, P = 0.006, 95% CI (1.18, 7.11)] (Figure 3b), indicating a substantial increase in LVEF in the experimental group at 6 months following treatment.
For LVEF assessed at 12 months, seven studies compared LVEF between experimental (227 cases) and control groups (223 cases). The results indicated heterogeneity for included studies (I² = 94%, P < 0.00001); thus, we used a random effect. Meta-analysis showed a notable difference in LVEF at 12 months between these groups [MD = 2.77, P = 0.006, 95% CI (0.78, 4.75)] (Figure 3c). This indicates a substantial elevation in LVEF in experimental group at 12 months following treatment compared with controls.
For LVEF assessed >12 months, three studies compared LVEF between experimental (86 cases) and control groups (85 cases). The results showed heterogeneity for included studies (I² = 98%, P < 0.00001); thus, we used a random-effects model. There was no significant difference in LVEF >12 months between these groups [MD = 3.50, P = 0.17, 95% CI (−1.51, 8.50)] (Figure 3d), indicating no changes in LVEF in experimental group comparing with controls at >12 months following treatment.
Funnel plots of the included studies did not show discernible asymmetry, indicating minimal evidence of publication bias (Supplemental Figures S1A–S1D); however, the small number of heterogeneous trials and variable reporting standards limit its sensitivity to detect publication bias.
Subgroup analysis based on administration route revealed that intracoronary administration of MSCs led to a significant enhancement in LVEF at 6 months [MD = 4.27, P < 0.0001, 95% CI (1.17, 7.37)] compared to the control group (Supplemental Figure S2A). Conversely, intravenous infusion did not lead to a substantial improvement in LVEF comparing with controls [MD = 2.55, P = 0.49, 95% CI (−4.76, 9.86)] (Supplemental Figure S2A). For the subgroup analysis of LVEF >12 months, intracoronary infusion of MSCs failed to significantly increase LVEF comparing with controls [MD = 3.12, P = 0.28, 95% CI (−2.56, 8.81)] (Supplemental Figure S2B). Consistent findings were observed in patients receiving intravenous infusion of MSCs [MD = 3.50, P = 0.17, 95% CI (−1.51, 8.50)] (Supplemental Figure S2B).
When stratified by cell origin, allogeneic MSCs, but not autologous MSCs, significantly improved LVEF at <6 months [allogeneic: MD = 4.40; P < 0.0001; 95% CI (3.96, 4.85); autologous: MD = 2.96, P = 0.05, 95% CI (–0.02, 5.94)] versus controls (Supplemental Figure S3A). In subgroup analysis, allogeneic MSCs continued to confer a significant benefit [MD = 5.04, P < 0.0001, 95% CI (4.10, 5.99)], whereas autologous MSCs did not reach significance (MD = 4.04, P = 0.08, 95% CI (–0.43, 8.51)) compared with controls (Supplemental Figure S3B). However, neither allogeneic [MD = 3.43; P = 0.11; 95% CI (–0.78, 7.64)] nor autologous MSCs [MD = 2.12; P = 0.05; 95% CI (0.04, 4.21)] achieved statistical significance versus controls at 12 months (Supplemental Figure S3C).
In a further analysis, combining all outcomes regardless of follow-up duration showed heterogeneity for included studies (I² = 97%, P < 0.00001); thus, we used a random-effects model. Meta-analysis demonstrated a notable difference in LVEF between these groups [MD = 3.52, P < 0.0001, 95% CI (2.39, 4.65)] (Table 2), indicating a substantial increase in LVEF in experimental group following MSC treatment comparing with controls.
Meta-analysis of LVEF, LVEDV, LVESV, and WMSI.

LVEDV assessment at different time points
For LVEDV assessed at <6 months, six studies compared LVEDV between experimental (183 cases) and control groups (175 cases). The results indicated heterogeneity for included studies (I² = 94%, P < 0.00001); thus, we used a random-effects model. Meta-analysis showed no difference in LVEDV <6 months between these groups [MD = −11.56, P = 0.11, 95% CI (−25.70, 2.59)] (Figure 4a), indicating no changes in LVEDV in the experimental group within 6 months following treatment compared to controls.

Forest plot of meta-analysis of LVEDV: (a) Forest plot of LVEDV <6 months; (b) Forest plot of LVEDV at 6 months; (c) Forest plot of LVEDV at 12 months; and (d) Forest plot of LVEDV >12 months.
For LVEDV assessed at 6 months, six studies compared LVEDV between experimental (147 cases) and control groups (146 cases). The results indicated heterogeneity for included studies (I² = 99%, P < 0.00001); thus, we carried out a random-effects model. Meta-analysis exhibited no difference in LVEDV at 6 months between these groups [MD = −21.10, P = 0.17, 95% CI (−51.54, 9.33)] (Figure 4b), indicating no changes in LVEDV in the experimental group at 6 months following treatment compared to controls.
For LVEDV assessed at 12 months, seven studies compared LVEDV between experimental (182 cases) and control groups (177 cases). The results indicated heterogeneity for included studies (I² = 94%, P < 0.00001); thus, we used a random-effects model. Meta-analysis showed no significant difference in LVEDV at 12 months between these groups [MD = −5.69, P = 0.23, 95% CI (−15.08, 3.69)] (Figure 4c), indicating no changes in LVEDV in the experimental group at 12 months following treatment compared to the control group.
For LVEDV assessed >12 months, two studies compared LVEDV between experimental (76 cases) and control groups (75 cases). The results indicated heterogeneity among studies (I² = 96%, P < 0.00001); thus, we used a random-effects model. No difference was detected in LVEDV >12 months between these groups [MD = −8.47, P = 0.12, 95% CI (−19.05, 2.11)] (Figure 4d), indicating no changes in LVEDV in experimental group compared to controls at >12 months following treatment.
The funnel plots for studies likewise appeared symmetrical, suggesting little publication bias (Supplemental Figures S4A–S4D). However, the limited number and heterogeneity of trials reduce the analysis’s power to detect it.
Subgroup analysis by cell origin confirmed that neither allogeneic [MD = −14.96, P = 0.11, 95% CI (–39.06, 9.15)] nor autologous MSCs [MD = −9.94, P = 0.20, 95% CI (–25.17, 5.30)] significantly reduced LVEDV versus controls (Supplemental Figure S5A). Subgroup analyses indicated that allogeneic and autologous MSCs trended toward a reduction [allogeneic: MD = −58.11, P = 0.07, 95% CI (–120.92, 4.17); autologous: MD = −3.85; P = 0.05; 95% CI (–7.70, 0.01)], but neither reached significance (Supplemental Figure S5B). Neither allogeneic [MD = −15.59; P = 0.23; 95% CI (–41.26, 10.09)] nor autologous MSCs [MD = −2.98; P = 0.08; 95% CI (–6.37, 0.41)] significantly altered LVEDV (Supplemental Figure S5C).
We also combined all outcomes regardless of follow-up duration. The results revealed heterogeneity for included studies (I2 = 98%, P < 0.00001); hence, we employed a random-effects model. Meta-analysis indicated a difference in LVEDV between these groups [MD = −13.19, P < 0.0001, 95 CI (−19.05, −7.33)] (Table 2). This suggests that, following MSC treatment, experimental group experienced a substantial reduction in LVEDV compared to controls (Table 2).
LVESV assessment at different time points
For LVESV assessed at <6 months, three studies compared LVESV between experimental (96 cases) and control groups (107 cases). Results showed heterogeneity for included studies (I2 = 93%, P < 0.00001); thus, we used a random-effects model. Meta-analysis indicated that experimental group exhibited a decrease in LVESV <6 months compared to controls [MD = −11.35, P = 0.11, 95% CI (−20.55, −2.16)] (Figure 5a).

Forest plot of meta-analysis of LVESV. (a) Forest plot of LVESV <6 months; (b) Forest plot of LVESV at 6 months; (c) Forest plot of LVESV at 12 months; (d) Forest plot of LVESV >12 months.
For LVESV assessed at 6 months, six studies compared LVESV between experimental (147 cases) and control groups (146 cases). The results revealed heterogeneity for included studies (I2 =100%, P < 0.00001); thus, we used a random-effects model. The meta-analysis showed no significant difference in LVESV at 6 months between these groups [MD = −18.23, P = 0.17, 95% CI (−48.24, 11.78)] (Figure 5b). This indicates that, at 6 months following treatment, experimental group showed no change in LVESV compared to controls (Figure 5b).
For LVESV assessed at 12 months, seven studies compared LVESV between experimental (182 cases) and control groups (177 cases). The results showed heterogeneity for included studies (I2 = 96%, P < 0.00001); thus, we used a random-effects model. Meta-analysis indicated no difference in LVESV at 12 months between these groups [MD = −7.62, P = 0.06, 95% CI (−15.71, 0.47)] (Figure 5c). This indicates that, at 12 months following treatment, experimental group showed no change in LVESV comparing with controls (Figure 5c).
For LVESV assessed >12 months, two studies compared LVESV between experimental (76 cases) and control groups (75 cases). The results revealed heterogeneity for included studies (I2 = 98%, P < 0.00001); thus, we used a random-effects model. Results showed no difference in LVESV >12 months between these groups [MD = −8.25, P = 0.13, 95% CI (−18.93, 2.43)] (Figure 5d). This indicates that, at >12 months following treatment, experimental group showed no change in LVESV comparing with controls (Figure 5d).
No obvious asymmetry was observed in the funnel plots shown in Supplemental Figures S6A–S6D, indicating minimal publication bias; however, the small, diverse sample of trials and uneven reporting standards constrain the sensitivity of this assessment.
Subgroup analysis based on cell origin revealed no significant LVESV reduction at <6 months for allogeneic [MD = −15.26, P = 0.05, 95% CI (–30.74, –0.22)] or autologous MSCs [MD = −8.36; P = 0.28; 95% CI (–23.65, 6.93)] versus controls (Supplemental Figure S7A). At 6 months, only allogeneic MSCs significantly reduced LVESV [MD = −49.08, P = 0.04, 95% CI (–96.84, 1.31)], whereas autologous MSCs did not [MD = −0.46; P = 0.74, 95% CI (–3.18, 2.26)] (Supplemental Figure S7B). Subgroup analysis demonstrated that neither allogeneic [MD = −17.73; P = 0.09; 95% CI (–38.11, 2.65)] nor autologous MSCs [MD −2.51, P = 0.07; 95% CI (–5.19, 0.17)] reached significance in reduced LVESV at 12 months (Supplemental Figure S7C).
In another analysis, combining all outcomes regardless of follow-up duration, results revealed heterogeneity for included studies (I2 = 97%, P < 0.00001); thus, we used a random-effects model. Results exhibited a difference in LVEF between these groups [MD = −12.40, P < 0.0001, 95% CI (−17.00, −7.79)] (Table 2). This suggests that, following MSC treatment, experimental group exhibited a substantial decrease in LVESV compared to controls (Table 2).
WMSI score assessment at different time points
For the WMSI assessed at <6 months, three studies compared WMSI between experimental (96 cases) and control groups (107 cases). The results showed no heterogeneity for included studies (I2 = 0%, P = 0.84); thus, we used a fixed-effects model. Results showed a notable difference in WMSI <6 months between these groups [MD = −0.06, P < 0.0001, 95% CI (−0.07, −0.05)] (Figure 6a). This suggests that, within 6 months following treatment, experimental group exhibited a substantial decrease in WMSI score compared to controls (Figure 6a).

Forest plot of meta-analysis of WMSI: (a) Forest plot of WMSI < 6 months; (b) Forest plot of WMSI at 6 months; (c) Forest plot of WMSI at 12 months; and (d) Forest plot of WMSI >12 months.
For the WMSI score assessed at 6 months, five studies compared WMSI between experimental (118 cases) and control groups (128 cases). The results revealed no heterogeneity for included studies (I2 = 0%, P = 0.60); thus, we used a fixed-effects model. Results indicated a notable difference in WMSI at 6 months between these groups [MD = −0.04, P = 0.006, 95% CI (−0.06, −0.01)] (Figure 6b). This suggests that, at 6 months following treatment, experimental group exhibited a substantial increase in LVEF compared to controls (Figure 6b).
For the WMSI assessed at 12 months, four studies compared WMSI score between experimental (111 cases) and control groups (125 cases). Results exhibited heterogeneity for included studies (I2 = 62%, P = 0.05); thus, we used a random-effects model. Meta-analysis indicated a notable difference in WMSI at 12 months between these groups [MD = 2.77, P = 0.06, 95% CI (−0.09, −0.03)] (Figure 6c). This suggests that, at 12 months following treatment, experimental group exhibited a substantial decrease in WMSI score compared to controls (Figure 6c).
For the WMSI assessed >12 months, two studies compared WMSI between experimental (76 cases) and control groups (75 cases). The results showed no heterogeneity for included studies (I2 = 0%, P = 0.62); thus, we used a fixed-effects model. Meta-analysis indicated a notable difference in WMSI at >12 months between these groups [MD = −0.03, P = 0.02, 95% CI (−1.51, 8.50)] (Figure 6d). This suggests that, at >12 months following treatment, experimental group exhibited a substantial decrease in WMSI score comparing with controls (Figure 6d).
Funnel plots of the included studies did not show discernible asymmetry, indicating minimal evidence of publication bias (Supplemental Figures S8A–S8D); however, the small number of heterogeneous trials and variable reporting standards limit its sensitivity to detect publication bias.
In a further analysis, combining all outcomes regardless of follow-up duration, results showed no heterogeneity for included studies (I2 = 30%, P = 0.14); thus, we used a fixed-effects model. Meta-analysis indicated a notable difference in LVEF between these groups [MD = −0.06, P < 0.0001, 95% CI (−0.06, −0.05)] (Table 2). This suggests that, following MSC treatment, experimental group exhibited a substantial decrease in WMSI comparing with controls (Table 2).
MACE assessment
For MACE, 10 studies compared MACE between experimental (268 cases) and control groups (247 cases). The results showed no heterogeneity for included (I2 = 0%, P = 0.92); thus, we used a fixed-effects model. Meta-analysis indicated no difference in MACE between these groups [OR = 1.61, P = 0.10, 95% CI (0.92, 2.83)] (Figure 7). This suggests that, at 12 months following treatment, experimental group exhibited no change in MACE comparing with controls (Figure 5c). Funnel plots of the included studies did not show discernible asymmetry, pointing to minimal publication bias (Supplemental Figure S9); yet the small scale and heterogeneity of included trials, together with variable reporting, may obscure subtle biases.

Forest plot of meta-analysis of MACE.
Subgroup analysis by administration route, intracoronary [OR = 1.62, P = 0.11, 95% CI (0.90, 2.92)] and intravenous [OR = 1.56, P = 0.64, 95% CI (0.24, 9.91)], also showed no effect on MACE (Supplemental Figure S10). Finally, cell-origin stratification confirmed that neither allogeneic [OR = 0.91, P = 0.05, 95% CI (0.30, 2.76)] nor autologous MSCs [OR = 1.98, P = 0.05, 95% CI (1.02, 3.85)] significantly affected MACE compared with controls (Supplemental Figure S11).
Discussion
Stem cell transplantation therapy has been a hot topic in recent years. Historically, stem cell transplantation was primarily used for hematological diseases, with over 40 years of clinical experience. Recently, numerous clinical trials both domestically and internationally have evaluated safety and efficacy of stem cell transplantation in patients with AMI post-PCI18,36. These studies have demonstrated that stem cell transplantation is effective to some extent in AMI patients and that intracoronary injection is a safe and feasible method. Certain animal experiments and clinical studies have suggested that the therapeutic efficacy of MSC transplantation for treating myocardial infarction might surpass that of bone marrow-derived mononuclear cells 37 . However, regarding long-term efficacy, especially benefits observed after 6 months, there are no significant differences compared to control groups 38 . Therefore, the clinical efficacy of MSCs for AMI requires extensive clinical trials to be substantiated. Our research shows that AMI patients who received MSC transplantation had an improvement in LVEF across different follow-up periods. For LVEF assessed at <6 months, meta-analysis of eight studies revealed a notable difference in LVEF between these groups, indicating a substantial increase in LVEF in experimental group within 6 months following treatment comparing with controls. Similarly, for LVEF assessed at 6 months, meta-analysis of 10 studies showed a significant difference, and for LVEF assessed at 12 months, meta-analysis of seven studies demonstrated a significant improvement. However, for LVEF assessed at >12 months, no difference was detected between these groups.
The mechanisms by which MSCs exert their myocardial repair effects have been widely confirmed, including promoting neovascularization, reducing myocardial fibrosis, restoring vascular contractile function through direct differentiation, and stimulating endogenous cardiac stem cells 39 . Paracrine mechanisms play a crucial role, whereby MSCs secrete various bioactive molecules that can inhibit fibroblast proliferation, promote the secretion of metalloproteinases, stimulate neovascularization, and mobilize endogenous cardiac stem cells to reduce fibrosis. Moreover, MSCs have the ability to differentiate into endothelial cells in vitro and cardiomyocytes in vivo. Furthermore, the transplanted MSCs influence the proliferation and differentiation of native cardiac stem cells and stimulate cell cycle of cardiomyocytes through cell-cell interactions, thereby repairing damaged myocardium, promoting angiogenesis, and achieving myocardial repair40,41.
Regarding left ventricular remodeling, our study found that intracoronary MSC transplantation does not effectively improve LVESV and LVEDV. For LVEDV assessed at <6 months, meta-analysis showed no difference between these groups. Similarly, no differences were detected at 6, 12, and >12 months. For LVESV, meta-analysis also showed no significant differences at <6, 6, 12, and >12 months. In terms of follow-up time for stem cell transplantation, previous studies have shown that MSC transplantation in AMI patients significantly increases LVEF in the short term (follow-up time < 6 months) compared to controls, with statistically significant differences. However, in the long term (follow-up time >6 months), the results did not show statistical differences.
Regarding the baseline levels of patients undergoing MSC therapy, previous studies by Tendera et al indicated that stem cell transplantation for AMI is more effective in patients with lower baseline LVEF levels 23 . However, recent large-scale RCT studies found that stem cell transplantation does not significantly improve outcomes in AMI patients with larger infarct areas and lower LVEF compared to control groups25,42. Since only a portion of MSCs injected via coronary arteries are retained in the myocardium, the dosage of MSCs used in the injection impacts the efficacy. Previous meta-analysis results by Meluzin et al 43 regarding bone marrow mononuclear cells for AMI patients indicated that only those receiving a high dose of MSCs showed sustained LVEF improvement, suggesting a dose-response relationship. Another meta-analysis concluded that only when the number of transplanted MSCs exceeded 107 could the patient’s LVEF improve 44 .
Furthermore, the WMSI assessment revealed significant improvements in the experimental group at different follow-up points. For WMSI assessed at <6 months, meta-analysis revealed a notable decrease. At 6 months, the WMSI also significantly improved, and similar positive results were observed at 12 months and >12 months. In contrast, MACE analysis indicated no difference between these groups, suggesting no change in MACE following MSC treatment. Subgroup analysis based on administration route also revealed no significant impact of intracoronary administration or intravenous infusion on MACE.
Although our meta-analysis involving a large number of subjects demonstrated that intracoronary MSC transplantation improves LVEF in AMI patients, this study also has its limitations. In the subgroup analysis, allogeneic MSCs demonstrated clear early benefits, significantly improving LVEF at both <6 months and 6 months, and reducing LVESV at 6 months. In contrast, autologous MSCs failed to reach statistical significance at these timepoints. By 12 months, gains in LVEF for both allogeneic and autologous cells had diminished to nonsignificant levels, and neither cell type produced meaningful changes in LVEDV or LVESV beyond this period. Importantly, there was no impact on MACE rates for either group. However, the allogeneic subgroup is based on relatively few studies, which may introduce bias and limit generalizability; larger, well-powered trials are needed to validate these early advantages. Across the trials included in our meta-analysis, cell doses varied widely, and emerging evidence suggests a clear dose–response relationship. Mechanistically, higher cell numbers may improve myocardial retention, enhance paracrine cytokine release, and more effectively attenuate adverse ventricular remodeling. Nonetheless, the small number of studies employing high-dose regimens introduces potential bias, and optimal dosing remains undefined. Future randomized trials should therefore incorporate multiple dose arms and standardized reporting of MSC viability and delivery techniques to establish the minimum effective dose and maximize therapeutic benefit. We also recognize that AMI subtype influences MSC efficacy: STEMI’s larger, transmural infarcts and earlier cell delivery generally yield greater LVEF improvements, whereas non-STEMI patients with smaller, subendocardial injuries, and preserved baseline function often show minimal gains. Differences in infusion timing and infarct characteristics likely contribute to these varied responses. Future trials should stratify by AMI subtype, standardize delivery windows, and report infarct size and transmurality to identify which patient groups benefit most from MSC therapy. Variability in imaging modalities introduces significant bias. Echocardiography can underestimate early LVEF gains due to operator dependence and poor acoustic windows; Cardiac magnetic resonance (CMR) delivers gold-standard volumetric accuracy but is limited by cost, scanner availability, and scan time; and SPECT offers high reproducibility at the expense of temporal resolution, potentially masking subtle functional improvements. These intermodality differences amplify between-study heterogeneity and can skew meta-analytic outcomes. To mitigate this, future MSC trials should predefine a single-core modality, which will enhance data comparability and more accurately reflect MSC-mediated cardiac repair. All included trials carry inherent bias. Many early studies were small, single-center with inconsistent randomization, introducing selection bias, and lack of blinding in cell delivery and assessment created performance and detection bias. Differential follow-up rates also raise attrition bias concerns. These factors may have inflated treatment effects relationships. Future multicenter, double-blind RCTs with standardized cell-processing and imaging protocols are needed to address these limitations.
Conclusions
MSC treatment significantly improves LVEF and enhances WMSI in patients with AMI, particularly within the first 12 months posttreatment. Intracoronary administration appears to be more effective than intravenous infusion. However, no significant effect on MACE was observed. Additional rigorous studies are required to validate these results.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251359773 – Supplemental material for Impact of mesenchymal stem cell therapy on cardiac function and outcomes in acute myocardial infarction: A meta-analysis of clinical studies
Supplemental material, sj-docx-1-cll-10.1177_09636897251359773 for Impact of mesenchymal stem cell therapy on cardiac function and outcomes in acute myocardial infarction: A meta-analysis of clinical studies by Mei Zhao, Yanpeng Xue, Qingqing Tian, Yan Deng and Tao Tang in Cell Transplantation
Footnotes
Acknowledgements
The authors acknowledge using ChatGPT-4 by OpenAI for English language editing and confirm the accuracy and originality of the AI-assisted content.
Ethical Considerations
Not applicable.
Statement of human and animal rights
There are no human subjects in this article and informed consent is not applicable.
Statement of informed consent
Not applicable.
Authors’ contributions
MZ, YD, and TT designed the whole study; MZ, YX, QT, YD, and TT analyzed the data; YD and TT prepared the figures; MZ, YX, QT, YD, and TT wrote the manuscript; all the authors approved the manuscript for submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availabilitystatement
The data are available upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
