Abstract
Background
With increasing life expectancy, dietary choices are crucial for healthy aging.
Objective
This study investigates associations between two dietary indices, the energy-adjusted Dietary Inflammatory Index (E-DII) and the healthful Plant-based Diet Index (hPDI), and biomarkers related to metabolic health and micronutrient status, focusing on the mediating role of gut microbiota.
Methods
We conducted a secondary, exploratory analysis of baseline data from 129 community-based participants aged ≥70 years (median age: 75 years). Dietary indices were calculated from 3-day food protocols, and biomarkers were measured in blood samples. Cognitive and physical function were assessed using validated questionnaires, body composition was determined via bioimpedance, and fecal gut microbiota composition through 16S rRNA gene sequencing. Statistical analyses included multiple linear regression models, Permutational Multivariate Analysis of Variance, and mediation analysis.
Results
We found no significant associations between the indices and gut microbiota diversity. However, red blood cell (RBC) folate and serum uric acid were significantly associated with E-DII (stand. β = −0.32 and 0.23, respectively,
Conclusions
This study suggests that gut microbiota composition does not mediate the associations between a healthy plant-based or anti-inflammatory diet and metabolic health markers in a healthy, older population. However, microbial genera were associated with uric acid and folate concentrations independent of the investigated dietary patterns, indicating potential roles in folate and uric acid metabolism.
Keywords
Introduction
Rising life expectancy in recent decades has increased the need to address age-related health challenges. 1 Promoting healthy aging, defined as preserving physical and cognitive function while extending lifespan, 2 is essential to combat diseases such as sarcopenia and cognitive decline, osteoporosis, kidney diseases, cancer, type 2 diabetes mellitus (T2DM), and cardiovascular diseases (CVDs). 3
Understanding the substantial role of diet as a modifiable factor influencing overall health is central to achieving healthy aging. Therefore, assessing dietary patterns and their impact on health is imperative for developing effective interventions. This study focused on two distinct dietary patterns: the inflammatory potential of the diet, as assessed by the energy-adjusted Dietary Inflammatory Index (E-DII), and a healthy plant-based dietary pattern, captured by the healthful Plant-based Diet Index (hPDI). The DII and its energy-adjusted version, the E-DII, developed by Shivappa et al., are well-established indices to assess the inflammatory potential of dietary components.4,5 They have been validated for their association with several inflammatory markers6–8 and metabolic and cardiovascular risk factors. 9 Recent research has linked the DII with the risk of sarcopenia and age-associated adverse effects such as disability and frailty in older populations. 10 Similarly, the hPDI, assessing the quality of a plant-based diet, has been associated with age-related ailments, including obesity, 11 cardiovascular events, 12 kidney disease, 13 and T2DM. 14
Gut microbiota play a crucial role in healthy aging and, conversely, are influenced by age-related physiological changes in the gastrointestinal tract alongside environmental factors. The gut microbiota of older individuals display a reduced microbial diversity, a decrease in short-chain fatty acid-producing bacteria, and changes in dominant bacterial genera, which can impact overall health. 15 Diet affects gut microbiota characteristics across different age groups, including older adults. 16 While research has linked diet patterns like the Mediterranean diet to health outcomes and gut microbiota composition, 17 studies particularly examining the relationship between the E-DII, hPDI, and microbiota in aging populations are limited. The Multiethnic Cohort–Adiposity Phenotype Study observed positive associations between the DII and total fat mass, mediated by specific bacterial taxa in a population aged 60–77 years. 18 Shen et al. reported that a diet rich in healthy plant-based foods positively correlates with microbiota alpha diversity and a higher abundance of polysaccharide-degrading bacterial species in an older population. 19
Given the limited number of studies exploring the correlation between E-DII or hPDI, gut microbiota, and health parameters within a healthy older population, our study aims to investigate associations between the diet indices and the composition of the gut microbiome. Additionally, we explore the potential involvement of the gut microbiota in mediating the relationships of the E-DII and hPDI with various markers associated with age-related ailments. Our study population focuses on home-dwelling individuals without severe pre-existing chronic conditions, allowing us to prioritize preventive measures for promoting healthy aging. By employing two indices with different focuses and assessing a broad range of health parameters, our study aims to provide insights into the interplay among diet, gut microbiota, and overall health in older adults.
Methods
Study design and participants
This study presents a secondary, exploratory cross-sectional analysis of baseline data from a double-blind, randomized, placebo-controlled trial conducted at the Institute of Food and One Health, Leibniz University of Hanover, Germany. The primary aim of the original trial was to evaluate and improve the status of essential nutrients in older adults. The trial included 134 home-dwelling participants aged ≥70 years. Full details of the study design and participants have been published elsewhere.20,21 Eligibility was determined through a phone interview, excluding individuals with a BMI >35 kg/m2, severe gastrointestinal or cardiovascular diseases, or those on immunosuppressants or chronic corticosteroids. Eligible participants attended a comprehensive clinical examination day, which included blood sampling, anthropometric measurements, and the completion of health questionnaires. Additionally, participants were instructed to provide a stool sample and complete a 3-day food diary in the week preceding the clinical examination.
The study adhered to the guidelines of the Declaration of Helsinki (revised version, October 2008, Seoul, South Korea) and was registered in the German Clinical Register (DRKS00021302). Written informed consent was obtained from all participants prior to their enrollment.
This present analysis includes data from 129 participants who provided a fecal sample before the start of the intervention (Figure 1). Flowchart of the study population. E-DII: energy adjusted Dietary Inflammatory Index; hPDI: healthful Plant-based Diet Index.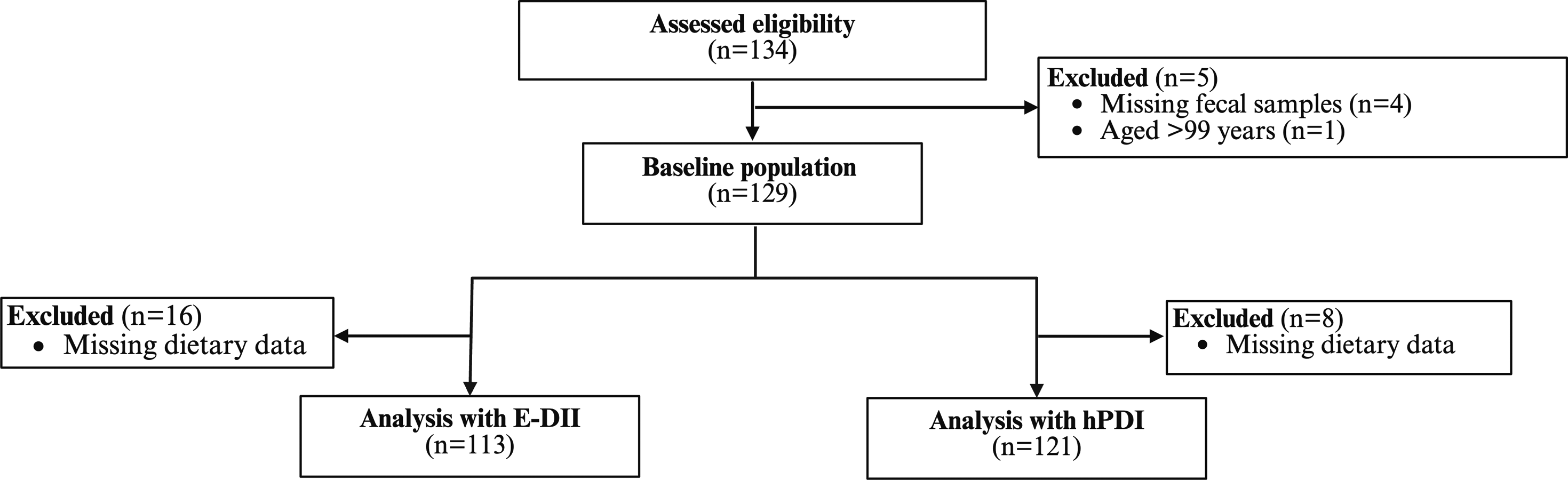
Clinical investigation day
On the clinical investigation day, participants arrived fasted (no food and caloric drinks for at least 12 hours prior) at the university. Blood samples were collected through venipuncture of the arm vein using Safety-Multifly® needles (Sarstedt, Nümbrecht, Germany). All samples were promptly stored at approximately 5°C and transferred to external laboratories on the same day or stored at −80°C for subsequent analyses. After the blood draw, measurements of body weight and height (Seca GmbH & Co. KG, Hamburg, Germany), waist and hip circumferences, and body composition were conducted. Analyses of body composition included fat mass (FM), lean body mass (BLM), total body water (TBW), and phase angle (PA), using an eight-point bioelectrical impedance analyzer (BIA, mBCA525, Seca Company, Hamburg, Germany). Participants completed a questionnaire regarding their medical history, medication use (frequency and dosage), and present health status. An optional assessment for cognitive impairment was conducted using the Saint Louis University Mental Status examination (SLUMS). 22 Physical function was evaluated using the Physical Functioning Scale of the 36-Item Short-Form Health Survey (SF-36). 23
Dietary intake and dietary indices
Participants documented their dietary intake through 3-day food protocols, encompassing two consecutive weekdays and one weekend day. A trained nutritionist analyzed the recorded dietary information using PRODI6.4® dietary software, aligning with the German Federal Food Code 3.02 by Nutri-Science GmbH in Freiburg, Germany. To assess distinct dietary patterns, we chose two indices: the energy-adjusted Dietary Inflammatory Index (E-DII) and the healthful Plant-Based Diet Index (hPDI).
E-DII
The DII is a population-based dietary index developed from existing literature to quantify the overall inflammatory potential of an individual’s diet. Detailed information about the development and scoring algorithm is available elsewhere.4,5 To calculate the DII for each participant, food and nutrient intake data from dietary protocols were linked to the corresponding inflammatory effect scores designated in the DII. A z-score was created for each food component and converted to a centered proportion score. We employed 30 out of a potential 45 components to derive the DII score for our analytical purposes. The included parameters were alcohol, vitamin B12, vitamin B6, β-carotene, caffeine, carbohydrate, cholesterol, energy, total fat, fiber, folate, iron, magnesium, monounsaturated fatty acid (MUFA), niacin, omega-3 fatty acid, omega-6 fatty acid, onion, protein, polyunsaturated fatty acid (PUFA), riboflavin, saturated fat, thiamin, vitamin C, vitamin D, vitamin E, zinc, and green/black tea. Recognizing the link between overall energy consumption and inflammation, we applied the nutrient residual model described by Willet et al. 24 to adjust the nutrient intake date for energy, thereby calculating the E-DII. A higher E-DII score indicates a more pro-inflammatory diet, while a lower score suggests a diet with a more anti-inflammatory profile.
hPDI
The hPDI was calculated according to Satija et al. 25 Food items were grouped into 18 groups based on nutritional and culinary similarities by summing intakes of all food items in each food group. Groups were divided into healthy plant foods (e.g., whole grain, fruits, and vegetables), less healthy plant foods (e.g., sugar-sweetened beverages or refined grains), and animal food groups (e.g., animal fats, dairy, and eggs). The food groups (servings/day) were categorized into quintiles, and each quintile was assigned a score between 1 and 5. Participants received a score of 1–5 for each healthy plant food group (1 for the lowest quintile, 5 for the highest), and a reversed score for animal and unhealthy plant food groups (1 for the highest quintile, 5 for the lowest). The scores were summed across the 18 food groups to derive the hPDI score, ranging from 18 to 90. A higher hPDI score reflects a higher intake of healthy plant-based foods, a lower intake of animal products, and less healthy plant-based food. 25
Biochemical analysis
The analytical procedures for 25-hydroxycholecalciferol (25-(OH)D), holotranscobalamin (holoTC), homocysteine, and red blood cell (RBC) folate were conducted according to the methodologies outlined in the works of Kerlikowsky et al. 20 and Savic-Hartwig et al. 21 Glucose, creatinine, uric acid, triglycerides, high-density lipoprotein (HDL), low-density lipoprotein (LDL), and cholesterol concentrations were determined using a photometric method (Beckman Coulter GmbH, Krefeld, Germany). HbA1c analysis was performed using high-pressure liquid chromatography (Bio-Rad Laboratories GmbH, Feldkirchen, Germany). Ferritin concentration was assessed using the chemiluminescence immunoassay method (CLIA) with DXI800 instrumentation (Beckman Coulter GmbH, Krefeld). Transferrin and CRP levels were measured using turbidimetry (Beckman Coulter GmbH, Krefeld). Insulin concentrations were determined employing the electrochemiluminescence immunoassay method (ECLIA) with cobas 801e instrumentation (Roche Diagnostics GmbH, Mannheim, Germany). The Omega-3 index (O3I) was determined by calculating the sum of eicosapentaenoic acid (EPA) to docosahexaenoic acid (DHA) in total red blood cell membranes relative to total fatty acid content, using dry blood spots and gas chromatography in a certified laboratory (OmegaQuant Analytics, Sioux Falls, SD, USA). Further methodological details are described elsewhere. 26
Fecal sample collection and 16S rRNA gene sequencing
Stool samples were collected at home before the examination using a fecal collection kit from Süsse Labortechnik, Gudensberg, Germany. The collection tubes contained 3.5 mL of RNASepar stabilizer. Upon arrival at the university, fecal samples were promptly stored at −80°C. Participants documented stool consistency using the Bristol Stool Chart (BSC). 27 DNA was extracted using the ZymoBIOMICS Miniprep Kit (ZYMO, USA). Amplification of the V3/V4 region and sequencing on Illumina MiSeq (2 × 300 bp) were conducted as previously described. 28 Sequences were processed via the DADA2 pipeline (v1.20) and annotated based on RDP’s taxonomy. Chimera was eliminated, and sequences with counts ≥5 that were annotated at the phylum level were included in follow-up analyses. All raw data are available at the European Nucleotide Archive (PRJEB74908).
Statistical analysis
Justification of sample size
The sample size for the original randomized controlled trial was based on multiple nutritional biomarkers (RBC folate, 25(OH)D, holoTC, O3I), with the parameter requiring the largest sample size used for the calculation. Assuming a two-sided significance level of 0.05, a statistical power of 80%, and a 10% dropout rate, 120 participants (60 per group) were required. For this exploratory secondary analysis, the sample size of 129 participants reflects those who met the inclusion criteria and provided fecal samples at baseline.
Associations between diet indices and health outcomes
We examined associations between E-DII and hPDI and health outcomes using multiple linear regression models with the
Associations between dietary indices, health outcomes, and gut microbiota composition
We processed the gut microbiota data using the absolute abundance table of amplicon sequencing variants (ASVs) and analyzed sequences matched to taxa and health outcomes through the
To investigate associations among taxa abundances, diet indices, and health outcomes, we employed
Mediation analysis
We conducted a mediation analysis using the Mediation model: The relation between dietary indices and metabolic biomarkers through the indirect effect of gut microbiota genera.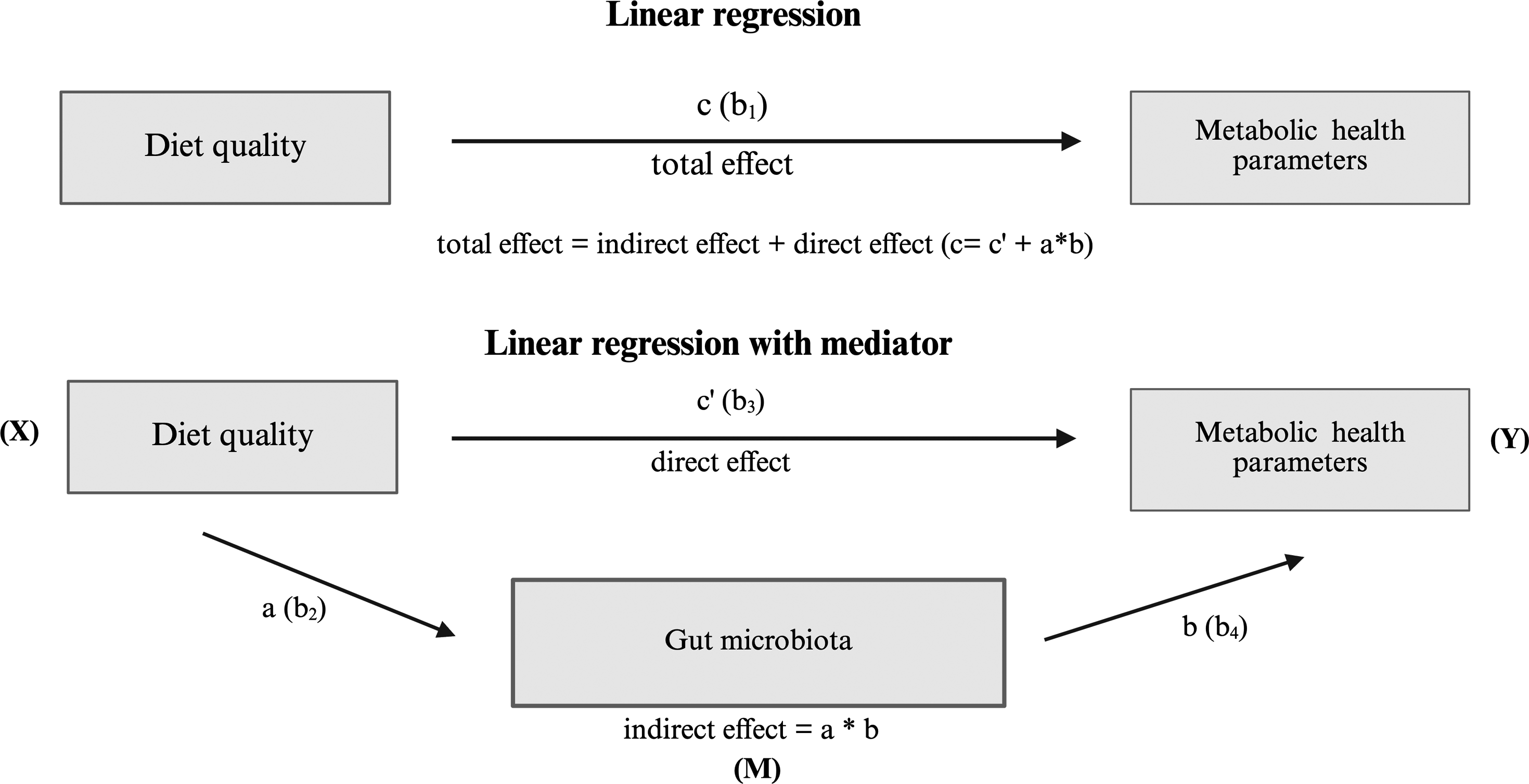
All analyses, including microbiome data, were adjusted for the same covariates as the models examining associations between the indices and health markers, with the additional adjustment for BSC. Visualizations were created using the
Results
Characterization of the study population
A total of 134 eligible participants were enrolled in the study, with five individuals subsequently excluded due to the lack of fecal samples. One participant aged 100 years was further excluded from the analyses due to extreme outlier status within our predominantly 70- to 80-year-old cohort, which disrupted statistical models. The final cohort comprised 129 participants, 36 males and 93 females. Sixteen participants were excluded from the calculation of E-DII and eight from the hPDI calculation due to incomplete dietary data (Figure 1).
Detailed anthropometric, demographic, lifestyle, and health behavior characteristics of the baseline study population have been comprehensively documented and reported by Kerlikowsky et al.
20
Supplemental Table 1 provides additional characterization of the study population concerning the metabolic health markers and dietary indices used in this study. The median and interquartile range (IQR) for E-DII was 0.17 (−0.62, 1.13), and for hPDI 56 (50, 60). Linear regression analysis showed a strong negative correlation between E-DII and hPDI (β = −0.55, 95% CI: −1.57, 0.47,
Associations between diet indices and health biomarkers
Associations between dietary indices, main characteristics, and metabolic biomarkers.
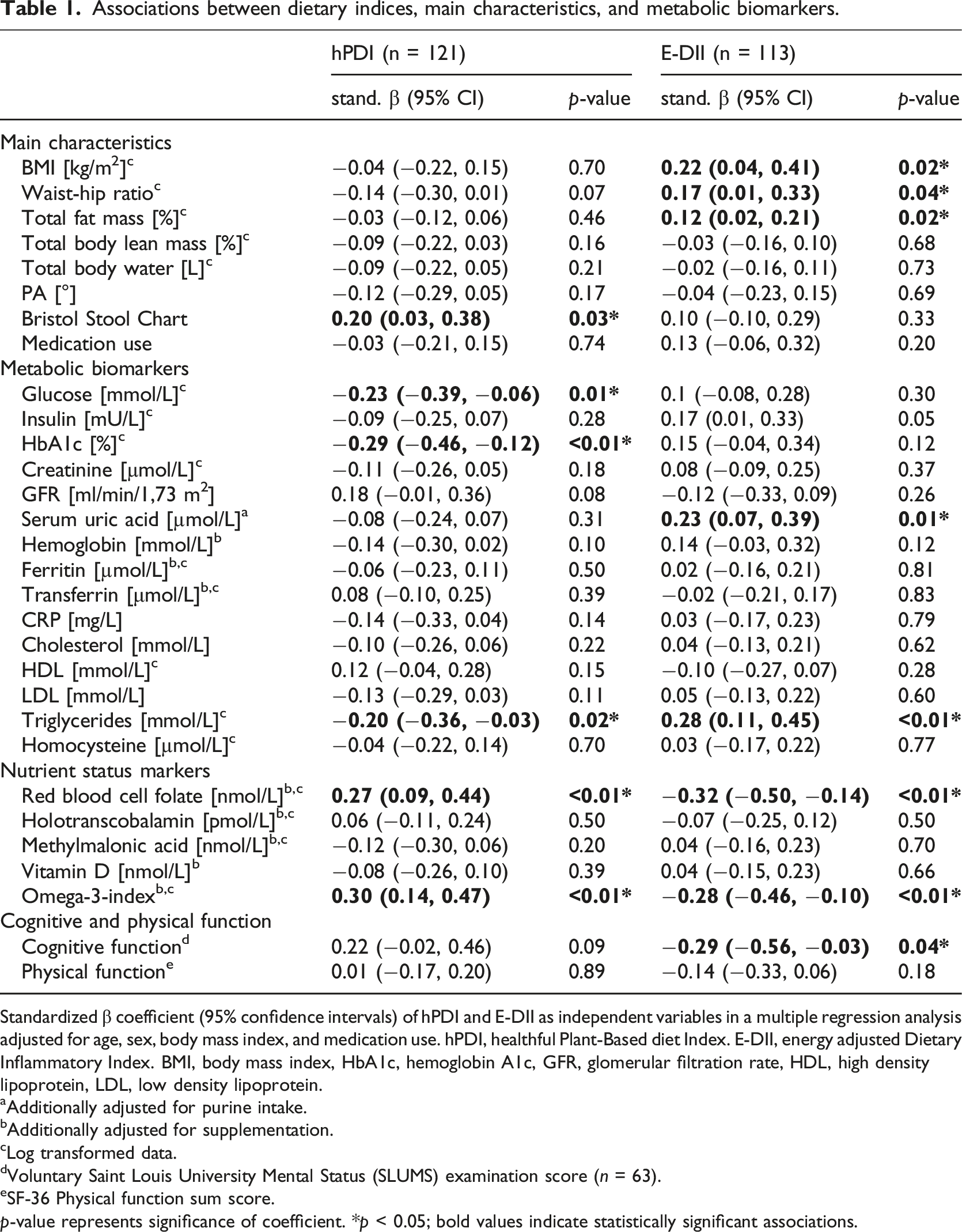
Standardized β coefficient (95% confidence intervals) of hPDI and E-DII as independent variables in a multiple regression analysis adjusted for age, sex, body mass index, and medication use. hPDI, healthful Plant-Based diet Index. E-DII, energy adjusted Dietary Inflammatory Index. BMI, body mass index, HbA1c, hemoglobin A1c, GFR, glomerular filtration rate, HDL, high density lipoprotein, LDL, low density lipoprotein.
aAdditionally adjusted for purine intake.
bAdditionally adjusted for supplementation.
cLog transformed data.
dVoluntary Saint Louis University Mental Status (SLUMS) examination score (
eSF-36 Physical function sum score.
Microbiota composition in relation to dietary indices and metabolic biomarkers
No significant associations were found between the dietary indices and the alpha diversity index Shannon (E-DII: β = −0.02, 95% CI: −0.20, 0.17, Principle coordinate analysis (PCoA) of all samples using Bray-Curtis dissimilarity and quantified by Permutational Multivariate Analysis Of Variance (PERMANOVA). (a) PCoA based on energy adjusted Dietary Inflammatory Index. (b) PCoA based on healthful Plant-based Diet Index. (c) Proportion of variation in taxonomy explained by diet indices, dietary factors, biomarkers and covariates measured by PERMANOVA (based on Bray-Curtis dissimilarity). Analyses were adjusted for age, sex, BMI, medication use, and Bristol Stool Chart. E-DII: energy adjusted Dietary Inflammatory Index; hPDI: healthful Plant-based Diet Index; HbA1c: hemoglobin A1c; BMI: body mass index. 1Voluntary Saint Louis University Mental Status (SLUMS) examination score (
While neither E-DII nor hPDI emerged as major drivers of overall variation in the gut microbiota, PERMANOVA analysis indicated that specific dietary factors significantly accounted for variance in microbiota composition. The consumption of the food groups nuts, whole grains, and sweets and desserts (R2 = 0.01,
After identifying variables significantly associated with the dietary indices and contributing to variation in microbiota composition, we examined specific bacterial genera potentially correlating with these variables. Filtering for abundance (≥1%) and prevalence (≥10%) retained 52 genera for inclusion in linear mixed model analysis. Confounder-adjusted MaAsLin2 analysis did not reveal significant associations between the dietary indices and individual bacterial genera ( Relationships between, dietary patterns, microbial genera, and blood biomarkers. Heatmap colors represent coefficients from linear mixed models in MaAsLin2. Models adjusted for age, BMI, Bristol Stool Chart, medication use, and sex. Additional adjustment included energy intake,1 purine intake,2 and folate supplementation.3 Genera were filtered ≥10% prevalence and ≥1% abundance. Statistical significance was determined using the Benjamini-Hochberg method for multiple comparison adjustment, with a target rate of 0.25 for Q-values. *Q < 0.25.
Investigating the relationship between dietary indices, health parameters, and microbiota composition, we found associations, particularly between E-DII and RBC folate, as well as serum uric acid concentration and specific bacterial genera. To explore whether the microbiota influenced the impact of E-DII on folate and uric acid, we performed mediation analyses. Using the 52 genera identified after prevalence and abundance filtering in MaAsLin2 analysis, we filtered for undefined taxa, resulting in 44 genera for mediation analysis with a focus on genera associated with folate (
In our mediation models:
The study discovered a direct effect of E-DII on serum uric acid, considering the
Additionally, a direct effect of E-DII on RBC folate was observed, controlling for Mediation analysis of the association between energy adjusted Dietary Inflammatory Index and red blood cell-folate concentration and serum uric acid concentration. E-DII: energy adjusted Dietary Inflammatory Index; RBC folate: red blood cell folate. Figure presents the average direct effect (ADE) and average causal mediation effects (ACMEs). *
Discussion
This study investigated the associations between distinct dietary patterns, evaluated through the E-DII and hPDI, and microbiota composition and the potential mediating role of gut microbiota in linking dietary patterns to various health markers in older adults. While we did not observe direct correlations between the dietary indices and fecal microbial diversity, significant associations emerged between both indices and diverse health outcomes, encompassing body composition, metabolic biomarkers, cognitive function, and nutritional status markers. Notably, some of these associations significantly explained variations in microbial community composition. Furthermore, specific microbial genera were associated with serum uric acid and RBC folate concentrations, indicating potential independent effects beyond dietary factors, as shown by mediation analysis.
Numerous studies underscore the intricate interplay between dietary patterns and disease susceptibility, particularly through their influence on age-related changes in gut microbiota, such as reduced microbiota diversity and shifts from beneficial to pathogenic bacteria and metabolites.15,16 For example, the Mediterranean diet has been linked to mitigating aging processes by modulating the gut microbiota composition and promoting the production of short-chain fatty acids (SCFAs).
17
However, the potential associations between healthy plant-based diet patterns and anti-inflammatory diet patterns with the microbiota of older individuals remain largely unexplored. In this study, neither the E-DII nor the hPDI demonstrated significant associations with alpha diversity or substantial explanatory power concerning overall microbiota composition, contrasting with previous research, mainly due to different age demographics or underlying chronic conditions.41–43 For instance, the Multiethnic Cohort Adiposity study, which included 1655 individuals aged 60–77 years, highlighted positive associations between the E-DII and total fat mass, mediated by specific bacterial taxa like
Interestingly, despite the lack of direct correlations between diet indices and microbiota composition, specific dietary components, body composition markers, and metabolic, nutritional, and cognitive function markers appeared pivotal in explaining microbial composition variance. However, our differential abundance analysis revealed associations with only a few variables and microbial genera. Nevertheless, the analysis benefited from the robustness of the methodology, which accounted for confounders, prevalence, and abundance filtering and used a linear mixed model tailored to microbial community data.
We observed a positive link between the consumption of nuts and the abundance of
We also observed a positive association between the abundance of
In addition to food components, RBC folate was positively associated with the abundance of
Fecal cultures were incubated anaerobically for 24 hours, and folate concentrations were measured before and after. The increase in folate concentration was positively correlated with the abundance of
Besides RBC folate concentration, serum uric acid concentration was negatively associated with
Our study has limitations that warrant consideration. Firstly, relying on self-reported dietary data and short-term food protocols introduces the potential for inaccuracies in calculating dietary indices. We could not calculate all components of the E-DII, including some phenolic derivatives, which could influence microbiota composition. Nonetheless, the E-DII remains a valid instrument for assessing the inflammatory potential of the diet, even when fewer than 30 components are included in the calculation. 4 Additionally, the study’s limited sample size and cross-sectional design restrict its ability to establish causal relationships and discern long-term effects. Another limitation is using single time-point stool samples rather than multiple samples collected over time from the same individuals. This approach may not fully capture temporal variability in gut microbiota composition, potentially leading to an underestimation of the strength of the association between dietary factors and microbial populations. Furthermore, stool samples have inherent limitations, such as incomplete separation of fecal bacteria and intestinal flora that cannot be eliminated and limited information about the microbiota of the small intestine or mucosa-associated microbiota. 56 Moreover, 16S rRNA sequencing provides only genera-level resolution, which may obscure finer taxonomic distinctions within the microbiota. The study also needs detailed information on potential metabolites and metabolic pathways that could influence health outcomes, leaving hypotheses regarding the involvement of specific microbial genera in metabolic processes without definitive evidence. Finally, this study represents a secondary, exploratory analysis of baseline data from a randomized trial. Therefore, no dedicated power calculation was performed for the specific outcomes of this analysis, since the sample size was originally determined for the primary endpoint of the intervention study. Nevertheless, the integration of dietary pattern scores, microbiota profiles, and a broad range of metabolic and cognitive health markers in a healthy, older population provides a valuable contribution to the existing literature. Studies combining these interrelated domains in this age group remain scarce, underlining the relevance of our findings for future aging and microbiome research.
In conclusion, by examining a wide range of health markers, including body composition, metabolic and cognitive parameters, and nutritional status indicators, while accounting for various confounders, this study offers an integrative view of the interplay between distinct dietary patterns, health, and gut microbiota in healthy older adults. Although no associations were found between microbiota composition and the dietary indices, nor was a mediating effect of the microbiome observed, our results confirm previously reported associations between dietary pattern and health markers. Importantly this study extends existing knowledge to a well-characterized cohort of community-dwelling older adults without chronic diseases, a group that is often underrepresented in microbiome and nutrition research.
Our study suggests that a more anti-inflammatory and healthful plant-based diet correlates with favorable metabolic, cognitive, and nutritional status among older adults. Moreover, the observed associations between uric acid and RBC folate concentration with specific microbial genera suggest potential roles in folate and uric acid metabolism. Future research using advanced sampling methods, multi-omic integration, and longitudinal designs are needed to further elucidate the complex interaction between diet, gut microbiota, and healthy aging.
Supplemental Material
Supplemental Material - Exploring the relationship between dietary indices, gut microbiota, and health status in healthy, older individuals
Supplemental Material for Exploring the relationship between dietary indices, gut microbiota, and health status in healthy, older individuals by Madeline Bartsch, Felix Kerlikowksy, Andreas Hahn, Shoma Berkemeyer, Sabrina Woltemate, Marius Vital, Mattea Müller in Nutrition and Healthy Aging.
Footnotes
Acknowledgements
We would like to thank the participants who contributed their time to this study.
Author’s Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially funded by food federation Germany (Lebensmittelverband Deutschland e.V.), Claire-Waldoff-Straße 7, 10117 Berlin. The sponsor had no role in the design, collection, analysis, or interpretation of the data, the writing of the manuscript, or the decision to publish the results. MB was supported by the CARLA Talent Academy – Health and Living of Osnabrück University of Applied Sciences. MM was supported by the Joachim-Herz-Foundation (Add-on Fellowship for interdisciplinary Life Sciences).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
