Abstract
Hyponatremia is the most common electrolyte disorder in patients with cancer and is associated with significant morbidity and mortality. Innovation in cancer therapies has led to substantial improvement in cancer outcomes, but also to new therapy-related toxicities, including electrolyte disturbance. Improvement in clinicians understanding of hyponatremia may mitigate adverse outcomes and improve quality of life in cancer patients. In this case-illustrated review, we discuss the mechanisms underlying drug-induced hyponatremia both in “classical” antineoplastic drugs and novel cancer therapies. Via these clinical cases, we describe hyponatremia caused by conventional chemotherapies (e.g. platinum compounds, vinca alkaloid, and alkylating agents) as well as hyponatremia related to tyrosine kinase inhibitors and other targeted therapies. We also focus on checkpoint inhibitors-induced hyponatremia, as these agents are increasingly used for a wide variety of malignancies. Lastly, we summarize therapy-related hyponatremia among recipients of newer treatments for multiple myeloma.
Introduction
Hyponatremia, defined as a plasma sodium concentration lower than 135 mmol/L, is the most common electrolyte disorder found in hospitalized patients. 1 Its prevalence in cancer patients is even higher, affecting nearly half of patients. 2 Moreover, hyponatremia in cancer patients has been reported to negatively correlate with performance status and cancer outcomes, with prolonged hospital stays and increased mortality.2–5 In a recently published study, the prognostic impact of hyponatremia was assessed in 1025 hospitalized patients with metastatic solid tumors, of whom 279 (27%) were hyponatremic. 6 With a median follow-up of nearly 27 months, median overall survival was 2 months for hyponatremic patients compared to 13 months for control patients (p < 0.001). These results are even more remarkable given that the median sodium level of the hyponatremic cohort was 133 (IQR 131–134) mmol/L, and thus considered mild hyponatremia. Careful monitoring of hyponatremia in cancer patients is therefore necessary, particularly in patients receiving cancer treatment known to induce hyponatremia. Innovation in cancer treatments in the past decade have led to improvements in cancer outcomes, but also to new toxicity profiles. This review aims to provide a current portrait of drug-induced hyponatremia in cancer patients in the clinical setting. Both classical drugs associated with hyponatremia and newer anticancer therapies will be described in this case-illustrated review (Table 1).
Mechanisms of hyponatremia associated with cancer therapy.
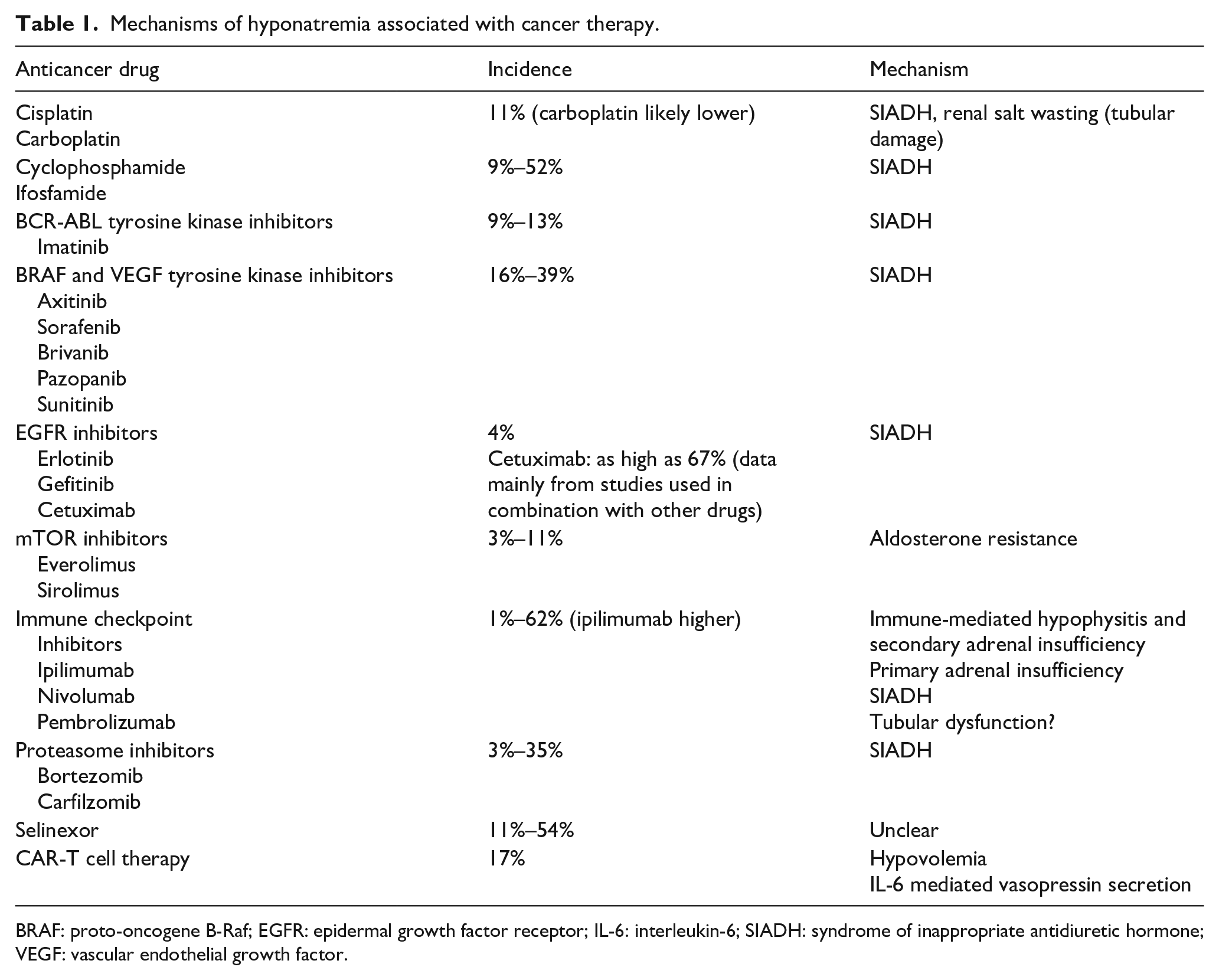
BRAF: proto-oncogene B-Raf; EGFR: epidermal growth factor receptor; IL-6: interleukin-6; SIADH: syndrome of inappropriate antidiuretic hormone; VEGF: vascular endothelial growth factor.
Case 1: A case of hyponatremia following cisplatin chemotherapy
A 64 year-old man with small cell lung cancer was treated with cisplatin chemotherapy with etoposide and concurrent chest radiotherapy. Serum sodium was 137 mmol/L prior to initiation of chemotherapy. He completed four cycles of therapy (with cumulative cisplatin dose of 300 mg/m2) with mild nausea, but no emesis or diarrhea. After the fourth cycle, he was noted to have presyncopal symptoms and significant orthostatic hypotension. A repeat serum sodium was 124 mmol/L, serum potassium was 3.4 mmol/L, and serum magnesium was 0.57 mmol/L. Urine sodium concentration was 143 mmol/L and urine osmolality was 484 mOsm/kg. Serum creatinine was 1.5 mg/dL and blood urea nitrogen (BUN) was 15 mg/dL (from 0.9 to 8 mg/dL pre-treatment, respectively). Clinical examination was suggestive of mild hypovolemia. Serum cortisol and thyroid function testing were normal. A presumptive diagnosis of renal salt-wasting syndrome (RSWS) was made. Treatment with intravenous normal saline resulted in initial resolution of orthostatic symptoms, with improvement in serum sodium to 129 mmol/L. Hypomagnesemia was corrected with intravenous supplementation. Following withdrawal of intravenous fluid replacement, mild orthostasis recurred. Oral salt tablets and increased fluid intake allowed for resolution of orthostatic hypotension, return of serum creatinine to baseline and normalization of serum sodium to 138 mmol/L 2 weeks later.
Hyponatremia related to platinum chemotherapy
Hyponatremia has been a recognized complication of platinum-based chemotherapy for decades.7,8 There is limited data to quantify the overall incidence of hyponatremia in patients receiving platinum-based anticancer agents; however, in a recent retrospective study of 472 patients who were treated with cisplatin for various cancers, 8.9% and 1.7% developed grade 3 (serum sodium 120–129 mEq/L) and grade 4 (<120 mEq/L) hyponatremia, respectively. 9 Risk factors for grade 3 and 4 hyponatremia determined via multivariable regression included the presence of small cell lung cancer (adjusted odds ratio, 3.26; 95% confidence interval (CI), 1.07–10.00)) and lower baseline serum sodium (<138 mEq/L, (adjusted odds ratio, 6.18; 95%CI, 3.21–11.90)). Carboplatin and oxaliplatin may have a lower incidence of hyponatremia relative to cisplatin containing regimens. 10 Of course, malignancy-specific risks of hyponatremia may be difficult to distinguish from treatment-related factors. In particular, small cell lung cancer has been associated with ectopic arginine vasopressin (AVP) production, whereas head and neck cancers have been strongly associated with malignancy-related syndrome of inappropriate antidiuretic hormone secretion (SIADH). 11 It should be noted in general that many patients with malignancy may be prone to SIADH-mediated hyponatremia as a result of lower osmolar intake, higher fluid intake, and other strong stimuli for ADH (e.g. nausea, pain, etc.) (Box 1).
Two primary mechanisms of hyponatremia have been implicated in patients receiving platinum chemotherapy (Table 2). The more frequent of these etiologies is likely platinum-associated SIADH.7,12 While the exact mechanism underlying SIADH in this setting has not been entirely elucidated, this presentation is distinct from RSWS in that hyponatremia develops within a few days of cisplatin administration, and resolves following chemotherapy cessation. Also, clinically these patients are euvolemic and have urinary findings in keeping with SIADH (e.g. higher relative urine osmolality, and the absence of low urine sodium/fractional excretion of sodium). 12 The other cause of hyponatremia is RSWS, as described in the clinical case above. In this clinical scenario, platinum-chemotherapy related tubular necrosis results in renal sodium, magnesium, potassium, and calcium loss. These patients may have significant volume depletion, with volume responsive orthostasis and substantial requirement for intravenous fluid repletion. 13 Associated hyporeninemic hypoaldosteronism (in the absence of hypokalemia) has also been reported. 14 Experimental studies have also suggested the cisplatin may inhibit the epithelial sodium channel (ENaC), which may also contribute to sodium wasting. 15 The onset may be variable and occur days to months after initiation of platinum-based therapy. Similarly, recovery may be variable, with some patients have persistent RSWS, while other may have resolution in days to months after platinum cessation. 7
Features of renal salt-wasting syndrome (RSWS), syndrome of inappropriate antidiuretic hormone (SIADH) and adrenal insufficiency.

N: normal; RSWS: renal salt-wasting syndrome; SIADH: syndrome of inappropriate antidiuretic hormone.
Essential parameters for hyponatremia workup and diagnosis.

Hyponatremia associated with vinca alkaloid and alkylating agents
The vinca alkaloids, particularly vincristine, have long been recognized to associate with hyponatremia which is mediated via an SIADH mechanism.7,10 The incidence of vinca alkaloid-related hyponatremia has not been well-characterized, but risk may be potentiated by concomitant use of antifungal azoles, which inhibit the metabolism of vincristine. 16 It is hypothesized that vinca alkaloids cause SIADH via toxicity to the hypothalamus and neurohypophysis, which leads to osmoreceptor dysregulation and ADH secretion.17,18 Some case reports have demonstrated elevated serum and urine ADH levels to substantiate this proposed mechanism. Hyponatremia has been described as occurring 1–2 weeks after receipt of vincristine and may present with other associated neurologic features such as paresthesias. 19
Alkylating agents such as bulsulfan, chlorambucil, cyclophosphamide, ifosfamide, and melphalan have also been associated with hyponatremia. Cyclophosphamide, in particular, has been associated with hyponatremia in a wide range of doses.20,21 In one retrospective cohort of 69 patients receiving high-dose cyclophosphamide (>2.5 g), the incidence of hyponatremia (defined as serum sodium <135 mEq/L) was 52%, with 8.9% having symptomatic hyponatremia. 22 Onset has been described in 4–12 h of cyclophosphamide administration and usually resolves within 24–48 h. 7 Again, SIADH has been implicated as the cause of hyponatremia with alkylating agents. In particular, busulfan, ifosfamide, and melphalan have been associated with central SIADH as a result of possible increased hypothalamic release of ADH.23–25 Aggressive intravenous fluid administration accompanying administration of alkylating agents (e.g. to reduce the risk of bladder toxicity) may increase risk of SIADH-mediated hyponatremia. 26
Case 2: A case of hyponatremia in a patient receiving anti-VEGF cancer therapy
A 72-year-old man with a history of clear cell carcinoma of the kidney and prior left nephrectomy was admitted to the hospital with severe hyponatremia; serum sodium of 114 mmol/L. This was associated with the use of maintenance sunitinib, a multi-targeted receptor tyrosine kinase inhibitor approved for use in advanced renal cell cancer, gastrointestinal stromal tumor (GIST) and pancreatic cancer. 27 The serum creatinine was 1.52 mg/dL, slightly increased from the prior baseline of 1.24 mg/dL. Sunitinib had been started 20 months earlier when his renal cell cancer had progressed to stage 4 with metastasis to bone and lung. The urine osmolality was 447 mOsm/kg with a urine sodium level of less than 20 mEq/L. Clinical examination indicated hypovolemia. The clinical history indicated long-standing issues of diarrhea since starting sunitinib therapy, but with a recent worsening coupled together with nausea. Intravenous crystalloid volume replacement was initiated. Sunitinib therapy was held during the admission. Diarrhea slowly improved with the serum sodium levels improving to 134 mmol/L during the seven-day admission. After discharge, the patient was initiated on pazopanib maintenance therapy with better tolerance. No further episodes of hyponatremia has occurred in the subsequent 6 months.
Hyponatremia associated with tyrosine kinase inhibitors and other targeted therapies
Targeted therapies have recently become valuable additions to improving cancer treatment outcomes. These small-molecule or antibody treatments have improved cancer survival while being able to decrease some of the toxicities of traditional chemotherapies by targeting specific biological targets instead of rapidly dividing cells broadly. Receptor tyrosine kinase inhibitors (TKIs), such as imatinib targeting the BCR-ABL oncogene, 28 have become the initial standard therapy for chronic myeloid leukemia (CML). Hyponatremia has been described in early cancer clinical trials. The prevalence of hyponatremia for imatinib 29 in phase 2 clinical trials was 12.5%. To date, the best characterized mechanism of TKI-associated hyponatremia is SIADH.30,31
Agents targeting v-Raf murine sarcoma viral oncogene homolog B (BRAF) have also been associated with hyponatremia. Sorafenib is a multikinase inhibitor of BRAF and c-Raf kinases and of the kinase domain of the vascular endothelial growth factor (VEGF) receptors 2 and 3, platelet-derived growth factor receptor, FLT3, p38 and c-Kit. 32 It’s role in anti-cancer therapy is primarily to alter angiogenesis. Phase II trial of sorafenib in recurrent head and neck cancer demonstrated a 39% prevalence of hyponatremia. 33 Sorafenib as an adjunct to gemcitabine and cisplatin in biliary adenocarcinoma resulted in a hyponatremia prevalence of 16%. 34 A recent review of the FDA adverse event reporting system also demonstrated hyponatremia occurrence with the BRAF inhibitors vemurafenib and dabrafenib. 35
Anti-angiogenesis agents targeting the VEGF pathway have been shown to be associated with hyponatremia as well. Brivanib is a tyrosine kinase dual-inhibitor of fibroblast growth factor (FGF) and VEGF signaling. Brivanib therapy in patients with advanced hepatocellular carcinoma had more frequent hyponatremia (23%) than sorafenib (9%). 36 Hyponatremia was common with pazopanib (31%), an oral angiogenesis inhibitor targeting VEGF, platelet-derived growth factor receptor and c-kit, in patients with locally advanced and metastatic renal cell carcinoma. 37 Axitinib, another inhibitor of VEGF receptor, demonstrated a 9% hyponatremia prevalence in phase 2 trials of advanced non-small cell lung cancer. 38 Sunitinib, as described in the above case, has been associated with hyponatremia. 39 The mechanism of hyponatremia with sunitinib is not clearly established. TKIs are mostly associated with SIADH-related mechanisms as described. The aforementioned clinical case portrays sunitinib-related hypovolemic hyponatremia. Diarrhea is common with several anti-angiogenesis agents, highlighting the importance of careful clinical history together with laboratory investigations for evaluating hyponatremia in this context.
Erlotinib and gefitinib, small molecule inhibitors of epidermal growth factor receptor (EGFR) have a low prevalence (4%) of hyponatremia. 40 However, when cetuximab, a human/mouse monoclonal antibody inhibiting EGFR, was used together with pazopanib in colorectal carcinoma, the prevalence of hyponatremia was substantially higher at 67%. 41
Inhibitors of mammalian target of rapamycin (mTOR) everolimus and temsirolimus have been linked to hyponatremia. The mechanism is not clear, but thought to be due to aldosterone resistance.42,43 Other classes of targeted therapies whereby hyponatremia has been described includes immunomodulators. Levamisole has been shown in case report to cause SIADH-related hyponatremia. 44 Interferon-alpha treatment in CML has also been linked to hyponatremia via similar SIADH mechanisms. 45
Case 3: A case of hyponatremia associated with immune checkpoint inhibitors adverse event
A 79-year-old woman with a history of mucosal melanoma for which a surgical resection was performed 2 years ago presented with recurrent disease and pulmonary metastases. She was started on first-line combined immunotherapy with ipilimumab and nivolumab. Nine days after she received her first dose of immunotherapy, she was admitted for progressive cough and dyspnea and was diagnosed with immune-related pneumonitis. Lab investigation was also significant for low sodium level of 127 mmol/L and a serum creatinine of 2.19 mg/dL (increase from her baseline of 1.25 mg/dL). Urine osmolality was 445 mOsm/kg. Morning serum cortisol level was 388 nmol/L (normal range 101–536 nmol/L). She was treated with IV steroids followed by prednisone 1 mg/kg and her clinical state gradually improved with resolution of her dyspnea and acute kidney injury, and normalization of her serum sodium. Her prednisone was tapered over a 12-weeks period. Due to disease progression, she was restarted on single-agent nivolumab about 1 month later with no complications.
Immune checkpoint inhibitors-induced hyponatremia
Immune checkpoint inhibitors (ICI) are increasingly being used in the treatment of several malignancies. Anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and anti-programmed death-1 (PD-1) act by activating the immune system and enhancing anti-tumor T-cell immunity. 46 Ipilimumab is a monoclonal antibody targeting CTLA-4, while nivolumab and pembrolizumab are directed against PD-1. PD-1 also has a ligand, PD-L1, that is targeted by checkpoint inhibitors atezolizumab, avelumab, and durvalumab. ICI have shown significant clinical benefits in tumor regression and survival for several malignancies including advanced melanoma, lung cancer, renal cell carcinoma, urothelial bladder cancer and lymphomas. 47 However, the robust immune response generated by ICI can be complicated by several autoimmune adverse effects, including dermatitis, pneumonitis, colitis, nephritis, and various endocrinopathies. Immune-related toxicities involving the kidneys, the most described being acute interstitial nephritis, occurs in about 2%–4% of patients.48–51 ICI have also been associated with electrolytes disturbances, the most frequent being hyponatremia. 52 A meta-analysis of 48 clinical trials of PD-1 inhibitor therapy in different cancer types showed an overall hyponatremia incidence of 1.2% and hyponatremia accounted for more than 53% of Grade 3–5 reported electrolytes abnormalities. 48 Another meta-analysis of six randomized clinical trials in patients with advanced non-small cell lung cancer (NSCLC) treated with ICI reported an incidence of hyponatremia of 8.7%, and treatment with ICI was found to increase the overall risk of all-grade hyponatremia (RR 1.78). 53 However, significant heterogeneity existed between studies evaluating anti-PD-1/PD-L1 and anti-CTLA-4 therapy, with the risk of hyponatremia being higher for the latter. This incidence value in NSCLC was also higher than previous reports and might be explained by incidence heterogeneity between cancer types. Recently, Seethapathy et al. published “real-world” data for incidence and risk factors of hyponatremia in a large cohort of 2458 patients receiving ICI therapy and followed for 12 months after initiation of therapy. 54 The overall incidence of any-grade hyponatremia in this cohort was 62%, with 6% of patients developing severe hyponatremia (Na < 124 mEq/L), which is much higher than reported in clinical trial setting. In their adjusted multivariate model, anti-CTLA4 therapy was associated with a higher risk of severe hyponatremia, but malignancy type was not.
The specific mechanism by which ICI increase the risk of hyponatremia remains unclear. Several case reports have described ICI-induced hyponatremia due to hypophysitis and secondary adrenal insufficiency.55–58 In this setting, hypophysitis causes low corticotropin (ACTH) levels and reduced cortisol secretion, and serum sodium is then decreased secondary to increased vasopressin levels resulting from reduced inhibition by cortisol (Table 2). The incidence of immune-related hypophysitis in patients treated with ICI varies between 0.5% and 9% in clinical trials 59 and was 0.3% in a recent large cohort study. 54 A recent meta-analysis of 38 clinical trials with ICI therapy in patients with advanced solid cancers showed an overall hypophysitis incidence of 1.3%. 60 Among these cases, 89% (76/85) occurred in patients with melanoma. The observed incidence was greatest with combination therapy (6.4%), and it was higher with CTLA-4 inhibitors (3.2%) than with PD-1 inhibitors (0.4%) and PD-L1 inhibitors (<0.1%). Since hypophysitis occurs mainly with anti-CTLA4 and is rarer with anti-PD1/PD-L1, this is consistent with the higher incidence of treatment-related hyponatremia reported with anti-CTLA-4 regimen. Moreover, CTLA-4 expression has been demonstrated on pituitary endocrine cells, suggesting a pathogenic role. 61
Though more rare, primary adrenal insufficiency following treatment with ICI has also been described. A meta-analysis of 38 clinical trials revealed an overall primary adrenal insufficiency incidence of 0.7% in patients receiving ICI for advanced solid cancers. 60 Again, incidence was higher in patients on combination therapy (4.2%). Less is known about ICI-induced primary adrenal insufficiency pathophysiology. A case report on hyponatremia in a 43-year-old man with metastatic melanoma on nivolumab revealed primary adrenal insufficiency cause by likely immune-related adrenalitis, with PET scan demonstrating bilateral increased FDG activity in the adrenals. 62 To date, few case reports have been published on this type of immune-related side effect of ICI. As with hypophysitis and secondary adrenal insufficiency, most cases responded to steroids therapy and hormone replacement.
In summary, the presence of unexplained hyponatremia in a patient receiving ICI should prompt further endocrine evaluation to assess for secondary or primary adrenal insufficiency. However, not all cases of ICI-induced hyponatremia are related to immune endocrinopathies. As in our patient case, hyponatremia can be seen in the setting of SIADH in absence of cortisol deficiency. In a recently published large cohort study, SIADH was the most common etiology of ICI-related hyponatremia, accounting for 35% of severe hyponatremia cases. 54 Hyponatremia might also be related to renal tubular dysfunction, as PD-L1 is expressed on renal tubular epithelial cells and may play a role in immunopathology of ICI-induced kidney injury. 63 However, to date, CTLA-4 expression has not been described in kidneys, and incidence rates of ICI-induced acute kidney injury are generally comparable between anti-CTLA-4 and anti-PD-1/PD-L1 regimens. 52
Case 4: A case of hyponatremia associated to selinexor therapy
A 59 year-old man was diagnosed with kappa light chains multiple myeloma with 13q deletion. He was first treated with CYBOR-D chemotherapy followed by autologous stem cell transplant and was kept on maintenance lenalidomide. Unfortunately, his disease progressed and he was subsequently treated with lenalidomide, cyclophosphamide and dexamethasone, daratumumab monotherapy, and carfilzomib-based regimen. Upon last progression, he was started on selinexor 100 mg weekly in combination with bortezomib and dexamethasone. Shortly after his first dose of selinexor, the patient was unwell with abdominal pain, nausea, diarrhea and dizziness. On day 7 after his dose of selinexor, his sodium level was 131 mmol/L in clinic, from baseline 140 mEq/L. His serum creatinine was 1.87 mg/dL, increased from baseline 1.14 mg/dL. Potassium was 3.5 mmol/L and total plasma protein 70 g/L (normal range 65–80 g/L). Unfortunately, urine studies were not performed. Due to the significant side effects, the patient refused to continue with selinexor. On next visit 5 days later his sodium level was back to normal, but his kidney function had worsen due to disease progression with serum kappa light chains at 3248 mg/L. He is currently receiving therapy with pomalidomide-based regimen.
Hyponatremia in multiple myeloma patients
In patients with multiple myeloma, hyponatremia was usually not a common clinical finding. However, several recent case reports have described hyponatremia in multiple myeloma patients receiving proteasome inhibitor bortezomib therapy.64–69 SIADH was the usual hyponatremia mechanism in these case reports. In prior phase 1 and 2 clinical trials assessing efficacy and safety of bortezomib in the treatment of multiple myeloma and other hematologic malignancies, incidence of hyponatremia ranged from 3% to 35%.70–72 In one study hyponatremia was found to be a dose-dependent side effect, substantiating the probable hyponatremic effect of bortezomib. 70 Hyponatremia was also reported with carfilzomib, another proteasome inhibitor, with an incidence around 10% in patients with hematologic malignancies, suggesting a class effect.73,74 However, hyponatremia was not consistently reported in later phase 3 trials with proteasome inhibitors in multiple myeloma patients.75,76 It is likely that proteasome inhibitors-induced hyponatremia may be underreported in the clinical setting, as clinicians are usually not aware of this possible side effect.
Selinexor is an oral selective inhibitor of exportin 1 (XPO1) that has recently been approved in the treatment of relapsed or refractory multiple myeloma and diffuse large B-cell lymphoma. This new therapeutic agent has been strongly associated with hyponatremia. In a phase 2 study with oral selinexor (80 mg twice weekly) and dexamethasone in 123 patients with refractory myeloma, 45 (37%) developed hyponatremia during treatment. 77 Of these, 27 (22%) had grade 3 or 4 hyponatremia, leading to treatment interruption and dose reduction. Another study again using oral selinexor in patients with refractory myeloma reported an overall hyponatremia incidence of 42% (33/79 patients), with 17 patients (22%) presenting grade 3 event. 78 In this study, selinexor-induced hyponatremia was found to be dose-dependent, with an incidence of 35% in patients receiving six doses per cycle and 54% in patients receiving eight doses per cycle. Results were similar in a study in patients with myelodysplastic syndromes and acute myeloid leukemia, with an incidence of 44%. 79 In a study with refractory large B-cell lymphoma population, selinexor-induced hyponatremia appeared to be less frequent, with an overall incidence of 11%. 80 In this study however, the selinexor dose was lower, with a median dose of 50 mg twice weekly. In a meta-analysis of 437 patients who received selinexor in different clinical trials, the overall incidence of hyponatremia was 32% and the median time to onset of hyponatremia was 8 (IQR 5–18) days. 81 The majority of cases, including grade 3 severity, were asymptomatic and reversible. Median time to resolution of hyponatremia was 7 days for patients who were treated with sodium chloride tablets, and 12 days for non-treated patients. In keeping with these results, monitoring sodium level at baseline and throughout treatment in patients receiving selinexor is mandatory. Recently published clinical guidelines recommend to hold selinexor therapy when sodium level falls below 130 mmol/L and resumed one dose level lower when hyponatremia is corrected. 82 Early referral to nephrology for evaluation and treatment of hyponatremia to prevent potential serious complications and avoid other hyponatremic drugs should also be recommended.
The mechanism by which selinexor induces hyponatremia is not well understood and is speculated to be multifactorial. In the majority of published data, no urinary workup was performed. A recently published series of four cases assessed by a nephrology team showed elevated urine sodium and osmolality, consistent with possible SIADH; but also reported nausea, hypovolemia and other medications as possible contributory factors. 83 The potential effect of selinexor on kidney tubular cells as a cause of hyponatremia needs further investigation.
Chimeric antigen receptor T-cell (CAR-T) therapy has been approved in the past 3 years for the treatment of acute lymphoblastic leukemia and aggressive B-cell lymphomas. It is now being studied for the treatment of refractory multiple myeloma. 84 There are very few data regarding CAR-T therapy-associated hyponatremia. A recent retrospective study of 78 adult patients who received CAR-T cells for the treatment of diffuse large B-cell lymphoma was published with the aim of characterizing associated kidney toxicities and electrolyte disorders. 85 This study reported significant hyponatremia (Na < 130 mmol/L) in 13 (17%) patients. Incidence of cytokine release syndrome was 85% in this study, with 30 (45%) patients having clinical manifestations of third-spacing. For this reason, the proposed mechanism of hyponatremia was intravascular volume depletion or IL-6 mediated increase in vasopressin secretion in the context of cytokine release syndrome. Hyponatremia has been previously shown to correlate with the severity of cytokine release syndrome, 86 as well as with the peak serum level of IL-6. 87 Because the number of patients receiving CAR-T therapy is likely to expand in the near future, nephrologists need to be aware of this possible complication.
Conclusion
The clinical cases presented in this review highlight the complexity and multifactorial etiology of hyponatremia in patients with cancer. Antineoplastic drugs may contribute to hyponatremia in these patients via a variety of mechanisms including toxic tubular damages, hypovolemia, SIADH and immune adverse events in the case of ICI therapy. Incidence of hyponatremia may also increase when anticancer drugs are used in combination. Collaboration between oncologists and nephrologists is essential to better understand hyponatremia in cancer patients and mitigate the associated adverse outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
