Abstract
Background:
Immune checkpoint inhibitors (ICIs) are currently the standard of care for metastatic urothelial cancer (mUC) after the failure of previous platinum-based chemotherapy. The choice of further therapy after ICI progression is a new challenge, and scarce data support it. We aimed to examine the outcomes of mUC patients after progression to ICI, especially when receiving chemotherapy.
Methods:
Data were retrospectively collected from clinical records of mUC patients whose disease progressed to anti-programmed death 1 (PD-1)or programmed death ligand 1 (PD-L1) therapy at 14 Italian centers. Patients were grouped according to ICI therapy setting into SALVAGE (ie, ICI delivered ⩾ second-line therapy after platinum-based chemotherapy) and NAÏVE (ie, first-line therapy) groups. Progression-free survival (PFS) and overall survival (OS) rates were calculated using the Kaplan-Meier method and compared among subgroups. Cox regression assessed the effect of treatments after progression to ICI on OS. Objective response rate (ORR) was calculated as the sum of partial and complete radiologic responses.
Results:
The study population consisted of 201 mUC patients who progressed after ICI: 59 in the NAÏVE cohort and 142 in the SALVAGE cohort. Overall, 52 patients received chemotherapy after ICI progression (25.9%), 20 (9.9%) received ICI beyond progression, 115 (57.2%) received best supportive care only, and 14 (7.0%) received investigational drugs. Objective response rate to chemotherapy in the post-ICI setting was 23.1% (28.0% in the NAÏVE group and 18.5% in the SALVAGE group). Median PFS and OS to chemotherapy after ICI-PD was 5 months (95% confidence interval [CI]: 3-11) and 13 months (95% CI: 7-NA) for the NAÏVE group; 3 months (95% CI: 2-NA) and 9 months (95% CI: 6-NA) for the SALVAGE group, respectively. Overall survival from ICI initiation was 17 months for patients receiving chemotherapy (hazard ratio [HR] = 0.09, p < 0.001), versus 8 months for patients receiving ICI beyond progression (HR = 0.13, p < 0.001), and 2 months for patients who did not receive further active treatment (p < 0.001).
Conclusions:
Chemotherapy administered after ICI progression for mUC patients is advisable irrespective of the treatment line.
Keywords
Introduction
Nowadays, the cornerstone of systemic treatment for urothelial cancer (UC) patients is still represented by platinum-based chemotherapy in the first-line setting. 1 For those patients who progress after (or even during) this primary treatment, and for those who are not eligible for cisplatin, immune checkpoint inhibitors (ICIs) represent the new standard of care. 1 Before ICIs approval, salvage therapy for metastatic urothelial cancer (mUC) was a largely unmet clinical need. Single-agent chemotherapeutic approaches, using taxanes or vinflunine, allowed to reach median progression-free survival (PFS) and median overall survival (OS) of approximately 3 and 8 months, with objective response rate (ORR) rarely over 10% and short duration of response.2-4
The advent of anti-programmed death ligand 1 (PD-L1; atezolizumab, durvalumab, and avelumab) and anti-programmed death 1 (PD-1; pembrolizumab and nivolumab) ICI antibodies opened revolutionary roads in the setting of mUC, showing higher response rates and improved survival, with a durable response.5-10 Currently, pembrolizumab is approved by the Food and Drug Administration (FDA) and European Medicines Agency (EMA) for mUC patients who are not eligible for cisplatin-containing chemotherapy and with PD-L1 expression (combined positive score, CPS ⩾ 10), or who are not eligible for any platinum-containing chemotherapy regardless of PD-L1 status (FDA only), as well as for mUC patients with disease progression during or following platinum-containing chemotherapy.9,11,12 Similarly, atezolizumab is approved for mUC patients after prior platinum-containing chemotherapy and for mUC patients who are considered ineligible for platinum-based chemotherapy and whose tumors present PD-L1 expression ⩾5%.5,13
Despite the great benefit obtained from introducing ICI agents in the armamentarium against mUC, a non-negligible proportion of patients still recur or progress after ICIs. In this clinical scenario, the management of the ICI-progressive disease is a new clinical challenge. The benefit of subsequent chemotherapy or ICI beyond progression is yet to be proven, with conflicting evidence reported up today.14-16 The introduction of ICI in the treatment landscape, mainly providing a second-line option, shifted the choice of single-agent chemotherapy in the third-line setting, possibly changing the risk-benefit ratio and the expected activity and toxicity of the drug. On the contrary, chemotherapeutic proposals can be even more challenging for patients progressing to first-line ICI since previously considered as not eligible for platinum-based chemotherapy.
Hypothesizing that chemotherapy can have a role after prior immunotherapy in mUC, we investigated the outcomes of mUC patients who progressed to immunotherapy with anti-PD-1/PD-L1 ICI in the context of salvage therapy (ie, ⩾ second-line therapy after platinum-based chemotherapy) or in the naïve setting (ie, as first-line therapy), analyzing the progression and survival rates according to the subsequent treatment received, and focusing on the subset of patients receiving chemotherapy after ICI.
Patients and Methods
Patient populations
We generated a national platform including posttreatment and follow-up data of all patients with mUC who have progressed to anti-PD-1 or PD-L1 therapy between September 2014 and December 2018, in the context of the MeeT-URO network (Italian Network for Research in Urologic Oncology). Progression to first-line or second-line ICI, irrespectively of ICI continuation or not, was the principal inclusion criterion. Data were retrospectively collected from 14 participating centers in Italy. Information about the immunotherapy compound was blinded (only the class of agent, anti-PD-1 vs anti-PD-L1, was provided). The overall study population was stratified into 2 cohorts: patients who received ICI as first-line therapy (NAÏVE cohort) and patients who received ICI as salvage therapy (SALVAGE cohort). Treatment patterns after progression to ICI were analyzed within these 2 cohorts, with a particular focus on patients receiving chemotherapy. The MeeT-URO 1 study was approved by the Ethics Committee of Fondazione IRCCS Istituto Nazionale Tumori of Milan (Coordinating Institution; Protocol Number INT 136/17). Following the approval from the coordinating institution, ethics committee of each participating center had endorsed the deliberation of the coordinating center. A deailed list of the participating institution is provided Supplementary Table S1.
Outcomes of interest
The study aimed to quantify the rate of patients who received any subsequent therapy after disease progression to ICI to provide elements to drive informed patient selection for other therapies after anti-PD-1 or PD-L1 therapy, to describe the outcome of patients undergoing ICI beyond progression or undergoing best supportive care (BSC) in the NAÏVE and SALVAGE setting and to identify the reasons for not receiving any other active treatment after immunotherapy.
Patients’ outcome to chemotherapy after immunotherapy failure was analyzed in the overall study population and within the 2 cohorts (NAÏVE and SALVAGE). The impact of different clinical variables on the outcome of patients to the subsequent chemotherapy was investigated to explore prognostic factors in this setting.
Statistical analyses
The outcomes of interest were PFS, defined as the time from chemotherapy initiation to progression or death; OS, defined as the time from chemotherapy initiation and death by any cause or last follow-up; the ORR, defined as the rate of partial or complete response as the best response to chemotherapy according to RECIST 1.1 criteria, as per the clinical practice of the participating centers; the disease control rate (DCR), defined as the rate of partial or complete responses plus stable disease. 17
PFS-2, defined as the time from immunotherapy initiation and the second progression (confirmed progression) or death after the first progression, was calculated for the subgroup of patients continuing ICI beyond progression and for the treatment sequence of immunotherapy followed by chemotherapy. OS-2 was also calculated according to the post-ICI PD treatment pattern, defined as the survival time from immunotherapy initiation and death by any cause or last follow-up.
Descriptive statistics included frequencies and proportions for categorical variables. Medians and interquartile ranges (IQRs) were reported for continuously coded variables. For data representation purposes, patient characteristics were reported for the overall cohort and stratified according to the ICI delivering setting groups (NAÏVE vs SALVAGE). The statistical significance of differences in medians and proportions between the groups was respectively tested with the Kruskal-Wallis and chi-square tests. Survival rates (OS and PFS) were analyzed using Kaplan-Meier analyses. According to the setting of ICI delivering, univariate and multivariate Cox regression models examined OS rates after systemic treatments. The factors considered for the univariate and multivariate models were age, sex, chemotherapy administration after ICI progression, smoking status, genitourinary tract of origin of the tumor, prior primary surgery, and baseline Eastern Cooperative Oncology Group performance status (ECOG-PS) at ICI progression.
All statistical tests were 2-sided with a level of significance set at p < 0.05. Analyses were performed using the R software (version 3.6.1, R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
The overall study population included 201 patients who progressed to ICI therapy: 59 in the NAÏVE cohort and 142 in the SALVAGE cohort.
Patient characteristics and comparative data between the 2 cohorts are reported in Table 1. The median age was 63 years, with older patients in the NAÏVE group (65 vs 62 years, p = 0.04).
Characteristics of the overall study population (201 patients) and of the 2 cohorts of NAÏVE and SALVAGE patients (according to the setting of the immune checkpoint inhibitor therapy).

Abbreviations: AC, adjuvant chemotherapy; CHT, chemotherapy; ECOG-PS, Eastern Cooperative Oncology Group Performance Status; ICI, immune checkpoint inhibitor; LTUC, lower-tract urinary cancer; N, number of patients; NA, not available; NAC, neo-adjuvant chemotherapy; TCC, transitional cell carcinoma; UTUC, upper-tract urinary cancer.
p < 0.05.
According to ICI delivering setting, most patients (78.1%) received a single ICI agent (66.7% an anti-PD-L1 agent). Patients who received an ICI combination treatment (all within clinical trials) were 25.4% in the NAÏVE cohort and 16.2% in the SALVAGE group.
Outcome to chemotherapy after prior immunotherapy
The patient population receiving chemotherapy after post-ICI progression of the disease (PD) was 52 subjects overall (25.9%). Of them, 25 belonged to the NAÏVE group and 27 to the SALVAGE cohort.
The ORR to chemotherapy in the post-ICI setting was 23.1% (partial response in all cases) in the overall population. ORR was 28.0% in the NAÏVE group and 18.5% in the SALVAGE group. The response patterns in the overall population and among subgroups are reported in Table 2. The DCR was 50% overall (56.0% in the NAÏVE cohort and 44.4% in the SALVAGE cohort).
Best response (according to RECIST 1.1 criteria) in the overall population of patients treated with chemotherapy after disease progression to immune checkpoint inhibitors (ICI) and within the 2 cohorts receiving chemotherapy after first-line ICI (NAÏVE cohort) or in the salvage setting (SALVAGE cohort).

Abbreviations: N, total number of patients; NE, not evaluable; PD, progression of disease; PR, partial response; SD, stable disease.
Median follow-up time from chemotherapy initiation was 6 months (IQR: 3-9 months), including alive patients only. Median PFS to chemotherapy after ICI-PD was 3.5 months (95% confidence interval [CI]: 3-8; Figure 1A). Similar median PFS to chemotherapy was found for NAÏVE and SALVAGE patient cohorts, respectively of 5 months (95% CI: 3-11) and 3 months (95% CI: 2-not reached [NR], Figure 1B).
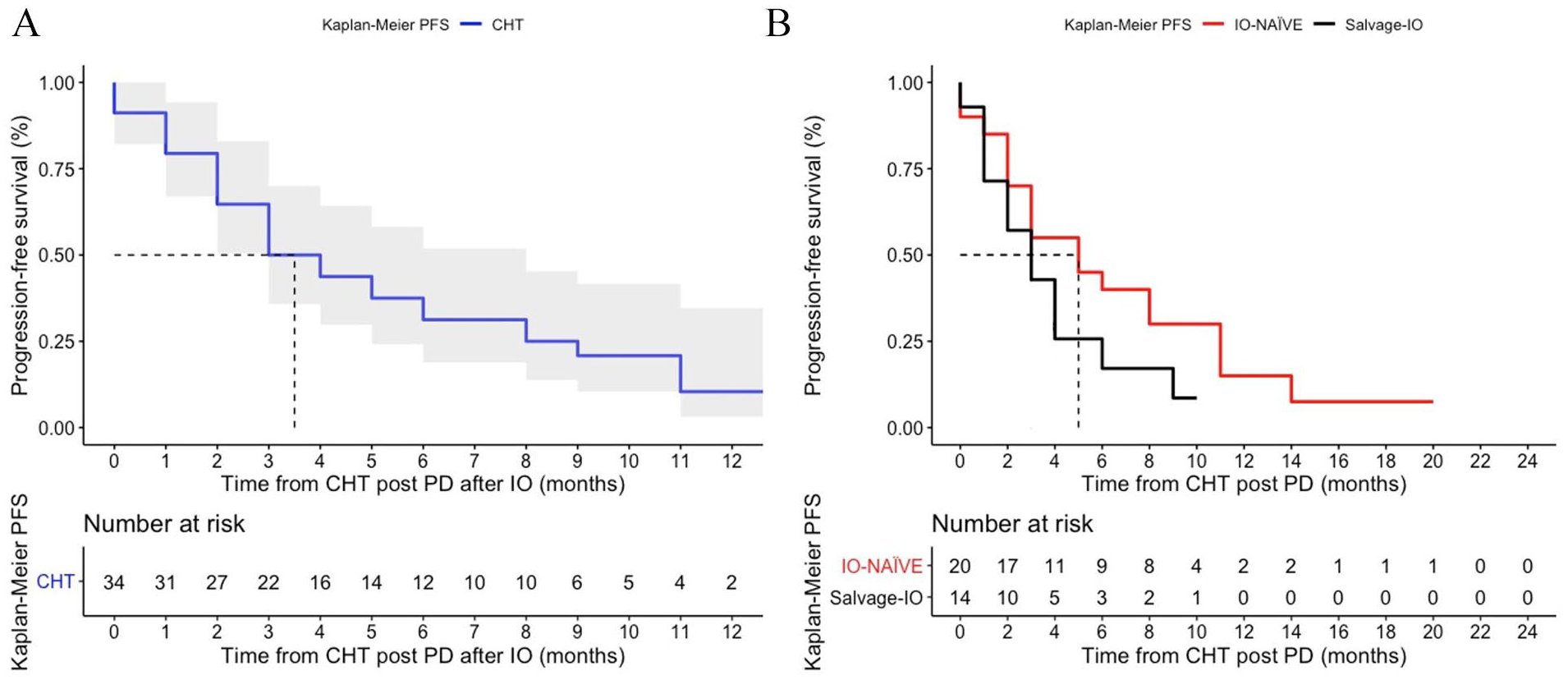
(A) Progression-free survival (PFS) of patients receiving chemotherapy after progression of disease (PD) to immune checkpoint inhibitor (ICI). Only 34 of 52 patients were evaluable for PFS, calculated from chemotherapy initiation, since a great number of patients lacked radiological assessments after chemotherapy initiation, mostly due to rapidly worsening conditions. (B) PFS of patients to chemotherapy received in the NAÏVE and SALVAGE cohort. Twenty and 14 patients were evaluable for PFS, respectively, in the 2 cohorts.
Median OS from chemotherapy after ICI-PD was 9 months (95% CI: 8-NR, Figure 2A). Similar median OS rates were found in NAÏVE and SALVAGE cohorts (Figure 2B), respectively of 13 months (95% CI: 7-NR) and 9 months (95% CI: 6-NR). ECOG-PS was the only significant predictor of progression to chemotherapy (hazard ratio, HR = 38.9, 95% CI: 6-248, P < .001) at the multivariate analysis (Supplementary Table S2) but not for OS. Conversely, the ICI class (anti-PD-1 vs anti-PD-L1), the ICI regimen (combinations vs single-agents), prior response to ICI, and ICI treatment lines were not associated with progression to subsequent chemotherapy.

(A) Overall survival (OS) of patients receiving chemotherapy after progression of disease (PD) to immune checkpoint inhibitor (ICI). Overall, 47 patients were evaluable for OS, calculated from chemotherapy initiation (5 patients were lost at follow-up). (B) OS of patients to chemotherapy received in the NAÏVE and SALVAGE cohort. 24 and 23 patients were respectively evaluable for OS.
Treatment following disease progression to immunotherapy
After PD to immunotherapy, the treatment pattern was heterogeneous across the study population, including patients continuing immunotherapy beyond the first PD, patients discontinuing treatment and not receiving any further active therapy (BSC only), and patients discontinuing ICI but receiving subsequent systemic treatments.
After PD to ICI, beyond the already described 25.9% of patients (52 cases) receiving chemotherapy, 57.2% of patients (115) did not receive any subsequent active systemic treatment, and 9.9% (20 patients) received ICI beyond progression due to clinical benefit; the remaining 6.9% (14 patients) received further active treatments other than chemotherapy, for example, targeted therapies within clinical trials (Table 3). Considering the 2 cohorts of the study, the rate of subsequent treatment received after ICI progression was different since chemotherapy was more frequently administered in the NAÏVE group (42.4% of cases) than in the SALVAGE group (19.0% of patients). The choice of continuing ICI beyond progression was similar in both cohorts; no further objective responses were recorded in patients treated with ICI beyond progression. In SALVAGE patients, the most frequent choice was BSC only (64.8% of cases vs 38.9% for NAÏVE patients).
Treatment received after progression of disease to immune checkpoint inhibitors.

Abbreviations: CHT, chemotherapy; ICI, immune checkpoint inhibitor; N, number of patients; PD, progression of disease.
The variables significantly related to the administration of further active treatments after ICI were as follows: a longer time from ICI initiation to PD (p < 0.001) and a lower number of metastatic sites at ICI initiation (p = 0.002).
Prognostic elements and their impact on survival
The impact of receiving chemotherapy on OS after ICI failure was significant in the overall population both at the univariate and multivariate analyses, with HR = 0.09, 95% CI: 0.05-0.16, p < 0.001 in the multivariate model (Supplementary Table S3).
Also, the impact of chemotherapy was significant in both cohorts at the univariate (HR = 0.26, 95% CI: 0.12-0.56, p < 0.001 and HR = 0.29, 95% CI: 0.16-0.54, p < 0.001, in the NAÏVE and SALVAGE ICI groups, respectively) and in the SALVAGE cohort at the multivariate analysis. In the SALVAGE setting, chemotherapy use was the only significant factor for OS after post-ICI PD (HR = 0.22, 95% CI: 0.10-0.48, p < 0.001, Table 4) at the multivariate analysis. In the NAÏVE cohort, baseline ECOG-PS (at chemotherapy initiation) was the only further variable resulting significantly related to OS in the multivariate analyses (HR = 0.09, 95% CI: 0.03-0.33, p < 0.001) aside from prior primary surgery (HR = 2.96, 95% CI: 1.05-8.34, p = 0.004, Table 4).
Univariate and multivariate Cox regression analyses testing the effect of chemotherapy after disease progression to immune checkpoint inhibitors according to the 2 cohorts of NAÏVE and SALVAGE patients.

Abbreviations: CHT, chemotherapy; CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group Performance Status; it was referred to baseline at chemotherapy initiation; HR, hazard ratio; ICI, immune checkpoint inhibitor; LTUC, lower-tract urinary cancer; PD, progression of disease; UTUC, upper-tract urinary cancer.
p < .05.
In the whole population, the multivariate analysis showed that, aside from receiving chemotherapy or ICI beyond progression, the only further significant predictor of OS was represented by the tumor histology (Supplementary Table S3).
Survival outcomes of patients according to the treatment sequence
Overall, the median follow-up time from ICI start was 7.5 months (IQR: 3-17 months), including alive patients only. Among patients receiving ICI beyond PD, the median PFS-2 (mPFS-2), calculated from ICI start to the second PD (confirmed PD), was 6 months (95% CI: 4-NR, Figure 3A). The mPFS-2 was 10 months (95% CI: 7-13) for patients receiving chemotherapy (Figure 3B).

(A) Second progression-free survival (PFS-2) of patients receiving immune checkpoint inhibitors (ICI) beyond progression of disease (PD), calculated from ICI start to the second PD confirmation. Of 20 patients treated with ICI beyond PD, 17 were evaluable for PFS by RECIST 1.1 criteria. (B) PFS-2 of patients receiving chemotherapy (CHT) after PD to ICI, calculated from ICI start to the second PD (PD to chemotherapy). Of 52 patients treated with CHT after ICI-PD, only 34 were evaluable for PFS. In spite of a likely positive selection bias of patients receiving ICI beyond PD, mPFS-2 was better for patients receiving CHT, suggesting that the sequence of ICI and CHT can be a favorable choice.
In the NAÏVE cohort, patients undergoing chemotherapy reached median OS (calculated from ICI-PD) of 15 months (95% CI: 9-NR), versus 1 month (95% CI: 1-10) for patients without chemotherapy after ICI-PD (p < 0.001, Figure 4A).

(A) Overall survival (OS) of patients in the NAÏVE cohort (59 patients) according to the treatment received after progression of disease (PD) to immune checkpoint inhibitor (ICI). There were 25 evaluable patients receiving chemotherapy (CHT) and 32 evaluable patients not receiving CHT; OS was calculated from ICI-PD. (B) OS of patients in the SALVAGE cohort (142 patients) according to the treatment received after progression of disease (PD) to immune checkpoint inhibitor (ICI). There were 27 evaluable patients receiving chemotherapy (CHT) and 105 evaluable patients not receiving CHT; OS was calculated from ICI-PD.
In the SALVAGE cohort, patients undergoing chemotherapy reached median OS (calculated from ICI-PD) of 10 months (95% CI: 9-NR), versus 1 month (95% CI: 1-2) for patients without chemotherapy after ICI-PD (p < 0.001, Figure 4B).
Considering the survival time from immunotherapy initiation, namely median OS-2 (mOS-2), it was 16 months (95% CI: 13-NR) for patients receiving chemotherapy post-ICI, versus 8 months (95% CI: 6-NR) for patients receiving ICI beyond PD, versus 2 months (95% CI: 2-3) for patients not receiving further active treatment, with significant survival difference in favor of chemotherapy (p < 0.001, Figure 5).
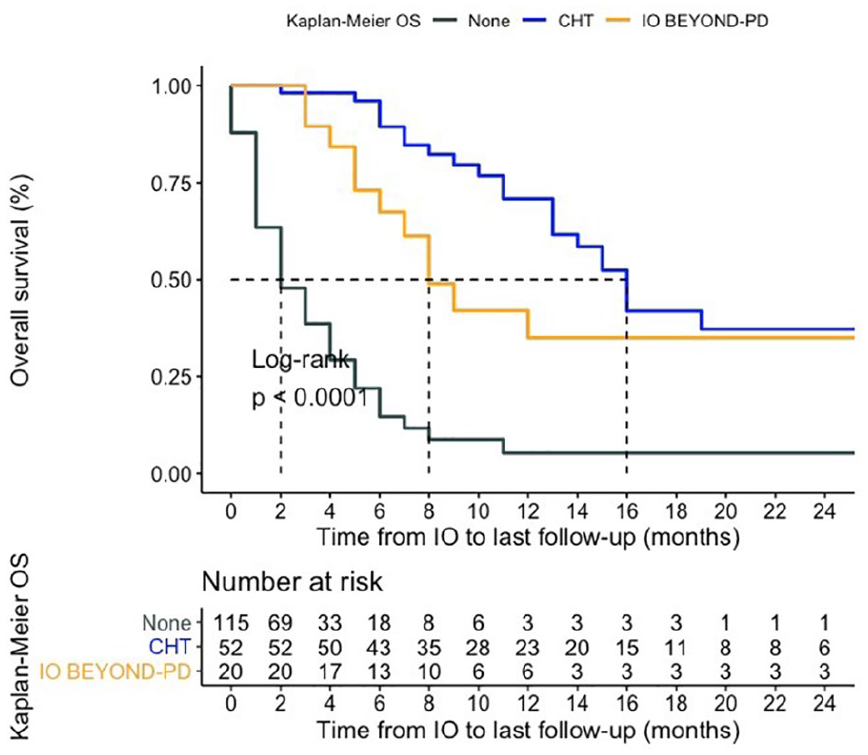
Second overall survival (OS-2) from immunotherapy initiation basing on the postimmune checkpoint inhibitor treatment. Median OS-2 was 16 months for patients receiving active treatment after ICI (52 evaluable patients), versus 8 months for patients receiving ICI beyond PD (20 patients), versus 2 months for patients not receiving further active treatment (115 patients), with significant difference in favor of chemotherapy (p < 0.001).
Discussion
This study is the result of the first Italian national registry of post-ICI treatment in mUC. Similar multicenter experiences previously reported in the literature suggested that patients progressing to frontline ICI are not likely to receive or benefit from subsequent chemotherapy.14,18 With the MeeT-URO 1 population, we obtained different results, showing a quite favorable outcome of patients treated with chemotherapy after progressing to ICI. Indeed, the outcome in terms of median PFS (3.5 months) and median OS (9 months) in the overall population was not inferior to that expected from salvage chemotherapy after the first-line polychemotherapy regimen.2-4 Furthermore, the outcome was significantly better than expected in the NAÏVE setting, with mPFS of 5 months, mOS of 13 months, and ORR of 28.0%. These data suggest that the sequence of immunotherapy followed by chemotherapy can be favorable even in patients not eligible for cisplatin.
On the contrary, the SALVAGE group’s outcome to third-line chemotherapy was similar to that expected from second-line chemotherapy in the preimmunotherapy era,2-4 as if the immunotherapeutic treatment line would have provided a time out to the disease. Even more, the ORR in the SALVAGE group (18.5%) was intriguing, considering the scarce evidence in favor of offering chemotherapy in the third-line setting for mUC patients. 19 This result could be subtended by ICI’s potential ability to restoring platinum sensitivity in tumors being refractory to chemotherapy at the first line. Similar data have been previously reported both in urothelial and lung cancer, suggesting that the sequence of immunotherapy followed by chemotherapy might be better than the opposite, at least in terms of response.20,21
On the contrary, in a very recent retrospective analysis, Sonpavde and coauthors evidenced a higher ORR for immunotherapy in the second line after prior chemotherapy than as the first treatment line. Nevertheless, ICI followed by carboplatin-based chemotherapy and the reverse sequence had comparable OS in the same report. 22
The reliability of such clinically relevant results in our population is due to our case series’s homogeneity since all patients belong to the same national health care system. Moreover, our patients receiving further active treatments after ICI-PD were about a quarter of the whole population, with similar treatment rates irrespective of the cohort. Conversely, a previously cited population from the literature 14 had higher rates of patients treated with chemotherapy after ICI failure, more than doubled compared to ours in the chemo-naïve setting (57.6%). This element suggests that a more robust selection of patients could have had an impact on our results.
Patient selection is probably the key to a favorable outcome of complex therapeutic sequences, especially when involving heavily pretreated subjects or considering cases with doubtful progression and clinical benefit. Concerning this issue, the only reliable factor with particular relevance for the patient selection for chemotherapy resulted in being the ECOG-PS, with significant prognostic value confirmed at the multivariate analyses irrespective of the treatment setting. Neither any other clinical element among those available nor the prior ICI treatment response was related to chemotherapy treatment outcome. As previously suggested by single-center reports, a lack of cross-resistance between chemotherapy and ICI seems to be confirmed.15,16 According to our results, ECOG-PS should be considered before offering other treatment lines to our patients progressed to ICI, as the only element at our disposal to evaluate the likeliness of benefit from chemotherapy. On the contrary, the elements related to the chance of receiving chemotherapy, namely the time of exposure to ICI and the number of metastatic sites, were probably the result of a positive selection bias.
The survival impact of receiving chemotherapy was strongly significant irrespective of the treatment cohort. Nevertheless, this result could also be due to a positive selection bias since the long survivors are more likely to receive further active treatments.
Interestingly, chemotherapy was more frequently administered in the NAÏVE cohort, namely to patients previously considered ineligible for chemotherapy in the first treatment line, demonstrating that contraindications to chemotherapy are often relative and likely to be reconsidered.
Eventually, this study aimed to offer valuable hints about the optimal therapeutic sequence once multiple agents likely become available for mUC patients worldwide. Considering mPFS-2, it was significantly longer in the case of immunotherapy-chemotherapy sequence when compared to that of immunotherapy beyond progression (10 months vs 6 months, see Figure 3B). Moreover, there was a significant benefit in terms of mOS-2, even doubled for the immunotherapy-chemotherapy sequence compared to that with immunotherapy continuation beyond progression (16 months vs 8 months, see Figure 5). Such results could suggest a particular caution about offering ICI continuation beyond clear radiological progression, even in the absence of evident clinical condition deterioration.
The worse prognosis was for patients not receiving any treatment after ICI-PD (2 months), obviously negatively selected as per the retrospective study limitations.
Although with cautious interpretation, the present results seem to testify for a potential augmentation of chemotherapy efficacy post-PD-1/PD-L1 inhibitor therapy. This hypothesis is even more interesting when considering that the new standard of care is now represented by first-line chemotherapy followed by switch maintenance with avelumab. 23 One of the hypotheses for the underlying mechanisms could be the persistence of a delayed immune response instigated by ICI, as suggested by the good ORR in both this study cohorts and in previous populations from the literature. 14 Another justification could be due to a synergism of chemotherapy with the post-ICI immunological context of the patient. Positive immunomodulatory effects have been found for standard chemotherapy, which can induce immunogenic cell death and modulate the activity of distinct immune cell subsets through multiple mechanisms. 24 On the contrary, aside from ORR’s intrinsic meaning, more reliable outcomes such as PFS and OS are quite aligned to historical data for salvage chemotherapy,2-4 suggesting a careful reflection about the cost-effectiveness of our treatment choices, especially in such advanced oncological settings.
The main limitations of the MeeT-URO 1 study are the retrospective nature, with consequent selection bias (patients selected to receive active treatment could be more fit than patients intended to receive BSC only), and the lack of a control group with new therapeutic options, such as the new drugs enfortumab vedotin 25 and erdafitinib, 26 currently representing new treatment options recently approved by FDA, respectively, for all-comers patients progressing to chemotherapy and immunotherapy and patients with FGFR-driven tumors. Further minor limitations were the anonymization of the immunotherapeutic compound (only ICI class was provided); the relatively high number of not evaluable patients; the lack of functional prognostic chemotherapy baseline elements, such as the metastatic sites and their number; and the relatively small sample size, for which subgroup analyses according to the chemotherapeutic drug and schedule would not have been reliable.
Conclusions
In conclusion, we could suggest considering chemotherapy at the first progression to ICI for mUC patients as an alternative to the preferable option of enrollment in clinical trials when available, especially in the case of ECOG-PS preservation, rather than other customized strategies such as ICI continuation beyond progression.
Supplemental Material
sj-pdf-1-onc-10.1177_11795549211021667 – Supplemental material for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study)
Supplemental material, sj-pdf-1-onc-10.1177_11795549211021667 for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study) by Melissa Bersanelli, Sebastiano Buti, Alessio Cortellini, Marco Bandini, Giuseppe Luigi Banna, Filippo Pederzoli, Elena Farè, Daniele Raggi, Patrizia Giannatempo, Ugo De Giorgi, Umberto Basso, Tania Losanno, Daniele Santini, Claudia Mucciarini, Marcello Tucci, Rosa Tambaro, Azzurra Farnesi, Orazio Caffo, Antonello Veccia, Emanuele Naglieri, Alberto Briganti, Giuseppe Procopio, Sandro Pignata and Andrea Necchi in Clinical Medicine Insights: Oncology
Supplemental Material
sj-pptx-2-onc-10.1177_11795549211021667 – Supplemental material for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study)
Supplemental material, sj-pptx-2-onc-10.1177_11795549211021667 for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study) by Melissa Bersanelli, Sebastiano Buti, Alessio Cortellini, Marco Bandini, Giuseppe Luigi Banna, Filippo Pederzoli, Elena Farè, Daniele Raggi, Patrizia Giannatempo, Ugo De Giorgi, Umberto Basso, Tania Losanno, Daniele Santini, Claudia Mucciarini, Marcello Tucci, Rosa Tambaro, Azzurra Farnesi, Orazio Caffo, Antonello Veccia, Emanuele Naglieri, Alberto Briganti, Giuseppe Procopio, Sandro Pignata and Andrea Necchi in Clinical Medicine Insights: Oncology
Supplemental Material
sj-pptx-3-onc-10.1177_11795549211021667 – Supplemental material for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study)
Supplemental material, sj-pptx-3-onc-10.1177_11795549211021667 for Clinical Outcomes of Patients With Metastatic Urothelial Carcinoma After Progression to Immune Checkpoint Inhibitors: A Retrospective Analysis by the Meet-Uro Group (Meet-URO 1 Study) by Melissa Bersanelli, Sebastiano Buti, Alessio Cortellini, Marco Bandini, Giuseppe Luigi Banna, Filippo Pederzoli, Elena Farè, Daniele Raggi, Patrizia Giannatempo, Ugo De Giorgi, Umberto Basso, Tania Losanno, Daniele Santini, Claudia Mucciarini, Marcello Tucci, Rosa Tambaro, Azzurra Farnesi, Orazio Caffo, Antonello Veccia, Emanuele Naglieri, Alberto Briganti, Giuseppe Procopio, Sandro Pignata and Andrea Necchi in Clinical Medicine Insights: Oncology
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB received honoraria as a speaker at scientific events by Bristol-Myers Squibb (BMS), Novartis, and Pfizer; as a consultant for advisory role by Novartis, BMS, IPSEN, and Pfizer; she also received fees for copyright transfer from Sciclone Pharmaceuticals and research funding from Seqirus, Pfizer, Novartis, BMS, Roche, AstraZeneca, and Sanofi. SB received honoraria as a speaker at scientific events and advisory role by BMS, Pfizer, MSD, Ipsen, Roche, Pierre-Fabre, AstraZeneca, and Novartis. AC received grant consultancies/speaking fees from MSD, BMS, Roche, AstraZeneca, Novartis, Astellas, and Sun Pharma.
Authors’ Note
Marcello Tucci is now affiliated with Medical Oncology Unit, Ospedale Cardinal Massaia di Asti, Asti, Italy.
Author Contributions
MB, SB, AC, MB GLB and AN contributed to study conception and design, acquisition of data, analysis, interpretation of data, drafting of the manuscript and critical revision. All other authors contribuited to acquisition and interpretation of data, read and approved the submitted version of the manuscript.
Data Access and Responsibility
MB, SB and MB had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Ethics
The principles outlined in the Declaration of Helsinki have been followed for this work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
