Abstract
Introduction:
Compliance of community pharmacies with the national medicines law for proper storage and dispensing of medicines is crucial for ensuring the quality and safety of medicines. Community pharmacies in Bhutan are of two types: retail pharmacies and wholesale pharmacies.
Objective:
This study was aimed to assess the compliance of community pharmacies with the Medicines Act of the Kingdom of Bhutan 2003 and Bhutan Medicines Rules and Regulation 2012.
Methods:
This is a retrospective cross-sectional study from the inspection reports of community pharmacies inspected from January 2015 to December 2017. Descriptive statistics in frequency and percentage was used to report the results.
Results:
The overall compliance level for the regulatory provisions under critical and minor categories was 90.4% followed by 86.74% for major category. Around 32.99% of community pharmacies were found without standard operating procedures for storing and dispensing of medicines while 100% of community pharmacies had technical authorization from the Drug Regulatory Authority.
Conclusion:
Although the compliance level of community pharmacies in Bhutan for storing and dispensing of medicines was found to be above 80%, there are several areas that need to be corrected to ensure full compliance. This study will serve as a baseline for policymakers, managers, and researchers in developing designs for future interventions.
Introduction
Private healthcare is growing in low- and middle-income countries, 1 and as a result community pharmacies (CPs) are becoming a source of healthcare in many parts of the world. CPs act as the first point of healthcare for the majority of the population living in the developing countries. 2 The quality of medicine is important due to the increasing prevalence of substandard and falsified medicines worldwide. 3 CPs play a key role in ensuring the quality of medicines for the public; however, the medicines quality cannot be ensured if they are not stored or distributed properly. 4
Healthcare in Bhutan is provided free by the government, 5 but the growing number of CPs indicates increased demand for private healthcare services. CPs in Bhutan are of two types: retail pharmacies (RPs) and wholesale pharmacies (WPs), 6 and they are governed by the Medicines Act of the Kingdom of Bhutan 2003 (Act) and Bhutan Medicines Rules and Regulations 2012 (Regulations). The Drug Regulatory Authority (DRA) is responsible for licensing and monitoring the compliance of CPs in Bhutan. All CPs in Bhutan are authorized by the DRA and must be operated under the supervision of a competent person certified by the DRA. Competent person is a qualified personnel usually with a background in pharmacy or medicine and certified by the DRA to handle medicines. CPs are inspected to verify their regulatory compliance for dispensing and storage of medicines, and necessary regulatory actions are taken by the DRA to encourage compliance. CPs are inspected on risk basis and the frequency of inspection varies among the CPs. The noncompliant CPs are inspected more often than the more compliant ones.
Inadequate compliance of CPs with the medicines law has been reported in other developing countries. A study conducted in Vietnam reported that more than 90% (n = 1833) of prescription-only-medicines were dispensed without prescriptions. 7 In line with this, 62.7% of Malaysians (n = 314) took at least one prescription-only-medicine in the past week without prescription 8 and the prevalence of self-medication was reported to be 77.5% (n = 405) in southwest Ethiopia. 9 Another study from Malaysia suggested that community pharmacists (n = 50) and general practitioners (n = 736) are responsible for irrational dispensing of antibiotics. 10 A study from Pakistan reported that none of the CPs (n = 371) fully complied with the legal requirements and necessary regulatory interventions. 11 Similar findings reported some deficiencies in the storage of medicines in CPs in Riyadh, Saudi Arabia and Nigeria.12,13
Despite the existence of regulatory system, CPs in Bhutan failed to comply with regulatory requirements for storage and dispensing of medicines. 14 The noncompliance of CPs with the Act and Regulations is an issue in Bhutan, like in many other developing countries.11,15 The effectiveness of inspections should be monitored to study if it contributes to improve the compliances. 16 It is therefore, important to study the compliance of CPs to understand the effectiveness of regulatory actions taken and develop appropriate protocols to improve compliance.
This study is expected to enrich the body of work and literature of pharmacy practice in developing countries. It also provides an interesting insight into Bhutan’s pharmacy policy focusing on proper storage and dispensing of medicines from which smaller developing nations can learn. It is first such article on compliance of CPs in Bhutan which would serve as a source of information for future research on this subject.
Research question
What is the compliance level of CPs with the provisions of the Act and Regulations and the effectiveness of the action taken by the DRA?
Objective
The study was aimed to investigate the compliance level of CPs with the regulatory requirements for proper storage and dispensing of medicines and assess the impact of the regulatory actions in improving the compliance of CPs.
Ethical approval
The Research Ethics Board for Health (REBH), Bhutan has exempted the protocol from ethics review vide letter no REBH/PO/2018/035 dated 21 May 2018 since this study did not involve any human subjects.
Materials and methods
Study design
This was a retrospective cross-sectional study aimed to explore the compliance of CPs with the Act and Regulations and study the effectiveness of regulatory actions. Compliance to each of the regulatory provisions which were broadly classified into critical (which indicates a potential risk for patient/product), major (which indicates an indirect potential risk for patient/product), and minor (which indicates general feedback for improvement) was assessed using the inspection guideline, DRA.
Study setting
This study was conducted at the DRA Office in Thimphu, in which approximately 50% of CPs in Bhutan are located. 17
Study population and sample size
Data were collected retrospectively from inspection reports between January 2015 and December 2017. Universal sampling was used for the study, that is, all 97 CPs inspected during the study period were included in the study. All CPs are required to comply fully with all the relevant provisions of the Act and Regulations to be considered compliant. Any CPs noncompliant to any of the regulatory provisions were considered noncompliance.
Data analysis
The data were coded into Epi-Data entry (Epi-Data, version 3.1), and Epi-Data Analysis version 2.2.2.182 was used for statistical analysis. Descriptive statistics in frequency and percentage was used to report the results.
Results
A total of 97 CPs were inspected from January 2015 to December 2017, and all the secondary data of inspection reports were included in the study. The number of RPs inspected increased from 12 in 2015 to 34 in 2017. In contrast, the number of WPs inspected increased from 6 in 2015 to 8 in 2016 but reduced to 4 in 2017 as shown in Table 1.
Year-wise Inspection of Community Pharmacies (n = 97).

The compliance level of CPs toward each regulatory provisions classified under the critical, major, and minor category is shown in Table 2. The overall compliance level for the regulatory provisions under the major category was the lowest with 86.74% followed by 90.4% each for critical and minor categories.
Frequency and Percentages of Compliance to Regulatory Provisions.
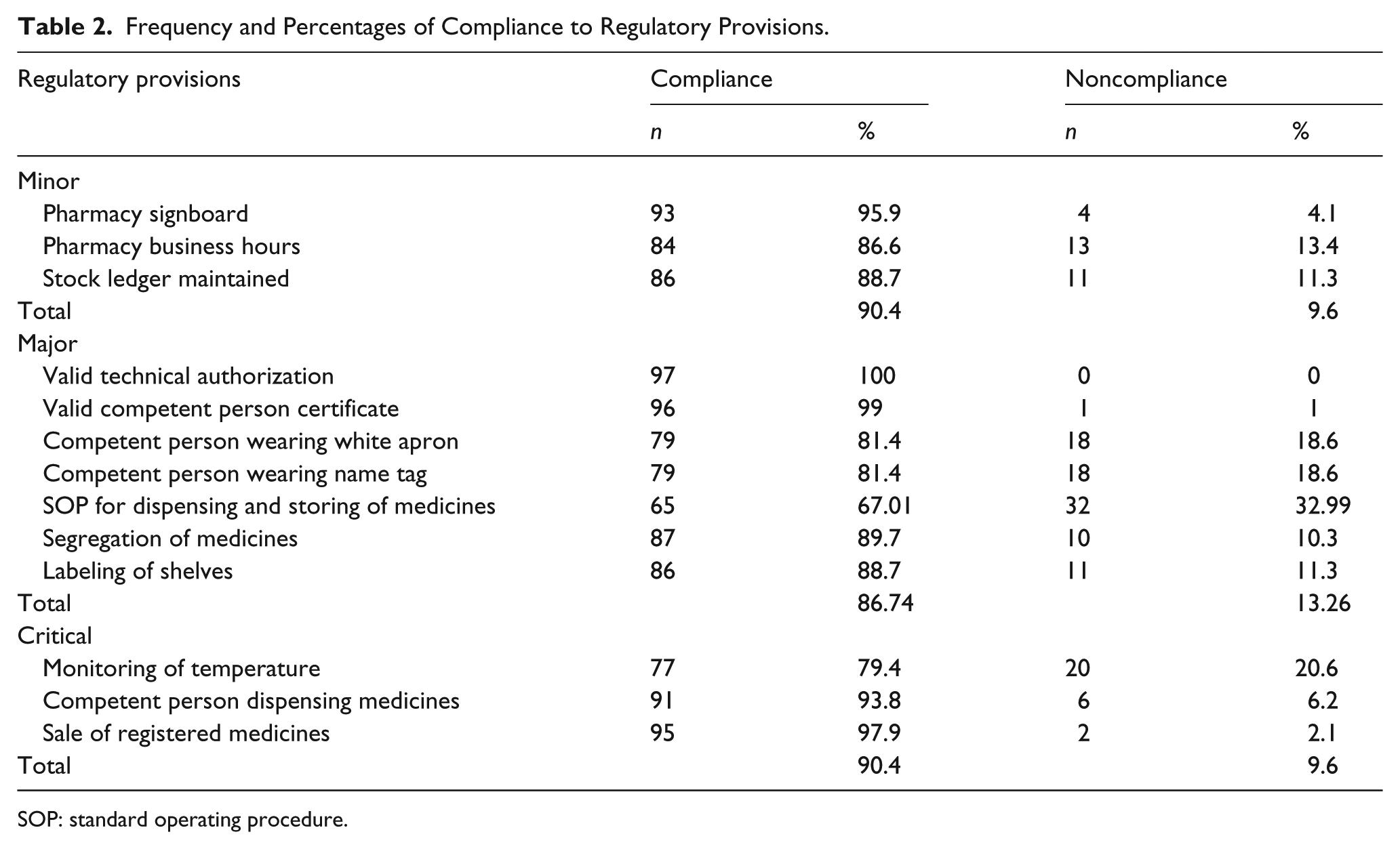
SOP: standard operating procedure.
The types of regulatory actions taken by the DRA are shown in Table 3. The number of warning letters reduced from 18 to 11 during the study period, while the highest number of fines was imposed in 2016.
Types of Regulatory Actions for Noncompliance.

The trend of compliance level of RPs and WP is shown in Figure 1. Over the years, from 2016-2017, the compliance rate of WPs increased from 25% to 100%, while it remained constant for RPs, with only a slight increase from 48.5% to 50%.

Trends of Compliance level among Retail and Wholesale Pharmacies inspected.
The compliance level of CPs of different Districts in the country is shown in Figure 2. CPs in Samdrup Jongkhar and Dagana Districts had the highest compliance level, whereas the CPs in Wangduephodrang, Trongsa, and Samtse Districts had the lowest compliance.

Compliance rate of Retail and Wholesale Pharmacies of different Districts.
Discussion
Compliance of CPs to the regulatory provisions of dispensing and storage of medicinal products were always reported as a problem in the developing countries.11,15,18–20 According to a study conducted in Malaysia, 47%–50% of CPs were reported as not fully compliant with the law and regulations. 16 This study found that the overall noncompliance rate was 13.26% for major regulatory provisions, followed by 9.6% each for the critical and minor regulatory provisions. The main noncompliance issues were lack of standard operating procedures (SOPs) for dispensing and storing of medicines followed by monitoring of temperature and competent persons wearing white coat and name tag.
SOPs for dispensing and storing of medicinal products
All CPs in Bhutan are required by the law to develop and implement the SOPs for storing and dispensing of medicines in their pharmacies. 6 SOPs for dispensing and storing of medicines are critical for ensuring the quality and safety of medicines. Studies have also shown that improper, inconsistent, and incomplete labeling of dispensed medicines is the major cause of medication errors.21,22 This study revealed that 32.99% of CPs in Bhutan did not have SOPs for storing and dispensing of medicines, thereby increasing the risk of dispensing errors. The reason for this noncompliance could be due to competent persons not knowing how to write SOPs.
Monitoring of temperature in CPs
Literature shows that stability of pharmaceutical products is essential to maintain their therapeutic efficacy, 23 and temperature plays a key role in maintaining the required efficacy of the medicinal products.15,24 Pharmacists must ensure that product integrity is not compromised by exposure to undesirable temperature or humidity. 25 However, past studies have shown that storage practices were very poor and only few CPs monitored temperature in their pharmacies. 26 Such findings were congruent with the findings of this study and revealed that the CPs did not either have temperature-monitoring devices in their pharmacies or failed to record the temperature. In addition, a uniform temperature-monitoring device can be recommended to be used in the CPs.
Competent persons wearing white coat and name tag
A study done in the Indian States of Kerala and Tamil Nadu reported RPs operating without a pharmacist.20,27 Our study also showed that 6.2% of CPs operated without a competent person which could be due to non-availability of competent persons in the market and thereby not allowing the CPs to replace the competent persons leaving their pharmacies. The white coat of a pharmacist is not a measure of the character but is a representation of what a pharmacist means to the patients they serve. 28 According to Cretton-Scott et al., 29 a pharmacist’s attire affects patient–pharmacist relationship, as it conveys a friendly and inviting message to the patients besides showing a message of professional competence. A study reported that 94% of CPs had pharmacist not wearing their white coats 20 which is in line with the findings of this study where 18.6% of CPs had competent persons not wearing white coat and name tag. The reason for this could be due to not understanding the importance of wearing white coat and name tag. Hence, the competent persons must be sensitized on the importance of wearing white coat and a name tag.
Effectiveness of regulatory actions
A study conducted in Malaysia identified warning letter as the most effective method to improve compliance as compared to follow-up inspection. 16 Our study also showed that the regulatory actions were effective in improving the compliance of CPs. Thus, it is suggested that most effective technique should be employed by the DRA to tackle the critical and more persistent noncompliance. A study done by Cabanam et al. 30 suggested that awareness of competent persons with the regulations is one of the contributing factors for reduction in noncompliance. Although the competent persons are required to be well-versed with all the regulatory sections related to their practice, it is not sure whether they are aware of the underlying principle behind such regulations and the importance of complying with them. Thus, to increase their compliance with the law, awareness programs and refreshers courses emphasizing on the rationale of each provisions could be useful.
Limitations of the study
This study included only those regulatory requirements relevant for proper dispensing and storage of medicines by CPs. Hence, other regulatory provisions not applicable to dispensing and storing of medicines were excluded. Although this study reports the effectiveness of regulatory actions in general, it failed to evaluate the effectiveness of individual regulatory actions.
Recommendations
Support from the government and collaboration with the relevant stakeholders is critical for effective enforcement of medicines laws.19,31 Training is crucial for improving the compliance of CPs and the competent persons must be trained on writing SOPs, monitoring temperature, and keeping records among many others. To enhance the compliance of CPs with the law, awareness campaign programs are essential to convey the regulatory principles and the consequences of not complying with the laws. Moreover, monitoring the effectiveness of inspection using key indicators like compliance rate would make it possible to gauge and re-strategize current inadequacies of the inspection activities to improve compliance of CPs. A regulatory impact assessment to assess the practical applicability of the regulation would be helpful in ensuring compliance of CPs.
Conclusion
This study reports the compliance level of CPs with the regulatory requirements for proper storing and dispensing of medicines and the effectiveness of regulatory actions taken by the DRA. The overall compliance level for the regulatory provisions classified as critical was 90.4%, while it was 86.74% for the major category. However, there are several areas that need to be corrected and improved to ensure full compliance. Furthermore, this study also provides baseline information for future research on this subject.
Footnotes
Acknowledgements
The authors would like to acknowledge Mr Kunzang Dorji and Tashi Dhendrup, Inspection Division, DRA for his support in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human or animal subjects performed by any of the authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
