Abstract
Substandard and falsified (SF) medicines pose a significant global public health threat, disproportionately affecting low- and middle-income countries (LMICs). While quantitative studies have documented their prevalence, a gap exists in the systematic synthesis of qualitative evidence exploring the underlying contextual, systemic, and behavioral drivers of the burden of SF medicines. A systematic review of qualitative studies was conducted. The study used PubMed and Google Scholar as databases, which were searched from June to October 30 for literature published between 2012 and 2025. The methodological quality of included studies was evaluated as per eight criteria of the Critical Appraisal Skills Program (CASP) qualitative checklist. Studies exploring drivers or mitigation strategies for SF medicines were included. Two reviewers conducted the study selection, quality appraisal, and thematic analysis. The thematic analysis in this review was conducted systematically and iteratively, following a well-established qualitative synthesis approach. After data extraction, the included studies were organized based on the review questions. Through collaborative discussion among authors, the evidence categories were refined and grouped into two overarching themes: drivers and mitigation strategies. A total of 19 studies were included. The review identified four key thematic drivers, such as market and economic drivers (profit-driven cost cutting and price arbitrage), weak regulatory and legal enforcement (inadequate oversight, corruption, and lenient penalties), complex supply chain and demand issues, and critical gaps in knowledge and perception (public and professional misconceptions, particularly regarding generic medicines). In addition, the review identified that emphasizing the need for strengthening regulatory systems, technological innovations (track and trace), public and professional education, and addressing underlying socioeconomic drivers are among the main actions used as mitigation strategies. This qualitative systematic review concluded that interconnected market pressures, weak regulation, fragile supply chains, and knowledge gaps are significant issues. Hence, effective action requires integrated policies that strengthen enforcement capacity, secure supply chains through affordable track-and-trace technologies, and launch context-specific education campaigns to combat public mistrust in quality-assured generics. These lead to translating systemic insights into practical policy implementation for medicine quality and positive public health outcomes.
Plain Language Summary
Drivers and Strategies to Combat Substandard and Falsified Medicines in Low- and Middle-Income Countries: A Systematic Review
This review summarizes what studies have found about why substandard, and falsified medicines are a problem in low- and middle-income countries and what can be done about it. It brings together findings from 19 qualitative studies. The key reasons identified include market pressures to cut costs, weak laws and enforcement, complex supply chains, and a lack of public and professional awareness, particularly a distrust of cheaper generic medicines. To fight this problem, the review highlights the need for stronger regulation and enforcement, better technology (like tracking systems), education campaigns, and improving access to affordable, quality-assured medicines. The findings show that a coordinated approach is needed to protect public health.
Keywords
Introduction
As per the World Health Organization definition, poor-quality drugs, often referred to as substandard, falsely labeled, falsified, or counterfeit (SFFC) medicines, can lead to treatment failure and, in severe cases, result in death. 1 In 2017, the counterfeit term was not further worked on and was replaced by “falsified” according to the WHO report. The report indicated 1 in 10 medical products in developing countries is substandard or falsified. 2 In the current context, there are growing concerns regarding the quality of medicines globally. 3 The Poor quality of medicines that includes substandard, spurious, falsely labeled, falsified and counterfeit medicines has resulted serious impacts. 4 They are a prominent issue in both developed and developing countries. 5 , 6 Access to quality life-saving medicines is one of the indicators of the quality of health services and a major target area of universal health coverage. 7 However, the right of access to quality lifesaving medicine being compromised due to the high prevalence of defective products circulating in the global market. 8
Due to inadequate regulatory oversight, the widespread availability of low-quality drugs has become a universal concern. 3 The evidence indicated that medicines in Africa failed at least one quality test. 9 It points out poor registration systems, poor import standards, and market regulatory permission as part of the problem. Due to weak regulatory enforcement, evidence from various parts of Africa indicates that many medicine retail outlets operate in non-compliance with established standards. 10 Such outlets frequently dispense or store drugs under improper conditions that fail to meet regulatory requirements, contributing to the circulation of medicines that do not pass quality control tests. 10
Limited access to accredited facilities, unregulated outlets, challenges in medicine manufacturing and supply chains, and low public awareness are the main contributors to the prevalence of poor-quality drugs.11–13 In addition, evidence indicate that, drug shortages further exacerbate this vulnerability by forcing rapid sourcing substitutions, fragmented procurement, and reliance on informal or non-verified supply channels, conditions that increase the risk of substandard and falsified (SF) medicines entering the market. 14 Due to these gaps, SF medical products place a significant burden on global health particularly affecting low- and middle-income countries across, Africa, Asia, and Europe. The WHO reports that, since 2013, have received 1500 reports of cases of substandard or falsified products. Among these, antimalarials and antibiotics are the most commonly reported. 2 The majority of the reports (42%) come from the WHO African Region, 21% from the WHO Region of the Americas, and 21% from the WHO European Region. 2 The presence of low-quality drugs can deplete the economic resources allocated for restoring health system. These substandard drugs not only endanger patient health but also place significant strain on healthcare systems and economies. 15 This evidence suggested that the economic strain from SF medicines in developing and middle-income countries justifies a qualitative study with strong policy relevance to identify systemic barriers and enable the combating of the economic burden of the health budget. 16
As the WHO report states, SF medicines waste limited healthcare resources by leading to ineffective treatments, additional diagnostics and repeat care, and prolonged illnesses, which increases costs for households and overstretched health systems in LIMICs and costs countries billions annually (US$30.5 billion globally) and diverts scarce funds from essential services. 2 Evidence suggests that socioeconomic factors influence policy and regulatory enforcement, which in turn may facilitate the circulation of SF medicines in health systems. Understanding how economic pressures, patient behaviors, informal markets, and trust in the health system open the way of the circulation and use of SF medicines. This requires qualitative insights to inform effective regulation, enforcement, and education strategies. 17 Qualitative data is needed, as it can contextualize statistical findings and support tailored policy responses in the local context. 17 Economic strain from SF medicines not only imposes direct costs but also undermines confidence in healthcare systems and providers, shaping treatment-seeking behavior and compliance, which are critical issues that qualitative methods are well suited to explore for policy design. 2 Currently, regulatory authorities and organizations like the WHO, and AUDA-NEPAD, 18 need baseline information for policy interventions aimed at combating SF medical products. There is limited documented evidence from qualitative studies that assesses drivers, contributing to the presence of poor-quality drugs. It is essential to understand these drivers in order to effectively defend against them. The findings might contribute to global knowledge for professionals and users to identify SF medical products and enable them to act as stakeholders against them.
Added value
The global health threat posed by SF medicines has been extensively examined through systematic reviews and meta-analyses, providing quantitative insights into their prevalence, driving factors, and potential prevention strategies. 9 , 15 , 19 However, there is a notable gap in qualitative systematic reviews that explore the underlying contextual, regulatory system failures, supply, and demand, as well as, social and behavioral factors influencing this issue.
The previous systematic reviews and meta-analyses have primarily focused on providing quantitative insights into the prevalence, distribution, and economic impact of SF medicines. However, there is a lack of evidence exploring the underlying contextual and systemic factors that drive their persistence. This review takes a unique approach by using a qualitative evidence synthesis to uncover the complex social, regulatory, and market dynamics that enable the production and circulation of SF medical products. By integrating findings from various qualitative studies across different regions, it identifies root causes that quantitative research alone cannot fully explain, such as weak governance structures, supply chain vulnerabilities, and behavioral factors that influence medicine use.
In addition, this review adds significant value to global health and policy discourse by synthesizing qualitative insights that inform practical and context-sensitive interventions. The findings provide a deeper understanding of how local regulatory failures, socioeconomic pressures, and public perceptions interact to sustain the SF medicines crisis. These insights can guide policymakers, regulators, and international health organizations in designing evidence-informed strategies and strengthening enforcement frameworks to safeguard medicine quality and ensure equitable access to safe and effective healthcare products worldwide.
Review questions
This qualitative systematic review is guided by two primary questions:
Research Question 1 (RQ1): what are the key drivers contributing to the prevalence of SF medicines in various global contexts?
Research Question 2 (RQ2): What strategies have been implemented to combat SF medicines, and how effective are they?
Based on these questions, the review has two objectives: to explore and synthesize qualitative evidence on the underlying drivers of SF medicines globally and to identify the mitigation strategies implemented to address them. By synthesizing existing literature, the review aims to uncover the factors facilitating the circulation of SF medicines and evaluate interventions, policies, and enforcement measures designed to mitigate their impact. The findings provide a comprehensive understanding of both the causes and solutions, offering insights to inform policy, regulation, and public health strategies worldwide.
Plain language summary
This review summarizes what studies have found about why SF medicines are a problem in low- and middle-income countries and what can be done about it. It brings together findings from 19 qualitative studies. The key reasons identified include market pressures to cut costs, weak laws and enforcement, complex supply chains, and a lack of public and professional awareness, particularly a distrust of cheaper generic medicines. To fight this problem, the review highlights the need for stronger regulation and enforcement, better technology (like tracking systems), education campaigns, and improving access to affordable, quality-assured medicines. The findings show that a coordinated approach is needed to protect public health.
Methodology
Literature search strategies
This qualitative systematic review was conducted in accordance with the preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) 2020 guidelines 20 (Supplementary File 1). A comprehensive literature search was conducted using the databases PubMed and Google Scholar from June to September 2025. The search employed a combination of keywords using Boolean operators, including “substandard medicines” OR “falsified medicines” AND “counterfeit medicines,” coupled with terms such as “drivers” OR “causes,” “factors,” AND “strategies,” OR “interventions,” OR “policies,” specifically focusing on qualitative studies. The search was limited to studies published from 2012 to 2025 to ensure the inclusion of contemporary evidence on the factors contributing to poor-quality medicines and the interventions employed to address them.
Inclusion, and exclusion criteria
The review included qualitative studies, such as interviews, focus groups, and ethnographies, that specifically addressed drivers or strategies related to SF medicines. Only studies published in English and conducted in any global context were considered. Studies were excluded if they were quantitative in nature, focused solely on pharmaceutical quality without addressing SF medicines, or were non-peer-reviewed articles. Mixed-method studies were included only if their qualitative components were substantial and could be meaningfully extracted and analyzed separately. The studies were included if they employed qualitative methodologies to explore drivers or mitigation strategies related to SF medicines, regardless of whether quantitative data were also collected. The qualitative findings from these mixed-methods studies were extracted and synthesized alongside those from purely qualitative studies, ensuring that the review captured rich, context-specific insights while maintaining methodological coherence.
Study selection process
The study selection process was conducted using a systematic approach to ensure rigor and minimize bias. Initially, two independent reviewers (Y.T.M. and A.A.T.) screened the titles and abstracts of all identified studies to assess their eligibility. Studies that met the initial criteria then underwent a full-text review to confirm their inclusion. In cases where disagreements arose between the reviewers, these were resolved through discussion. If consensus could not be reached, a third reviewer was consulted to make the final decision.
Data extraction and analysis
Data were systematically extracted from each study, encompassing details on study characteristics such as author, year, country, and setting, as well as the research design and methodology employed. Key findings relevant to the identified drivers and mitigation strategies were also captured, along with contextual factors that may have influenced the outcomes. Following data extraction, the studies were organized both categorically based on review questions, and thematically.
The synthesized evidence from the included studies was grouped into two major thematic outcomes: (1) Drivers and (2) Mitigation Strategies. After thorough discussion among the authors, the major thematic category “Drivers” was further divided into four subthemes: (1) Market and Economic Drivers, (2) Regulatory and legal enforcement, (3) Complex Supply Chain and Demand Issues, and (4) Critical Gaps in perception (Figure 1). In addition, mitigation strategies were grouped into eight subthemes based on the research questions and insights from published work, particularly from the perspective of policymakers. These subthemes include: (1) Addressing Underlying Socioeconomic Drivers, (2) Strengthening Regulatory Systems and Enforcement, (3) Professional Practice and Public Awareness, (4) Digital and Technological Innovations, (5) Stakeholder Engagement and Feedback Mechanisms, (6) Emergency Preparedness, (7) Improving Testing and Quality Assurance Infrastructure, and (8) Storage and Logistics Management.

Thematic presentation of drivers and Mitigation strategies of SF medicine (Green color: drivers or cause while blue color mitigation strategies).
Methodological quality of the included study
To evaluate the methodological quality of the studies included in this review, the Critical Appraisal Skills Program (CASP) Qualitative Checklist (Supplementary File 2) was used. 21 This ensure a thorough assessment of the rigor and trustworthiness of the evidence presented. Each study was evaluated based on eight CASP criteria: (1) Clear statement of the research aims, (2) appropriateness of qualitative methodology, (3) suitability of the research design, (4) adequacy of recruitment strategy, (5) appropriateness of data collection methods to address research objectives, (6) consideration of the researcher-participant’s relationship, (7) attention to ethical considerations, and (8) Sufficiency and rigor of data analysis. The risk of bias was meticulously assessed to ensure that the extracted information was evidence-based and representative. All 19 studies evaluated by the reviewer met the eight CASP guidelines set for qualitative studies. 21
Results
Figure 2 presents the PRISMA flow diagram detailing the systematic study selection process. In this study, a total of 3156 records were initially identified from PubMed (n = 456) and Google Scholar (n = 2700). After duplicate removal, 1586 studies underwent title and abstract screening, leading to the exclusion of 1275 records. The remaining full-text articles were assessed for eligibility, with 292 excluded for being review articles, meta-analyses, economic modeling studies, systematic reviews, or laboratory quality control reports. Ultimately, 19 studies were included for the actual qualitative data synthesis. This rigorous and transparent process underscores the methodological robustness of the review while indicating the relative scarcity of dedicated qualitative evidence on the drivers and mitigation strategies for SF medicines.
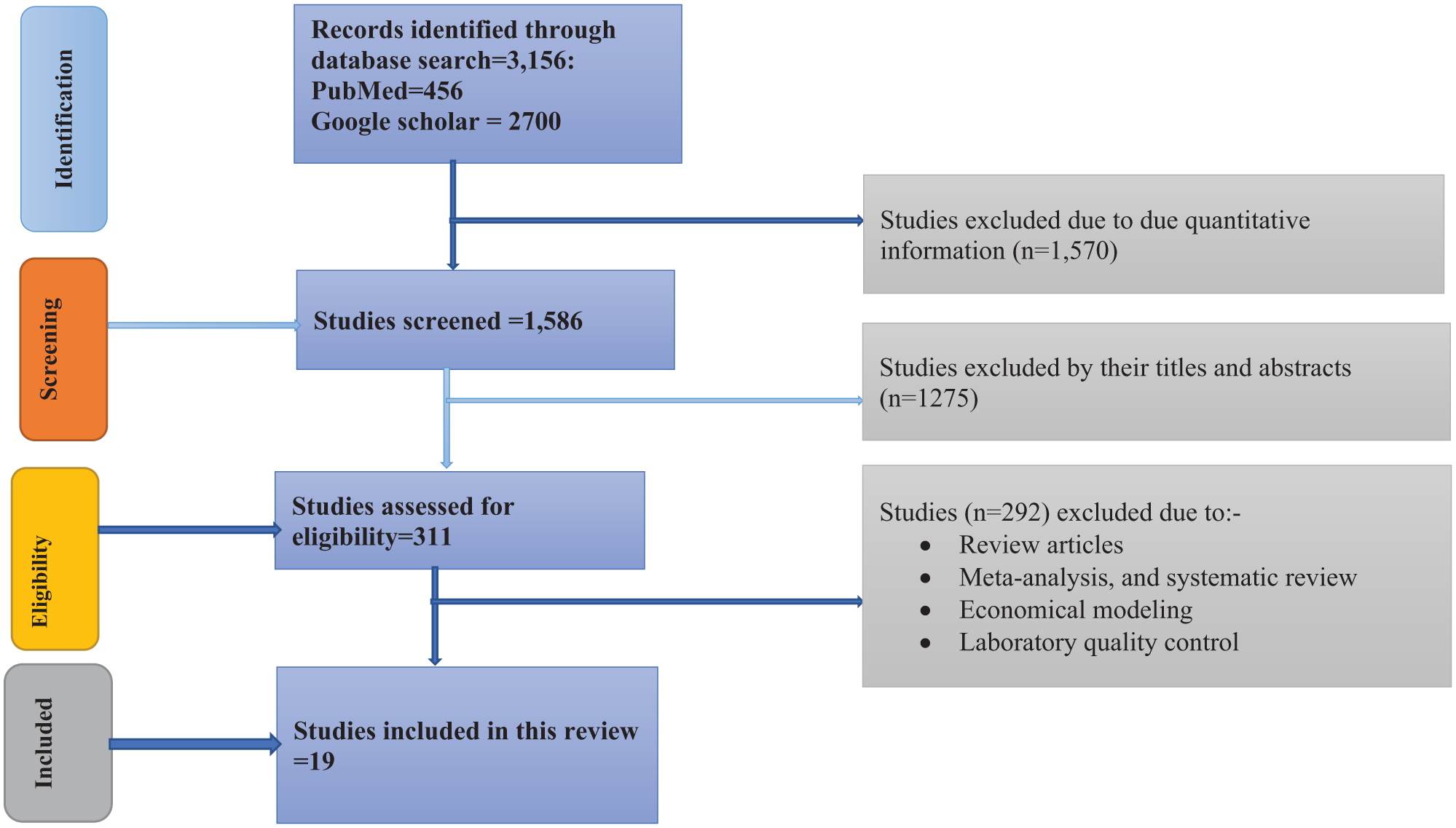
PRISMA flowchart indicate the screening process of included study (N = 19).
General description of included studies
The reviewed studies indicate that investigators explored the perceptions, experiences, and practices of diverse stakeholders involved in medicine regulation, distribution, and use across different countries. Most of the studies utilized semi-structured interviews, key informant interviews, and focus group discussions, allowing for an in-depth understanding of regulatory challenges and gaps in pharmaceutical systems. Participants commonly included regulators, pharmacists, healthcare providers, manufacturers, and community members, reflecting a multi-stakeholder approach to addressing medicine quality and regulatory enforcement.
The included studies were published from 2012 to 2025 (Table 1), showing a wide temporal and geographical distribution. Early studies were conducted in South Africa (2012), Sudan (2013), and Yemen (2014), followed by Nigeria (2019) and multi-country collaborations involving China, Indonesia, Turkey, and Romania (2019). Research activity increased after 2021, with notable contributions from Pakistan (2021), Tanzania (2021 and 2023), and Cameroon and Senegal (2023). Additional report from India (2023), Zimbabwe (2023), and Jordan (2024) were included. Ethiopia demonstrated the highest frequency of publications, with studies published in 2018, 2024, and 2025, while Ghana (2025) and Pakistan (2025) also contributed recent research. The overall trend indicates a noticeable rise in publications between 2023 and 2025, reflecting growing scholarly attention toward regulatory and pharmaceutical governance challenges in recent years.
Summary of the included study characteristics.

Across the studies, a consistent theme emerges concerning the complexity of medicine regulation and the interplay between policy, enforcement, and stakeholder collaboration. The predominance of qualitative and mixed-method approaches underscores the need to capture nuanced socio-political and institutional dynamics influencing medicine regulation in low- and middle-income countries. Moreover, the inclusion of mixed-method designs in countries such as Tanzania, Yemen, and Cameroon demonstrates a growing trend toward integrating quantitative insights with qualitative narratives to strengthen evidence-based policy recommendations. Collectively, these studies emphasize the global recognition of regulatory system challenges and the critical role of stakeholder engagement and qualitative inquiry in developing more effective pharmaceutical governance frameworks.
Drivers of SF medical products
This qualitative systematic review explores the key drivers contributing to the prevalence of SF medical products. The discussion is organized by themes based on the similarity of study objectives, as identified and analyzed by the primary investigator.
Reports from countries such as China, Indonesia, Turkey, and Romania indicate that legitimate, profit-driven pharmaceutical companies often protect their profit margins by cutting production costs or withdrawing from less profitable markets, while distributors engage in price arbitrage. At the same time, some healthcare providers seek to maintain profitability by upselling patients to medicines not covered by insurance. Such cost-cutting practices can undermine quality assurance systems, resulting in the production and circulation of substandard or degraded medicines. As noted by study participants,
Manufacturers are going to look for the very cheapest active pharmaceutical ingredients (APIs) and the least expensive suppliers. They may also alter their packaging, for instance, shifting from blister packs to cheaper strip packaging—all in pursuit of greater profit margins.
24
A report from India has highlighted some of the challenges that facing API manufacturers and pointed out potential vulnerabilities that could influence medicine quality. 26 Authors reported that cost-cutting measures driven by profit motives are contributing to the deterioration of medicine quality in India. They said,
while the full effects of these practices are not yet completely understood, the combination of such cost-reduction strategies particularly in areas like the use of lower-quality packaging may have a substantial cumulative impact on the overall quality and stability of medicines. This issue is further compounded by persistent weaknesses in India’s pharmaceutical regulatory framework and the inconsistent standards applied to medicine quality across the country.
26
However, a study from Nigeria revealed that open markets also play a significant role in facilitating the circulation of SF medicines. 11 Participants noted that patent medicine vendors continue to sell prescription drugs as over-the-counter (OTC) products largely due to the presence of these open markets large-scale wholesale and retail centers that drive the importation and distribution of medicines in major cities. Operated primarily by patent medicine vendors, these markets often serve as key retail points for substandard or falsified medicines, many of which enter the country through illegal channels. 11 Evidence from Jordan indicates that economic constraints exacerbate the problem, increasing the risk of exposure to SF medicines. 23
A report from Pakistan highlights that, according to legal experts, substandard, spurious, and adulterated (CSSA) drugs pose a serious threat to public health. Participants noted that, “to date, the regulatory authorities within the healthcare system have failed to effectively address and eliminate this pressing issue.” 28 In addition, participants highlighted several significant flaws in the legislation. One participant noted that “the penalties prescribed by law were illogical, pointing out that neither the severity of the punishment outlined in Section 27 of the Drugs Act 1976 nor the corresponding sentencing guidelines were appropriate or well-defined.” 28
In addition evidence from Tanzania indicate, significant weaknesses in deterring misconduct and improving the regulatory function within Tanzania’s pharmaceutical sector, primarily due to the ineffectiveness of enforcement and the minimal perceived risks of penalties. Regulators reported that the “vast majority of violations go unpunished, with few successful prosecutions and penalties that are either lenient or inconsistently applied such as short-term shop closures or fines that are too low to act as a meaningful deterrent.” 29 This is compounded by systemic issues like potential corruption, political interference, and a lack of inter-agency coordination, which further undermine enforcement efforts. As a result, the current monitoring and reporting system fails to create a credible threat of punishment, thereby offering little incentive for compliance and ultimately limiting its capacity to deter the circulation of SF medicines. 29
Summary of drivers of Substandard and Falsified medicines from global perspectives.

Tackling the threat of SF medicines: strategies for effective mitigation
Tackling the threat of SF medicines requires comprehensive, multi-level strategies that address the weaknesses in regulatory systems, supply chains, and public awareness. Evidence indicate that effective mitigation begins with strengthening the capacity of national regulatory authorities to ensure rigorous pre- and postmarketing surveillance as shown in Ghana’s approach where FDA conducts continuous inspection, market surveillance and digital tacking systems to verify medicine authenticity. 25 Similarly, enforcing good manufacturing practices, promoting generic substitutions backed by bioequivalence data, and ensuring transparent supply chains are critical steps toward minimizing infiltration of falsified products. The authors suggested investment in laboratory infrastructure and human resource development, particularly in regulatory science and quality control. 25 This can empower low- and middle-income countries to detect and respond to quality lapses rapidly.
Evidence from Ethiopia shown education and collaborative interventions are equally critical that enhances stakeholders motivation and awareness. 22 Ethiopia’s experience illustrates that improving trust in local generics depends not only on increasing production but also on ensuring bioequivalence testing and clear communication about medicine quality. This evidence indicate, public education campaigns, targeting consumers and prescribers can reduce demand for unverified drugs and promote rational use. Evidence from Ghana suggest, technology driven tools such as mobile authentication system and digital reporting platforms strengthen community participation and regulatory oversight. 25 Collectively, integrating these measures regulatory strengthening, technological innovation, stakeholder collaboration and behavioral changes provides a sustainable framework to mitigate the circulation of SF medicines and safeguard public health.
In addressing the threat of SF medicines, existing evidence proposes a multi-faceted mitigation strategy that emphasizes regulatory reinforcement, public and professional education, and systemic transparency. 40 A study from South Africa highlights the disconnect between public perception and actual drug quality, advocating for improved communication and participatory policy-making to build trust in generic medicines. 35 In addition, evidence from Jordan identified low awareness and inadequate reporting mechanisms, urging nationwide educational campaigns, structured pharmacovigilance systems, and economic interventions to reduce reliance on risky sources. 23 Similarly, a study from Yemen points to weak regulation, unqualified dispensers, and widespread counterfeit medicines, calling for updated curricula, stricter licensure, continuous professional development, and stronger enforcement to safeguard pharmaceutical practice. 33 Together, these stress that effective SF medicine mitigation requires coordinated action across education, regulation, and socio-economic support to protect public health.
One of bottle neck for quality of medicines is supply and demand issues. Evidence from Pakistan indicated, on supply side, strengthening regulatory frameworks and enforcement mechanism is critical. 28 This include enhancing post-marketing surveillance, improving the capacity and reliability of the drug testing laboratories and securing the pharmaceutical supply chain through track, and trace technologies such as barcoding and serialization. In addition, effective prosecution of offenders also depends on the proper training of drug inspectors, regulators, and law enforcement to correctly interpret and implement drug laws. 28 On the demand side, efforts must focus on improving access to affordable and quality medicines, as high drug prices and limited availability are key drivers of SF medicines consumption. 17 , 37 Public awareness campaigns, led by trained healthcare professionals, can help educate consumers about the risks of SF medicines but these must be coupled with systemic improvements in medicines affordability and availability. In many low- and middle-income countries, the informal medicine market thrives because the formal system failed to meet patient needs. 41 Therefore, policies that ensure a consistent supply of essential medicines through better procurement system, local production and price control are essential. 17 Without addressing the structural and economic factors that fuel the demand for SF medicines, regulatory measures alone will remain insufficient.
Strengths and limitations of the review
This review has several remarkable strengths. As per existing evidence, this is the first systematic review to synthesize qualitative evidence specifically on the drivers and mitigation strategies for SF medicines. This may fill a critical gap in the existing literatures, which is dominated by quantitative prevalence studies. By integrating rich, contextual insights from diverse low- and middle-income countries, the review provides a nuanced understanding of the complex socio-economic, regulatory, and behavioral factors that quantitative methods alone cannot fully capture. Methodological rigor was ensured through a comprehensive search strategy across multiple databases, a transparent study selection process following PRISMA guidelines, and a systematic quality assessment of all included studies using the established CASP checklist. Furthermore, the thematic analysis yielded a robust framework of interconnected drivers, and multi-prolonged mitigation strategies, providing a holistic evidence-base for policymakers and regulators
Despite its strength, this review is subjected to certain limitations. The inclusion criteria were restricted to studies published in English, which may have led to the omission of relevant qualitative research published in other languages, potentially introducing a language bias and limiting the geographical scope of the findings. A remarkable methodological limitation was also observable due to the absence of a formal assessment of synthesis confidence using the GRADE-CERQual (Confidence in the Evidence from Reviews of Qualitative Research) approach. As with any systematic review, the findings are constrained by the scope and quality of the primary studies available. In addition, the review findings were inherently limited by the scope and quality of the primary studies available. In this review, the lack of qualitative research on SF medicines resulted in the inclusion of only a small number of studies for data synthesis. The inherent nature of qualitative research, while providing rich and in-depth insights, limits the generalizability of findings beyond the specific contexts studied and may not fully represent all low- and middle-income countries. Moreover, this review synthesizes perceived drivers and proposed strategies but does not empirically assess the real-world effectiveness or practical feasibility of the identified mitigation interventions.
Conclusion and outlook
This review demonstrates that combating SF medicines requires moving beyond isolated interventions to address a synergistic network of drivers. The most salient evidence points to the compounding effects of economic pressures incentivizing poor quality, inadequate regulatory oversight failing to deter it, fragile supply chains enabling its circulation, and public misconceptions sustaining its demand. As a result, effective action demands a coordinated policy response that strengthens enforcement to counter regulatory weakness, employs digital tools to secure supply chains, and invests in public and professional education to fill knowledge gaps and rebuild trust, particularly in affordable, quality-assured generics. The outlook for progress hinges on translating this systemic understanding into tangible action. Future initiatives should focus on implementing integrated strategies with dedicated funding and cross-sectoral collaboration. To support this, a critical need exists for further applied qualitative research to evaluate the feasibility and effectiveness of proposed mitigations in specific local contexts and to understand the barriers to their adoption. In addition, by grounding policy in the tangible context of specific evidence, the stakeholder can develop more resilient, adaptive, and ultimately successful frameworks to ensure access to safe, effective medicines and achieve positive public health outcomes worldwide.
Supplemental Material
sj-docx-1-map-10.1177_27550834261435773 – Supplemental material for Drivers and strategies to combat substandard and falsified medicines in low- and middle-income countries: A systematic review of qualitative evidence
Supplemental material, sj-docx-1-map-10.1177_27550834261435773 for Drivers and strategies to combat substandard and falsified medicines in low- and middle-income countries: A systematic review of qualitative evidence by Yesuneh Tefera Mekasha, Melese Tilahun, Addisu Afrassa Tegegne, Mikael Abraham, Abibo Wondie Mekonnen and Melaku Getahun Feleke in The Journal of Medicine Access
Supplemental Material
sj-docx-2-map-10.1177_27550834261435773 – Supplemental material for Drivers and strategies to combat substandard and falsified medicines in low- and middle-income countries: A systematic review of qualitative evidence
Supplemental material, sj-docx-2-map-10.1177_27550834261435773 for Drivers and strategies to combat substandard and falsified medicines in low- and middle-income countries: A systematic review of qualitative evidence by Yesuneh Tefera Mekasha, Melese Tilahun, Addisu Afrassa Tegegne, Mikael Abraham, Abibo Wondie Mekonnen and Melaku Getahun Feleke in The Journal of Medicine Access
Footnotes
Ethical Considerations and Consent to Participate
Ethical approval and informed consent were not required for this review.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data sets generated during and/or analyzed during the current study are available from the corresponding authors or upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
