Abstract
The trait-based tendency to respond rashly to emotions is robustly tied to many forms of psychopathology and poor behavioural outcomes, including aggression and suicidality. Researchers have found associations between response inhibition and emotion-related impulsivity; however, effect sizes are often small. Because emotion-related impulsivity emerges in the context of heightened positive and negative emotions, arousal is a candidate trigger of impulsivity. The goals of the present study were to (1) replicate the association between emotion-related impulsivity and response inhibition, and (2) test whether emotion-related impulsivity is associated with arousal-induced decays in response inhibition performance. Participants (N = 55) completed a self-report measure of emotion-related impulsivity, and then completed a computer-based response inhibition task (the antisaccade task, in which participants must make a rapid saccadic eye movement away from a cue rather than toward it) before and after a well-validated stress induction (the Trier Social Stress Test). Psychophysiological indices of arousal were measured throughout the session. Findings provide partial support for the association between emotion-related impulsivity and pre-stress response inhibition. Contrary to hypotheses, emotion-related impulsivity did not interact with arousal to predict post-stress response inhibition performance after controlling for pre-stress response inhibition performance. Future research is needed to consider clinical samples and to assess whether emotion-related impulsivity is related to deficits in other facets of cognitive control and decision-making.
Emotion-related impulsivity is the trait-like tendency to respond to emotion with rash speech or behaviour. Early work identified the trait Negative Urgency, the tendency to react rashly to negative emotion (Whiteside et al., 2005; Whiteside and Lynam, 2001). Researchers extended work to include Positive Urgency, the tendency to react rashly to positive emotion (Cyders and Smith, 2007). Evidence from factor and network analyses across clinical and non-clinical samples supports combining Positive and Negative Urgency to form a higher order factor, referred to as emotion-related impulsivity (Billieux et al., 2021; Carver et al., 2011; Cyders et al., 2007; Sperry et al., 2018).
Emotion-related impulsivity correlates with many forms of psychopathology and problematic behaviours (Bari and Robbins, 2013; Johnson et al., 2013). In a meta-analysis with 40,000 people, emotion-related impulsivity, when compared with other aspects of impulsivity, was the strongest predictor of every form of psychopathology studied, including depression, eating disorders and suicidality (Berg et al., 2015).
Emotion-related impulsivity appears distinct from emotional reactivity. A growing body of work using diverse inductions and indicators has not found correlations between emotion-related impulsivity and emotional reactivity (Cyders and Coskunpinar, 2010; Johnson et al., 2016). Rather than reflecting emotional reactivity, it is likely emotion-related impulsivity involves neurocognitive processes leading to lack of control in response to emotion (e.g. Clark, 2005). Nonetheless, we considered whether emotion-related impulsivity, or Negative Urgency specifically, relates to reactivity given prior work linked Negative Urgency to greater reactivity to a stress induction (Owens et al., 2018).
Many have suggested emotion-related impulsivity may involve poor response inhibition (Bechara and Van der Linden, 2005; Carver et al., 2008; Johnson et al., 2016), a facet of cognitive control that corresponds to the ability to override an action or prepotent response (Miyake and Friedman, 2012). Response inhibition tasks are often used as a behavioural measure of impulsivity (Cyders and Coskunpinar, 2011), and meta-analytic evidence indicates that high emotion-related impulsivity significantly relates to poor response inhibition (Cyders and Coskunpinar, 2011; Johnson et al., 2016).
A handful of studies have examined response inhibition after altering emotional valence through mood inductions. In one study, persons high in emotion-related impulsivity, as compared with controls, showed relatively greater recruitment of the frontoparietal network when completing trials of a response inhibition task involving negative images, suggesting the need for compensatory effort to obtain comparable performance (Chester et al., 2016). In studies examining response inhibition after inducing positive or negative affect, higher emotion-related impulsivity is related to poorer response inhibition after the affect induction (Dekker and Johnson, 2018; Johnson et al., 2016), though the minor effects of the affect inductions suggest a need for additional research. Naturalistically occurring variations in arousal have been found to predict decays on subsequent cognitive control trials for those with higher emotion-related impulsivity, but not for those with lower levels (Pearlstein et al., 2019). Because these studies only considered modest shifts in arousal, the current investigation was designed to determine the effects of high arousal, induced using a stress manipulation.
In this study, participants completed a response inhibition task, underwent an arousal induction and then completed a second iteration of the response inhibition task. We hypothesised that (1) higher emotion-related impulsivity will relate to poorer overall response inhibition performance, and (2) higher emotion-related impulsivity will interact with arousal, such that higher emotion-related impulsivity will relate to greater arousal-induced decay in response inhibition performance. Arousal included measures of autonomic nervous system activation (heart rate) and hypothalamic–pituitary–adrenal (HPA) axis activation (cortisol). Because Negative Urgency is specifically characterised by responses to negative affect, we also conducted post hoc analyses examining Negative Urgency.
Method
The university Institutional Review Board approved all study procedures before data collection (University of California Berkeley Center for the Protection of Human Subjects Protocol ID Number 2016-01-8316). Before conducting analyses, a pre-registration was submitted to the Open Science Framework (osf.io/tkh72).
Participants
Participants (N = 59) were recruited from psychology courses at a large public university and were offered research participation credit in exchange for their participation. Five participants were excluded from analyses: one for withdrawing consent during the stress induction, three for failing to complete self-report measures (two failed to complete sufficient self-report items and one failed to accurately answer attention catch trials), and one for not successfully obtaining 50% accuracy on prosaccade trials of the response inhibition task. The analysis sample (N = 54) was 67.9% female, age M = 20.13, SD = 3.56. Participants reported their ethnicity as 31% European/Caucasian, 33% Asian, 17% Hispanic/Latino and 9% Other (10% chose not to disclose).
Procedures
Potential participants provided informed consent and then completed an online survey assessing exclusion criteria, including confounds of cardiovascular response, such as physician-diagnosed hypertension, heart murmur, presence of a pacemaker or other implanted cardiovascular device, and use of medications affecting cardiovascular system responses (e.g. beta-adrenergic blocking agents). Participants who provided cortisol (n = 25) met additional exclusion criteria to rule out potential confounds that could influence salivary hormone levels, including Cushing’s disease, pregnancy, gum disease and the use of steroids or chewing tobacco. Participants who met study criteria completed a measure of emotion-related impulsivity. The online questionnaires included attention check items (e.g. ‘Please answer “Agree” to this item’) and participants were required to accurately answer 75% of attention check items. Eligible participants were invited to attend an in-person session. Participants were scheduled between 9:00 a.m. and 5:00 p.m. and were asked to avoid eating and moderate-to-heavy exercise within 2 h of their session, with additional restrictions for the cortisol subsample, who were asked to consume sufficient calories at lunch and to avoid high sugar food or drink. Participants who gave cortisol samples completed their experimental laboratory sessions from 2:00 p.m. to 5:00 p.m. to control for diurnal effects of cortisol.
At the start of the in-person laboratory session, signed informed consent was obtained and possible confounds were assessed, including prior night and typical night hours of sleep; past 24-h nicotine, caffeine, alcohol, marijuana, allergy or cold medication use; and food and drink consumption. Participants provided baseline physiological measurements, completed the antisaccade task as an index of response inhibition, underwent the Trier Social Stress Test (TSST) and completed a second iteration of the antisaccade task to assess arousal-induced effects on response inhibition. Participants completed self-rated measures of affect at baseline and after the TSST. A subset of participants had salivary cortisol collected; because salivary cortisol response is delayed (Dickerson and Kemeny, 2004), salivary cortisol was measured at three time points: before the stress induction, 15 min post-stressor and 25 min post-stressor to assess stress reactivity and recovery. At the end of the session, participants were asked to what extent they were aware the study sought to elicit stress. Participants were debriefed about the stress induction procedures and reconsent was obtained.
Measures
Three-factor impulsivity index
The three-factor impulsivity index was derived from multiple scales and novel items designed to measure heterogeneous forms of impulsivity (Carver et al., 2011). The measure includes three factor-analytically derived subscales, including two factors covering emotion-related impulsivity (Feelings Trigger Action and Pervasive Influence of Feelings) and a third factor of items covering impulsiveness without an emotional antecedent (Lack of Follow Through). Feelings Trigger Action covers the tendencies to engage in regrettable speech or action in response to positive or negative emotions, including items drawn from the Positive and Negative Urgency scales (e.g. ‘When I feel a desire, I act on it immediately’ and ‘When I feel bad, I will often do things I later regret in order to make myself feel better now’). Pervasive Influence of Feelings refers to unconstrained effects of (mostly negative) emotion on cognition (e.g. ‘My feelings greatly affect how I see the world’) and motivation (e.g. ‘When I feel sad, it paralyzes me’). Lack of Follow Through refers to impulsiveness interfering with attention and with completion of intended actions, and consists of items without reference to emotions (e.g. ‘I am easily distracted by stray thoughts’). The emotion-related impulsivity factors have been shown to relate to early adversity, as well as symptoms of depression, anxiety, mania, externalising disorders and suicidality (Auerbach et al., 2017; Johnson et al., 2013a, 2013b). Lack of Follow Through enables tests of discriminant validity of emotion-related as compared to non-emotion-related impulsivity, and has demonstrated diminished correlations with psychopathology and neurocognitive indices (Pearlstein et al., 2019). In this sample, the three subscales have high internal consistency (Feelings Trigger Action: α = .88, Pervasive Influence of Feelings: α = .89 and Lack of Follow Through: α = .92).
Self-reported negative affect (Positive and Negative Affect Schedule – PANAS-S)
Before and after the arousal manipulation, we assessed high arousal negative affect to verify the effect of the TSST. High arousal negative affect was measured using seven items (e.g. ‘distressed’ and ‘upset’) selected from the Positive and Negative Affect Scales (Watson et al., 1988). Each item was rated on a 1 = ‘Very Slightly or Not at All’ to 5 = ‘Extremely’ scale. Scores on these items were summed. The negative affect items had high inter-item reliability both before (α = .88) and after (α = .85) the stress induction.
Antisaccade task
The antisaccade task (Kane et al., 2001) is a well-validated measure of response inhibition (Miyake and Friedman, 2012). The antisaccade task used in this study was designed in compliance with recommended standards (Antoniades et al., 2013). Prosaccade trials begin with a fixation cross in the centre of the screen (randomly varying between 200 and 1800 ms), followed by a blank screen (10 ms), and then a cue (=) on either the left or right side of the screen at a visual angle of 8.68° (250 ms). A randomly selected target (B, P or R) is presented on the same side of the screen as the cue (100 ms), immediately followed by the letter H as a mask (50 ms), and then the number 8. Participants are instructed to indicate which letter they saw before the mask and are given up to 10 s to respond by button press. Trials were separated by a 400-ms inter-trial interval. Antisaccade trials are identical to prosaccade trials, differing only in whether the cue and target letter appear on the same (prosaccade) or opposite (antisaccade) side of the screen as the target letter. An accurate response on an antisaccade trial requires inhibiting the reflexive and automatic response to look toward the cue and, instead, to override that prepotent response in order to look to the opposite side of the screen for the letter. Before the stress induction, participants received instructions, completed prosaccade and antisaccade practice trials, and then 10 prosaccade and 40 antisaccade trials. After the stress induction, participants received reminder instructions and then completed 40 antisaccade trials.
Consistent with previous work, we operationalised response inhibition as average accuracy on antisaccade trials (i.e. proportion of correct trials), controlling for average accuracy on prosaccade trials (Roberts et al., 1994). Participants were given a score of 1 when they correctly identified the target letter and a score of 0 when they failed to identify a letter or identified the incorrect letter. Accuracy scores were the average across the trials in that block (prosaccade, pre-stress antisaccade or post-stress antisaccade). Because the interval for responding is very brief, reaction time showed little variability and was not used in analyses.
TSST
The TSST is a well-validated laboratory task designed to induce acute psychosocial stress (Frisch et al., 2015; Kirschbaum et al., 1993). The TSST has been shown to consistently induce self-reported and physiological stress, including increased activation of the HPA axis as indexed by cortisol response as well as increased activation of the sympathetic nervous system (SNS; Allen et al., 2014). During the TSST, participants are given 5 min to prepare and 5 min to perform a speech about their qualifications for a prospective job for a panel of evaluative judges. Then they are asked to complete a 5-min arithmetic task consisting of serial subtractions. We used a modified version of the task that involves a single evaluator as opposed to a panel of evaluative judges; multiple studies have confirmed that this modified TSST induces comparable levels of physiological and psychosocial stress (Frisch et al., 2015). TSST evaluators were trained and selected based on their ability to provide ambiguous non-verbal feedback and assertive, terse and non-reassuring responses.
Physiological measurement
Stress is known to elicit responses in the autonomic nervous system and HPA axis. We assessed autonomic nervous system and HPA-axis activation (cortisol) during the TSST. Due to limitations in sample size (n = 20) in cortisol collection, cortisol findings must be interpreted with caution.
Autonomic nervous system
To measure autonomic activation, electrocardiography (ECG) was collected continuously during a 5-min baseline period, throughout the TSST procedures, and during the second block of the response inhibition task at a sampling rate of 1 kHz. ECG recordings were obtained using a Standard Lead II configuration. Signals were integrated and recorded with a Biopac MP150. Heart rate was used as a measure of arousal. Heart rate was calculated by measuring the magnitude between successive R spikes in the ECG. Artefacts were flagged by statistical algorithms (e.g. Berntson et al., 1990), and were then checked visually and edited as necessary according to the guidelines of the Society for Psychophysiological Research Committee Report. Although higher heart rate is predominantly associated with lower parasympathetic system activation (Mendelowitz, 1999), sympathetic activation can also contribute to increased heart rate (Wang et al., 2018). Mindware Technologies Ltd. was used to analyse and inspect the data in 1-min epochs. Values across the 5-min baseline period were averaged to yield a mean baseline value. Values during the TSST were averaged to obtain a mean stress reactivity value.
HPA axis
Salivary cortisol is used to measure activity of the HPA axis, a neuroendocrine system that reacts to stress and energetic demand (Dickerson and Kemeny, 2004; Kozlov and Kozlova, 2014). Cortisol promotes energy expenditure during high metabolic states, such as arousal. A subset of participants (n = 25) provided three cortisol samples. Saliva was collected using passive drool kits and analysed using ELISA (Enzyme-Linked Immunosorbent Assay) assay kits from Salimetrics. Samples were stored in a −80°C freezer, thawed and centrifuged before assaying. Each sample was assayed twice for reliability, and the average concentration of cortisol across the two measurements was used. Samples with intra-assay values (i.e. the estimate of similarity across the two samples) greater than 20% were treated as missing data. Cortisol data from five participants were unusable (e.g. insufficient saliva produced) and excluded from analyses. Cortisol reactivity (n = 20) was operationalised using area under the ruve in respect to increases (AUCi; Pruessner et al., 2003) to provide a single score to capture the level of increase during the TSST as compared to baseline and the extent to which that increase is sustained over time.
Data analysis plan
Preliminary analyses assessed potential confounds. Effectiveness of the experimental manipulation of arousal was verified by assessing change in negative affect ratings and physiological arousal. To test the first hypothesis, we considered partial correlations between emotion-related impulsivity with baseline antisaccade accuracy, controlling for prosaccade accuracy; parallel tests were conducted with non-emotion-related impulsivity as a control comparison. To test the second hypothesis, we conducted a hierarchical linear regression model to consider the main effects and interaction of emotion-related impulsivity and arousal on post-stress response inhibition controlling for pre-stress response inhibition. For control comparison, analyses included the non-emotion-related impulsivity subscale. Post hoc analyses assessed the effects of Negative Urgency specifically.
Results
Analyses were completed using SPSS, Version 24, using two-tailed tests with alpha = .05. All variables were normally distributed, with skew and kurtosis estimates <1.5. As shown in Table 1, variability was adequate for each key variable. Also shown in Table 1, on average, participants showed improved response inhibition performance in the block after the stressor as compared to pre-stress (paired t = 4.67, df = 54, p < .001).
Descriptive characteristics of key variables (N = 54).

TSST: Trier Social Stress Test.
AUCi refers to area under the curve in respect to increases and values reflect reactivity during the TSST compared to baseline and the degree to which that change was sustained (n = 20).
Potential confounds
Baseline response inhibition (antisaccade) scores and physiological arousal were not related to age, caffeine, alcohol, medications, nicotine use nor hours of sleep, all |rs| < .25, ps > .12. As such, these variables were not included in subsequent analyses. In post-task debriefing, 74% of participants stated that they were aware the study was related to stress reactivity. Self-reported awareness of our focus on stress reactivity did not relate to arousal (r = .15, p = .48), indicating awareness about the study procedures did not influence effectiveness of the manipulation.
Task engagement and efficacy of arousal induction
We verified the TSST successfully elicited arousal across measures, as indicated by significant increases in self-reported high arousal negative affect (t = 6.55, p < .01), SNS activation (heart rate: t = 6.16, p < .001), and HPA-axis activation (cortisol: t = 9.55, p < .001). To determine whether the stress induction elicited comparable physiological reactivity across levels of impulsivity, we examined correlations of impulsivity scores with the degree of change in arousal during the TSST. As shown in Table 2, none of the impulsivity factors significantly correlated with heart rate or self-reported negative affect (|rs| < .18, ps > .17). HPA-axis activation also appeared comparable across levels of impulsivity for Feelings Trigger Action, Pervasive Influence of Feelings and Lack of Follow Through (|rs| < .32, ps > .16). Also shown in Table 2, we did not find associations between Negative Urgency and heart rate or negative affect (|rs| < .14, ps > .33). Findings suggest comparable levels of arousal and negative affect during the TSST across levels for the three impulsivity factors and Negative Urgency.
Pearson correlations between key variables (N = 54).

TSST: Trier Social Stress Test.
p < .05, **p < .01.
Hypothesis 1: Emotion-related impulsivity and overall response inhibition
We examined partial correlations between the three impulsivity factors and pre-stress antisaccade accuracy, controlling for prosaccade accuracy. Although three of the partial correlations were in the hypothesised direction and the bivariate correlation of Feelings Trigger Action with pre-stress antisaccade accuracy was significant (see Table 2), none of the partial correlations were significant (Feelings Trigger Action: partial r(52) = −.24, p = .08; Pervasive Influence of Feelings: partial r(52) = −.14, p = .31; Lack of Follow Through: partial r(52) = −.18, p = .18). We did not observe logarithmic or polynomial relationships between emotion-related impulsivity and pre-stress response inhibition (|rs| < .23, ps > .09). Contrary to hypotheses, partial correlations between emotion-related impulsivity and pre-stress response inhibition were not significant; however, effects were in the hypothesised direction.
Hypothesis 2: Emotion-related impulsivity and arousal-induced effects on response inhibition
To test our main hypothesis that emotion-related impulsivity would interact with arousal to relate with post-stress response inhibition, we conducted a hierarchical linear regression model. Block 1 controlled for antisaccade pre-stress response inhibition performance, Block 2 included main effects for arousal (heart rate) and the three impulsivity factors (Feelings Trigger Action, Pervasive Influence of Feelings and Lack of Follow Through) and Block 3 included the three 2-way interaction terms for arousal with each form of impulsivity. As shown in Table 3, only the first block containing pre-stress antisaccade was significant, with a very large effect size (β = 0.78, p < .001). Adding the impulsivity factors and arousal (Block 2) and their interactions (Block 3) did not significantly improve the model (
Hierarchical linear regression relating arousal, impulsivity and their interactions to post-stress response inhibition (antisaccade) performance, controlling for pre-stress response inhibition performance (N = 54).
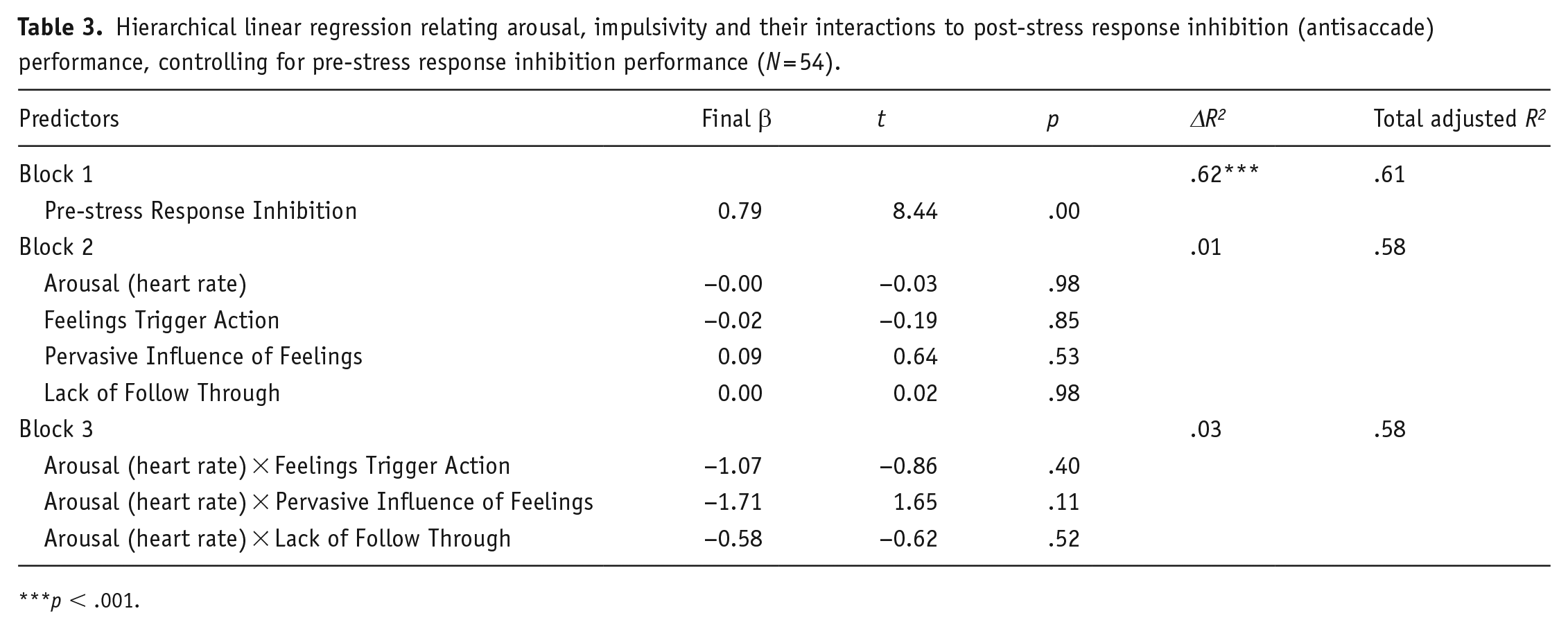
p < .001.
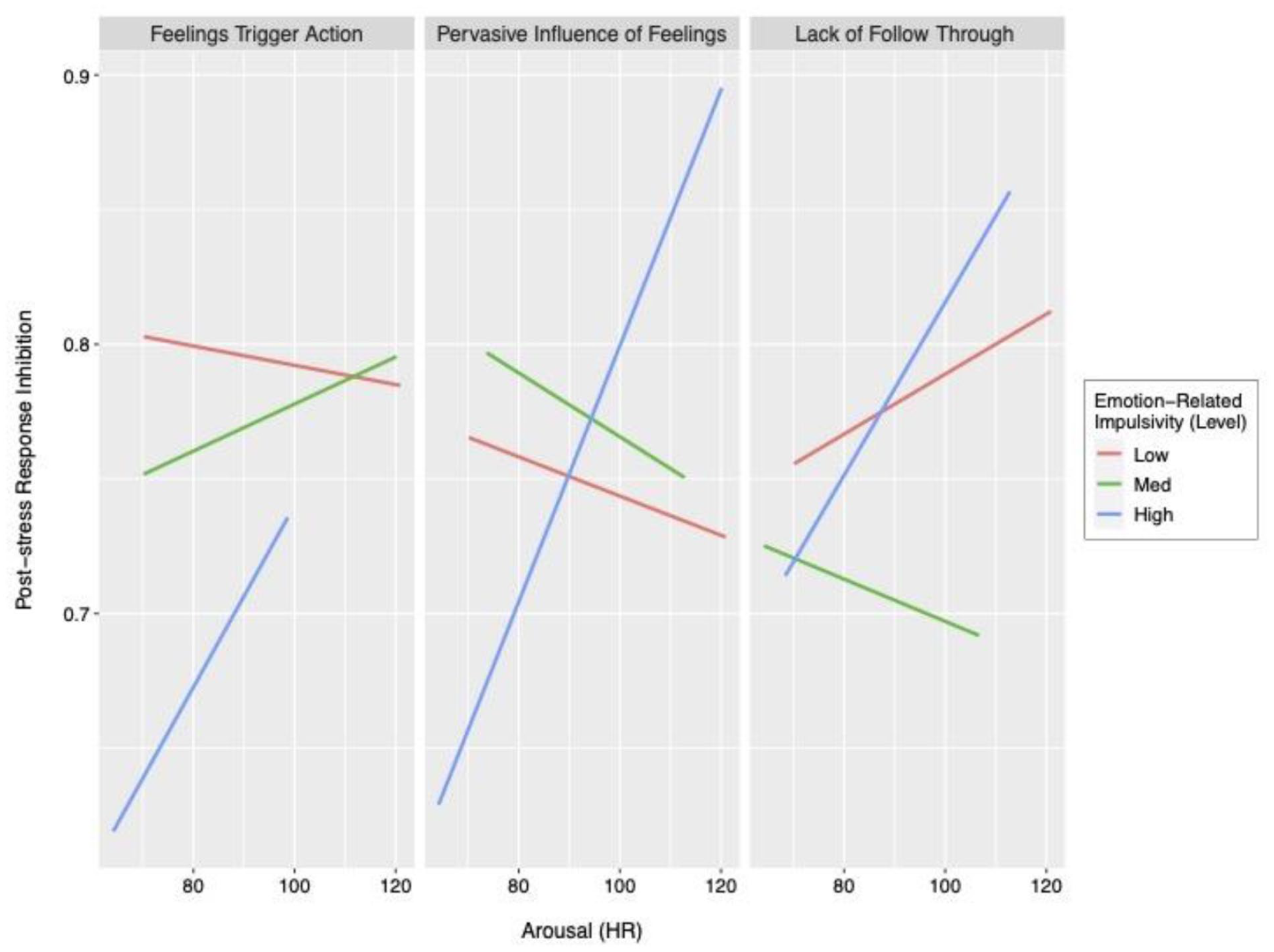
Interaction of the three impulsivity factors and arousal (heart rate) in relation to post-stress response inhibition performance.
Hierarchical linear regression model relating cortisol, impulsivity and their interactions to post-stress response inhibition performance, controlling for pre-stress response inhibition performance (n = 20).

TSST: Trier Social Stress Test.
Cortisol is measured using AUCi, which refers to area under the curve with respect to increases and corresponds to reactivity during the TSST compared to baseline and the degree to which this change was sustained.
p < .001.
We conducted a parallel hierarchical linear regression model to assess the potential role of HPA-axis activation (cortisol). Block 1 controlled for antisaccade pre-stress response inhibition performance, Block 2 included main effects for arousal as measured by cortisol AUCi and the three impulsivity factors (Feelings Trigger Action, Pervasive Influence of Feelings and Lack of Follow Through) and Block 3 included the three 2-way interaction terms for arousal with each form of impulsivity. As shown in Table 3, only the first block containing pre-stress antisaccade was significant, with very large effect sizes (βs > 0.75, ps < .001). There was a main effect for arousal such that greater cortisol reactivity was associated with poorer post-stress response inhibition (β = 0.34, t = 3.27, p < .05).
Exploratory post hoc analyses
We conducted parallel analyses to test the specific role of Negative Urgency for Hypotheses 1 and 2. The pattern of null results observed for Feelings Trigger Action and Pervasive Influence of Feelings was similarly observed for Negative Urgency.
Discussion
Emotion-related impulsivity is associated with diverse forms of psychopathology and a range of poor behavioural outcomes, making it important to understand neurocognitive correlates related to this tendency. This study offers a test of a putative model of emotion-related impulsivity by examining whether those with high emotion-related impulsivity exhibit deficits in response inhibition during periods of high arousal. Previous work on this form of impulsivity and neurocognition has been hampered by relatively low levels of arousal. We used a well-validated stress induction technique, and we successfully induced high arousal, as evidenced by increases in subjective affect, heart rate and cortisol. The success of the induction did not appear confounded by levels of emotion-related impulsivity. We were also able to rule out many confounds, through sample selection and statistical analyses. We aimed to test whether people with high emotion-related impulsivity showed poor response inhibition overall, and whether they showed a decay in response inhibition performance during high arousal.
We partially replicated previous findings relating emotion-related impulsivity to poor response inhibition (Cyders and Coskunpinar, 2012; Johnson et al., 2016). The bivariate correlation between pre-stress response inhibition and Feelings Trigger Action, but not Pervasive Influence of Feelings, was moderate and statistically significant. Nonetheless, contrary to our predictions, partial correlations controlling for prosaccade performance were not significant – suggesting that difficulties related to emotion-related impulsivity were closely tethered to performance on the more basic prosaccade task, rather than specific to cognitive control. The limited support for the relationship between emotion-related impulsivity and pre-stress response inhibition dovetails mounting evidence that this relationship may only emerge consistently in samples with higher emotion-related impulsivity. Indeed, one meta-analysis concluded the relationship between emotion-related impulsivity and response inhibition is evident in clinical samples but not in community or student samples (Johnson et al., 2016). Although studies conducted with larger samples of undergraduates found a curvilinear relationship in which emotion-related impulsivity and response inhibition were related at higher levels (Dekker and Johnson, 2018; Johnson et al., 2016), we were unable to replicate that pattern in this small sample. The inability to replicate the association between self-reported and behavioural impulsivity coheres with an abundance of studies documenting poor convergence between self-report and behavioural impulsivity (Duckworth and Kern, 2011; Mazza et al., 2020).
Our primary focus, though, was to extend this work by considering response inhibition during states of high arousal. Contrary to hypotheses, we did not find that emotion-related impulsivity, whether measured using Feelings Trigger Action, Pervasive Influence of Feelings or Negative Urgency, is related to individual differences in arousal-induced decay in response inhibition. Cortisol reactivity is related to poorer post-stress response inhibition; however, there were no interactions with the emotion-related impulsivity factors as hypothesised. Whereas this finding suggests that cortisol may impair response inhibition performance, that contradicts meta-analytic findings (Shields et al., 2016) and may be spurious given the very small sample size for cortisol analyses. Notably, post-stress response inhibition was robustly predicted by pre-stress response inhibition, leading to little variability to account for after controlling for pre-stress response inhibition performance. None of the other parameters we examined explained additional variance after accounting for pre-stress response inhibition.
Numerous models have been posited to explain how stress impacts cognitive control, and here we discuss two that are relevant for interpreting the present findings. First, the Yerkes–Dodson law (Yerkes and Dodson, 1908) proposes that stress enhances cognitive control to a certain extent, and then at high levels, stress impairs performance on cognitively complex tasks, forming an inverted-U relationship between arousal and task performance. Using this model, it is possible we did not elicit sufficiently high arousal to elicit cognitive performance decays, or the task was not sufficiently challenging to be sensitive to arousal. Because we found evidence supporting arousal manipulation and variability in task performance, we believe results are instead due to the task selected and the domain of cognitive control studied. We observed overall improvement in response inhibition performance, which is consistent with previous work showing a practice effect on this task (Ettinger et al., 2003). The improved response inhibition post-stress also fits with meta-analytic evidence suggesting that in non-clinical samples, response inhibition may be a facet of cognitive control that is enhanced, rather than hindered, by stress (Shields et al., 2016).
Second, neurobiological models (e.g. Arnsten et al., 2015; Hermans et al., 2014) indicate stress leads to excessive threat-related subcortical activity, such as the amygdala and basal ganglia, and the reduction of prefrontal cortical activity essential for cognitive control. These models posit that stress elicits high levels of catecholamines (i.e. noradrenaline and dopamine) that ‘turn off’ the prefrontal cortex and reallocate activity to more primitive subcortical networks. Although neurobiological models have garnered significant support, these models do not explain why stress enhances response inhibition performance (e.g. Schwabe et al., 2013). These models also suggest that greater physiological arousal leads to greater cognitive control decay via catecholaminergic systems, suggesting that the lack of support for this model using heart rate or cortisol as indices of arousal may be because other pathways are more relevant. For example, previous research has found the norepinephrine activity, as indexed through pupil dilation, relates to arousal-induced decays in response inhibition for those high in emotion-related impulsivity (Pearlstein et al., 2019). Norepinephrine appears to more directly and immediately influence cognitive control than cortisol (Shields et al., 2016). Therefore, it is possible that the physiological measures used were poor indicators of the neurobiological pathway by which arousal influences response inhibition.
Study findings are also limited by sample characteristics. We were limited in only having cortisol data on a small subset of the sample and have provided these data only to inform future research. Participants were primarily cis-gendered women (67.9% identified as female) and were all undergraduates at a large public university. Additional work is needed to ensure these findings replicate and generalise.
This study provides evidence to evaluate a model explaining how emotion-related impulsivity emerges. Our findings provide partial support replicating the relation between emotion-related impulsivity and response inhibition but raise questions about whether that effect is specific to response inhibition versus performance on other cognitive control tasks. Of more import, we did not find support for the idea that emotion-related impulsivity relates to arousal-induced decays in response inhibition. Future research is warranted to evaluate other physiological mechanisms, such as norepinephrine, alternate high arousal inductions, and other tasks and facets of cognitive control and decision-making that may be more sensitive to arousal-induced decays. It is also possible that individuals differ in the mechanisms through which emotions shape impulsive responses. More research is warranted to better characterise the neurocognitive processes associated with emotion-related impulsivity.
Footnotes
Authors’ note
Portions of these findings were presented as a conference presentation at the 2019 Association for Behavioral and Cognitive Therapies (ABCT) in Atlanta, Georgia.
Data availability statement
This study was pre-registered prior to analyses, and anonymised data and analytical code are available on the Open Science Framework (osf.io/tkh72).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data collection and preliminary analysis were supported by the National Science Foundation’s Graduate Research Fellowship Program and the National Institute of Mental Health (R01 MH110477).
