Abstract
The proliferation of harmful gases is a major global problem caused by anthropogenic activities. This study hypothesized that the emission of noxious gases varies seasonally in indoor and outdoor environments. Air samples were collected from 40 georeferenced residential areas (N = 40) in Port Harcourt using Aeroqual 500 monitor to measure six gases: CH4, CO, NH4, NO2, O3, and SO2. Samples were collected at different times, locations, and seasons. The analysis of variance (ANOVA) indicates a significant difference in concentration between gases, times, and locations at p < 0.001. But the interaction effect shows that times and seasons significantly affect noxious gas concentration compared to location. In general, the concentration of CO was the highest, while that of O3 was the lowest. The order of concentration is CO > SO2 > NO2 > CH4 > NH4 > O3. The concentrations of NO2 and SO2 were higher than the WHO limit, while CO, CH4, NH, and O3 were below the WHO limit. Furthermore, CO concentration was highest in the morning, while NO2 and SO2 concentrations were highest in the evening. For seasons, the CH4, NH4, NO2, and SO2 were higher during the dry season, while CO concentration was higher during the wet season. Pollutant levels should be regularly monitored to help provide exposure advisory to city dwellers on the impact of atmospheric pollution on their health.
Introduction
Life has significantly increased in abundance, complexity, and diversity over the earth’s history. Still, human activities have continuously altered the earth’s environment, posing a serious threat to the inhabitants of the earth. 1 The earth’s system is comprised of the atmosphere, hydrosphere, and lithosphere. The gaseous components of the atmosphere are nitrogen (78%), oxygen (21%), argon (1%), and some trace elements. 2
Anthropogenic activities that release poisonous gases into the atmosphere include industrialization, urbanization, deforestation, vehicular transport, building, agriculture, road construction, and traffic congestion. These activities are known to have impacted the eco-environment negatively. The primary sources of indoor air pollution worldwide include the combustion of fuels, tobacco, 3 coal, ventilation systems, emissions from furnishings, and construction materials. 4 Indoor fires can produce black carbon particles, nitrogen oxides, sulphur oxides, and mercury compounds, among other emissions. 5 Over 1.6 million people died from cooking stove fumes globally, 6 and about 396,000 of these deaths occurred in sub-Saharan Africa, with Nigeria’s contribution to this death statistics being the highest. Similarly, studies in Port Harcourt reveal that gaseous pollutants from automobiles are the major cause of air pollution, 7 and most analyzed air samples show CO, SO2, and NO2 above the WHO air quality standards. 8
Global data shows that indoor air pollution (IAP) is far more lethal than outdoor air pollution (OAP)9. Health complications from IAP include pneumonia in children, asthma, tuberculosis, upper airway cancer, and cataract. 9 Seventy-eight percent (78%) of the population uses biomass burning for cooking, contributing to IAP (IAP). 10 Sources of IAP include mosquito repellent fumes, electricity generator fumes, and smoke from cigarettes. 6 There has been a rapid increase in the use of generators over the past decade as an alternative power source for homes and commercial activities in Nigeria. Many people are often ignorant of the hazard generators cause, as they habitually expose themselves to the harmful effects while utilizing the power equipment. 11 High concentrations of carbon monoxide (CO), nitrogen dioxide (NO2), sulphur dioxide (SO2), and benzene are responsible for sickness in children. 12 Carbon monoxide (CO) is a very hazardous, colorless, and odorless gas emitted from the incomplete combustion of fuel in power generator sets, automobiles, and firewood. Health challenges that may arise from CO poisoning include vision and hearing impairment, 13 cerebral congestion, 14 fainting, headache, 15 dizziness, asphyxia, 16 oedema, and death. 17 This study aims to determine the concentration of indoor and outdoor air pollutants around Port Harcourt because the city is s a host to a major oil refinery, the Port Harcourt refining company, where gases are flared daily. Secondly, there is a spike in atmospheric soot from numerous artisanal refineries along the surrounding creeks of the city.
The objectives of the study, therefore, were (1) to assess the concentration of harmful gases inside and outside residential buildings; (2) to determine the concentration between different collection times (i.e., frequency of collection); and (3) to evaluate the seasonal difference in air quality at different residential areas.
Materials and methods
Description of study area
Port Harcourt is one of the major cities in Nigeria (Figure 1) and serves as the capital of Rivers State.18,19 It is the fourth most populated city in Nigeria, with many industries around it. It lies along Bonny River, part of the eastern tributary of the Niger River, 66 km upstream from the Gulf of Guinea, located in Nigeria’s coastal region. Port Harcourt metropolis is partly situated in a wetland ecosystem between Latitudes 4° 45′ N and 4° 55′ N and Longitudes 6° 55′ E and 7° 05′ E with 15.83 m elevation above sea level.
20
Map of study area showing areas of air sample collection around Port Harcourt, Rivers State, Nigeria.49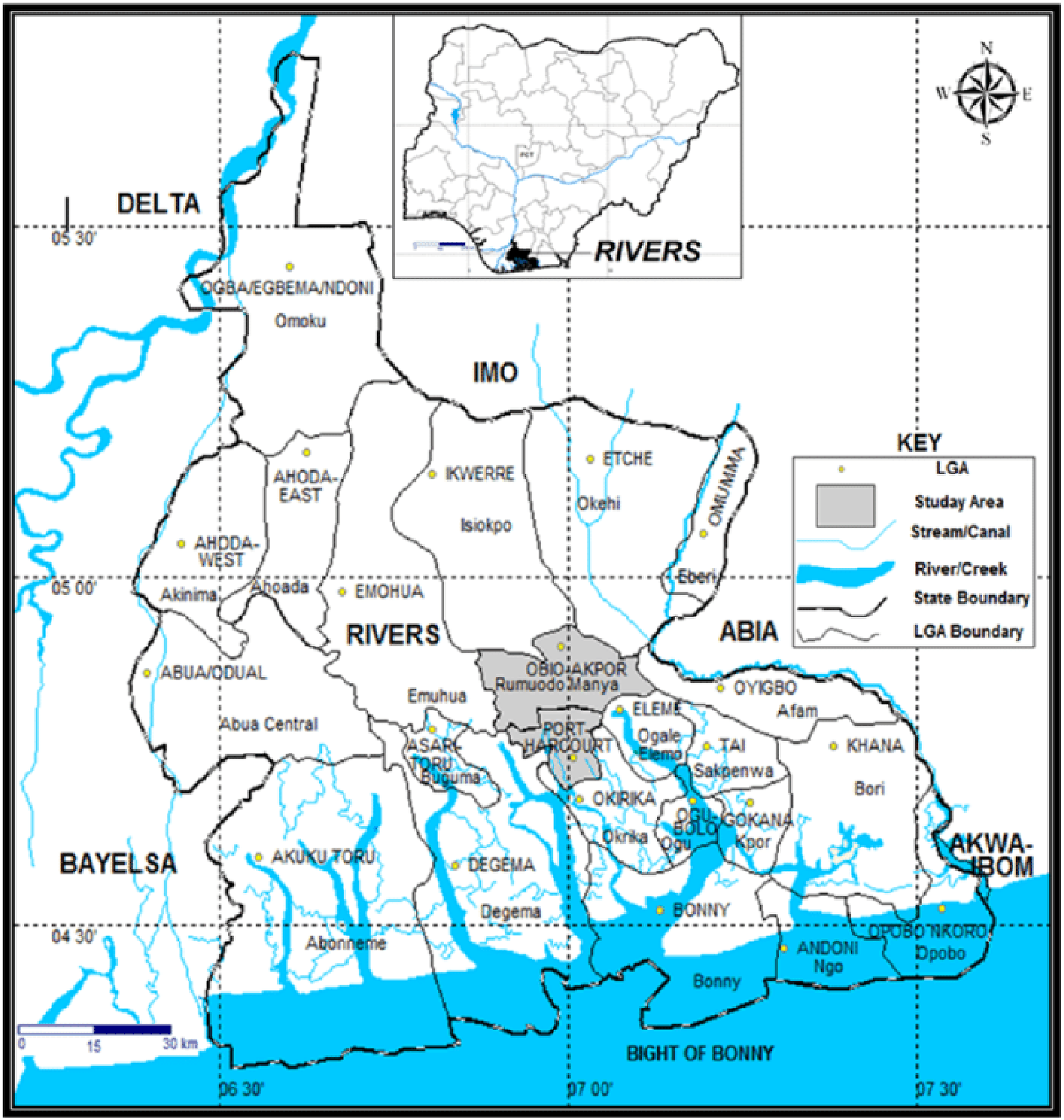
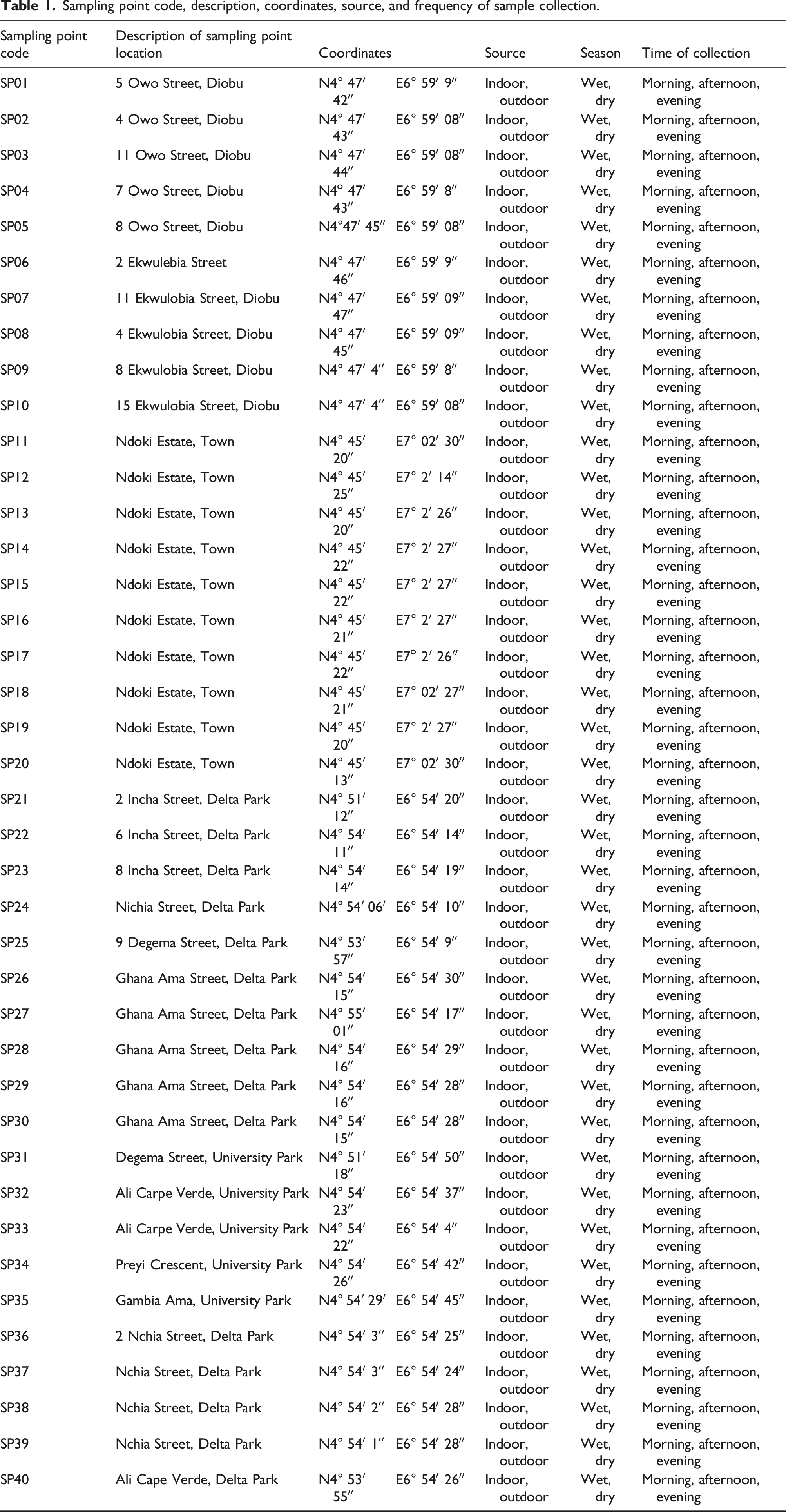
Sampling point code, description, coordinates, source, and frequency of sample collection.
Experimental design
The conceptual model below shows a hypothetical distribution of atmospheric gaseous pollutants inside and outside residential areas (n = 40) (Figure 2). The independent variables (CH4, CO, NH4, NO2, O3, and SO2) are influenced by the time of sample collection, seasons, and collection point, that is, indoor and outdoor (dependent variables). For instance, seasons affect gaseous concentration and determine whether it will be high or low. Experimental design of the study of noxious gases done in indoor and outdoor residential areas in Port Harcourt, Nigeria.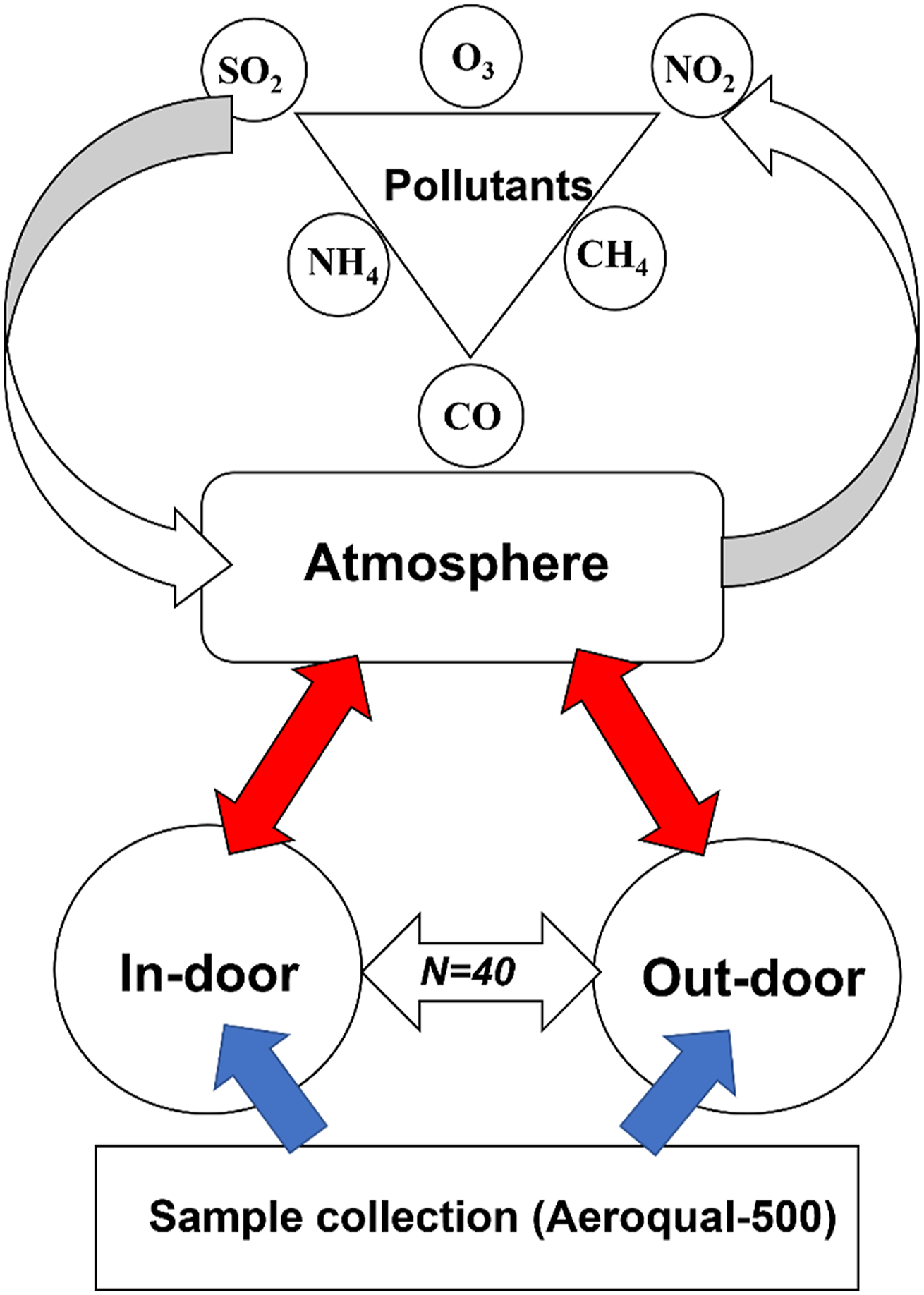
All 40 locations were sampled in indoor and outdoor areas at three time periods (morning, afternoon, and evening) during two seasons (dry and wet), giving a total of 480 observations (i.e., n = 2 × 3 × 2 × 40) using the GPS Model 76×. The air quality parameters measured were Sulphur dioxide (SO2), Carbon Monoxide (CO), Nitrogen Dioxide (NO2), Methane (CH4), Ozone (O3), and Ammonia (NH4).
Stratified random sampling was used to delineate the study area into low and high-density areas.
Sample collection
Multiple air samples were collected between April 2021 (wet season) to October 2021 (dry season) after the calibration of the sensor on 30 April 2021. The measuring instruments were placed in a strategic location of each building; the readings were carried out in situ to measure the number of air parameters found in the building. Samples were collected three times on each sampling day, that is, morning, afternoon, and evening. We visited each location three times each day. The morning, afternoon, and evening samples were collected from 7 to 10 a.m., 12 a.m. to 2 p.m., and 4 to 7 p.m. Diobu, Town, and University of Port Harcourt Choba’s sampling points cover over 15 km.
Sampling instruments
The instruments used for this study included a GPS (Model 76Cx), which was used to determine the coordinates of the sampling points. An Aeroqual series 500 gas monitor was used to assess all the gaseous pollutants in the study area. The gas monitor is a portable meter with highly sensitive replaceable sensors for different gaseous air pollutants.
Sensor calibration
Detection limit and resolution of gas monitor (Aeroqual series 500) sensor.

†1 ppm = 1 mg/kg.
(website: http://wwwgassensing.com/downloads/aeroqual/Aeroqual_Handheld_Manual.pdf).
Geospatial analysis
The indoor air quality contours were made using ArcGIS 10.2 software. This software is a Geographic Information System program that integrates spatial data and attributes (indoor air quality values), and stores and analyzes the input variable for graphic presentation.
Modeling of indoor air quality
This indoor modeling uses a mass balance approach to estimate indoor air pollutant concentrations in the study area.
24
In this modeling approach, a house was considered a simple box, as shown in Figure 3. The model does not indicate an equilibrium but simply shows the pattern of gaseous movement in and out of an apartment. It shows that the rate of pollutant increase in the box depends on the difference between the cumulative effect of pollutants entering and leaving the box. Meteorological parameters collected in the study include air pressure, relative humidity, wind speed, wind direction, and temperature. Mass balance for indoor air quality modeling.
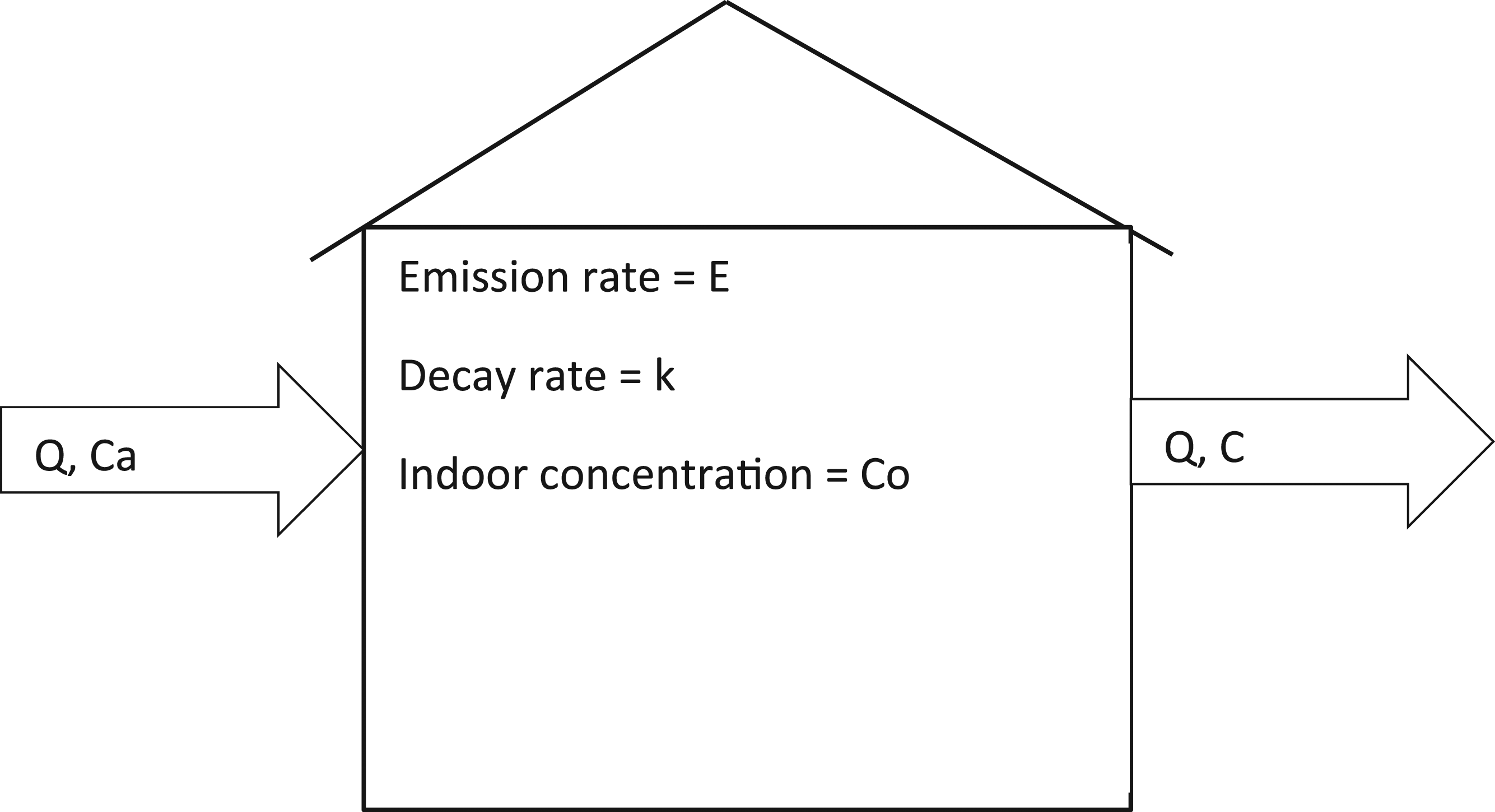
The governing mass balance model for indoor air pollution, as contained in Davis and Cornwell,
25
is expressed as given in equation (2) C = concentrations (μg/m3) Ca = ambient concentrations (μg/m3) Q = rate of infiltration of air into and out of the box (m3/s) V = volume of the box (m3) E = emission rate of pollutant into the box from indoor source (g/s) k = pollutant decay constant or rate of reaction coefficient (/s)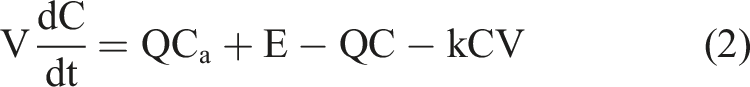
Statistical analysis
An analysis of variance (ANOVA) was conducted to determine whether there was a significant difference in gas concentration inside and outside residential buildings at different collection times and seasons. A four-way ANOVA was conducted to determine whether there is any interaction effect. The data was initially log-transformed to ensure they were normal and had equal variances. 26 Later, bar graphs were then used to illustrate the results. 27 A posthoc Tukey’s HSD test was done to investigate pair-wise mean differences between groups. All analysis was performed in R statistical environment. 28
Results
Interaction effect
Four way-ANOVA of the interaction between sampling parameters and noxious gas concentration in residential areas around Port Harcourt, Nigeria.
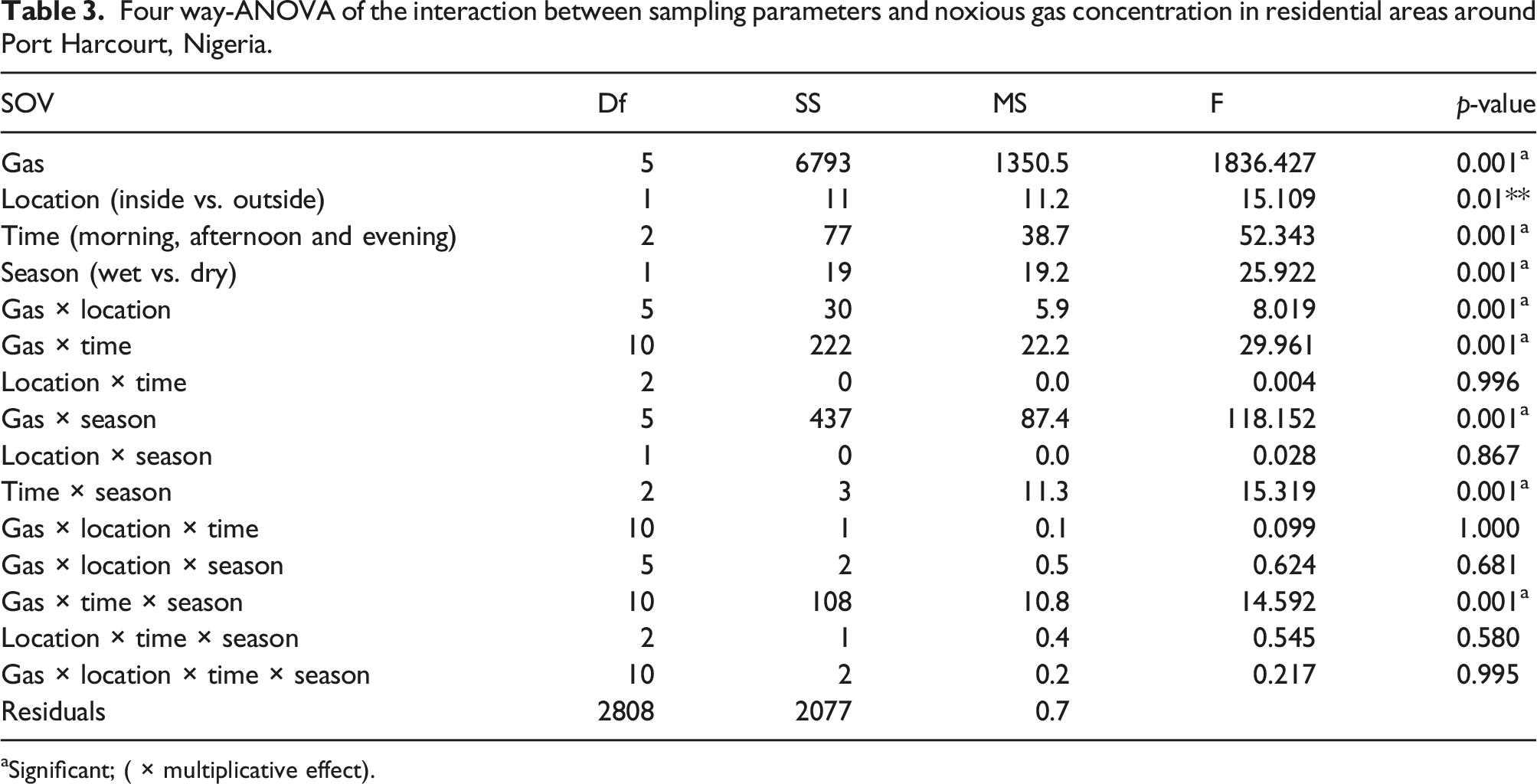
aSignificant; ( × multiplicative effect).
In general, the two- three- and four-way interactions show that the concentration of gases in the atmosphere was mainly influenced by time and season (p < 0.001) and not location (p > 0.05) (Table 1).
Assessment of the concentrations of different gases and between inside and outside areas (i.e., location) of residential buildings
Tukey, multiple comparisons of means of some gases, analyzed in some households around Port Harcourt, Niger Delta, Nigeria.
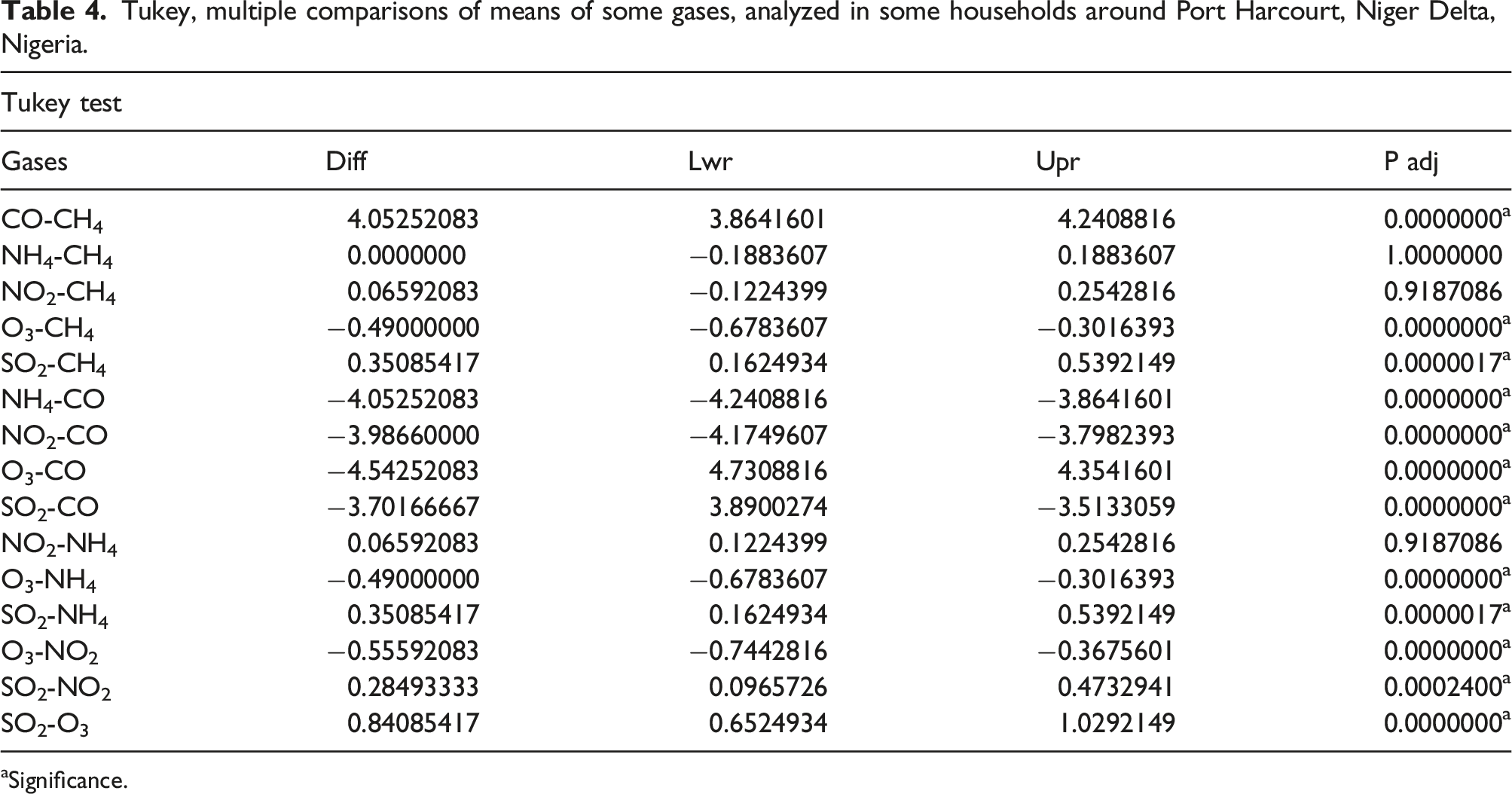
aSignificance.
Mean levels of noxious gases ±1 SE in inside and outside environments in some communities in Port Harcourt, Niger Delta Nigeria.
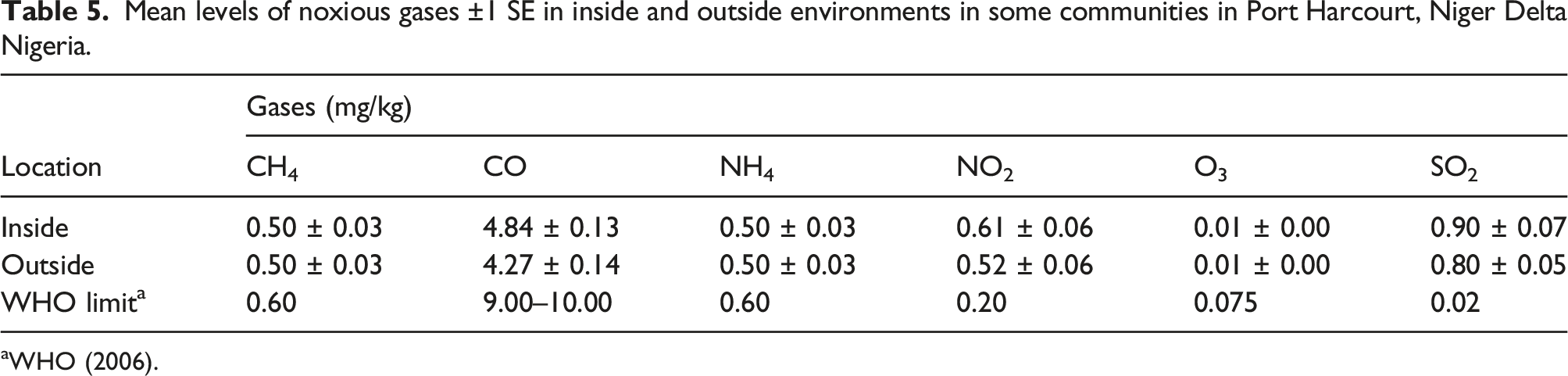
aWHO (2006).
The concentration of noxious gases inside versus outside of residential areas is further illustrated in a bar graph (Figure 4). The concentration of some noxious gases between indoor and outdoor environments around Port Harcourt, Rivers State, Nigeria. The legend shows increasing concentration. At the WHO limit of <0.02 mg/kg, NO2, O3, and SO2 are above the limit, while at 0.60 mg/kg CH4 and NH4 are within the limit. Similarly, at >5.0 mg/kg CO is within the limit.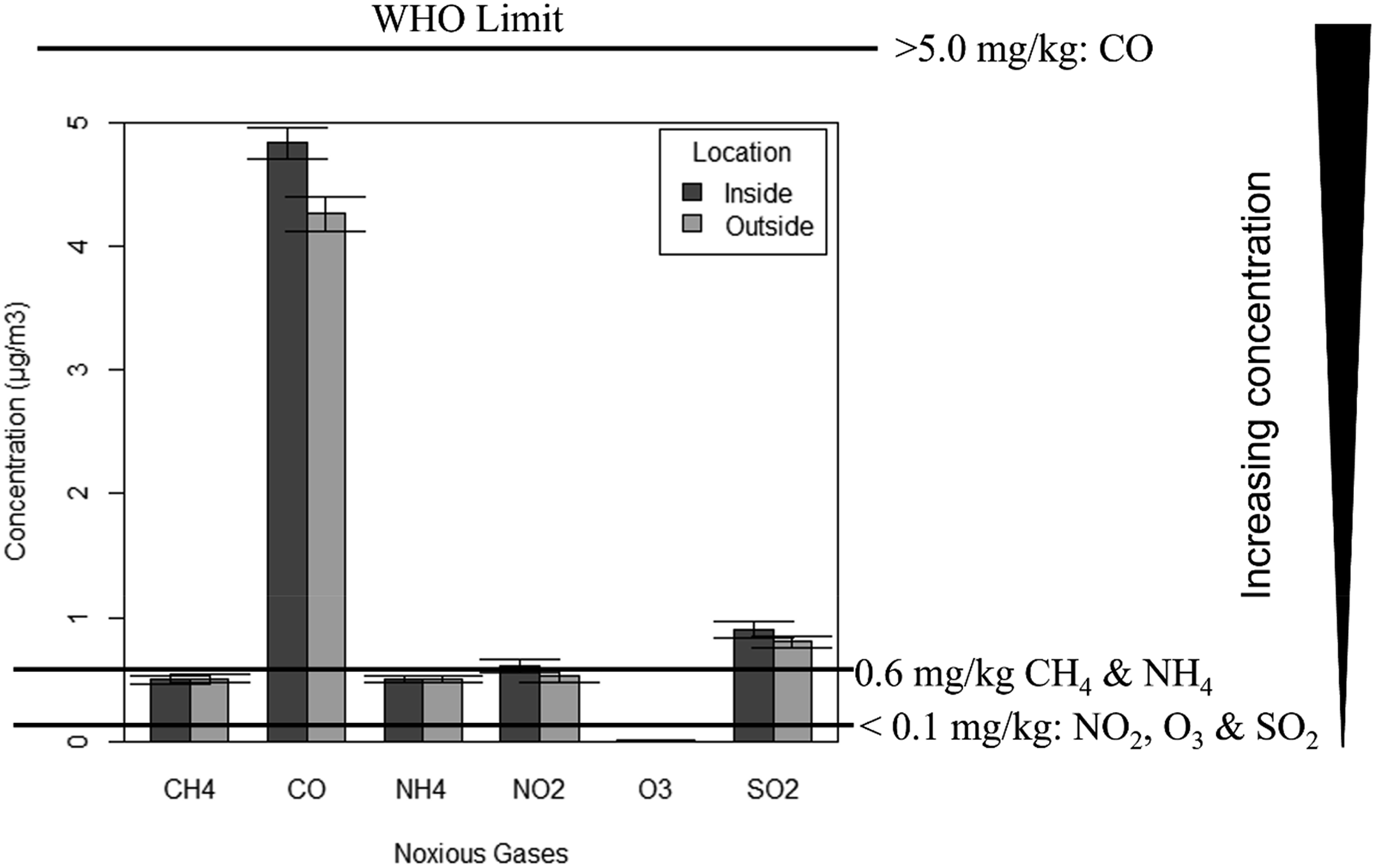
Determination of the concentration between different collection times
Mean levels of noxious gases ±1 SE at different collection times in some communities in Port Harcourt, Niger Delta Nigeria.
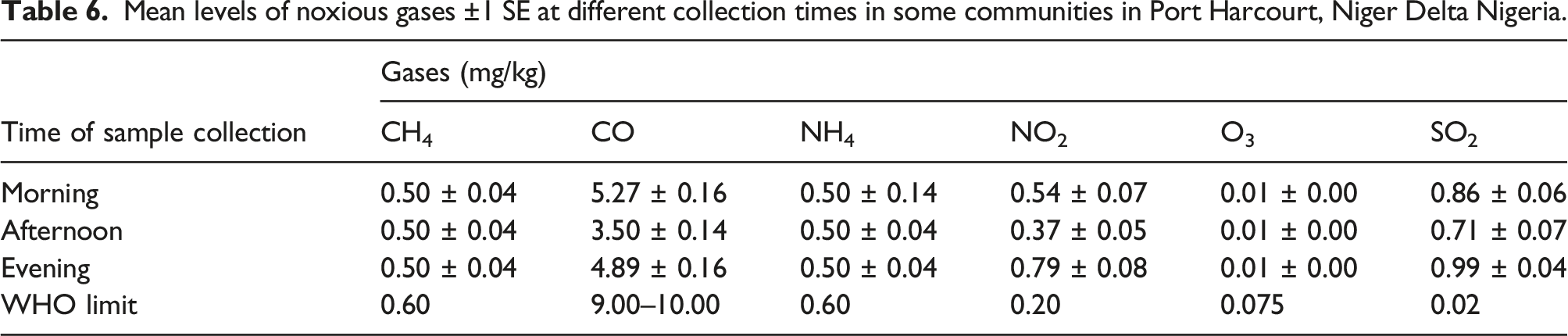
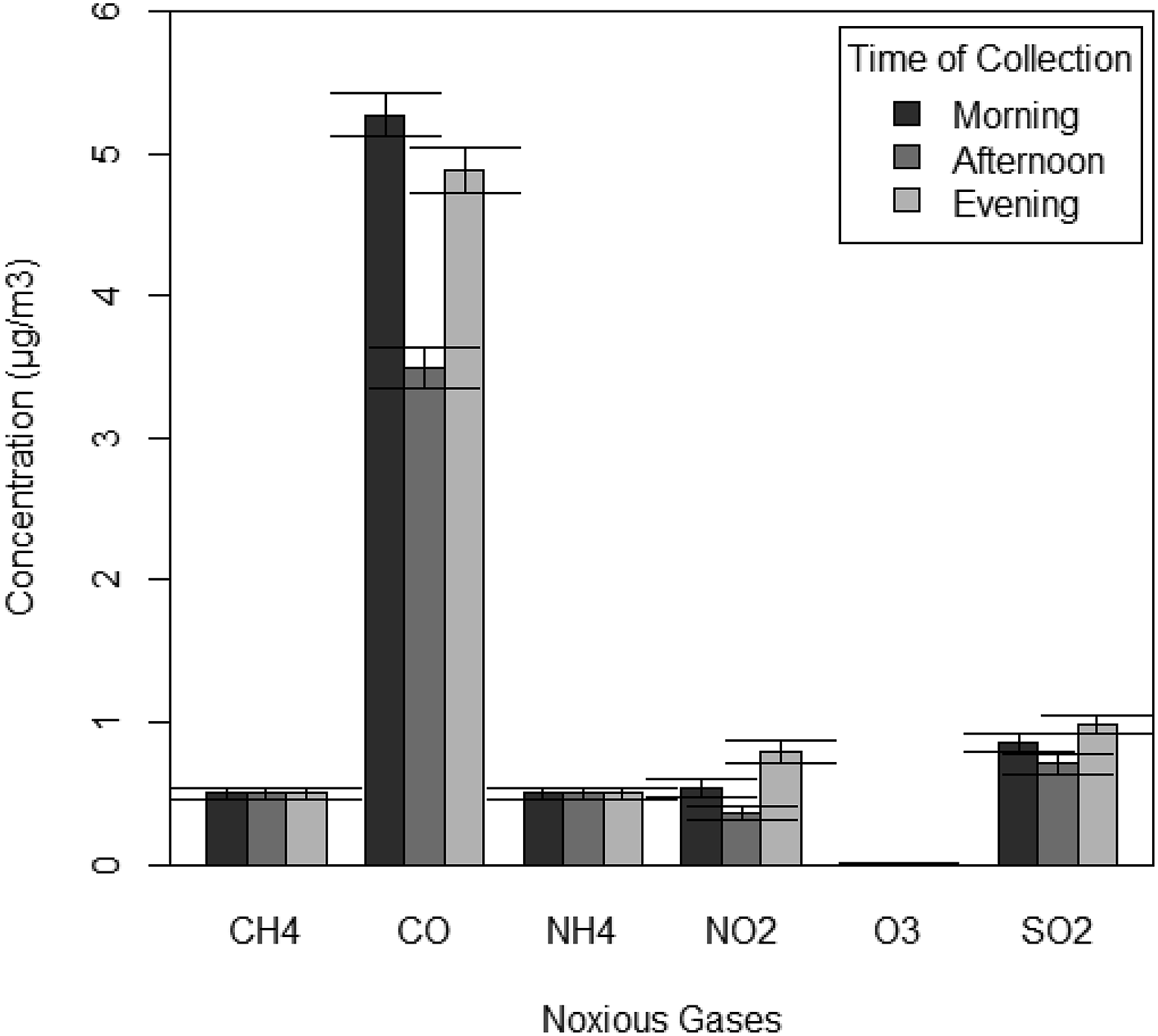
Concentration of some noxious gases at different collection times (i.e. collection frequency) around Port Harcourt, Rivers State, Nigeria. (Apply the legend of Figure 3 here).
The concentration of noxious gases across the time of sample collection is further illustrated in a bar graph (Figure 5).
Evaluation of the seasonal difference in the air quality in different residential areas
The ANOVA results indicate a significant difference between the dry and wet seasons (F1, 2808 = 5.64. p = 0.02, Figure 6). The CH4, NH4, NO2, and SO2 were higher during the dry seasons, while CO was higher during the wet season. However, the concentration of O3 was not significantly different (Table 7, Figure 6) in both seasons. The concentration of some noxious gases during wet and dry seasons around Port Harcourt, Rivers State, Nigeria. (Apply the legend of Figure 3 here). Mean levels of noxious gases ±1 SE at different seasons in some communities in Port Harcourt, Niger Delta Nigeria.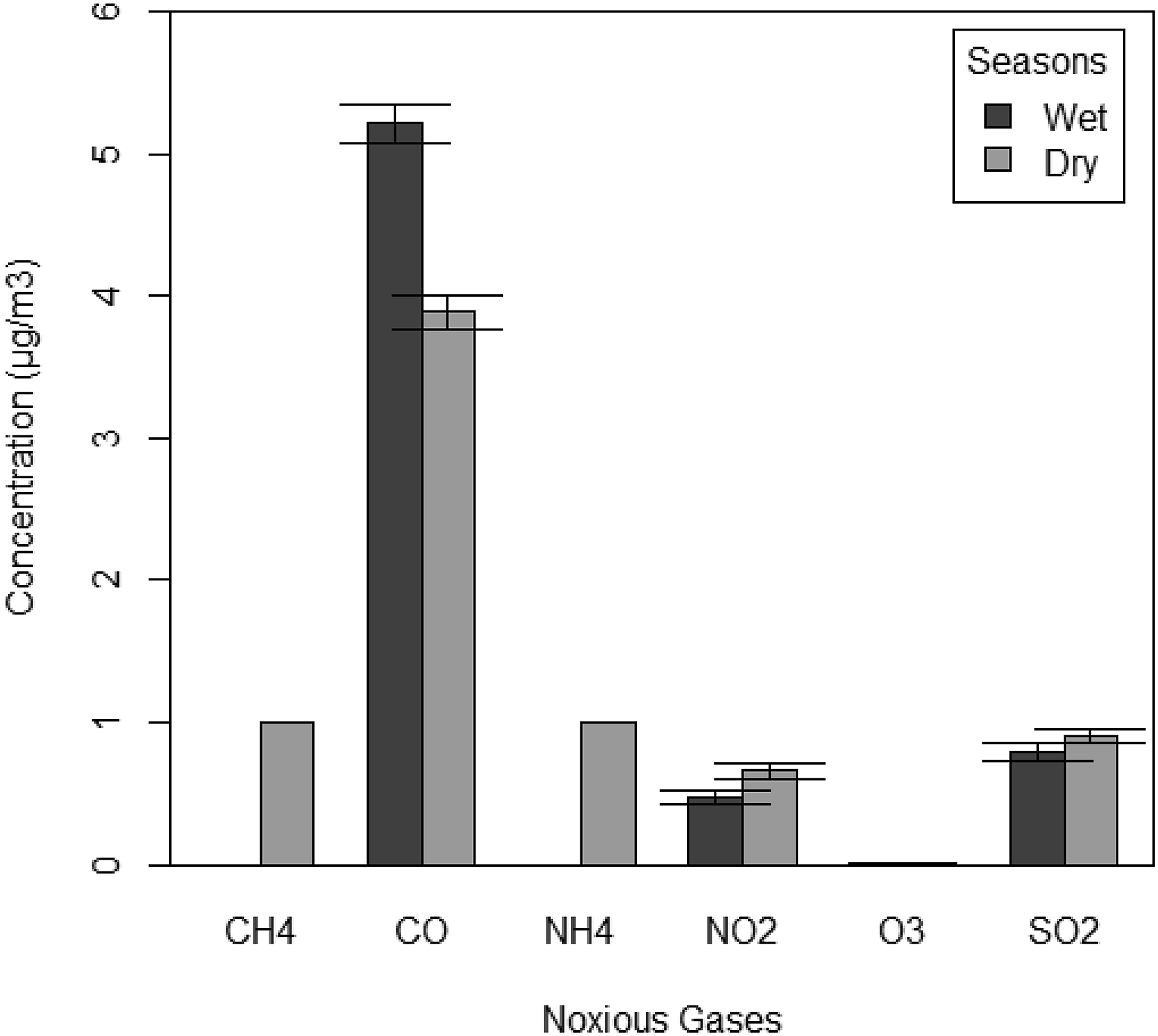

The concentration of noxious gases in wet versus dry seasons is further illustrated in a bar graph (Figure 6).
Spatial interpolation of CO concentrations in the study area
Since CO concentration is significantly higher than other noxious gases, a spatial interpolation map for high and low-population-density areas was drawn (Figure 7). The maps revealed a low CO concentration in the low population density area during the dry season. While during the wet season, there was high CO concentration in the low population density area. Spatial interpolation of indoor SO2 for dry and wet seasons (a and b); NO2 for wet and dry Season (c and d), and CO for dry and wet season (e and f) in some localities around Port Harcourt, Rivers State, Nigeria.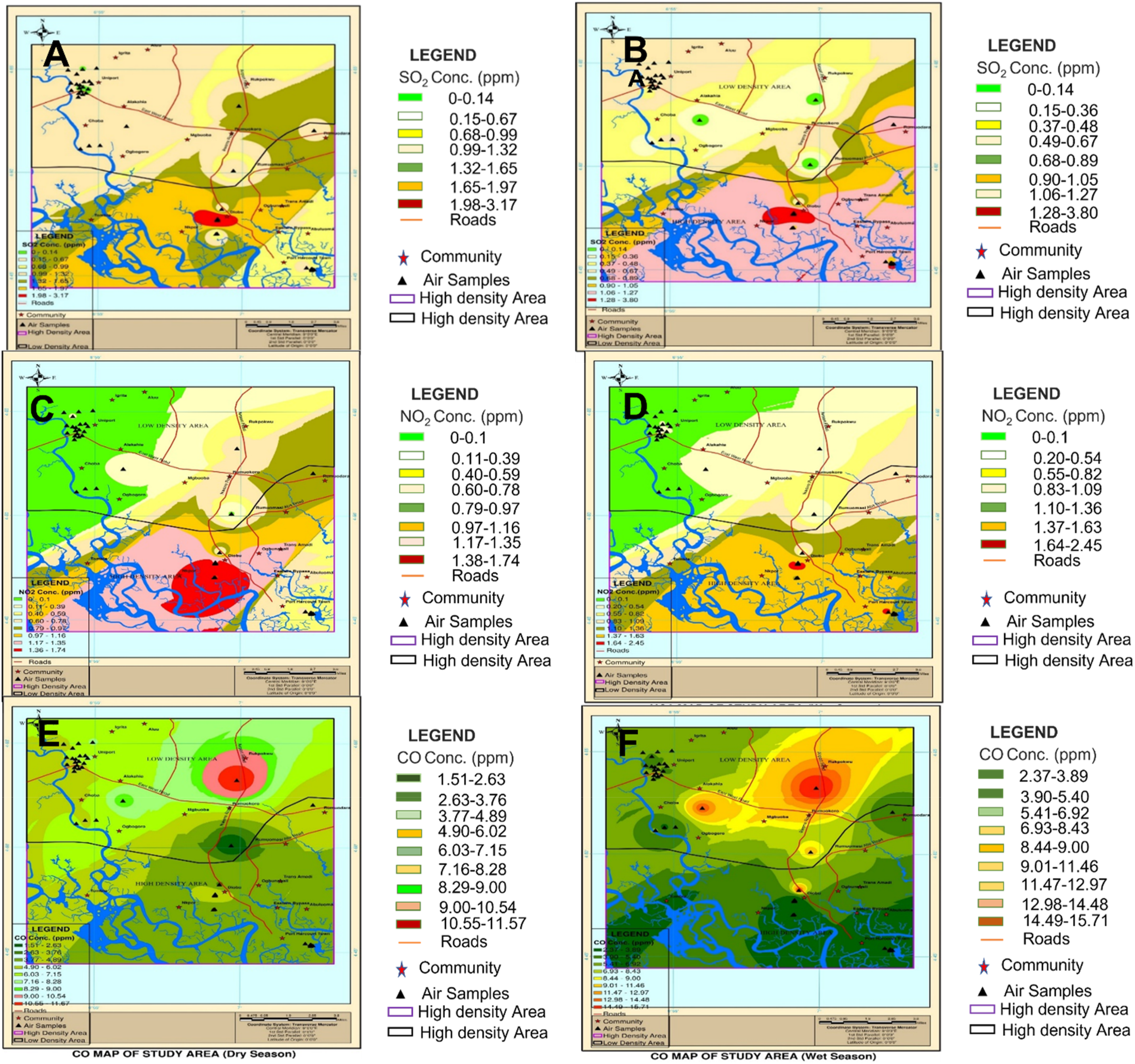
Discussion
The objective of our study was to determine the concentration of noxious gases inside and outside residential buildings. And to determine the influence time, season and location have on poisonous gas concentration individually and interactively. Our study revealed that noxious gases in indoor and outdoor areas are mostly below the WHO limit except SO2 and NO2, which were above the WHO limit (Table 5). Carbon dioxide had the highest concentration in the indoor environment (Table 5) but was well below the WHO limit. Indoor pollutants are more harmful to health than outdoor pollutants, 9 though, in terms of concentration, the previous study has shown that outdoor pollution is generally greater than indoor pollution. 29 Our study showed that the concentration of CO inside buildings was significantly higher than outside buildings. This reflects the use of generators, cooking stoves, and cigarette smoking, which produce CO. Carbon monoxide (CO) reduces the blood’s oxygen-carrying capacity, causing nausea, 30 unconsciousness, and death. 31 This occurs because CO binds more readily to hemoglobin than oxygen and results in carboxyhemoglobin (COHb) formation, which leaves less hemoglobin available for transferring oxygen around the body. EPA (Environmental Protection Agency of US) set an 8-hour primary standard for CO at nine parts per million (ppm). 32 However, in the absence of emission sources, concentrations of CO in indoor environments are generally lower than those outdoors.
On the other hand, NOx is a combination of NO2 and NO pollutant concentrations. 33 The combustion of fossil fuels mainly produces nitric oxide. The primary sources of NOx are vehicle exhausts like diesel, petrol, liquefied petroleum gas (LPG), and compressed natural gas (CNG). 34 The concentration of NO2 is also slightly higher inside residential buildings because of the accumulation of indoor and outdoor gases inside buildings. Nitrogen dioxide gas is mostly derived from generating sets used to provide electricity in buildings. 35 This is because of the irregular electricity supply by the electric power authority in many areas around the city. Almost every house has a power generating set that provides electric power. This generating set releases smoke into living homes, thus contaminating the indoor environment with other noxious gases, for example, SO2. In addition, vehicle emissions also migrate into houses to increase the pollutants load, especially in dwellings near major roads in the city. Here, NO2 is produced as a combustion product from fossil fuel cars. This gas is also produced from industrial emissions, as observed in areas near the Port Harcourt refinery that receive pollutants from gas flaring activity. Increased indoor and outdoor pollutants increase the risk of different types of cancers.35–37 Harmful atmospheric gases have increased in the city because of the proliferation of artisanal refineries in the creeks around the city. 38
The higher concentrations of NO2 and SO2 in the evening are because of the “rush-hour” period, where there is an increase in vehicular traffic on the road. Concentrations of NO2 and SO2 are above the WHO limits of 0.2 mg/kg, and 0.02 mg/kg, respectively (Tables 6 and 7), and are mostly the components of vehicle exhaust. Increased traffic causes slow movement of vehicles and results in more gases from car exhaust. Another factor is the increased use of generating sets to supply electricity during the evenings. Sulphur dioxide (SO2) is readily soluble in water, can be oxidized within airborne water droplets, and is formed because of the combustion of fuels like petrol, diesel, and coal containing sulphur. 5 Thermal power plants and diesel vehicles are also major sources of SO2. 39 Our study shows that SO2 has a higher concentration inside (0.90 ± 0.07 mg/kg) than outside (0.80 ± 0.05 mg/kg) buildings (Table 6). In contrast, studies sulphur dioxide concentration is generally higher outdoors than indoors, especially during the evenings. 40 Absorption of SO2 affects the respiratory system 41 A higher concentration of CO in the morning might be because of higher cooking activities by various families in the morning.
Higher concentrations of most of the gases (i.e. CH4, CO, NH4, NO2, and SO2) during the dry season is because of the low humidity and dryness of the atmosphere, which facilitates the atmospheric distribution of the gases. Harmattan wind causes an increase in wind action, which enables the movement and distribution of pollutants in the air. During the wet season, most atmospheric gases are washed down as “acid rain” onto the land surface. This is shown in Figure 5, where the wet season concentrations of CH4 and NH4 are low compared to the dry season. A similar result is replicated by high concentrations of SO2 and NO2 during the dry season than in the wet season. The concentration of CO is higher during the wet season than during the dry season because of increased cooking with firewood and stove during the festive period (October–February). Sociocultural activities such as new yam festivals, child naming ceremonies, and traditional weddings lead to the use of firewood for cooking, which inevitably increases the CO level around urban and rural environments. Also, Studies show that CO is an acute indoor toxic air contaminant caused by increased cooking with a kerosene stove, smoking, and use of generating set to supply the electricity in indoor and outdoor environments. 42 These gases have the potential to cause public health problems 43 and pollution-related mortality.44–46
Our results indicate that the season affected CO concentration in the indoor environment (Figure 6). There was higher CO concentration indoors during the wet season because the CO concentration outside is washed by rainfall to form “acid rain.”
Our study also revealed that time and season (independent variables) have a more significant effect on noxious gas concentration than location (inside or outside residential buildings). This is supported by the fact that the two and three-way ANOVAs having location as a factor has a non-significant p-value (Table 5). Collection time determines if there is high traffic on the road and, thus, high exhaust emission. Similarly, the time of the year determines the increase or decrease in environmental parameters such as humidity, temperature, etc., which will impede or accelerate the proliferation of gaseous pollutants at the time of sample collection. Future studies will consider environmental and epidemiological data, which will be correlated with zones of high noxious gas concentrations around the study areas. This is because biomass burning during cooking increases atmospheric particulate matter, which results in increased deaths and diseases. 47 It is also reported that environmental factors cause 24% of the global disease burden and 23% of all deaths. 48
Conclusions
The time of sample collection, season, and location are essential factors in toxic gas concentration in the atmosphere. Samples collected in the morning and evening show high concentrations of NO2, SO2, and CO, which are poisonous gases that affect health if found indoors or outdoors. This is because, in the morning, there is a spike in the concentration of CO due to an increase in traffic, while in the evening, there is an increase in NO2 and SO2 because of the rush home traffic and roadside cooking activities. Since time and season are influenced, gaseous concentration city dwellers should be careful about how they expose themselves to pollutants by avoiding highly polluted environments. They can also wear a nose mask to prevent inhaling toxic gases.
This study indicates that it is essential to investigate and monitor the concentrations of poisonous gases and other particulate matter and their interactive effect on residential areas. The result shows that most of the gases are not at a hazardous concentration; however, if they are not monitored, they may rise to a level that is harmful to human health.
Footnotes
Acknowledgment
We thank the Director of the Institute of Natural Resources, Environment, and Sustainable Development (INRES), A.I. Hart, for providing a supporting letter for our lead researcher. Arc. M.A. Ibom for facilitating the approval of the letter from the University and E. Oti of the Ministry of Urban Development for facilitating access to residential settlements.
Author contributions
FMO: carried out the sampling, and wrote the manuscript, AON: did statistical analysis and wrote manuscript, AO: supervised the project, DSO; conceptualized the project and edited the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/ or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The Institute of natural resources and sustainable development was informed of the use of questionnaires.
Availability of data and materials
Data would be available on request through the corresponding author
