Abstract
Some vegetable oils show beneficial effects in modulating neurodegeneration; in this work, we evaluated the therapeutic potential of corn and olive oils against neurodegenerative processes using the acute parkinsonism murine model induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in C57BL6 mice. The effects of corn and olive oils were quantified by the performance of mice in the open field and rotarod, and grasp strength tests and neuronal survival in the substantia nigra and striatum were determined by immunohistochemistry. Extra-virgin olive oil decreased the toxicity induced by MPTP administration judged by the performance in the behavioral motor tests and the number of total neurons in the analyzed brain regions. In contrast, corn oil only produced discrete changes in the behavioral and histological evaluations. Despite the numerous benefits of olive oil, its active substances that confer desirable effects and their mechanism of action remain unclear. Our observations can help to understand the ameliorative effects of some natural oils on neurodegeneration induced by some toxins, particularly the attenuation of neural damage related to toxin-induced parkinsonism or other pathologies that comprise neuronal death and motor disruption.
Introduction
The etiology of most neurodegenerative diseases has not been completely elucidated; however, neurotoxicity is a common factor observed in Parkinson’s disease (PD) and Alzheimer’s disease. Despite the theories to explain the pathogenesis of these diseases, researchers have focused on the balance between genetic predisposition, environmental neurotoxins, and diet. 1
PD is a neurodegenerative disorder, clinically characterized by bradykinesia, rigidity, resting tremor, and some nonmotor symptoms, which affects approximately 1–2% of the population above the age of 65 years. Pathological hallmarks include the loss of dopaminergic neurons within the substantia nigra pars compacta, as well as the intraneuronal accumulation of proteins forming Lewy bodies. 2
Although the pathogenesis of PD remains elusive due to the global increase of the aging population, there is an urgent need for discovering novel preventive and therapeutic approaches against PD. Despite the efforts of researchers and promising strategies worldwide, no cure for PD currently exists. Moreover, although the promising results of preclinical studies investigating various neuroprotective agents, all current clinical trials testing potential disease therapies against PD have failed. Thus, the therapy for PD based on levodopa as a dopamine precursor remains as the gold standard. 3
Previous studies have associated some specific toxins with neurodegeneration. 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), paraquat (N, N-dimethyl-4-4-bipiridinium), and rotenone are prototypic toxins known to exert deleterious effects and motor disruption through oxidative stress related to mitochondrial biotransformation and interactions. Due to their lipophilicity, they can be significantly distributed into the brain, particularly MPTP, which can induce biased damage in the dopaminergic systems of some mammals; additionally, other toxins induce motor disruption, including some heavy metals. In contrast, other molecules obtained from the diet have been associated with protective effects against neurodegenerative processes. 4,5 For instance, many studies have associated the consumption of olive oil with a better overall health. 6 –8 Conversely, only a few studies have focused on the beneficial effects of other vegetable oils on degenerative disorders, such as corn or perilla oil. 9
Previous assays have investigated the beneficial effects of olive oil in murine models of neurodegenerative diseases related to cognitive deficit. 10 –12 Therefore, the information about the beneficial effects of olive oil on neurotoxicity models related to motor disorders is scarce. 13,14
To the best of our knowledge, this is the first report that focuses on the effects of olive and corn oils on motor disorders using a murine model of parkinsonism induced by MPTP. In fact, MPTP is a small molecule which acts as a neurotoxin, predominantly in the substantia nigra, which affects dopaminergic pathways. 15 At the cellular level, MPTP, as well as its active metabolite 1-methyl-4-phenylpyridinium (MPP+), interferes with the mitochondrial respiratory chain and causes the redistribution of vesicular dopamine to the cytoplasm in neurons, which leads to the generation of MPTP-induced toxic reactive oxygen species. 15 –17 Due to its toxic effects, MPTP has been widely used to induce parkinsonism and neuronal damage in some mice strains and primates. 15,17 This study aimed to evaluate the potential of corn oil and olive oil to reduce both neuronal damage and behavioral changes related to motor disruption in models of parkinsonism induced by MPTP.
Material and methods
Drugs and chemicals
MPTP (M0896), corn oil (C8267), extra-virgin olive oil (O1514), and all the reagents and chemicals for the solutions used in this study were purchased from Sigma Aldrich Co. (St Louis, Missouri, USA).
Animals
All experiments were performed according to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines. Male C57BL6 mice (6-week old and weighing 20–23 g) were obtained from the vivarium of UAM-Xochimilco, Mexico City. Free of known viral, bacterial, or parasitic pathogens, animals were kept under standard conditions with access to food and water ad libitum and housed in polystyrene cages under controlled environmental humidity, temperature (18–25°C) and lighting (12-h light/dark cycle, lights on at 7 am). All the experiments were carried out between 09:00 hand 15:00 h. The current protocol was approved by the Ethics Committee and Institutional Animal Care and Use Committee of the Escuela Superior de Medicina, Instituto Politécnico Nacional, in accordance with the guidelines established by the Mexican Secretary of Agriculture and Animal Breeding (NOM-062-ZOO-1999, SAGARPA), and with the guide for the Care and Use of Laboratory Animals of the National Research Council.
Treatments
Animals (N = 40) were randomly divided into five groups. Group 1 (control group; n = 8), were non-intoxicated C57BL6 mice administered intraperitoneally (i.p.) with saline solution (SS, NaCl 0.9% 10 ml kg−1 × 4 doses within 1.5 h intervals); groups 2 to 5 (n = 8) were exposed to MPTP (18 mg kg−1 × 4 doses within 1.5 h intervals administered i.p. to induce parkinsonism). 18 –20 MPTP-treated groups (2 to 5) were injected i.p. (a volume of 10 ml kg−1) 1 h after the last administration of MPTP and every 24 h for 4 days as follows: group 2 received normal saline solution (MPTP + SS); group 3 received corn oil (MPTP + Corn); group 4 received extra-virgin olive oil (MPTP + Olive); and group 5 received levodopa dissolved in normal saline (MPTP + LD). Animals were euthanized 7 days after the last treatment and brain tissues were dissected and processed for analysis.
Motor tests
Before the treatments, basal motor tests were performed to discard any motor disturbance. Only animals with normal behavior (motor performance between percentile 25 to 75 according to previous reports) 18,20 were included in the subsequent evaluations.
All the tests were performed five times: after the initial MPTP-intoxication, 2 h (day 1), 24 h (day 2), 48 h (day 3), and 72 h (day 4) after the treatments with either normal saline, corn oil, olive oil, or levodopa.
Spontaneous locomotor activity
After the treatments, mice were placed in motor-activity measuring cages (50 × 50 × 50 cm3) with detectors each 2.5 cm (OA-BioMed OMNIALVA®, Mexico) to determine the number of total movements, distance traveled, highest speed, and vertical movements. Open field tests were performed for 5 min. 20
Rotarod test
Muscle strength, coordination, and balance were assessed through the rotarod test, as determined elsewhere. 21 An Ugo Basile® (Italy) apparatus, which consisted of a horizontal cylinder (diameter 6 cm) elevated 15 cm above the switch floor, was used for the test. Each animal was placed on the cylindrical rod rotating at a constant speed of 25 r min−1, and the fall-off time was recorded. This test was performed for 2 min.
Grasp test
Muscle strength and balance were assessed by this test, as determined elsewhere. 22 A forepaw grip strength test was performed using a device described previously. 23 Subsequently, mice were placed on a horizontal aluminum wire fixed to two metallic poles elevated 15 cm above the floor to hold on using only their forelimbs, for which their tail was restrained to prevent the use of their hind limbs. The time mice were able to hang in was monitored for each group. This test was performed for 90 s.
Immunohistochemistry assays
After the motor behavioral evaluation, mice were decapitated and transcardially perfused with 200 ml of ice-cold (4°C) sodium phosphate buffer (PBS) 0.1 M, pH 7.4, and a freshly prepared fixative (4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4). The brain was removed immediately after the perfusion and postfixed in ice-cold (4°C) 4% paraformaldehyde, followed by 48 h in ice-cold (4°C) 30% sucrose in 0.1 M phosphate buffer for cryoprotection. Coronal slices of the brain (30 µm thick) were cut on a freezing microtome and stored at −20°C in a solution containing 30% ethylene glycol, 30% glycerol, and PBS until immunohistochemical analysis.
Free-floating sections were incubated in PBS containing 3% H2O2 for 10 min and rinsed three times with PBS for 5 min. Subsequently, sections were incubated in 0.1% Tween 20 in PBS (T-PBS) for 10 min, then incubated in 0.5% bovine serum albumin (BSA; GE Healthcare, UK) in T-PBS for 2 h. Finally, sections of the substantia nigra (pars compacta) and the striatum were incubated with the primary antibody anti-NeuN (1:1000, rabbit monoclonal to NeuN from Abcam, Cambridge, UK (Cat. PR12763)) at 4°C for 48 h. The following day, sections were rinsed three times in T-PBS for 5 min and incubated with a biotinylated secondary antibody and a peroxidase-labeled streptavidin reagent (LSAB System-HRP Kit; DAKO, Carpintería, California, USA) for 30 min each. Antibody labeling was visualized by exposure to 3–3′-diaminobenzidine (DAKO). Omission of primary or secondary antibodies as negative controls resulted in the absence of immunolabeling. Finally, sections were rinsed twice in PBS and mounted in glass slides with Entellan solution (Millipore, Danvers, Massachusetts, USA). The number of NeuN-positive neurons was calculated through random sampling bilaterally in two sections per animal under a light microscope (magnification 40×) by two independent trained observers who were blind to the treatment to avoid biased appraisal. Cell counts from the right and left hemispheres of each two sections were averaged to provide a single value for each animal. From these data, the mean of these labeled neurons in substantia nigra and striatal areas (n = 4) was calculated for each experimental group, as this method is used to estimated neuron survival related to toxicity or protective effects in neurodegenerative diseases. 20,24
Statistical analysis
Data were expressed as the mean ± standard error mean. For comparison between groups, a one-way analysis of variance followed by Tukey’s post hoc test were used as data were parametric. All statistical analyses were performed using Sigma Plot 12.0 and attached tools (Systat Software, San Jose, California, USA). Statistical significance was set at a p-value <0.05.
Results
Behavioral changes by the administration of MPTP and treatments
MPTP induced motor disruption in C57BL6 mice; these findings were similar to previous reports. 18 MPTP administration diminished the performance in the open-field, rotarod, and grasp tests. No observable changes were induced in the first (immediately after the last MPTP administration) or the second evaluation (2 h after the last MPTP administration). However, the performance was different after the first day of evaluation (Figures 1 to 3). A notable improvement was observed after the administration of levodopa or olive oil, but not for the groups treated with saline solution or corn oil.
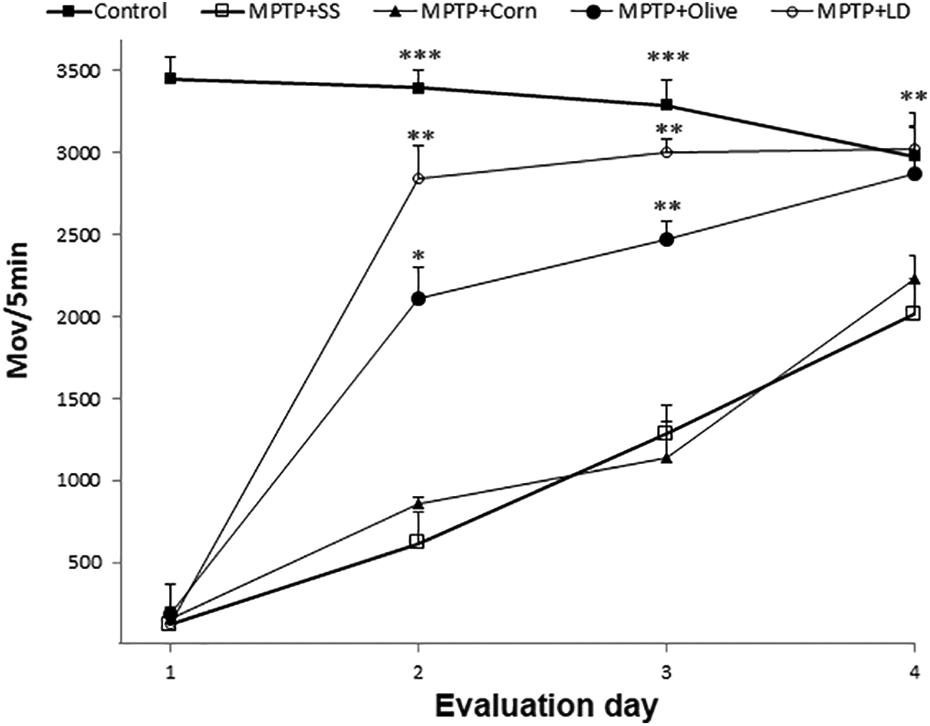
Open-field test evaluation posttreatment. Results are expressed as mean ± SE (n = 8). *p ≤ 0.05; **p < 0.01; ***p < 0.001 compared to the MPTP + SS group. SE: standard error; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; SS: saline solution; LD: levodopa.
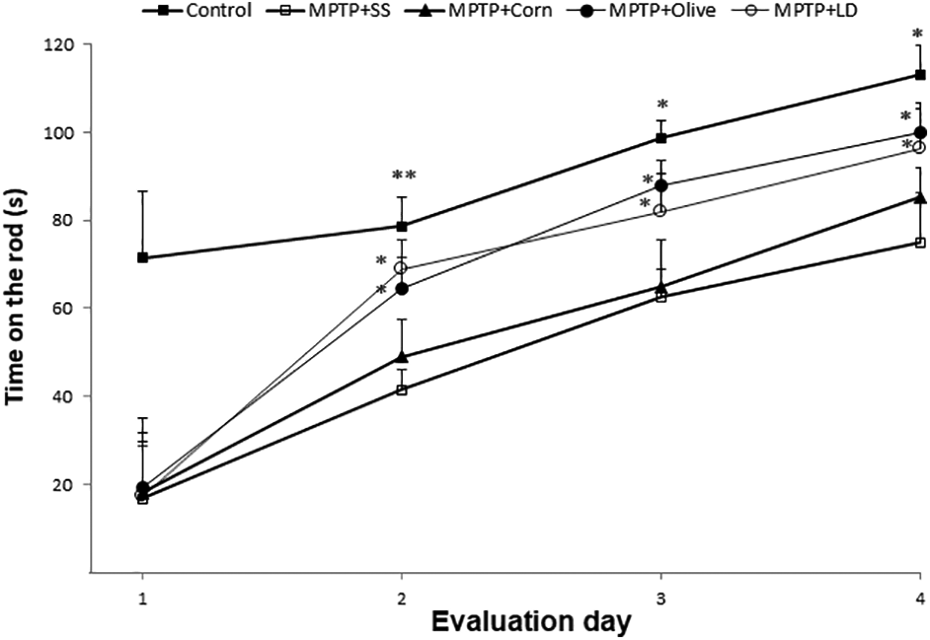
Rotarod test evaluation posttreatment. Results are expressed as mean ± SE (n = 8). *p ≤ 0.05; **p < 0.01 compared to the MPTP + SS group. SE: standard error; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; SS: saline solution; LD: levodopa.
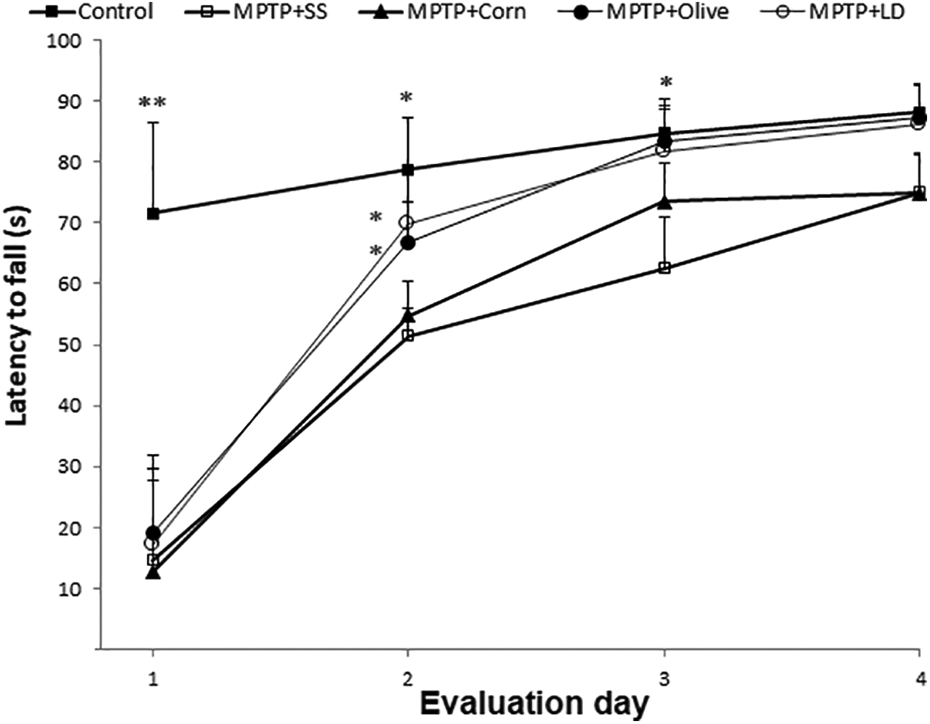
Grasp-test evaluation posttreatment. Results are expressed as mean ± SE (n = 8). *p < 0.05; **p < 0.01 compared to the MPTP + SS group. SE: standard error; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; SS: saline solution; LD: levodopa.
The spontaneous locomotor activity showed a significant difference between the MPTP + SS group and the groups treated with levodopa and olive oil, which showed more movements from day 2 to day 4, and similar values on the third and fourth days of evaluation when compared to the control group (Figure 1).
Animals treated with levodopa or olive oil remained on the rod longer than animals treated with saline solution (MPTP + SS). These results were significant since day two (p < 0.01), and this difference remained until the third and fourth days of evaluation (p < 0.05). Levodopa and olive oil groups showed similar values on the third and fourth days of evaluation in comparison with the control group (Figure 2).
Finally, to evaluate forelimb muscular strength, differences were observed only between the MPTP + SS group and those groups treated with levodopa or olive oil on the second and third days (p < 0.05) in the grasp test (Figure 3).
Effects of the treatments on neuronal survival in the substantia nigra and striatum
A significant difference (p < 0.038) was observed between the control and the treated groups in the number of neurons by immunohistochemistry. MPTP + SS and MPTP + Corn groups showed a reduced neuronal survival in the substantia nigra (p < 0.01; Figure 4) and the striatum (p < 0.01; Figure 5). In contrast, the groups treated with olive oil and levodopa showed a higher number of NeuN-positive cells in comparison with the MPTP + SS group; apparently, these treatments prevented the neuronal loss by MPTP administration in both areas (p < 0.05).
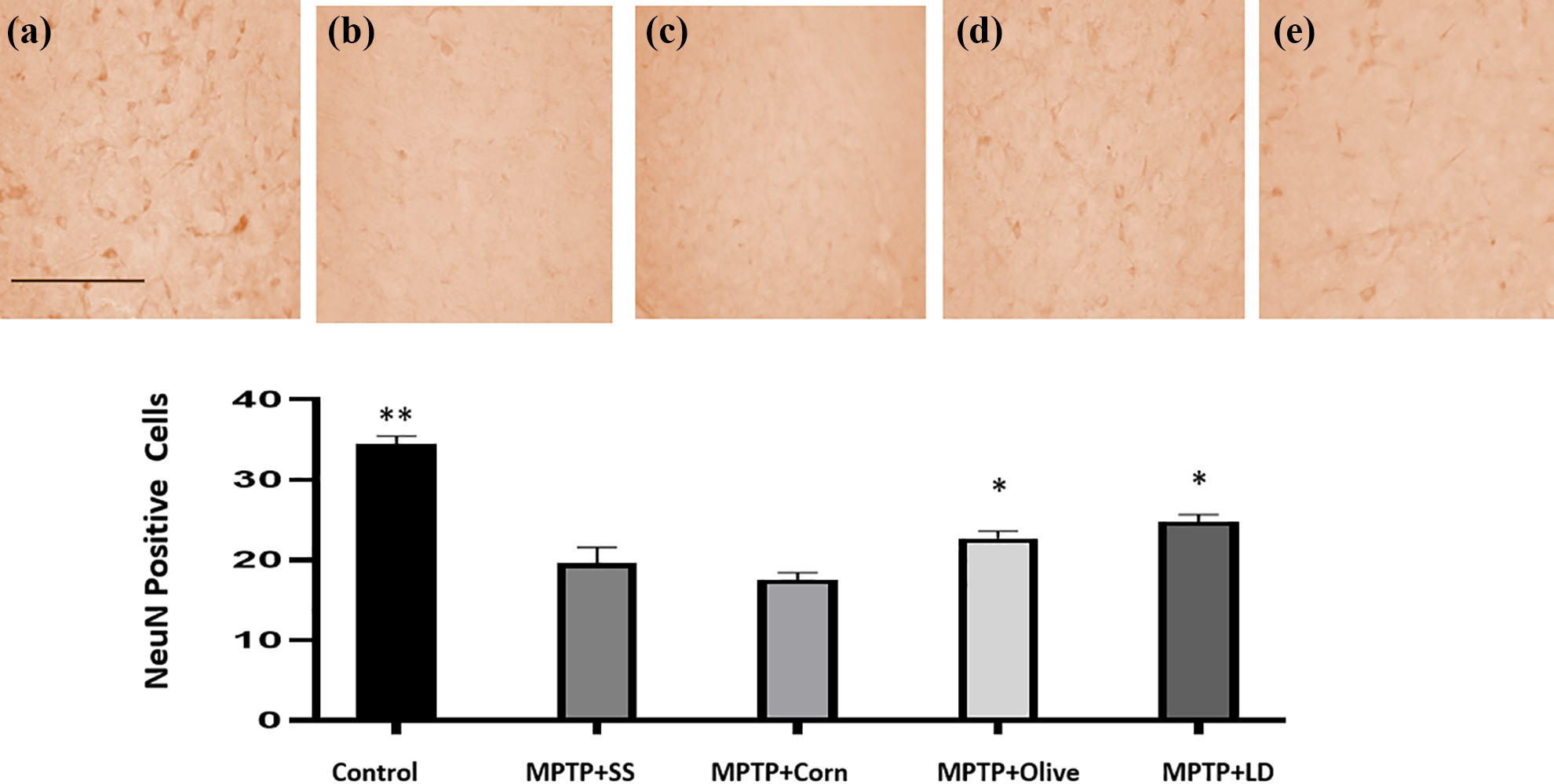
Photomicrographs of NeuN-positive labeling in the substantia nigra of C57BL6 mice. Control (a), MPTP + SS (b), MPTP + Corn (c), MPTP + Olive (d), MPTP + LD (e). Magnification ×40. Scale bar represents 100 µm. Bars represent the number of NeuN-positive neurons ± SEM (n = 4). *p < 0.05; **p < 0.01 compared to the MPTP + SS group. SEM: standard error mean; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; SS: saline solution; LD: levodopa.
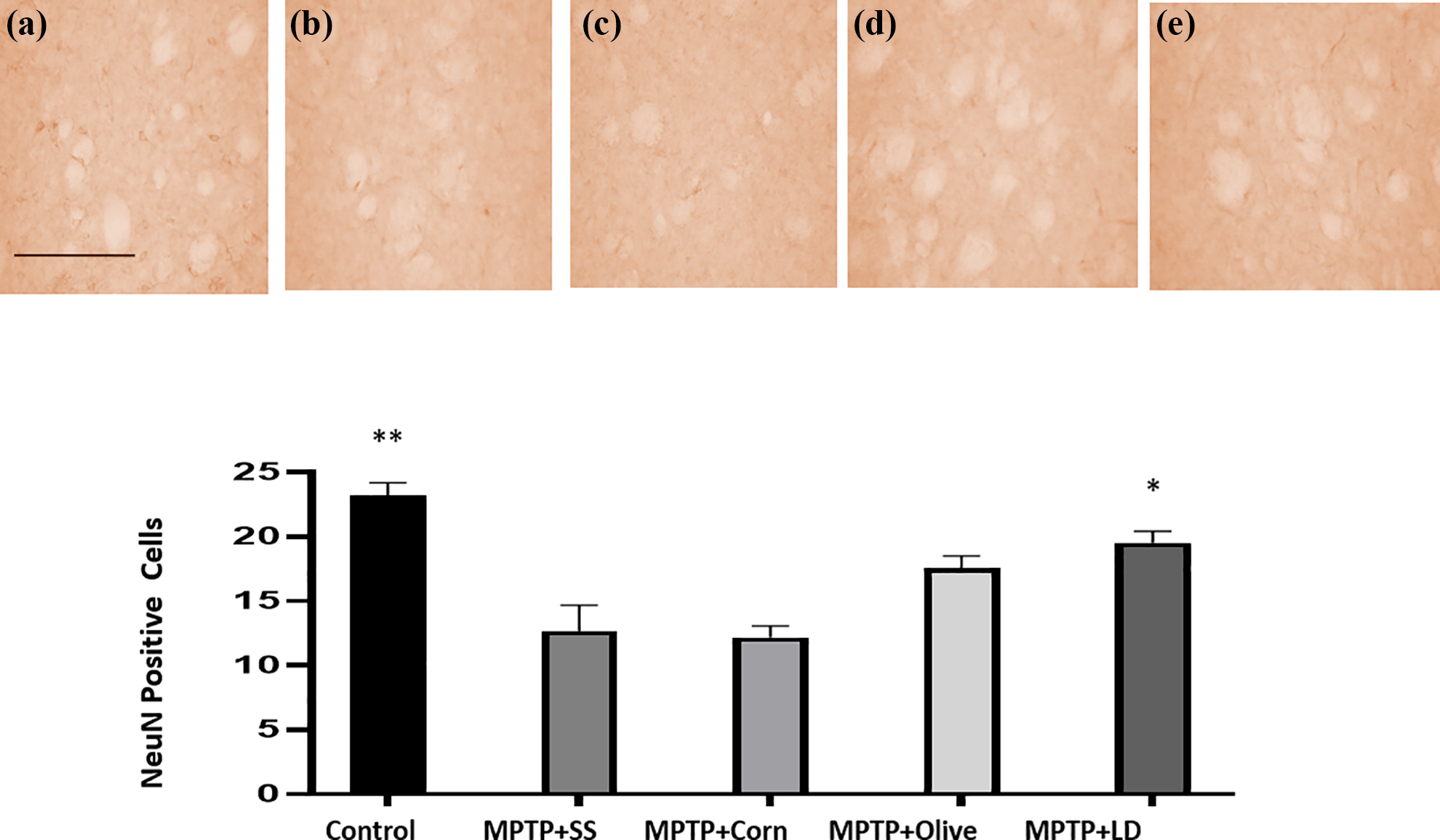
Photomicrographs of NeuN-positive labeling in the striatum nuclei of C57BL6 mice. Control (a), MPTP + SS (b), MPTP + Corn (c), MPTP + Olive (d), MPTP + LD (e). Magnification ×40. Scale bar represents 100 µm. Bars represent the number of NeuN-positive neurons (mean ± SEM (n = 4). *p ≤ 0.05; **p < 0.01 compared to the MPTP + SS group. SEM: standard error mean; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; SS: saline solution; LD: levodopa.
Discussion
A beneficial effect of olive oil administration on motor behavior has been described in nonintoxicated C57BL6 mice. Pitozzi et al. 25 described that olive oil prevented the age-related impairment in motor coordination in the rotarod test when administered orally. These authors suggested that this effect was probably related to reduced lipid peroxidation in the central nervous system (CNS). Furthermore, Luceri et al. 26 showed that an intervention with olive oil modulates the expression of genes and miRNAs involved in neuronal function and synaptic plasticity, along with cognitive, motor, and emotional behaviors. Moreover, the frontal cortex of old mice fed with olive oil exhibited miRNA expression profiles similar to those observed in young mice. 26
In the current work, our findings show that MPTP induced motor disruption in C57BL6 mice. These results are consistent with those reported in previous investigations. 18,20,27 Despite the acknowledged limitations of the model, this finding, in turn, is consistent with most studies that describe parkinsonian-like motor deficit after the administration of this neurotoxin. 27
Several tests are used to obtain a motor profile; for instance, locomotor activity in an open field is a good way to evaluate gait and coordination; 28 the performance in the rotarod test requires a variety of proprioceptive, vestibular, and fine-tuned motor abilities, 21 and the grasp test is clearly dependent of muscle strength. 29
With the current experiments, a constant diminished motor activity was registered. An acute motor disruption including tremor and ambulatory limitation was observed as elsewhere. 20,30 In this MPTP acute administration protocol, the performance recovered sequentially in some motor tests during the following days. However, neuronal damage was significant after a week of MPTP administration. 18 Our results showed notable motor disruption in the first day, which involved limitation in every motor ability evaluated (gait, motor balance, coordination, muscular strength). 27
The recovery of the locomotor ambulatory activity is well-known. 18 However, in this work, the diminished performance in open-field and rotarod tests allowed to notice the limited coordination within several days of evaluation; although muscle strength loss was noticeable within the first hours, 31 it recovered faster than coordination, as it is often described when MPTP or analogues are used to induce motor disruption. 27,32,33 This observation requires special attention as muscle strength is related to different neuronal networks and nuclei from those related to coordinated movements of the limbs. 34 Additional studies are required to establish if the damage is a partial judging from the behavioral evaluation. As expected, our findings showed a clear levodopa recovery effect on motor behavior. Moreover, olive oil showed a similar effect, while no recovery was observed in the performance of any of the tests with corn oil or SS treatments. The beneficial effect of levodopa administration is mainly because it is a dopamine precursor, which acts on dopamine receptors to revert motor disruption. Additionally, its administration modulates monoamine levels and enhances antioxidant mechanisms involved in the limitation of MPTP-toxic effects. 35,36
Regarding the histological analysis, anti-NeuN staining is extensively used as a marker of mature cells in many fields, including neurosciences, developmental biology, diagnosis, or animal models of neurodegenerative diseases, since the RBfox3/NeuN protein is distributed in the nuclei of mature neurons in most vertebrates, including neurons in the spinal cord, cerebral cortex, hippocampus, dorsal thalamus, caudate/putamen, and cerebellum. 24 In the present study, MPTP induced a significant reduction of stained neurons in the striatum and substantia nigra (Figures 4 and 5) and discrete changes in the frontal cortex and cerebellum (data not shown), as elsewhere. 20,31 Both olive oil and levodopa groups showed a higher number of NeuN-positive cells in comparison to the group treated with MPTP + SS, in which neuron loss was observed in both areas.
A limitation of this study was the identification of active substances and mechanisms involved in behavioral and neuronal changes. However, the use of SS as a polar vehicle, as well as levodopa as a known active substance in this model, 30,35,36 and two different oils suggests possible molecules and mechanisms of action involved in the amelioration of neurotoxicity.
In this context, it should be mentioned that several diet components have been associated with effects limiting the progression of neurodegenerative processes. 4,37 Vegetable oils often used in diets are basically composed of fatty acids. For example, the common percentage of saturated and unsaturated acids in corn oil is 15% and 85%, respectively, from which oleic acid (45–60%), linoleic acid (40–55%), palmitic acid (7–8%), stearic acid (2–3.5%), arachidic acid (0.5–1%), and lignoceric acid (0.2–0.4%) are the main fatty acids. 37 –39 Conversely, oleic acid, a monosaturated omega-9 fatty acid (55–83%), linoleic acid (3.5–21%), palmitic acid (7–20%), stearic acid (0.5–5%), arachidic acid (0.5%), and linolenic acid (0.2–1.5%) are the main fatty acids of olive oil. 40 –42 The proportion of different fatty acids in oils has been proposed as a neuroprotective factor. 43
Moreover, some other components are particularly relevant in olive oil composition. 43 –45 Thus, some phenolic compounds (phenolic acids, phenolic alcohols, secoiridoids, hydroxy-isocromans, flavonoids, and lignans) in olive oil (5mg 10 g−1 of olive oil) such as polyphenols are important. 46 –49 Mainly, tyrosol, hydroxytyrosol, oleuropein, and oleocanthal structures are related to neurotransmitters and catabolites produced in the mammal brain, particularly dopamine (see supplemental Figure 1). 50 –52 These compounds show their effects in a dose-dependent manner since they exhibit their activity in the micromolar range and are considered potent antioxidants as well. 53 Polyphenols exert antioxidant, anti-inflammatory, anticancer, antimicrobial, antiviral, antiatherogenic, hypoglycemic, hepato-, cardio-, and neuroprotective effects. 53,54 These compounds have demonstrated cell protective effects due to their ability to scavenge free radicals and inhibit low-density lipoproteins oxidation, as well as the activation of specific signaling pathways, that is, the Nrf2-Keap1 pathway. 48,49,53 This ability could interfere with the MPTP-toxicity mechanism since this process is dependent on the oxidative pathways. 15,33
Additionally, hydroxytyrosol and oleuropein contain catechols and other chemical moieties in their structure, which confer the potential capacity to act as disrupters of catecholamine transporters or enzymes involved in the action of endogen neurotransmitters, their receptors, and the toxicity of MPTP. 55 –57 Moreover, additional mechanisms of oleuropein have been associated with neuroprotective effects, such as the decrease in the production of leukotrienes and the oxidized forms of lipoproteins; the attenuation of tumor necrosis factor-α and interleukin-1β expression, and, consequently, the expression of inducible nitric oxide synthase and cyclooxygenase-2; and the reduction of the fragmentation of amyloid precursor proteins. 56 In 2014, Sarbishegi et al. reported that intraperitoneal administration of oleuropein once a day for 6 months in aged rats improved the antioxidant enzyme activities in midbrains in comparison to the control group. Furthermore, treated rats showed more neurons in the substantia nigra, revealing that oleuropein protects against dopaminergic neuron loss. This result provides a new insight into the prevention or attenuation of damage or loss of dopaminergic neurons associated with PD. 57
Oleocanthal is structurally related to ibuprofen and its anti-inflammatory activity through cyclooxygenase inhibition as well. 58 Moreover, this compound inhibits tau fibrillization in vitro by forming an adduct with peptides related to detrimental phosphorylation and enhances beta-amyloid clearance, which is often associated with a potential protective effect on Alzheimer’s disease. Similarly, oleocanthal and other iridoids have shown beneficial effects on PD. 59,60 Some other bioactive compounds present in lower concentrations in olive oil (1–100 ppm), such as vitamins E and K, squalene, and pigments like chlorophyll, pheophytin, and carotenoids, have shown less evidence as neuroprotectors. 43
Not only the direct interactions of these compounds with some proteins should be considered to explain the observed effects. Emerging evidence encompassed by individual genomics and responses to dietary patterns is also being included in basic and clinical research. Therefore, changes in gene and protein expression, specifically those downstream signaling components, should be also analyzed after an agent exposition. 61
Another limitation of the current study was the use of only one neurotoxin to induce parkinsonism, for which only acute toxicity and short-time evaluation were performed. Further studies are necessary to characterize each component of olive oil 41,62,63 and asses every pharmacological effect. Also, it would be interesting to investigate the effects of olive oil—or some of its components—over time and via alternate routes of administration. That is noted since a model that utilizes intraperitoneal delivery of both the toxicologic challenge as well as the damage-attenuating agent limits the generalizability of the results to actual parkinsonism in humans. Thus, additional approaches could enrich the knowledge on the role of olive oil in the amelioration of MPTP toxicity and its neuroprotective effects, as well as the neuromodulation of specific agents obtained from olive oil.
Conclusions
In the present study, we evaluated the potential of two vegetal oils to diminish the acute toxicity in the CNS induced by MPTP administration, which emulates parkinsonism in mice. A clear attenuation of toxicity and damage on motor function and neuronal integrity was produced by olive oil but not by corn oil. After the administration of olive oil in mice with MPTP-induced toxicity, the behavioral tests showed notable improvement; however, the immunohistochemistry results were less noticeable. Additional studies are required to obtain more insight into the mechanisms involved in the observed effects, as well as the possible implications for parkinsonism, PD, or other pathologies involving neuronal death and motor disruption. Moreover, the evaluation with different MPTP dosages and other PD models is also necessary to support possible therapeutic benefits for PD. Also, further studies are required for the identification of specific compounds in olive oil with potential to ameliorate the incidence and progression of PD. Those results could lead to the development of synthetic polyphenols structurally related to some of the components found in olive oil which can ameliorate parkinsonism.
Supplemental material
Supplemental Material, Suppl._Fig.1 - Olive oil limited motor disruption and neuronal damage in parkinsonism induced by MPTP administration
Supplemental Material, Suppl._Fig.1 for Olive oil limited motor disruption and neuronal damage in parkinsonism induced by MPTP administration by Eunice D. Farfán-García, Antonio Abad-García, Alberto Alatorre, Teresa Pérez-Capistran, Enrique Querejeta and Marvin A. Soriano-Ursúa in Toxicology Research and Application
Footnotes
Acknowledgments
The authors thank Dr. Ana Karen García-Avila for her technical and academic contribution in preliminary stage of this research work. The authors also thank Dr. Julia J. Segura Uribe and Nielsen Line Nielsen Maldonado for proofreading on this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Comisión de Operación y Fomento de Actividades Académicas, the Secretaría de Investigación y Posgrado del IPN (SIP M1930), and the Consejo Nacional de Ciencia y Tecnología (CB235785).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
