Abstract
A large volume of antibiotics is used in fish farms to treat diseases because the farmed fish are fully affected by diseases and parasites in the aquaculture and particularly in the ocean environment where disease pathogens multiply quickly. The frequent use of these antibiotics in aquaculture has resulted in animal; stress, infection, and their dissemination in the form of antibiotic resistant genes to other bacteria including human and animal pathogens. The problems arising with antibiotics can be overcome by using silver nanoparticles (AgNPs) due to their physiochemical properties and low toxicity. So AgNPs could be combined with antibiotics to induce infections in fish cell lines and to protect dissemination of antibiotics in the form of antibiotics resistant genes. We expose AgNPs on fish cell lines as a new nano-antibacterial agent to investigate and obtain findings in terms of the cell viability and toxicity. The experimental data is analyzed to examine the antibacterial effects of nanosilver as a replacement agent and discuss the complex scenario, drawbacks, techniques, methods, main mechanisms, and procedures to perform antibacterial tests of exposed AgNPs. There would be an attempt to deal with the AgNPs antibacterial therapies for the fish cell lines.
Introduction
Commercially, nanotechnology has been proved the most viable and potentially emerging technology with new features and advanced procedural approaches to fight and prevent diseases including marine toxicology. To improve the human and marine life, nanotechnology has introduced new methods, novel biomedical nano devices, and nanomedicine which has largely facilitated the health tools and medicines. 1 –3 Preventing diseases in the marine life play a turn key role in the socioeconomic sector of a country and in this regard, the role of nanotechnology seems indispensable; providing potential tools to come up with the situation for the possible solution. Nanotechnology mainly deals with the processes and manipulation of nano-products and nanomaterials where physiochemical properties become novel and size dependent. 4 –6 Due their size dependent properties, in biotechnology and life sciences, these nanomaterials have received considerable attention for creating new analytical tools. 7 It has been estimated that silver nanoparticles (AgNPs) have got the highest degree of commercialization in the consumer products. In addition to this, AgNPs have also shown great interest for applications such as catalysis, surface enhanced imaging, surface enhanced Raman spectroscopy (SERS), and enhanced florescence. 8 The nanomaterial of Ag acts as a nanochemical contaminant by entering in the sewage pipes and the waste water treatment plants they are causing toxic, antibacterial, and microbial effects.
The increased chemical activity and toxicity of a nanomaterial largely to depend on the particle size, concentration, solubility, and the chemical and biological properties. 9 NPs could cause overload ophagocytes, if inhaled, ingested, or by skin contact. The inhaled NPs diseases include asthma, bronchitis, and lung cancer but not limited. The adverse effects of a nanomaterial on the biological systems and the cellular components have been previously reported. 10 Though, there are wisely reported mechanisms for the cytotoxicity of engineered NPs such as; the physicochemical properties, contamination with the toxic element, fibrous structure, and the generation of high surface charge and radical species. However, a wide gap still exists which needs to fill up to understand the mechanisms for the toxicity. The bactericidal effects of a nanomaterial have great scope due to new resistant-strain of the bacteria against antibiotics as reflected by the reported studies. 10,11
There are several biocidal NPs such as CuO, ZnO, and Ag, however, among these, the AgNPs have shown significant toxicity particularly to the aquatic organisms, algae, fish, and crustaceans. 12,13 The toxicity of AgNPs depends on their size, morphology and their dispersion in the solution. For the same concentration, aggregated and non-monodispersed AgNPs have shown entirely different toxicity. 14,15 Some of the research groups have shown the bactericidal properties of Ag colloidal suspension as the health supplement. On the nanoscale, the Ag ions could cause bio-uptake and over toxicity. Perhaps, in the dissolved form, Ag ions are more toxic for fresh water and marine invertebrate. In water, it is rather a hard task to empirically test the toxicity of Ag ions due to their complex nature. Different biological models have been developed to predict the acute metal toxicity to aquatic vertebrates and invertebrates. 16 Yokel et al. performed a review on the behavior and biological effects of AgNPs in the aquatic environment by considering physiochemistry of the NPs. 17 They critically reviewed and evaluated the existing knowledge on AgNPs as a potential problem for the environmental health by taking into consideration the characteristics, behavior, bioavailability, biological effects, antibacterial effects, and the antimicrobial effects. From current information on AgNPs and their role in the environment, the existing gap of required key current knowledge could be identified. It is well-known fact that the accumulation of AgNPs in bacterial membrane causes permeability and results in the cell death. But it is impossible for researchers to explain the antimicrobial property of the positively charged AgNPs. However, the genetic differences in disease resistance have been determined on fish which has shown different resistance for one type of disease.
In fish, main disease is caused by the infectious organisms such as bacteria, virus, fungi and protozoans, metazoans and parasites. The prevalence and spread up of the disease problem has become the most serious concern in the fish farming and fish communicable diseases, intensely effecting our food sector and subsequently the local economy. In this regard, AgNPs are used as a medium for delivery of antibiotic and synthesized composites as disinfecting filters. The antibacterial property of AgNPs, anchoring and penetrating into bacterial cell wall, has been associated to the formation of pits in erectus coli cells. 18,19 The study by Ahamed et al. 19 describes the damaging response of deoxy-ribonucleic acid (DNA) in mammalian cells to the different surface chemistry of AgNPs. It is noteworthy to mention here that the antibacterial effect of NPs is independent of the acquisition of resistance by the bacteria against antibiotics. However, further studies are required to verify if the bacteria could develop resistance toward NPs. It would also be worth to examine the cytotoxicity of NPs toward fish cells before their therapeutic practice.
In antimicrobial utilization, AgNPs also have been effective and potent agents due to their potential antimicrobial properties and low toxicity toward fish cells. 17 The toxicity effect of AgNPs on microbes has been related to the interaction of Ag ions with the thiol group of vital enzyme and proteins; affecting the cellular respiration and the transport of ions across the membranes with the ultimate cell death. 20 To control microorganisms and infections, AgNPs can be applied for formulations of a new type of bactericidal material. Previously, the bactericidal efficacy of AgNPs has been investigated and their potential against a broad range of microbes has been proved. Ahamed et al. studied the bactericidal activity of AgNPs and demonstrated that the bacterial cells death could happen due to the interaction with the building elements of bactericidal cell membrane. 19
In this research work, we investigate the potential capability of engineered AgNPs to decrease the cell viability in the cultured fish cells. The purpose of the designed culture study was to confirm the toxicity of engineered AgNPs by reporting that the cytotoxicity of AgNPs leads to decrease the cell viability. We generated and analyzed a homogeneous set of experimental data on the toxicity of engineered AgNPs with the size of the particles in the range of 50–100 nm by performing Alamar Blue (AB) and Neutral Red (NR) Assays. AgNPs have been chosen for several rational reasons including; (i) these are widely used as antimicrobial in the medical equipments and cosmetic products, (ii) these have shown more toxic effects especially to aquatic organisms, algae, fish, and crustaceans. 21,22 The fish cell lines were exposed to different concertation of AgNPs to convey a meaningful information on the mechanisms of toxic actions which could be useful as an input tool to quantitatively model toxicity. Another important aspect of this contradictory data is that, AgNPs used for the toxicity tests were not monodispersed and theoretically seems to cause toxic effects of different magnitudes which has significant worth. 23,24
Materials and procedure
Characterization methods
Characterization is critical to accurately determine a dose (defined as the amount of particular agent that reaches the target organisms) required for diseases in marine life. For NPs, the number of particles or the total surface area could be important in spite of the total mass and is critical to measure the dose in traditional toxicology. Dissolved Ag speciation and bulk particles characteristics are also needed quantification. Characterization in all media which are demonstrated with extensive changes in aggregation state due to the addition of biological media, are potentially change due to PH (potential of hydrogen), ionic strength and specific counter ions. There are of a variety of analytical and metrological techniques which are used to measure the toxicity and interaction of NPs with biota. Previously, such studies have done and in the most cases been compared to either larger size (micron size), similar size particles not made of silver aggregates and or dissolved Ag ions. The antibacterial activity of Ag ions is determined by the release of Ag ions in the aquatic media. The concentration of Ag ions does lower the antibacterial activity of the Ag ions. AgNPs of 0.4 mg was characterized in 25 ml pure water under specified experimental conditions. UV–Vis. absorption spectrum of 100 µm AgNPs suspensions was scanned in the range of 250–800 nm as shown in Figure 1(a). The spectrum showed the usual surface plasmon resonance (SPR) absorption, peaked at 380–500 nm with a small kick that appears on the lower wavenumber side and the tail stretching in the infra-red regime. Basically, SPR is a non-radiative electromagnetic surface wave phenomenon arises due to the collective coherent oscillation of the conduction band free electrons in the metal NPs when optically is excited. The size distribution of AgNPs in the aqueous dispersion and culture media was characterized by using dynamic light scattering (DLS) also known as photon correlation spectroscopy (PCS) in the prepared AgNPs solution (0.025 mg/25 ml) for 24 h to check the stability. The Zeta average size measured by DLS is displayed in Figure 1(b). Briefly, DLS is a laser light scattering phenomenon caused by the randomly dispersed moving particles within a solution medium due to the Brownian motion to exploit the particle size. The rapid fluctuation in the scattered light by the dispersed particles, on the order of tens of nanoseconds to hundreds of milliseconds, are directly related to the motion and size of the particles.
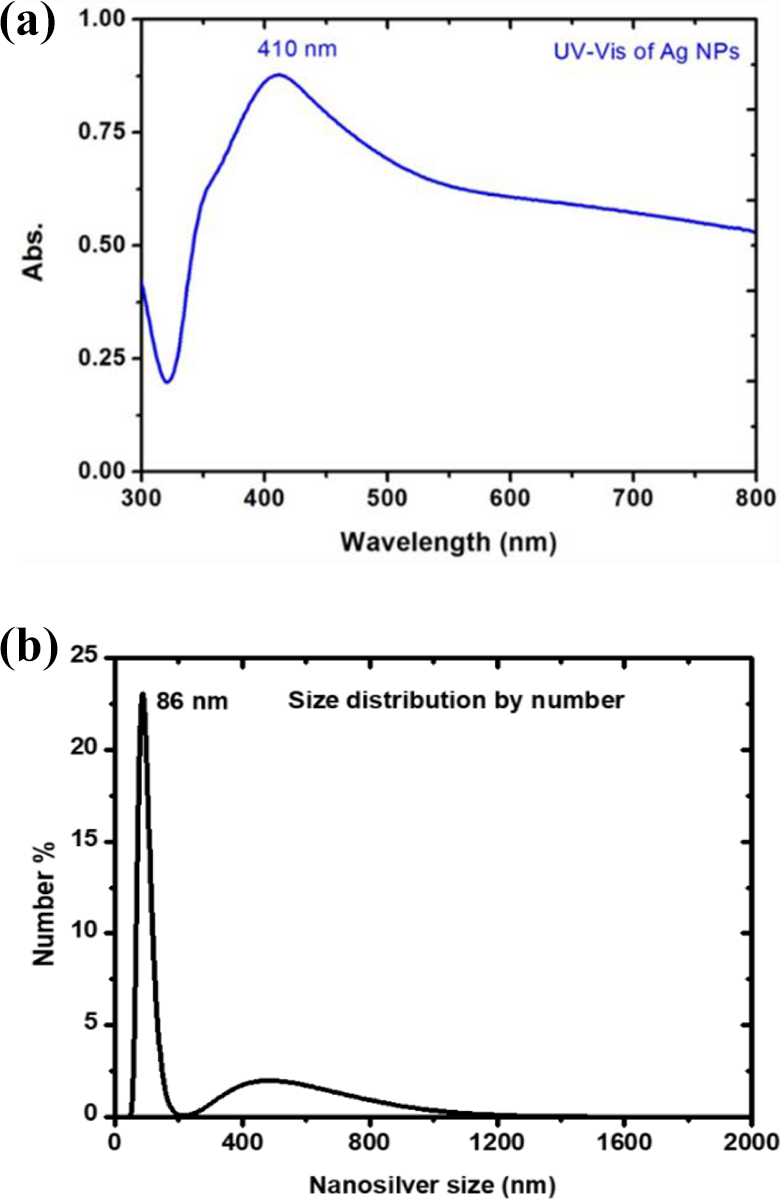
Surface plasmon resonance absorption (a) and dynamic laser scattering (b) spectra of commercial grade AgNPs solution in distilled water.
Cell culture
AgNPs can be toxic to fish cell lines depending upon whether the exposure was to an adult or juvenile fish. The AgNPs could also be cytotoxic and clastogenic to fish cells depends upon the concentration of the NPs. AgNPs have shown to be cytotoxic and genotoxic to fish cells. The PLHC-1 (Poeciliopsis lucida hepatocellular carcinoma) cell lines are co-cultured in 96 well coaster plates. This system allows the measurement of NPs across the polarized liver cells (HEPATIC) and at the same time the measurements of the cellular and molecular effects together. These studies yield valuable information on the effects of the engineered nanomaterials (ENMs) on the aquatic organisms especially on the molecular and cellular level.
As determined, during method development of RTG-2 (Rainbow trout gonad cells) media (5%), cell performance largely depends upon the culture propagation, percent confluence of flasks and cell passage. In all experiments conducted here, in the culture conditions, the media were contained 500 ml DMEM (Dulbecco’s Modified Eagle Medium), 50 ml fetal bovine serum, 5 ml penstrip, 5.6 ml non-essential aminoacids, 7.5 ml HEPES (4-(2-hydroxyethyl)-1-piperazine ethane sulfonic acid), and DMEM unsupplemented as an Assay medium.
Seeding of cells
For seeding of PLHC-1 cells, confluent cells (70–75%) T-75 flasks were detached using trypsin–versine solution followed by the addition of propagation medium to the neutral trypsin. Then cells were pelleted by centrifugation process at 2000 RPM for 3 min at 20°C. Supernatants were removed and cell pellets were resuspended in Assay medium and counted using haemocytometer (a calibrated microscope slide designed to quickly estimate the concentration of cells in a sample.). The cell densities were adjusted to 2 × 104 cells per 100 microliters (µl). Cells were seeded into 96 well coaster plates and were placed in an incubator for overnight to allow for attachment and recovery.
Methodology was developed for quantifying the photocytotoxicity of flouranthene to liver cells of fish cell line. Solubilization in a modified culture medium was achieved with DMEM as the positive control and AgNPs as a negative control using different Ag concentrations. In order to maximize the surface area available for the AgNPs uptake, RTG2 cell culture were trypnized and plated in 92 coaster plate treated DMEM and suspended in 100 µl.
Exposure of AgNPs
The AgNPs suspended in unsuplemented media at a stock solution of 5 mg/ml was vortexed to ensure maximal dispersion. The AgNPs were then added to the culture media to make different concentrations up to 3 ml. Different concentrations of 2.5, 2, 1.5, 1.0, and 0.5 were used for both Alamer Blue (AB) and Neutral Red (NR) tests to achieve the final concentration of cells harvested for 24 h after AgNPs exposure. After 24 h exposure, AgNPs induced apoptosis. Viable cell numbers were significantly decreased in the time dependent manner. Optical images of PLHC-1 cell lines before and after exposure are shown in Figure 2(a) and (b) respectively.

Optical images of PHLC-1 fish cells before exposure (a) and PHLC-1 fish cells after exposure (b) to commercial grade AgNPs.
Cytotoxicity assays
To assess toxicity of AgNPs in an exposed culture media, Alamar Blue Assay (ABA) and Neutral Red Assay (NRA) were performed. NRA is one of the most widely cytotoxicity tests used to provide an estimation of the number of viable cells in the culture. This test is based on the potential of viable cells, incorporated to bind a dye. ABA bio Assay is another important test used to evaluate the metabolic function and the cellular health. ABA is used to assess cell viability and cytotoxicity in a range of biological subsystems and cell types. We performed AB and NR Assays and measured the toxicity of AgNPs using fish cell line as a model and are described below.
Alamar Blue and Neutral Red Assays
After exposure, loss of monolayer changes in the shape in NR/AB solution 1.5/30 ml AB dye and 0.375/30 ml of NR stock for each 10 ml of unsupplemented media (no serum or antibiotics). Now we removed the media and washed the plate with 100 µl of sterile PBS (phosphate buffered saline) and added 100 µl AB/NR media solution to 96 well coaster plates to be tested and incubated for 3 hours and measured the AB fluorescence with a plate reader, using a 540 nm excitation wavelength and a 595 nm emission wavelength. After, the AB measurement the medium used was discarded. We used 100 µl PBS to wash plates, then 150 µl of NR fixative and shake the plate for 10 min at 240 RPM (rotation per min.), read NR fluorescence in plate using 540 nm excitation and 650 nm emission values. The sketches of the experimental fish model and procedure of the methods/materials is presented in Figure 3.
Experimental fish model used and a sketch of the methods and materials applied in the lab; showing cell culture, tests and chemicals.
Results and discussion
Several studies have shown that AgNPs are toxic, however, there have been no systematic studies to convey a solid analysis on the toxicity of varying amounts of Ag ions present in AgNPs suspension. So in the current study of our here, fish cells were exposed to AgNPs for 24 hours, and the toxicity effects were measured using potential tests; the Alamar Blue (AB) and the Neutral Red (NR). In both essays, AgNPs shows the high degree of toxicity at low particle concentrations of 0.5 and 0.4 mg/ml as described in Figures 4 and 5. The healthy looking cells were maintained a reducing state within their cytosol. The “Resurzine reduction test” has been used to assess the bacterial and yeast contamination of milk. As dye is a very simple way of measuring the cell proliferation and cytotoxicity.
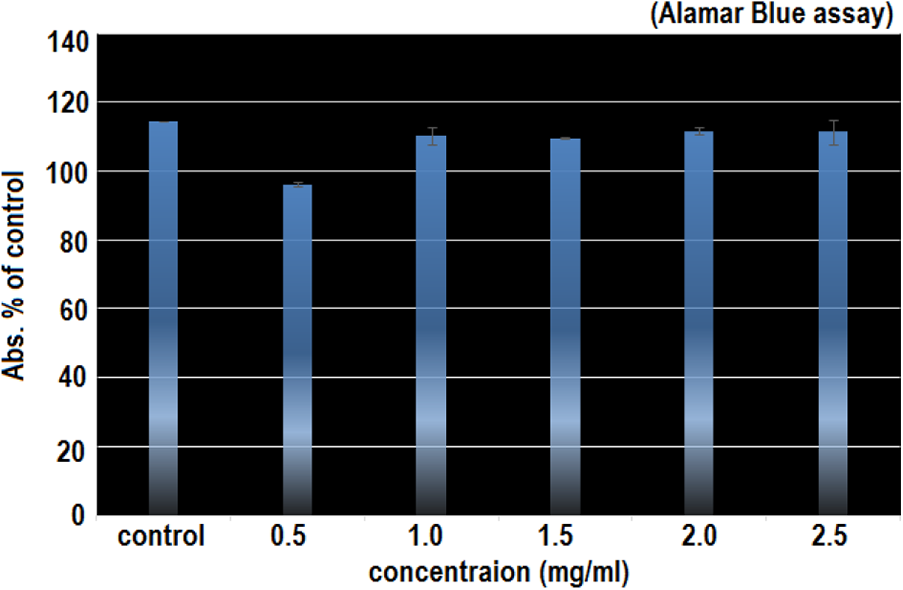
The Alamar Blue Assay to test the effect of AgNPs on the viability of fish cells. PLHC-1 cells have been treated with AgNPs at concentration of 0.5 mg/ml for 24 hours. Nanosilver toxicity was higher at the concentration of 0.5 mg/ml. AgNPs shows low cell viability of 98% for 0.5 mg/ml while for 1.0, 1.5, 2.0, and 2.5 mg/ml, more than 100% cell viability was observed.
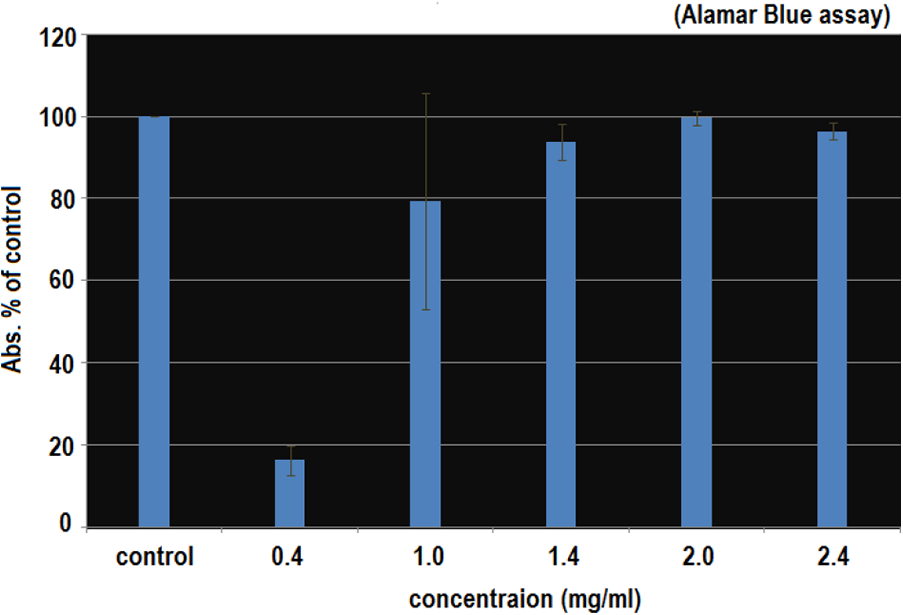
Alamar Blue Assay shows the effect of AgNPs on profiliration of fish cells. PLHC-1 cells were treated with AgNPs at concentration of 0.4 mg/ml for 24 hours. Nanosilver toxicity was higher at the concentration of 0.4 mg/ml. AgNPs effects shows low cell viability of 18% for 0.4 mg/ml, while for 1.0, 1.4, 2.0, and 2.4 mg/ml, cell viability was more than 80%.
The reducing potential of cells converts the Alamar Blue reagent into a detectable (absorbent) product. Since Alamar Blue reagent is nontoxic and can incubate cells with reagent and monitor fluorescence (or absorbance) over the time on the same sample. The absorbance of Alamar Blue reagent which changes is depending upon the cell viability and the proliferation.
It has to be kept in mind that the total amount of Ag particles added to the fish cells was in the range of 0.5–2.5 mg/ml in one case and 0.4–2.4 mg/ml in the second case to replicate the concentration of silver added up to the cells. So in the comparison, the viability of the cells exposed to AgNPs is lower at low concentration. The cell viability was 90% and 18% for the concentrations of 0.5 and 0.4 mg/ml respectively. Clearly, the toxicity of AgNPs was much higher at lower concentration of 0.5 and 0.4 mg/ml. With the increased concentration of AgNPs, the toxicity was decreased; showing the cell viability of more than 100%.
The Neutral Red uptake Assay provides a quantitative estimation of the number of viable cells in the cell culture and many biomedical and environmental applications. This Assay is based on the ability of cells to incorporate Neutral Red dye in lysosomes. It is weakly cationic dye and penetrates into the cell membrane by the non-ionic passive diffusion and concentrates in the lysosomes. When the cells are died out, the dye cannot retain inside it when the dye pH gradient is reduced to a certain level. The dye retained inside is proportional to the live cells. The procedure seems cheaper and the Assay can be completed in a shorter time of about <3 h. The fluorescent units obtained in the cell viability Assays were converted into percent viability of the control cells. Low or high uptake of the chemical affect the lysosomes so AgNPs exposure on cells avoid the interference produced by the cell proliferation. Since any chemical having a localized effect on the lysosome results in a low or high uptake and for this reason, we expose fish cell lines to AgNPs to check their viability without interference produced by the cell proliferation. The amount of retained in the dye is proportional to the number of viable cells thus survived. It is thus possible to distinguish between the viable, the damaged and the same time the dead cells.
In the Neutral Red Assay, the fish cells were treated for 24 hours. The cell viability was measured using the Neutral Red dye. The toxicity of AgNPs for the exposed fish cell lines were to depend on the total concentration of the AgNPs. The percentage of the Neutral Red dye absorbed at concentration of 0.5 is about 3% while that of the Neutral Red dye absorbed at a 0.4 concentration is about 18% (see Figure 6)
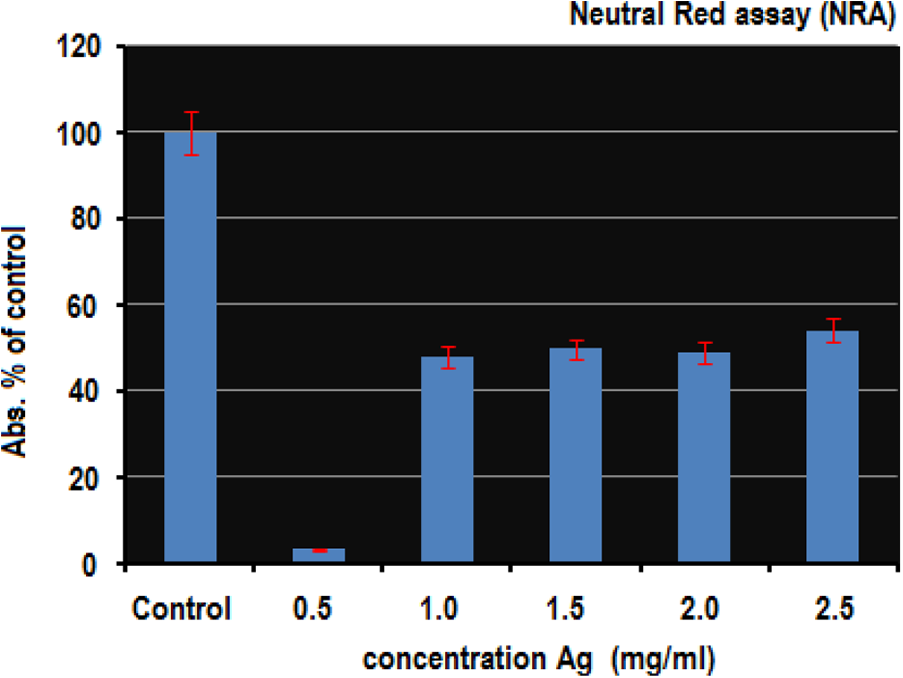
Neutral Red Assay (NRA) to test the effect of AgNPs on viability of fish cells. PLHC-1 cells were treated with AgNPs at concentration of 0.5 mg/ml for 24 hours. The cell viability was higher at the concentration of 0.4 mg/ml.
In Figure 6, the absorbance % of the control vs. Ag concentration has displayed. The cellular viability was checked with the help of Neutral Red Assay. In this case, nanosilver measures the cytotoxicity effect by killing the fish cells. Hence, the cytotoxic effects brought by the exposed nanosilver is the measure of the cytotoxicity with the help of cell viability. Clearly, the cell viability decreased as the cytotoxicity of effect of nanosilver was increased. The nanosilver toxicity was higher at the concentration of 0.5 mg/ml but with the increase in concentration of AgNPs, the viability of cells was also increased which shows less toxicity of the AgNPs at the high concentration. The AgNPs effect shows low cell viability of 5% at 0.5 mg/ml for 24 hours while for the other concentrations of 1.0, 1.5, 2.0, and 2.5 mg/ml the observed cell viability was 48%, 49%, 49% and 58% respectively.
Figure 7 also shows the cytotoxicity effect of AgNPs assessed by using Alamar Blue Assay test. The cellular viability has been determined for the different concentration of nanosilver is logical. Here, toxicity of the nanosilver was less at the concentration of 7 mg/ml which is about 69% and shows high viability of the fish cells exposed to the nanosilver.
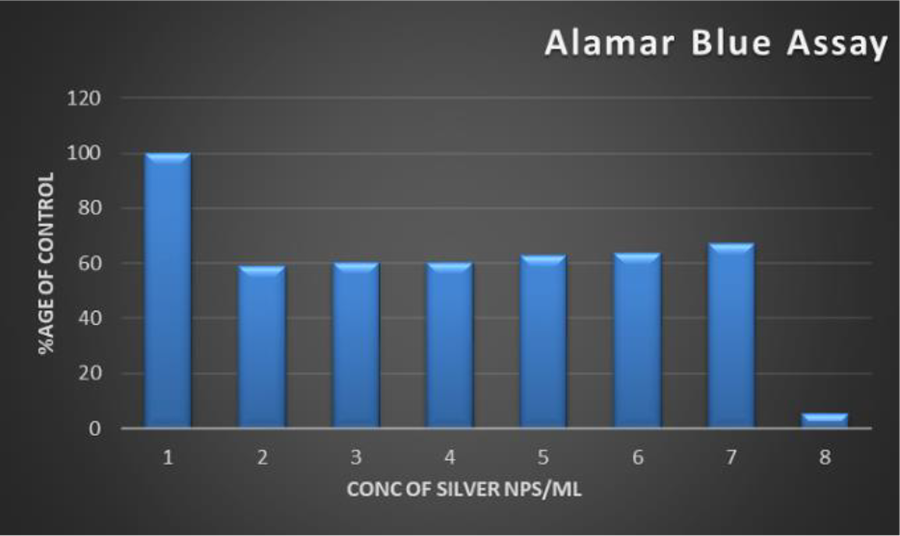
Alamar Blue Assay to test the effect of AgNPs on the viability of fish cells. PLHC-1 cells were treated with AgNPs for 24 hours. Nanosilver toxicity was less at the concentration of 2 mg/ml.
Conclusion
Aquaculture study on fish cell lines was carried out to confirm and estimate the toxicity of engineered AgNPs. The fish cell lines were exposed to non-monodispersed AgNPs with size in the range of 5–100 nm and the viability of cell was tested using the Alamar Blue (AB) and Neutral Red (NR) Assays. Two conditions were considered; in the first, the amount of silver added to the cells was ranged up from 0.5–2.5 mg/ml, and in second case, the replicate was generated and has a concentration in the range of 0.4–2.4 mg/ml. In both essays, AgNPs shows high toxicity at low particle concentrations of 0.5 and 0.4 mg/ml. The healthy looking cells were maintained a reducing state within their cytosol. So in comparison, at low concentration of 0.4 and 0.5 mg/ml, the cells viability was 18% and 90% respectively; the toxicity of AgNPs was higher at the lower concentration of 0.4 compared to 0.5 mg/ml. With the increased concentration of AgNPs, toxicity was decreased; showing more than 100% cell viability. The percentage of neutral red dye absorbed at a 0.5 mg/ml concentration was about 3% and that of the Neutral Red dye absorbed at concentration 0.4 mg/ml is about 18%.
Studied showed that nanotechnology related toxicological studies provide basis for the human health, aquatic animals and environment. By the expansion of the experimental data sets, test compounds can be accurately visualized on living systems to detect lethal edge with multiple toxicity to screen out the results. The Alamar Blue Assay provides an accurate time course measurement. However, in addition to the standard tests, there is need further to develop better and rapid model systems to superiorly judge the toxicity of a nanomaterial on fish cell line. Nanotechnology provides a good platform to overcome the problem of resistance with the help of engineered AgNPs.
Footnotes
Acknowledgment
The authors are indebted to FOCAS (Nanao lab) of DIT, Ireland, for providing the lab facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by TUD Fiosraigh Scholarship, Ireland.
