Abstract
We investigated the effects of 7-day supplementation with three different dosages of citrulline nitrate (CN) on blood pressure at rest and after exercise, biochemical safety markers, and self-reported outcome measures of adverse events in healthy men. 12 apparently healthy young men (age 25.9 ± 4.0 years; weight 78.6 ± 10.0 kg, height 181.0 ± 7.0 cm) volunteered to participate in this double-blind, randomized, placebo-controlled cross-over trial. The dosages of CN were 1.5 g per day (low dose), 3.0 g per day (medium dose), and 6.0 g per day (high dose). No significant differences were found for systolic and diastolic blood pressure and heart rate at rest and after exercise between varying doses of CN and placebo (p > 0.05). In addition, hematological indices, biochemical variables, and clinical enzyme profiles were not affected by either intervention (p > 0.05), and the type and frequency of side effects were comparable to the placebo group. Citrulline nitrate was safe and well tolerated when administered for 7 days in dosages up to 6 g per day.
Introduction
Several plant-based and chemically synthesized vasoactive nutraceuticals have been identified during the past decade or so,1,2 with nitrate-containing foods and citrulline supplements often recognized for their notable role in regulating vascular tone.3-5 Citrulline nitrate (C6H14N4O6) recently emerges as a novel chemical mixture that combines two vasoactive components into one molecule, suggesting a summative effect of two components on vascular function via both nitric oxide synthase (NOS)-dependent and NOS-independent pathways. 6 Theoretically, this vasoactive amalgam could adversely affect vasodilation leading to a clinically relevant drop in blood pressure accompanied by headache, dizziness, fainting and other signs, and symptoms of hypotension. In this randomized placebo-controlled pilot trial, we examined the effects of 7-day supplementation with three different dosages of citrulline nitrate on blood pressure at rest and after exercise, biochemical safety markers, and self-reported outcome measures of safety events in healthy men in order to confirm that the dosages tested would be safe and well tolerated and that no adverse effects on blood pressure would be produced.
Methods
Subjects
Twelve apparently healthy young men (age 25.9 ± 4.0 years, weight 78.6 ± 10.0 kg, height 181.0 ± 7.0 cm) volunteered to participate in this double-blind, randomized, placebo-controlled cross-over trial that was conducted in the Applied Bioenergetics Lab at the University of Novi Sad from July to October 2020. The trial was conducted in compliance with the ethical standards laid down in the Declaration of Helsinki (7th revision) and the International Conference of Harmonization Efficacy Guidelines E6. All procedures involving human subjects were approved by the local IRB (# 401-TLI/2020). The sample size was calculated using G*Power 3.1 software, with the effects size set at 0.50, alpha error probability at 0.05, and power was 0.80, with four groups and five measurements. The primary outcome was the change in seated blood pressure (BP) from baseline to 7-day follow-up, while the secondary outcome was the change in standing BP following exercise from baseline to 7-day post-administration. Other outcomes included biochemical safety markers and self-reported outcome measures of safety events. The inclusion criteria for participants were age ≥18 years, body mass index from 18.9 to 29.9 kg/m2, no major chronic diseases or acute disorders (including all conditions with unstable BP), and resting BP < 160 mmHg systolic and/or <100 mmHg diastolic. Exclusion criteria were the use of dietary supplements within 30 days before the study commences (e.g., citrulline- or nitrate-containing products), the use of medications for erectile dysfunction within 4 weeks and/or pressure-lowering medications within 3 months before the study commences, and the unwillingness to return for follow-up analyses.
Experimental protocol
The volunteers were allocated to receive three different regimens of citrulline nitrate supplementation and placebo in a cross-over design, with each intervention administered for 7 days. The dosages of citrulline nitrate were 1.5 g per day (low dose), 3.0 g per day (medium dose), and 6.0 g per day (high dose), with the compounds supplied by ThermoLife International LLC (Phoenix, AZ). The participants were asked to ingest an intervention powder stirred in lukewarm water (200 mL) in the morning after an overnight fast, with the participants considered compliant only if all doses were consumed. All interventions were similar in appearance, smell, and texture. A wash-out period of 7 days was pre-defined to exclude any residual effects of interventions across the study periods. Both participants and testing personnel were blinded to participants’ treatments. The participants were asked to maintain their usual lifestyle (including diet and physical activity) and to abstain from using other dietary supplements or medication during the trial. The outcomes were assessed at baseline (pre-intervention) and after each 7-day regimen, with participants having visited the laboratory five times. All measurements were taken between 08:00 and 12:00 after at least 12 h of fasting. The venous blood was drawn from a peripheral antecubital vein and centrifuged immediately at 3000 g, with serum separated and analyzed for biochemical variables (see below) by an automated analyzer (RX Daytona, Randox Laboratories Ltd., Crumlin, UK). After blood sampling, resting blood pressure and heart rate were measured after 5 min of seated rest with an automated system (OMRON Hem 907XL IntelliSense, Kyoto, Japan). Following this, the participants were subjected to a 10-min treadmill walk at 6 km/h (Quasar, h/p/cosmos, Nussdorf-Traunstein, Germany), with post-exercise blood pressure measured immediately after exercise cessation while the participant rested in an upright position. Finally, the participants were asked to report any adverse effects (e.g., lightheadedness, fainting, palpitations, gut disturbances, headache) of either intervention through an open-ended questionnaire.
Statistical analyses
Initially, data were tested with the Shapiro–Wilk test for the normality of distribution and with Bartlett’s test for the homogeneity of the variances. When homogenous variances were verified for normally distributed data, summary measures were compared by one-way analysis of variance (ANOVA), with post hoc Tukey HSD test employed to identify the differences between individual interventions. When non-homogenous variances were identified, data were compared using the independent samples Kruskal–Wallis test, with Games-Howell post hoc test used to evaluate differences between individual interventions. The significance level was set at p < 0.05. The data were analyzed using the statistical package SPSS version 24.0 for Mac (IBM SPSS Statistics, Chicago, IL).
Results
A total of 12 participants completed the follow-up measures for all experimental regimens, with the adherence determined to be 100.0%, as calculated by the sachet count (336/336). The volunteers reported no major side effects that precluded their participation in the study. However, three out of 12 participants (25.0%) taking the highest dose of citrulline nitrate reported a single or multiple episodes of mild dizziness when standing up after sitting or lying down; this appeared at the beginning of the intervention, with no further symptoms after 2–3 days of treatment. In addition, seven participants (58.3%) reported an unpleasantly sour taste of high-dose citrulline nitrate. No self-reported side effects were recorded in low-dose and medium-dose citrulline nitrate, and placebo trials.
Changes in biochemical safety markers during the study (n = 12). Values are mean ± SD.
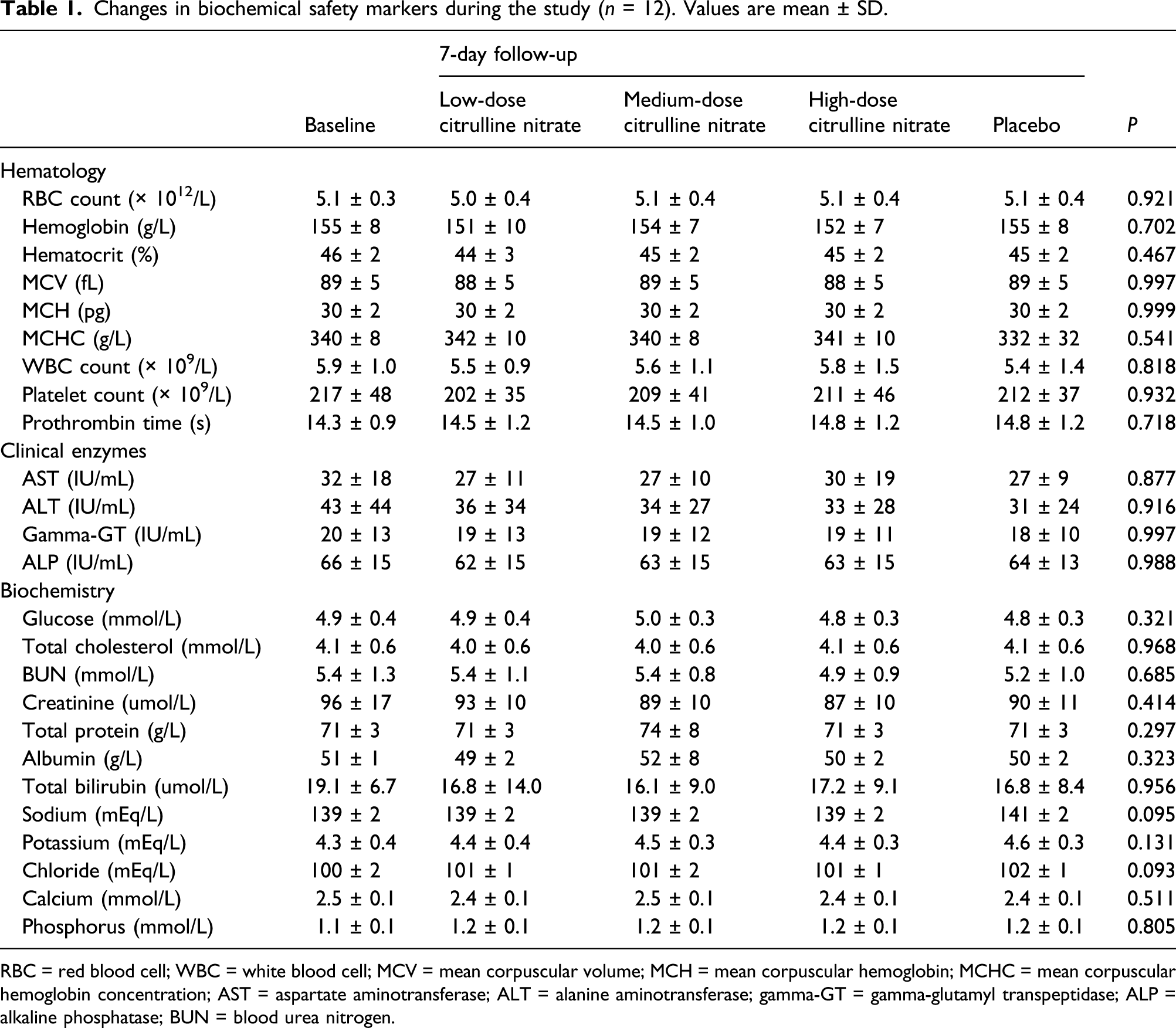
RBC = red blood cell; WBC = white blood cell; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; AST = aspartate aminotransferase; ALT = alanine aminotransferase; gamma-GT = gamma-glutamyl transpeptidase; ALP = alkaline phosphatase; BUN = blood urea nitrogen.
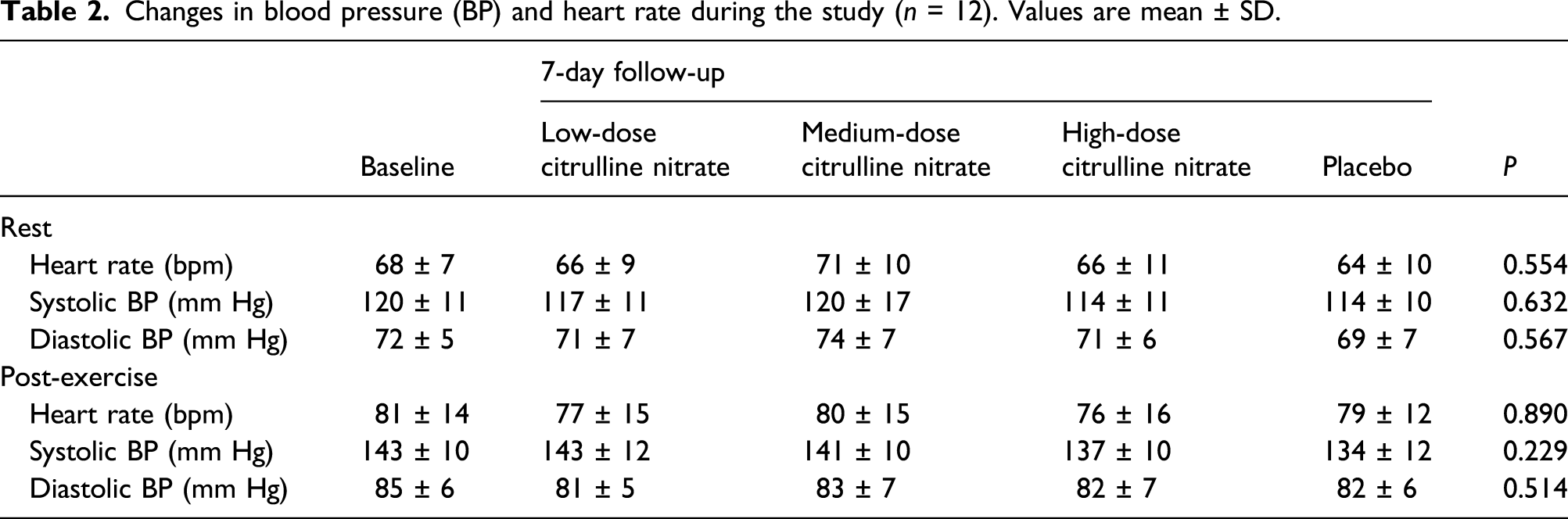
Changes in blood pressure (BP) and heart rate during the study (n = 12). Values are mean ± SD.
Discussion
This pilot trial demonstrated no statistically significant effect of short-term citrulline nitrate supplementation on resting and post-exercise BP and biochemical safety markers or self-reported side effects in healthy young men. Although there were reports from 3 out of 12 subjects in the high dose group of single or multiple episodes of mild dizziness, these symptoms were not noted after 2–3 days of treatment and there were no adverse effects seen on blood pressure.
Nitrate and nitrate-derived nitrite from various nutritional sources are precursors for nitric oxide synthesis exhibiting various effects on vascular function, including improved endothelial function, reduced arterial stiffness, and stimulated smooth muscle relaxation. 5 Besides its use in supporting cardiovascular disease therapies, vasoactive dietary compounds are often used by athletes and active individuals to improve training physiology. 4 For example, supplementation with citrulline salt improved maximal strength and anaerobic power in professional master athletes, 7 perhaps by mechanisms that involve vasodilation and increased skeletal muscle blood flow. 8 It has been reported that vasoactive dietary supplements can reduce BP in hypertensive patients 9 and normotensive young men. 10 While this could represent an effective intervention capable of reducing BP and improving cardiovascular health in a specific population, a dietary nitrates-mediated arterial hypotension might be an unfavorable side effect. 11
In this study, we found that citrulline nitrate, a novel vasoactive compound, shows no hypotensive effect when administered for up to 6 g/day during 7 days in healthy normotensive and pre-hypertensive young men. Although the combination is made of two active components that could affect BP via both nitric oxide synthase (NOS)-dependent and NOS-independent pathways, 6 no clinically relevant changes in blood pressure at rest and after exercise were recorded throughout each trial with citrulline nitrate. Also, no notable symptoms of low blood pressure were registered, except for transitory dizziness reported by 3 out of 12 participants who consumed the highest dose of the compound. Citrulline nitrate intake affected no safety biomarkers, with hematological indices, biochemical variables, and clinical enzymes remain essentially equivalent to pre-intervention values. These preliminary findings advance citrulline nitrate as a relatively safe dietary additive in healthy young men, with the risk of side effects similar to placebo.
We reported here that citrulline nitrate was well tolerated in terms of blood pressure fluctuations, yet several limitations should be considered when our findings are interpreted. Our sample was relatively small and consisted of normotensive and pre-hypertensive young healthy men, so whether citrulline nitrate differently affects other populations remain currently unknown. Second, we employed here a rather short-term protocol for citrulline nitrate intake (e.g., 7 days) and limited maximal dosage to 6 g per day; longer trials and escalating dosages above those used in our study are warranted to further assess its performance and safety. Further studies with citrulline nitrate should also extend safety profile evaluation and perhaps include additional biomarkers of cardiovascular safety, including the possible risk of fluid retention, accelerated thrombogenesis, and asymptomatic arrhythmogenesis. For instance, the U.S. Food and Drug Administration (FDA) Pharmacy Compounding Advisory Committee overviewed non-clinical and clinical safety of L-citrulline or L-citrulline preparations 12 and documented various adverse events through both the FDA and the Center for Food Safety and Applied Nutrition reporting systems. The Committee concluded that limited safety data are available at the moment, yet decades of citrulline use has not been associated with major safety concerns. Still, additional longitudinal and well-powered toxicologic trials with citrulline nitrate should corroborate safety of this novel nutritional compound.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study was partially supported by the Serbian Ministry of Education, Science and Technological Development (# 175037), and the ThermoLife International LLC. The funders played no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
