Abstract
The major factors (macro) which cause human cancer have been elucidated and include tobacco use, diet, infection, reproductive and sexual behavior and, to a lesser extent, alcohol consumption and occupational factors. Several reports have been published about endogenous chemicals made in humans which produce DNA adducts; however, few have linked them to possible carcinogenic activity. This paper discussed four chemicals made in humans (formaldehyde, acetaldehyde, isoprene and ethylene oxide), pathways of their formation, their animal carcinogenicity and questions about these and other endogenous chemicals’ possible role in human cancer. In addition, the author posits a simplified formula for development of cancer and a formula for causing mutations by various agents.
Introduction
The reports of Wynder and Gori, 1 Higginson and Muir 2 and of Doll and Peto 3 on cancer causation have been strengthened by research during the last three decades. These reports stated that the major factors (macro) which are the cause of cancer are tobacco use, diet, reproductive and sexual behavior, infection and, to a lesser extent, alcohol consumption and occupational factors. A recent review and synthetic analysis found that about one in six cancers worldwide were caused by various viruses, bacteria and parasites. 4
There are few reports about chemicals endogenously produced by humans, which might be carcinogens, but several reports on endogenous DNA adducts resulting from oxidative stress, lipid peroxidation, inflammation, some estrogen metabolites 5 –7 as well as from formaldehyde and ethylene oxide. 8
This commentary discusses four chemicals endogenously made in humans which are reported animal carcinogens. The endogenous formation of these carcinogens raises questions on their role, if any, in human cancer, and their importance in risk assessment. In addition, based on reported causes of cancer, theories of cancer 9 –11 and experimental cancer causation in non-human primates 12 –14 the author proposes a formula for the development of cancer and for agents to cause mutation resulting in a mutated cell that eventually leads to cancer.
Only a few animal chemical carcinogens have been reported to be endogenously produced by humans. However, it is possible that there are many more endogenous chemicals formed in humans that are known animal carcinogens than currently recognized. The endogenous production in humans of four compounds discussed in this article have been reported in the peer reviewed literature. Table 1 lists the four chemicals produced in humans and their carcinogenic evidence in animals and humans as reported by the International Agency for Research on Cancer (IARC) in their series of monographs. 15
Endogenous chemical carcinogens in humans.
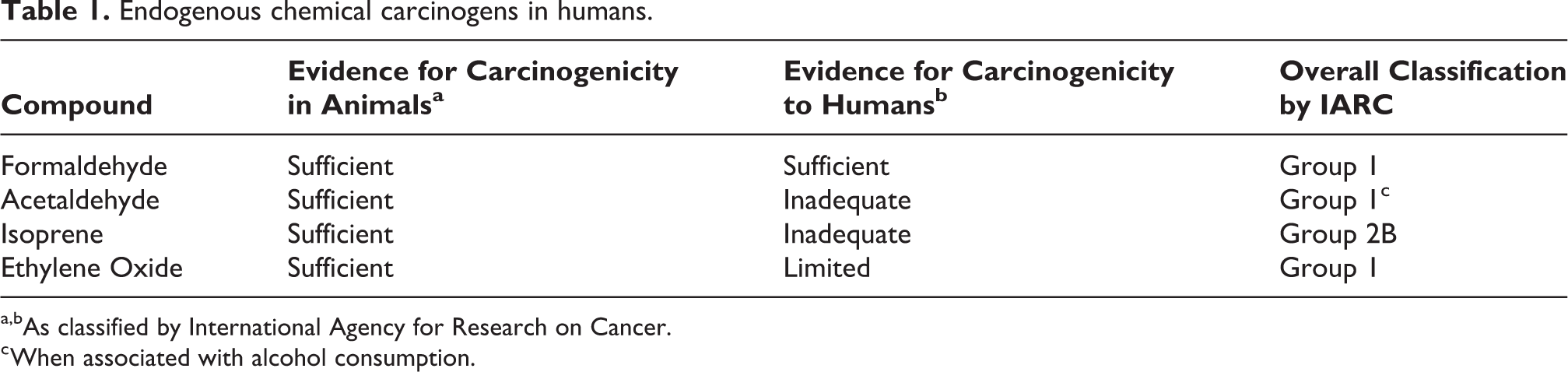
a,bAs classified by International Agency for Research on Cancer.
c When associated with alcohol consumption.
Chemicals and results
Formaldehyde
Formaldehyde is a ubiquitous chemical. It occurs naturally in foods, including fruits, vegetables and meat. It is used widely in industry and occurs in cigarette smoke. IARC reported there is sufficient evidence in experimental animals for carcinogenicity based on the induction of squamous cell carcinomas in rats and sufficient evidence for carcinogenicity in humans based on nasopharyngeal cancer and leukemia. Three mechanisms focused on genotoxicity that had moderate support were discussed for induction of hematological malignancies in humans from formaldehyde. 16 However, formaldehyde is also formed endogenously by humans as well as animals. Formaldehyde is an essential intermediate in the biosynthesis of various amino acids and purines and is formed from the metabolism of some amino acids, i.e. glycine, methionine and serine. 17 It has also been reported in a mouse model that formaldehyde is an important source of endogenous DNA damage that is counteracted in mammals by a conserved protection mechanism. 18
The amount of formaldehyde in human blood is reported to be 2.6 µg/g and is tightly regulated. Even exposure by inhalation to formaldehyde does not change the pre-exposure blood concentration, likely due to rapid metabolism by dehydrogenases in human erythrocytes. 19 –21 Although humans differ with respect to their blood formaldehyde concentration, it is unknown how much this variation is due to genetic, dietary, age or life style factors. Formaldehyde can induce DNA adducts which occur on the amino groups, participating in Watson-Crick base pairing and can be considered premutagenic. 8 Formaldehyde causes mutations in a variety of test systems, and also in laboratory animals. The formation of formaldehyde-DNA adducts has been reported in human leukocytes. 22 The authors reported N6-hydroxymethyldeoxyadenosine in leukocyte DNA samples of both smokers and non-smokers, although the level was higher and more frequent in smokers. 22 The amount of endogenous turnover of formaldehyde is estimated to be 878–1310 mg/kg bw per day assuming a half-life of 1–1.5 minutes. 23
It would be of interest to evaluate formaldehyde-DNA adducts in patients recently diagnosed with myeloid leukemia, both those with known exposure to formaldehyde and those with no known risk factors. The adduct reported by Wang et al. 22 might be a logical starting adduct to evaluate.
Acetaldehyde
Acetaldehyde was found to cause gene mutations in bacterial cells and mammalian cells and DNA damage in vitro in cultured mammalian cells. It was tested for carcinogenic activity following inhalation exposure in hamsters and rats. In hamsters, it produced laryngeal cancer and in rats, adenocarcinomas and squamous cell carcinomas of the nasal mucosa. 24 IARC stated there was sufficient evidence for carcinogenicity in experimental animals and possibly carcinogenic to humans. In humans, L-serine hydroxymethyl transferase converts threonine to glycine and acetaldehyde. Threonine aldolase, found in the liver and kidneys, can degrade threonine to acetaldehyde and glycine. 17 Since ethanol can be detected in the exhaled breath of mice, one can speculate that the intestinal flora of humans might produce ethanol. 25 As the major metabolite of ethanol is acetaldehyde, this is a potential additional source of acetaldehyde in humans not only following ethanol consumption but also even in the absence of ethanol intake.
Isoprene
Isoprene is produced naturally by many plant species, occurs in tobacco smoke, and it occurs from industrial pollution. Isoprene is the basic structural unit of several important compounds, including natural rubber, various vitamins and the steroid sex hormones. 26
Occupational exposure may occur from production of the monomers and from synthetic rubber.
Isoprene also occurs in human blood and human breath in quantities estimated to be as high as 4 mg over a 24-hour period. 27 –29 Isoprene in humans, especially in breath, may be the product of mevalonate metabolism and is possibly linked to cholesterol metabolism. 30 Isoprene, by inhalation, has been reported by the National Toxicology Program to cause several types of tumors in mice and rats. The tumors in rats were mammary gland fibroadenomas and carcinomas, renal tubular adenomas and testicular interstitial cell adenomas. IARC reported isoprene demonstrated sufficient evidence of carcinogenicity in experimental animals. Due to a lack of a biomarker of exposure to isoprene, there has been a paucity of molecular studies on this endogenous compound.
The author suggests it would be of interest to evaluate the amount of expired isoprene in non-smokers recently diagnosed with adenocarcinoma with no known risk factors compared to non-cancer matched age/gender patients.
Ethylene oxide
Ethylene Oxide is used in the chemical industry to produce a variety of substances and also it is used
IARC classifies ethylene oxide as a Group 1 substance (carcinogenic in humans) with sufficient evidence of carcinogenicity in experimental animals and limited evidence in humans (Table 1). Also, for several years, it has been known that the conversion of ethylene to ethylene oxide produces 2-hydroxethylation of DNA to form background levels of N7-(2-hydroxyethyl) guanine in unexposed humans. 33,34
Discussion of endogenous carcinogens
This commentary discusses four chemical compounds which are formed in humans and which are known to cause cancer in animals. In addition, one is reported by IARC as carcinogenic in humans (formaldehyde), and acetaldehyde, ethylene oxide and isoprene are reported as limited or inadequate evidence for human carcinogenicity. These four (formaldehyde, acetaldehyde, isoprene and ethylene oxide) as well as other endogenous DNA-damaging agents 6 are produced as a consequence of normal biochemical reactions. Among the questions that arise about potential endogenous carcinogens in humans are how tightly are they regulated and if they mutate normal cells? How many other potential endogenous chemical carcinogens are waiting to be identified? Can these compounds contribute to cancer causation because of faulty metabolism or genes or aberrant repair and are the endogenous chemical compounds and their concentration taken into account during risk assessment, especially linear risk extrapolation?
An important paper which demonstrated the difference between DNA adducts formed by exogenous and endogenous formaldehyde was published 35 and highlighted in a commentary. 36 That commentary entitled “Paracelsus and Formaldehyde 2010: The Dose to the Target Organ Makes the Poison” could be paraphrased to “the dose to the target organ induces the cancer.” Thus, it is not just the agent and the dose of the agent, but the dose at the site which initiates the cancer and/or promotion of the induced cell. If the endogenous carcinogens are rapidly used in normal metabolic reactions, then it is less likely they would cause mutations, but if there is abnormal metabolism due to diet, inflammation, repair enzymes that are unsuccessful or other factors or excess chronic production, then they could cause mutations, if not repaired would eventually lead to cancer. Also, the endogenous carcinogen must get to the site where it will cause the mutation and other cellular damage. The example of formaldehyde has previously been discussed. Whether endogenous chemicals in humans contributes/causes cancer needs to be thoroughly investigated.
Nevertheless, the current theories of cancer are based on the somatic mutation, clonal expansion and promotion hypothesis and a focus on genes that regulate cell growth and differentiation. 9,10,37 More recently, it has been suggested that inflammation can co-initiate and amplify activated stem cells. 11 The induction of cancer experimentally involves a multistep process involving cellular genes (initiation), clonal expansion, promotion, progression and then cancer. 38 –41 This multistep process has recently been elaborated on in a review. 42 Studies on induction of cancer in non-human primates for three decades with various chemicals has also demonstrated a dose–response, time to tumor, a possible threshold (especially to the carcinogen diethylnitrosamine) and the ability to predict that a specific non-human primate will develop cancer, from the alpha-fetoprotein level, even when microscopic examination of the organ that will be affected (liver) is without gross evidence or microscopic tumor detection and administration of the chemical carcinogen ceases. 12 –14 Of course, the number of non-human primates used per dose was too small to make a definite conclusion.
An algebraic cancer equation has been proposed to explain how cancer may arise with normal mutation rates. 43 and thus far 291 cancer genes have been reported. 44 In addition, a recent study reported that 1 to 10 mutations are needed to drive cancer varying across different cancer types. 45 However, a formula of minimal complexity is herein suggested.
Thus, the current theories of cancer causation and studies on cancer genes as well as non-human primate experiments allows the author to posit a formula for cancer development and for mutation.
The formula is uncomplicated and primarily applicable to experimental cancers, especially those involving the induction of cancer by DNA adduct/mutation carcinogens. It may also apply to development of experimental cancers that involve the two-stage system, i.e. a single application of a direct acting carcinogen and followed by a promoting agent. 46 Of course, this would necessitate the addition of a promotion variable (P). The proposed equation is not generalizable to all cancer development. Such a mathematical equation would be more complex and involve additional variables, including, but not limited to variables for different repair systems involving different cancer types and for immune-surveillance.
A formula for the development of cancer and agents that cause cancer
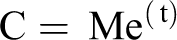
Cancer development (C) is the result of a cell with a minimum of two, and likely more mutated cellular growth and/or tumor suppressor genes (M) resulting in a mutated cell with abnormal growth, then clonal expansion (e) of the mutated cell and with time (t) promotion and progression to the tumor. Additional mutations of growth controlling genes may occur during periods of clonal expansion. The time (t) will be dependent on the species and organ involved, the number and type of mutations and the cell turnover rate. Also, one cannot exclude changes in DNA methylation which may result in increased expression of oncogenes or silencing of tumor suppressor genes.
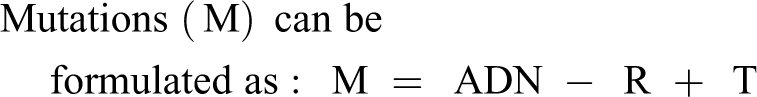
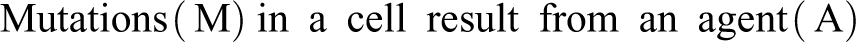
The dose (D) of the agent is important as is the number (N) of times the dose occurs, minus repair (R) and time (T).
The agent (A) can be a variety of factors including UV or gamma radiation, oxidative stress, various chemicals, cell proliferators, some viruses, and for some other potential cancer-causing agents.
The dose (D) of the agent that is important is that which gets to the critical site causing mutations. Also, of importance, is the number (N) of times the dose exposes the site in the cell to the agent.
The repair (R) can be one of several types including but not limited to DNA excision repair, nucleotide repair, nucleotide excision repair, double strand break repair and DNA mismatch repair.
Experimentally, the equation for cancer might be solved for example using diethylnitrosamine dosing in non-human primates. One can determine the mutation frequency in several growth genes since the dose, and the number of doses will be known. The time to tumor will be known. Also, it can be predicted which of the non-human primates will develop cancer from the amount of alpha-fetoprotein in the serum. Clinically, the equation might possibly be solved using retrospective information from dose exposure to a known therapeutic agent (one having known carcinogenic potential), genomic analysis and time to diagnosis of known cancer.
Summary
The major macro factors which are the cause of human cancer have been elucidated including tobacco use, diet, reproductive and sexual behavior, infection and, to a lesser degree, alcohol and occupational factors. There have been several reports on endogenous DNA adducts formed in humans from chemicals which are known animal carcinogens.
This paper discusses four chemicals made in humans (formaldehyde, acetaldehyde, isoprene and ethylene oxide) that are known animal carcinogens. Reasons for their formation and questions about their potential to cause mutations and cancer in humans are discussed. The question of whether their occurrence is factored into account during risk assessment is asked.
A simplified formula for the development of experimental cancer is proposed:
C= Me(t); cancer is the result of a cell with mutated (M) cancer growth or inhibitor genes, clonal expansion (e) of the mutated cell and time (t) for progression and detection.
Also, it is theorized that human cancer risk assessment might be refined by understanding the role of endogenous chemicals formed in humans which are known animal carcinogens.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
