Abstract
Exposure to chemicals produced by natural processes is ubiquitous. First, in addition to the products of normal metabolism produced in humans of normal body weight, adipose tissue produces a large number of chemicals, including estrogen, testosterone from the produced estrogen, thyroid-stimulating hormone, leptin and approximately 500 other molecules termed adipokines, and a large number of inflammatory mediators. Second, the gut biome contains approximately the same number of bacteria as cells found in the entire body and produces a large number of small molecules. Third, the overwhelming majority (99.9%) of pesticide exposure occurs during ingestion of natural plant pesticides from eating vegetables. Fourth, consumption of cooked muscles meats leads to significant exposure to mutagenic and carcinogenic heterocyclic amines, polycyclic aromatic amines, and nitropyrenes. Fifth, many common beverages, for example, beer, coffee, and tea contain organic chemicals that display mutagenic activity. As compared with man-made production levels, from 1945 to 2015, an estimated 5000-fold more organic compounds were produced by a variety of natural processes, including common wood-degrading and forest litter-degrading fungi, microorganisms in temperate and boreal forest soils, bacteria in marine sponges, marine macro-algae, volcanoes, and forest fires. Exposure to these naturally produced organic compounds occurs via inhalation of ambient air, ingestion of food and water, and contact with soil, freshwater, and seawater. Contact with several thousand different endogenous or exogenous chemicals per day is unavoidable. This understanding might assist in better allocating resources toward controlling exposures to agents of highest concern as determined by current concepts of chronic disease causation.
Introduction
The National Library of Medicine (NLM) issues collection development guidelines for each of the various fields of study related to biomedicine. 1 The NLM describes the focus of the field of toxicology as follows: “Toxicology is traditionally associated with chemical exposures, such as the effects of drugs, industrial chemicals, pesticides, food additives, household products, and personal care items.” The Swiss physician Paracelsus (1493–1541) is credited with the first expression of the opinion that the dose was the most important factor related to the toxicity of a substance. 2 Since the first exposition of the dose–response concept by Paracelsus, the dose–response relationship has become a central concept in toxicology. 2
Meeting the technical challenges inherent in designing and executing experiments on a particular chemical agent and in interpreting the results of the successfully completed experiment requires significant focus that generally occupies the attention of the investigators. In the 1970s, Bruce N Ames and his colleagues began a line of investigation that ultimately resulted in broadening the understanding of the effects of exposure to chemicals capable of producing mutations in in vitro assays 3,4 and tumors in chronic rodent bioassays. 5 A major and quite unexpected finding reported by Ames et al. in 1990 was that 99.99% (by weight) of the pesticides ingested in the American diet are chemicals produced by plants to prevent being eaten by insects or higher animals. 6 Also in 1990, Duke at the US Department of Agriculture Research Service in Oxford, Mississippi, 7 provided strong support for the contention made by Ames et al. 6 Duke reported that tens of thousands of chemicals have been identified in plants and that experts estimate the existence of hundreds of thousands of natural plant products. In addition, most of the tens of thousands of plant pesticides identified to date are natural plant pesticides. 7
The realization that humans were ingesting about 1.5 g per day of a huge variety of natural plant pesticides 6 raised the question as to whether these chemicals were toxic in standard toxicology tests. Up to 1990, 52 natural plant pesticides had been tested in chronic rodent bioassays with 27/52 (52%) displaying carcinogenicity. This carcinogenic percentage is comparable to that seen for synthetic chemicals. 6
As research on chemical products produced by the gut biome and adipose tissue accumulated, it became apparent that the natural pesticide case was not unique and that humans are exposed daily to small concentrations of an extremely large number of chemicals produced by natural processes. The background of natural chemical exposures has several possible implications for hazard and risk assessment. First, human exposure to a toxic synthetic chemical does not occur in vacuo. Factors like inflammatory cytokines released by excess adipose tissue (see “Exposure to chemicals produced by adipose tissue” section) or imbalances in the gut biome (see “Exposure to chemicals produced by gut bacteria” section) can increase risks as compared with normal-weight individuals or those with properly balanced gut bacterial colonization. Alternatively, in some cases, the contribution to hazard from the synthetic chemical might be less than that made by structurally similar chemicals of natural origin. In relation to a particular human health endpoint, the combined exposure from both synthetic and natural sources should be considered. The purpose of this review is to demonstrate that contact with several thousand different endogenous or exogenous chemicals per day is unavoidable. This understanding might assist in better allocating resources toward controlling exposures to agents of highest concern as determined by current concepts of chronic disease causation.
Exposure to chemicals produced by adipose tissue
Obesity has risen rapidly both worldwide and in the United States (US) over the last several decades. In 2014, it was estimated that more than 1.9 billion adults were overweight with over 600 million classifiable as obese. 8 Approximately two-thirds of US adults over 20 years of age are currently classified as overweight with about 35% classified as obese. 9 The obesity rate in the US is predicted to reach 42% by 2030 in people over 18 years of age. 10
Adipose tissue is composed of a number of different types of secretory cells, including adipocytes, precursor cells, endothelial cells, macrophages, foam cells, neutrophils, lymphocytes, fibroblasts, and other cells. 11 The range of biological activities, structural diversity, and sheer number of different molecules secreted by adipose tissue is enormous. Molecules secreted by adipose tissue include the following: (a) cytokine and cytokine-like proteins, including tumor necrosis factor alpha (TNF-α), interleukin 6 (IL-6), monocyte chemotactic factor 1, resistin, and progranulin; (b) proteins of the fibrinolytic system, including plasminogen activator inhibitor (PAI-1) and tissue factor; (c) complement and complement-related proteins, including adipsin, complement factor B, acylating simulation protein, and C1q/TNF-related proteins; (d) enzymes including dipeptidyl peptidase-4; (e) lipid transport molecules, including apolipoprotein E, cholesterol ester transfer protein, and lipoprotein lipase; (f) endocannabinoids and other lipids, including anandamide, 2-arachidonoylglycerol (2-AG), and free fatty acids; and (g) proteins of the renin–angiotensin system including angiotensinogen. 11
The class of molecules secreted by fat cells, termed adipokines, is sufficiently complex and important that it merits separate discussion. Approximately 500 adipokines have been discovered to date with the following examples demonstrating the extremely wide range of biochemical and physiological reactions reported in the literature: leptin, discovered in 1994, regulates appetite; adiponectin, discovered in 1995, enhances insulin sensitivity and lessens inflammation; ADAMTS1, discovered in 1997, affects fat stem cell differentiation, blood vessel formation, and ovulation; chemerin, discovered in 1997, increases inflammation and blood pressure; resistin, discovered in 2001, mediates insulin resistance; retinol-binding protein 4, discovered in 2005, affects insulin resistance; lipocalin-2, discovered in 2007, increases insulin resistance and inflammation; isthmin-1, discovered in 2014, improves fat metabolism in the liver, mediates immune function, and influences embryonic developmental patterning; asprosin, discovered in 2016, modulates glucose release from the liver; Slit2-C, discovered in 2016, stimulates glucose metabolism; and lipocalin-5, discovered in 2018, improves skeletal muscle respiration. 11,12
Adipose tissue is very hormonally active as demonstrated by possessing a large number of different receptors for traditional endocrine hormones 13 including the following: insulin receptor, glucagon receptor, growth hormone receptor, thyroid-stimulating hormone (TSH) receptor, gastrin/cholecystokinin B receptor (CCK-B), glucagon-like peptide-1 receptor, and angiotensin II receptors type 1 and 2. Adipose tissue possesses enzymes capable of activating, interconverting, and inactivating steroid hormones. 14,15 A number of different enzymes that produce steroids are expressed in adipose tissue, including cytochrome P450-dependent aromatase, 3-hydroxysteroid dehydrogenase (3HSD), 11HSD1, 17HSD, 7-hydroxylase, 17-hydroxylase, 5-reductase, and UDP-glucuronosyltransferase 2B15. 14,15
The large mass of adipose tissue leads to significant hormonal release with adipose tissue contributing up to 100% of circulating estrogen in postmenopausal women and 50% of circulating testosterone in premenopausal women. 14,15 Stromal cells and pre-adipocytes in adipose tissue highly express cytochrome P450-dependent aromatase and 17HSD. The aforementioned aromatase influences the conversion of androgens to estrogens, for example, androstenedione to estrone and testosterone to estradiol. The adipose tissue enzyme 17HSD regulates the conversion of the weak androgen and rostenedione to the strong androgen testosterone and concomitantly the conversion of the weak estrogen estrone to the strong estrogen estradiol. In subcutaneous adipose tissue, the expression of 17HSD is decreased relative to the cytochrome P450-dependent aromatase. In contrast, in visceral adipose tissue, the expression of 17HSD is increased relative to the cytochrome P450-dependent aromatase. 14,15 Deposition of body fat to the abdominal region in the pattern typically observed in males and postmenopausal females is associated with this increase in 17HSD expression relative to aromatase expression thereby implicating increased local androgen production in visceral adipose tissue. 14,15 These human data and supportive mechanistic data in mice demonstrate that adipose tissue is an important site for both metabolism and secretion of sex steroids. 16 –19
Estradiol is of particular importance due to its role in increasing risk for development of breast, endometrial, and ovarian cancers. 20 Estradiol is the most potent of the naturally occurring estrogens. 21 In postmenopausal women, estradiol is mainly produced in the mesenchymal cells of adipose tissue but also by osteoblasts and chrondrocytes of bone, vascular endothelium and aortic smooth muscle cells, and several sites in the brain. 22 This locally produced estradiol is the main driver of estrogenic action in postmenopausal women, as opposed to circulating estrogen levels. Therefore, locally produced rather than circulating estrogens play an important role in breast cancer development, bone mineral maintenance, and preservation of cognition. 22
The relationship between thyroid hormones and body weight is complex. 23 Basal metabolic rate (BMR) is decreased in patients suffering from hypothyroidism, and an underactive thyroid is usually associated with an average weight gain of 5–10 pounds. Most of this weight gain is due to excess accumulation of salt and water. Very large increases in body weight are usually not due to hypothyroidism. In contrast to a modest decrease in BMR seen in hypothyroidism, hyperthyroidism causes a larger increase in BMR with an associated loss of body weight. 23
In contrast to the observation that thyroid dysfunction can affect body weight, 23 several studies have demonstrated that obesity can conversely alter thyroid hormones. Karavani et al. 24 examined a large pediatric and adolescent database in Israel. The study subjects had either normal or slightly higher than normal TSH levels. 24 TSH is released by the anterior pituitary gland. Following TSH stimulation, thyroid follicular cells release thyroxine (T4) and to a lesser extent triiodothyronine (T3). 25 Karavani et al. 24 demonstrated that across normal weight, overweight, and obese groups T3 but not T4 increased in proportion with the TSH increase, supporting the hypothesis that TSH preferentially stimulates T3 rather than T4. As compared with the normal weight group, TSH and T3 levels were slightly higher in the overweight and obese groups. 24
Al-Musa correlated levels of serum thyroid hormones with body mass index (BMI) in 278 Saudi Arabian adults. 26 Slightly more than three-fourths of the study subjects were either overweight (31.3%) or obese (44.6%). Mean TSH serum levels showed a significant increase with increasing BMI, although T3 and T4 were not significantly related to BMI. In a Greek study on 736 euthyroid healthy subjects, Milionis and Milionis reported that TSH increased with BMI in women but not in men. 27 Bandurska-Stankiewicz has hypothesized that obesity-associated increases in TSH and peripheral hormone levels might be an adaption to increase energy expenditure thereby reducing further weight gain. 28
C-reactive protein (CRP) is a ring-shaped (annular) protein synthesized in the liver and composed of five subunits (pentameric). CRP plasma concentration increases in response to inflammation. 29 CRP production is stimulated by IL-6. 30 A large number of studies have reported elevations in CRP, 31 –34 IL-6, 35 –38 or both biomarkers of inflammation in association with obesity. 39 –42 Similar elevations in CRP levels are seen in active cigarette smokers. 43 –48
TNF-α is another important inflammatory mediator that is frequently elevated in association with obesity. 49 In obesity, macrophages invade the adipose tissue 49 and secrete TNF-α. 50 The degree of adiposity and insulin resistance correlates with the TNF-α level. 49,51,52 Anti-TNF-α antibody therapy is administered for a number of clinical conditions, including rheumatoid arthritis and Crohn’s disease. These therapies are associated with an increase in body fat suggesting that inflammation is caused by obesity in contrast to inflammation-inducing obesity. 53
In addition to systemic inflammation, obesity is associated with inflammatory changes in the brain. 54 The hypothalamus lies between the thalamus and the midbrain. Via regulation of the autonomic nervous system, the hypothalamus influences sleep cycles, body temperature, and appetite. 55 The original brain region where obesity-associated inflammation was observed was the hypothalamus. 56,57 Miller and Spencer 58 have reported that circulating cytokines, free fatty acids, and immune cells cross the blood brain barrier at the level of the hypothalamus. Once in the hypothalamus, these systemic factors induce local inflammation, including the proliferation of microglia. These authors 58 hypothesize that the localized inflammation remodels synapses and causes neurodegeneration within the hypothalamus thereby disrupting cognition in the hippocampus and amygdala. Recent research has demonstrated that obesity-related inflammation not only affects the hippocampus but also affects the brainstem, amygdala, and cortical structures. 59 Neuro-inflammation observed in these brain regions is associated with symptoms of depression and impaired cognition. 59
In conclusion, a majority of the adult population in the US is exposed to a biologically significant level of hormonal disruption and inflammation contributed by excess adipose tissue. Clinical epidemiology studies on the potential adverse effects of exposure to endocrine-disrupting chemicals should account for the large hormonal and inflammatory contribution made by excess adipose tissue.
Exposure to chemicals produced by gut bacteria
The healthy human gut usually contains between 300 and 1000 different species of bacteria, 60,61 with 30 to 40 species accounting for 99% of the total bacterial count. 62 The mass of bacteria residing in the gut of an average-sized (70 kg) man is estimated to weigh about 0.2 kg. 63 Sender et al. 63 estimate that the number of cells in an average man’s body and the number of bacteria in his gut are similar, that is, 3.0 × 1013 human cells and 3.8 × 1013 bacteria.
The huge number of bacteria in the human gut produces a large number of different chemicals.
64
Clostridial clusters IV and XIVa of firmicutes, including species of Eubacterium, Roseburia, Faecalibacterium, and Coprococcus produce short-chain fatty acids, including acetate, propionate, butyrate, isobutyrate, 2-methylpropionate, valerate, isovalerate, and hexanoate.
65
–68
Lactobacillus, Bifidobacteria, Enterobacter, Bacteroides, and Clostridium make bile acids, including cholate, hyocholate, deoxycholate, chenodeoxycholate, a-muricholate, b-muricholate, w-muricholate, taurocholate, glycocholate, taurochenoxycholate, glycochenodeoxycholate, taurocholate, tauro-a-muricholate, tauro-b-muricholate, lithocholate, ursodeoxycholate, hyodeoxycholate, glycodeoxylcholate, taurohyocholate, and taurodeoxylcholate.
69
–71
Faecalibacterium prausnitzii and Bifidobacterium produce choline metabolites, including methylamine, dimethylamine, trimethylamine, trimethylamine-N-oxide, dimethylglycine, and betaine.
72,73
Clostridium difficile, F. prausnitzii, Bifidobacterium, Subdoligranulum, and Lactobacillus synthesize phenolic, benzoyl, and phenyl derivatives and also make benzoic acid, hippuric acid, 2-hydroxyhippuric acid, 2-hydroxybenzoic acid, 3-hydroxyhippuric acid, 3-hydroxybenzoic acid, 4-hydroxybenzoic acid, 3-hydroxyphenylpropionate, 4-hydroxyphenylpropionate, 3-hydroxycinnamate, 4-methylphenol, tyrosine, phenylalanine, 4-cresol, 4-cresyl sulfate, 4-cresyl glucuronide, 4-hydroxyphenylacetate, 3,4-dihydroxyphenylacetate, phenylacetylglycine, phenylacetylglutamine, phenylacetylglycine, phenylacetate, phenylpropionate, phenylpropionylglycine, and cinnamoylglycine.
74,75
Clostridium sporogenes and E. coli produce indole derivatives including N-acetyltryptophan, indoleacetate, indoleacetylglycine, indole, indoxyl sulfate, indole-3-propionate, melatonin, melatonin 6-sulfate, serotonin, and 5-hydroxyindole.
76
–78
Vitamin K, vitamin B12, biotin, folate, thiamine, riboflavin, and pyridoxine are made by bifidobacterium.
79,80
Campylobacter jejuni and Clostridium saccharolyticum produce the polyamines putrescine, cadaverine, spermidine, and spermine.
81,82
Bifidobacterium, Roseburia, Lactobacillus, Klebsiella, Enterobacter, Citrobacter, and Clostridium manufacture lipids, including conjugated fatty acids, lipopolysaccharides (LPS), peptidoglycan, acylglycerols, sphingomyelin, cholesterol, phosphatidylcholines, phosphoethanolamines, and triglycerides.
83,84
In addition, Bacteroides, Pseudobutyrivibrio, Ruminococcus, Faecalibacterium, Subdoligranulum, Bifidobacterium, Atopobium, firmicutes, and Lactobacillus produce
Exposure to chemicals made by plants as natural pesticides
In 1990, Ames et al. reported that 99.9% of pesticide exposure experienced by humans comes from natural pesticides produced by plants in contrast to trace residues of synthetic pesticides. 6 This contention by Ames et al. 6 is supported by a comprehensive review by Duke. 7 Duke noted that tens of thousands of secondary products of plants have been identified. Plant-produced compounds and derivatives with herbicidal activity include the following: 1,8-cineole, cinmethylin, hypericin, and delta-aminolevulinic acid. 7 Plant-produced compounds with insecticidal activity include the following: camphene, nicotine, anabasine, and rotenone. 7 Plant-produced compounds with fungicidal nematicidal and rodenticidal activity include the following: pisatin, juglone, alpha-terthienyl, and strychnine. 7 It has been estimated that hundreds of thousands of these secondary plant products exist. The majority of this huge number of chemicals produced by plants “are involved in the interaction of plants with other species-primarily the defense of the plant from plant pests.” 7 Anyone who eats vegetables is exposed to a subset of these natural plant pesticides.
Exposure to potentially toxic chemicals in common foods and drinks
A very large number of chemicals of diverse structures reported as mutagens, rodent carcinogens, and endocrine disruptors are found in common foods and drinks. A number of different polycyclic aromatic hydrocarbons have been found in cooked fish, broiled hamburger, barley malt, puffed cereal, and other common foods. 87 –89 For example, benzo[a]pyrene and benz[a]anthracene are found in common foodstuffs, including fresh vegetables, vegetable oils, coconut oil, margarine, mayonnaise, coffee, tea, grain, oysters and mussels, smoked ham, smoked fish, smoked bonito, cooked sausage, singed meat, broiled meat, charcoal-broiled steak, broiled mackerel, barbecued beef, and barbecued ribs. 89
Several N-nitrosamines have been found in foods and beverages and reported on a ng nitrosamine/g food or beverage basis. The N-nitrosamines detected include N-nitrosodimethylamine, N-nitrosodiethylamine, N-nitrosopyrrolidine, and N-nitrosopiperidine. 89 Acetic acid, 2-methylnitrosamino)-[N-nitrososarcosine] was reported in meat 90 and beer (and malt) 91,92 ; 1-butanamine, N-butyl-N-nitroso-[N-nitrosodibutylamine] in fish 93 ; ethanamine, N-ethyl-N-nitroso-[N-nitrosodiethylamine] in bacon, 94,95 fish, 96 –98 cheese, 94,95 powdered milk, 99 beer (and malt), 100,101 gastric juices and nitrite 102,103 ; methanamine, N-methyl-N-nitroso-[N-nitrosodimethylamine] in meat and cured meat, 95,104 –109 bacon, 94,95,110,111 fish, 93 –98,112 –121 squid, 114,117 cheese, 94,95,107,108,122 –127 powdered milk, 99,128 –131 wheat flour, 132 beer (and malt), 100 –102,107,108,133 –152 Scotch whiskey, 134 French brandy, 135 other alcoholic beverages, 133,153,154 and water 155 –159 ; morpholine, 4-nitroso-[N-nitrosomorpholine] in fish 97,98 ; piperidine, 1-nitroso-[N-nitrosopiperidine] in meat and cured meat, 107,108 bacon, 94,95,110 fish, 94,95 squid, 102,117 and cheese 94,95 ; 1-propanamine, N-nitroso-N-propyl-[N-nitrosodipropylamine] in fish 93,96 ; pyrrolidine, N-nitroso-[N-nitrosopyrrolidine] in meat and cured meat, 107,108,160 –162 bacon, 89,110,162 –176 fish, 94,95,102,117 cheese, 94,95 and beer (and malt) 100 –102,152 ; 2-pyrrolidinecarboxylic acid, 1-nitroso-[N-nitrosoproline] in meat, 109,146,160,177 –179 bacon, 146,163,180,181 chicken, ham, toast, biscuits, cornflakes, 180 and beer (and malt) 91,92,180 ; diethanolamine, N-nitroso-[N-nitrosodiethanolamine] in meat and cured meat. 182
Mutagenicity has been measured in heated, grilled, and broiled foods, 183 –190 including beef extract 191 –196 ; broiled, fried, and/or charred beef 191,192,197 –205 ; broiled cuttlefish 206 ; eggs, fish, and meat 184 ; flour and rice 184 ; soy beans 184 ; broiled fish, 200,201,207 –209 including herring, mackerel, pike, and sardine 200 ; broiled sardine 196,197 ; protein pyrolysates of albumin 210 –213, and soybean globulin 214,215 ; calf thymus, egg white, and serum albumin 216 ; casein, collagen, gluten, histone, insulin, lysozyme, ovalbumin, and zein 217 ; peptide pyrolysates from polypeptides 218 ; carnosine, glycyl glycineb, glycyl glutamic acid, glycyl proline, glycyl tryptophan, leucyl glycyl phenylalanine, tryptophanyl alanine, tryptophanyl glycine, tryptophanyl tryptophan, and tryptophanyl tyrosine 219 ; amino acid pyrolysates 212,220 –223 from phenylalanine 223 and lysine 224 ; tryptophan, 225,226 –231 glutamic acid, 186,190,216,222 –233 histidine, 234 and 3-methylhistidine; alanine, arginine, asparagine, citrulline, cysteine, cysteine, glutamic acid, glutamine, histidine, lysine, methionine, ornithine, phenylalanine, serine, threonine, tryptophan, tyrosine, and valine 218 ; and beverages including roasted coffee, 235 –239 instant coffee, 238 tea, 236 and brandy and sake. 240
Exposure to chemicals made by bacteria, fungi, algae, and volcanoes
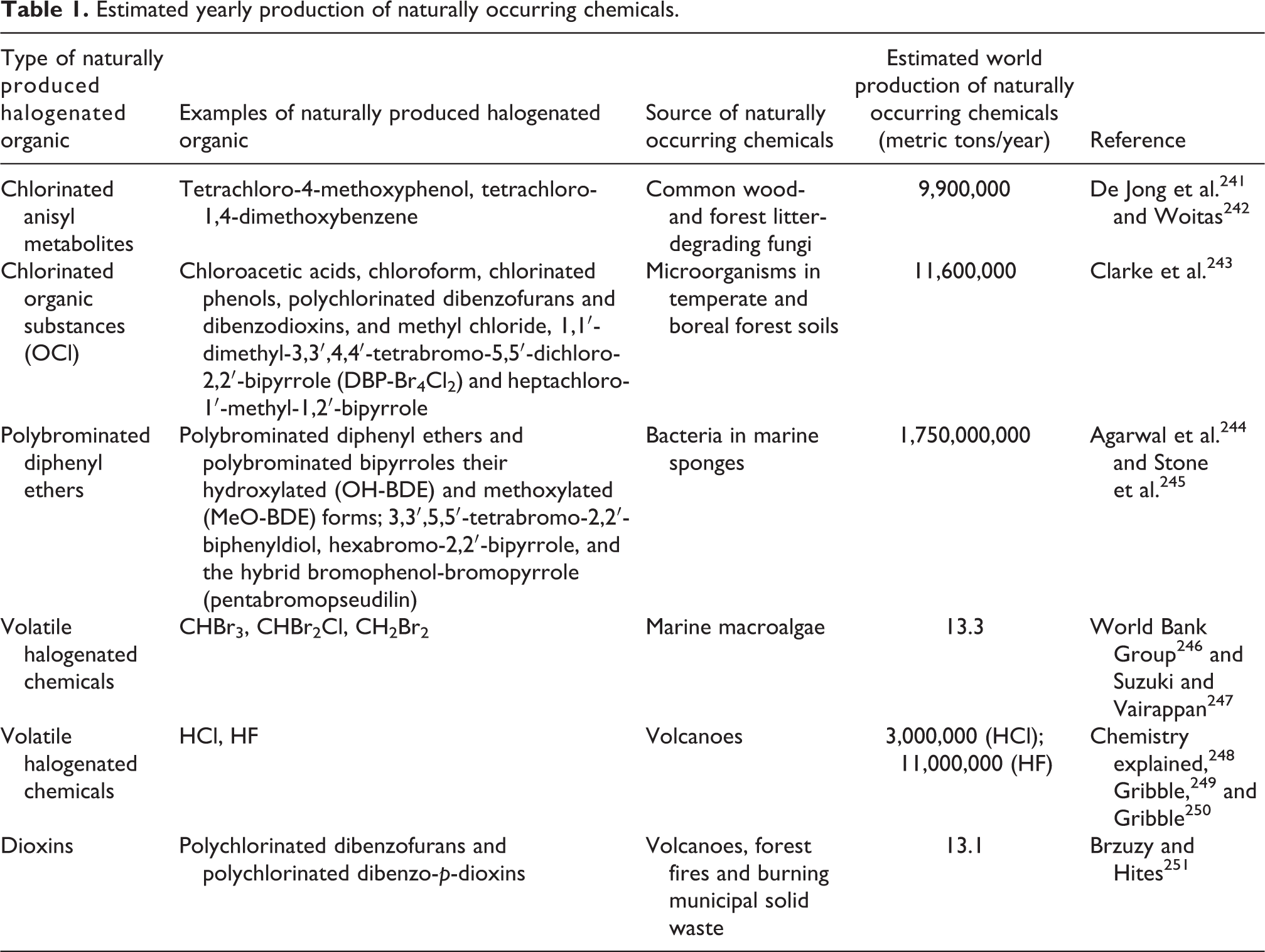
A number of natural processes produce comparatively large amounts of persistent organic compounds (POP-like chemicals) (Table 1). Common wood-degrading and forest litter-degrading fungi produce 9,900,000 metric tons yearly of chlorinated anisyl metabolites, including tetrachloro-4-methoxyphenol and tetrachloro-1,4-dimethoxybenzene. 241,242 Microorganisms in temperate and boreal forest soils produce 11,600,000 metric tons yearly of chlorinated organic substances (OCl), including the following: chloroacetic acids; chloroform; chlorinated phenols; polychlorinated dibenzofurans (PCDFs) and dibenzodioxins; methyl chloride; 1,1′-dimethyl-3,3′,4,4′-tetrabromo-5,5′-dichloro-2,2′-bipyrrole (DBP-Br4Cl2) and heptachloro-1′-methyl-1,2′-bipyrrole. 243 Bacteria in marine sponges produce 1,750,000,000 metric tons yearly of polybrominated diphenyl ethers (PBDEs); PBDEs and polybrominated bipyrroles and their hydroxylated (OH-BDE) and methoxylated (MeO-BDE) forms; 3,3′,5,5′-tetrabromo-2,2′-biphenyldiol; hexabromo-2,2′-bipyrrole; and the hybrid bromophenol-bromopyrrole (pentabromopseudilin). 243 –245,252 Marine macroalgae produce 13.3 tons yearly of the volatile halogenated chemicals CHBr3, CHBr2Cl, and CH2Br2. 246,247 Each year, volcanoes release 3,000,000 tons of volatile halogenated chemicals, including HCl, and 11,000,000 tons of HF. 248 –250 Volcanoes, forest fires, and burning municipal solid waste release 13.1 tons per year of dioxins, PCDFs, and polychlorinated dibenzo-p-dioxins. 251 The yearly quantity of organic compounds produced in nature that are either themselves relatively stable or can react (e.g. HCl, HF) with other naturally occurring compounds and form relatively stable compounds is astronomical at an estimated release into the environment of 1.79 billion metric tons yearly. Assuming that yearly worldwide releases of organic compounds into the environment by natural sources have remained approximately stable during the postwar period, the total tons of organic compounds released into the environment over the 70-year period from 1945 to 2015 is an astronomical 125 billion metric tons. Therefore, natural releases of organic compounds into the environment exceed synthetic production of chemicals over the same time period by 5000-fold. Human exposure to natural chemicals released into the environment will vary significantly from person to person based upon geography, occupation, and activity levels.
Estimated yearly production of naturally occurring chemicals.
Conclusions
Recently, Bruce Ames (developer of the Ames test for mutagenicity) addressed the widespread confusion regarding exposure to natural versus manmade chemicals.
252
Ames explained the following: In 1962, Rachel Carson published her book, Silent Spring. In this book, Rachel Carson said: ‘For the first time in the history of the world, every human being is now subject to contact with dangerous chemicals from the moment of conception until death.’ But Rachel Carson was made of chemicals. Everything is made of chemicals. In that book, Carson would only quote the results of experiments that showed that a chemical did something bad because that would fit her theory, and she would ignore experiments where it was found that that chemical had not done anything bad. She unscientifically assumed that everything ‘natural’ was beneficial. But she ignored a lot of available information. What about arsenic? or aflatoxin and other fungal toxins? or poisonous mushrooms? or many other “natural” toxic and carcinogenic compounds, and the relevant large literature on natural toxins in plants? Thus, Silent Spring was not a scholarly piece of work. Unfortunately, it got people on the wrong track.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
