Abstract
The physiological and health status of control animals may vary. Due to this variation, it is important to define acceptable ranges of control hematology parameters to gain a better understanding of adverse and non-adverse effects of test substances. After generating historical control data for two Wistar rat strains (RccHan™:WIST and Crl:WI(Han)) from different breeders, the data sets were statistically analyzed using Minitab®. After noticing that single outliers can affect the study control data set, the respective outliers were verified relative to the available histopathology findings, for example, inflammatory pulmonary lesions following vehicle aspiration or spontaneous sperm granuloma affecting the health status and hematology data of the respective animals. Such data points were excluded from the control data set. Comparing both data sets, it was obvious that different blood sampling and anesthesia methods as well as strain differences may result in slightly different values. After excluding the outliers, a data set from animals with presumably good health status was generated to define acceptable ranges and severity degrees. To evaluate effects, possibly influencing hematology parameters and defined acceptable ranges, selected vehicles and different study types were observed.
Keywords
Introduction
Toxicity studies can only be interpreted when appropriate reference values established from healthy control animals are available. Specific reference intervals are needed for each animal species being tested. Many physiological parameters vary with age, sex, housing, food/water consumption, circadian rhythm, increased or decreased activity, stress, or sexual cycle.
The use of historical control data in preclinical studies is discussed in detail by Keenan et al.,
1
whereas this publication, like several others,
2
–5
relates solely to proliferative lesions. Nevertheless, the base recommendations as summarized by the Historical Control Data Working Group of the Society of Toxicologic Pathology are also valid for historical control data other than proliferative lesions. When adapted, these recommendations
1
also easily apply to data other than pathology data and consist mainly of: the use of the concurrent control group as the most relevant comparator to determine treatment-related effects; the estimation of the potential impact of design-related parameters (e.g. laboratory, species/strain, administration route, vehicle, feed, feeding practices, study duration, housing) on study outcome, which underlies the appropriate selection of studies for the use of control data; standardized diagnostic practices, including sampling procedures, use of instruments, and application of diagnostic criteria, which can impact study and control data; use of control data from the laboratory that conducted the study under review, which will likely be more comparable than control data compiled from several laboratories; careful evaluation of published control data, which might be helpful in the interpretation of study data; and presenting control data as a range of incidences or percentages, mean, and standard deviation and/or minimum/maximum value ranges for any parameter.
Furthermore, recommendations for the evaluation of clinical pathology data have been published. 6,7
The evaluation of hematology parameters is standardized by several ICH (M3 R2 2009), 8 OECD (407/2008, 9 408/2018 10 ), and EPA (2000) 11 guidelines for toxicity studies. Standard parameters include red blood cell (RBC), hemoglobin (HB), hematocrit (HCT), total and differential leukocyte count (white blood cell (WBC)), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), reticulocyte (RET), and platelet count (PLT).
However, additional parameters such as erythrocyte indices methemoglobin (MET-HB) and Heinz body (HEINZBOD) may be included in the evaluation as appropriate to the testing of specific test items. Furthermore, there is no indication in the guidelines for an approach using absolute and/or relative differential leukocyte count values. Moreover, RET values can be split further into the maturity indices using the fluorescence behavior of these cells. Hence, there are different outlines for control data compilations in different laboratories.
Scarce data are published on historical control data for hematology parameters from toxicity studies performed in rodents. 12 –14 In some circumstances, the published data cannot be used for comparisons due to missing methodological data, for example, animal ages or study lengths (e.g. data published by Said and Abiola 15 ).
Material and methods
RccHan™:WIST
General:
Control data for hematology parameters were collected at RCC Ltd/Harlan Laboratories Ltd, Switzerland, over a 6-year period from 2002 to 2007. The animals originated from a single production site (RCC Ltd/Harlan Laboratories Ltd, Füllinsdorf, Switzerland) thus minimizing the influence of breeding and environment-related variables. Data were collected from control animals at ages from ≤7 weeks up to ≥71 weeks (including dose-range finding studies up to carcinogenicity studies); 5062 males and 5046 females are included in this survey.
Some hematology parameters (HEINZBOD, neutrophil (NEUT), and MET-HB) were compiled from fewer animals and studies because they were outside the range of standard laboratory evaluations. All studies were performed in compliance with the Swiss Ordinance relating to Good Laboratory Practice (SWISS GLP, 2005 16 ) based on the OECD Principles of Good Laboratory Practice (OECD, 1997) and/or the standard operating procedures of the testing laboratory. The studies were performed under permission of the ethical committee and Swiss authorities. RCC Ltd/Harlan Laboratories Ltd, Switzerland was accredited by AAALAC. Clinical laboratory data were generated at RCC Ltd (Füllinsdorf, Switzerland) in an accredited laboratory in accordance with the ISO/IEC 17025 standard under accreditation number STS 085 from the Swiss Accreditation Service.
The data described below were obtained from various types of repeated-dose toxicity studies (i.e. feeding, gavage, dermal, inhalation, or intravenous administration). Animals were housed individually or in groups of three or five (depending on the administration route) in Makrolon® cages with wire mesh tops and standardized softwood bedding (Lignocel, Schill AG, Muttenz, Switzerland). Rats had free access to standard rat maintenance diet 3433 (Provimi Kliba AG, Kaiseraugst, Switzerland) and to tap water. The animal facility was air-conditioned with 10–15 air changes per hour, temperatures of 22°C ± 3°C, and relative humidity of 30–70%. A 12-h fluorescent light/dark cycle was maintained with music during the light period.
The evaluations were generally performed during the pretest and at the end of the toxicity study prior to necropsy in fasted animals that had free access to water. Experienced technicians withdrew blood with heparin-coated glass capillaries from the retro-orbital plexus of animals under light isoflurane anesthesia (5% isoflurane/95% oxygen) into tri-potassium-ethylenediaminetetraacetic acid-coated anticoagulant tubes for complete blood cell count evaluation. Sampling was performed early in the working day prior to necropsy to reduce biological variations caused by circadian rhythms.
Hematology evaluation methods:
Commercially available test methods (flow cytometry) based on the ADVIA 120 hematology system (Bayer AG, now Siemens AG; including the reagents) were used to determine hematological parameters, and in-house procedures based on published scientific procedures 17,18 were used for microscopy, blood smear preparation, and staining (differential WBC count, red cell morphology, and HEINZBOD counts). MET-HB determination (spectroscopy) was based on a commercially available test method in conjunction with an OSM-3 (Radiometer) hemoximeter.
Crl:WI(Han)
General:
Control data on hematology parameters were collected at BSL BIOSERVICE Scientific Laboratories Munich GmbH (BSL), over a 10-year period from 2010 to 2019. The animals derived from a controlled, full-barrier maintained breeding system (Charles River, Sulzfeld, Germany) thus minimizing the influence of breeding and environment-related variables. The present data were collected from control animals at ages of 8–12, 13–18, and 19–40 weeks (including dose-range finding, subacute toxicity, and repeated-dose toxicity studies—OECD and pharma on the one hand and implantation and reprotoxicology studies on the other); 1636 males and 1596 females are included in this survey.
All studies were performed in compliance with OECD Principles of Good Laboratory Practice 19 (revised in 2018) and the testing laboratory’s standard operating procedures. In accordance with German animal protection law, the study types were reviewed and accepted by local authorities. Furthermore, the studies were subjected to an ethical review process and were authorized by the Bavarian animal welfare administration. Since 2014, studies were performed in an AAALAC-accredited laboratory.
The data described below were obtained from dose-range finding, repeated-dose toxicity, reprotoxicology, and implantation studies with various administration routes (i.e. feeding, gavage, dermal, intravenous, subcutaneous, or intramuscular). Animals were housed individually or in groups of two to five (depending on the administration route) in type III/H (approximately 800 cm2) or type IV (approximately 1800 cm2) cages (most commonly individually ventilated) on Altromin sawdust bedding. Rats had free access to standard rat maintenance diet 1324 (Altromin) and to acidified water. The animal facility was air conditioned with 10–15 air changes per hour, temperatures of 22°C ± 3°C, and relative humidity of 55% ± 10%. There was a 12-h artificial light/dark cycle. Makrolon® or cardboard tunnels, wood bricks, and sizzle material were used as environmental enrichment.
Blood sampling was generally performed at the end of the toxicity study prior to necropsy in overnight-fasted animals that had free access to water. Experienced technicians withdrew blood, with uncoated cannulas (length: 40 mm and diameter: 0.9 mm) from the abdominal aorta of animals under ketamine–xylazine anesthesia into EDTA-coated anticoagulant tubes for complete blood cell count evaluation. Sampling was performed early in the working day prior to necropsy to reduce biological variations caused by circadian rhythms.
Hematology evaluation methods:
Commercially available test methods (flow cytometry) based on the ADVIA 120 hematology system (Bayer AG, now Siemens AG; including the reagents) were used to determine hematological parameters.
Statistical analysis
General:
The values for each of the reported parameters were grouped by sex and age range derived from study type and duration. The number of animals, the respective mean, standard deviation, minimum and maximum were reported for all parameters. The data evaluated from Harlan Laboratories Ltd includes animals at ages of ≤7 weeks up to ≥71 weeks 13 in six different age-groups (≤7, 8–12, 13–18, 19–40, 41–70, and ≥71 weeks). Outliers were detected during the respective studies. No statistical outlier test was done with Minitab® (version 18, Minitab, LLC) because only summarized data was available, meaning no individual data and medians were available.
The data collected at BSL was evaluated including animals at ages of 8 to 40 weeks in three different age-groups (8–12, 13–18, and 19–40 weeks) to permit comparison of the two different data sets. All parameters were analyzed statistically using Minitab® (version 18, Minitab, LLC). Individual parameters for each of the three age-groups were checked for parametric/nonparametric distribution using the Anderson–Darling normality test to exclude all outliers before further analysis. All statistically identified outliers were then assigned to the respective study and were examined on available histopathology finding.
Comparison of different study types:
A data set for reproduction toxicity studies (OECD 422) and implantation studies (ISO 10993) was generated to consider differences between various study types with animals at the ages of 19–40 weeks using variance analysis.
Vehicle effects:
Further statistical data analysis was performed on the data set. Data from repeated-dose toxicity studies (oral route) from BSL at the ages of 8–12 and 19–40 weeks were analyzed to see if different vehicles may have an impact on hematological parameters by applying Bonett’s and Levene’s tests for variance analysis at a significance level of 95%. p Values below 0.05 were treated as significantly different.
Comparison of different rat strains:
Both data sets as well those collected from other related published data were compared, whereas Crl:WI(Han) and RccHan™:WIST were statistically analyzed in addition.
BSL and RCC Ltd/Harlan Laboratories Ltd, Switzerland, summarized data were compared in Minitab® with a two-sample t-test at a significance level of 95%. p Values below 0.05 were treated as significantly different. As the true values for the data sets were unknown, a possible offset between both laboratories was calculated by comparing the means of all three age-groups against each other.
Results
Setting acceptable ranges and grading deviations
For statistical correctness and data comparability, the acceptable ranges of both data sets were defined as mean value ± 2 standard deviations. In some cases, acceptable ranges started at 0.0% or 0.0 G/L, what does not mean that 0.0 values are really acceptable as it can be seen in the case of relative high fluorescence reticulocyte (HRET) and absolute NEUT values (see Tables 7 and 8 ).
The degrees of variation (decrease or increase) were estimated for three severity degrees (slight = grade 1, moderate = grade 2, severe = grade 3) using the mean and the minimum and maximum ranges obtained from control animal values. The severity degrees were defined random, for example, in RBC, HCT, red cell volume distribution width (RDW), MCV, MCH, MCHC, hemoglobin concentration distribution width (HDW), RET, low fluorescence reticulocyte (LRET), HRET, and WBC, as an increase or decrease of 10% (slight), 50% (moderate), and 70% (severe) referring to the respective acceptable ranges. For these parameters, the division of severity into 10%, 50%, and 70% was chosen because they can be correlated with pathological findings, for example, a deviation of 10% in HCT or HB usually has no effect in the histopathological examination. But a deviation of more than 10% becomes visible as either extramedullary hemopoiesis or bone marrow atrophy, or embolism. A deviation of 70% and more is usually associated with severe damage or even death. In the differential leukocyte count, the acceptable range for eosinophil (EOS), basophil (BASO) and monocyte (MONO) started at 0.0% or 0.0 G/L. Therefore, only increasing severity degrees were defined, as deviations of 10% to 20% is already extremely unusual for these parameters. The severity degree settings are necessary to interpret slight, moderate, and severe effects of test substances.
The acceptable ranges and grading deviations for RccHan™:WIST Rats are presented in Tables 1 to 15 and for Crl:WI(Han) in Tables 16 to 19 .
RccHan™:WIST (Harlan laboratories Ltd).
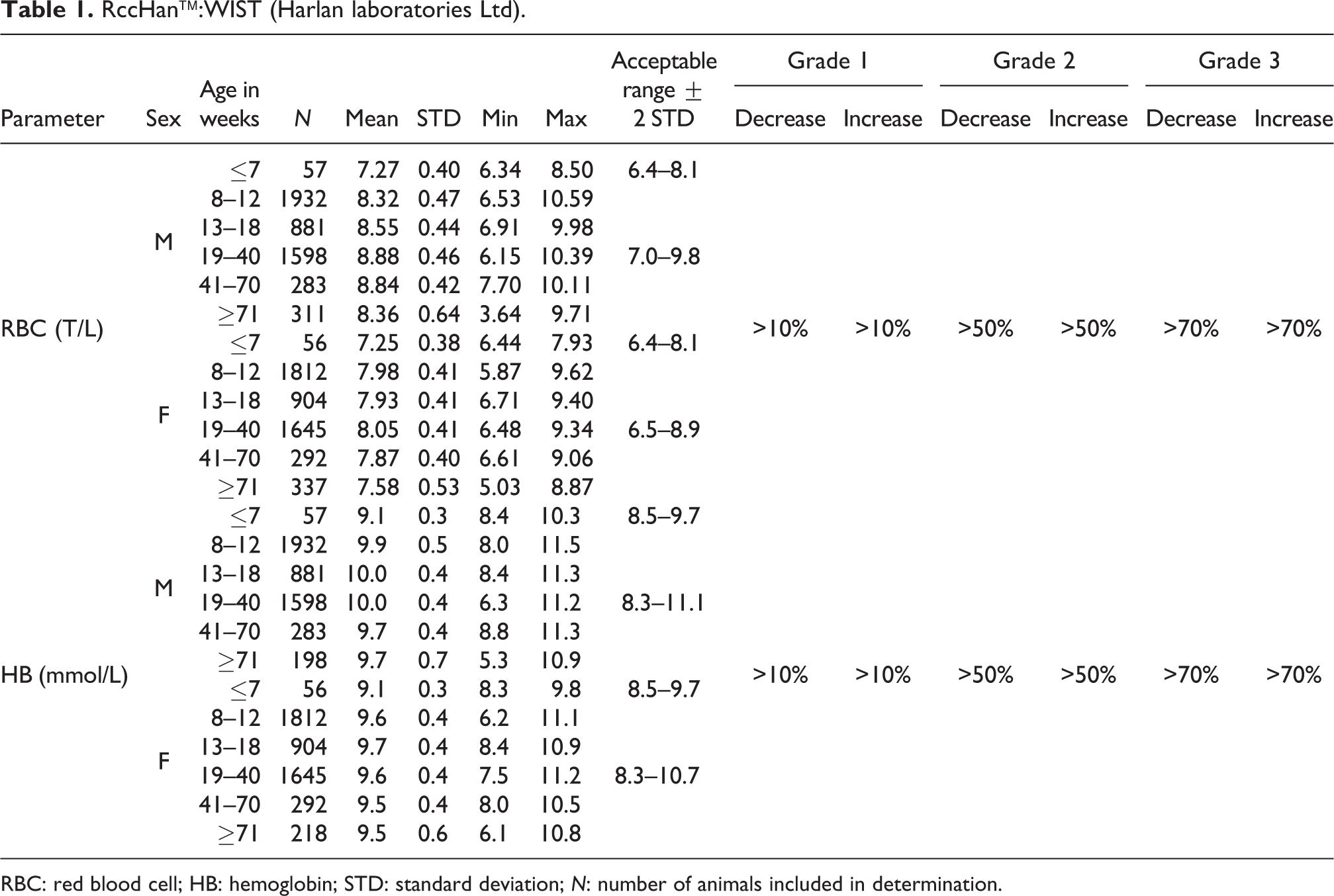
RBC: red blood cell; HB: hemoglobin; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
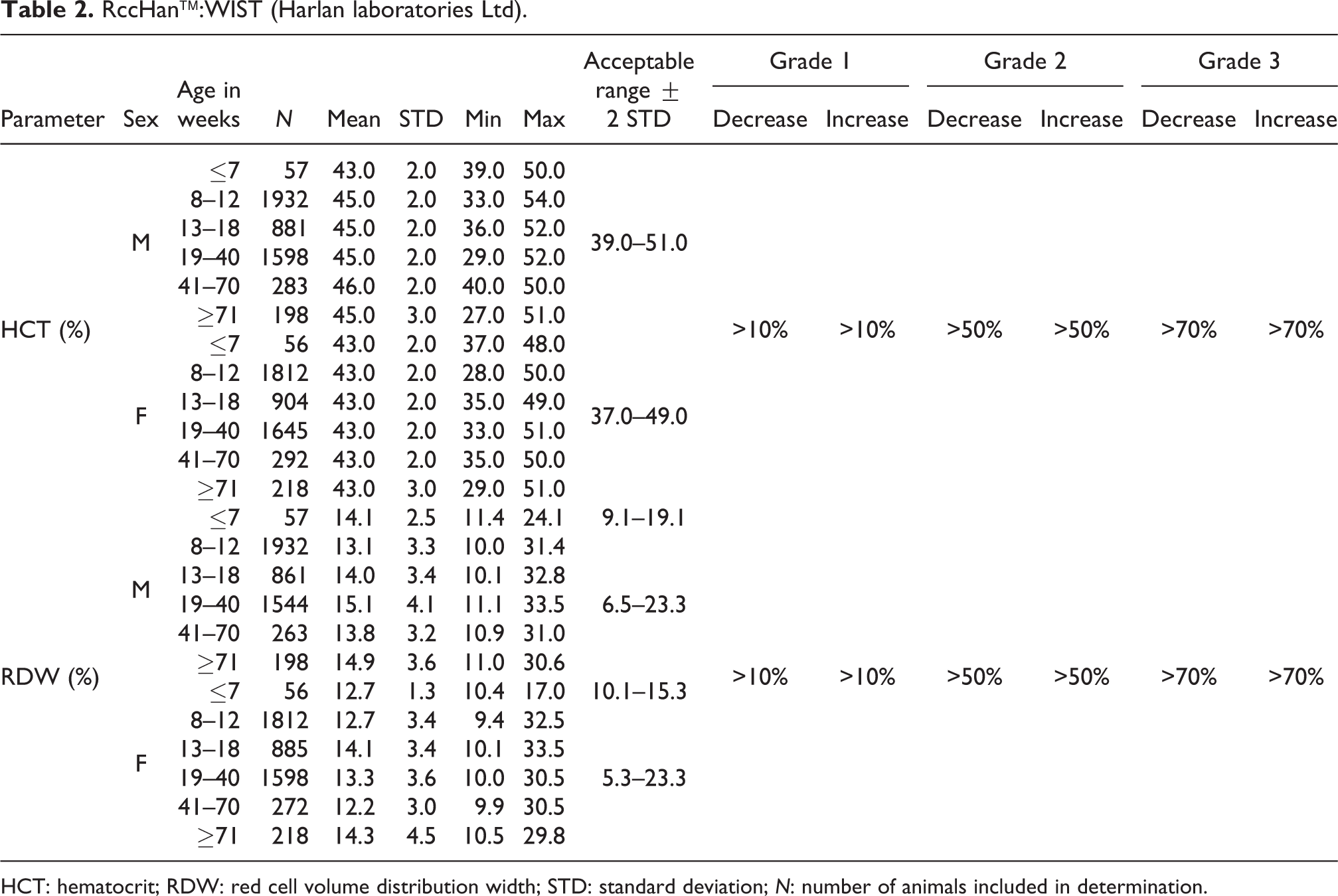
HCT: hematocrit; RDW: red cell volume distribution width; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
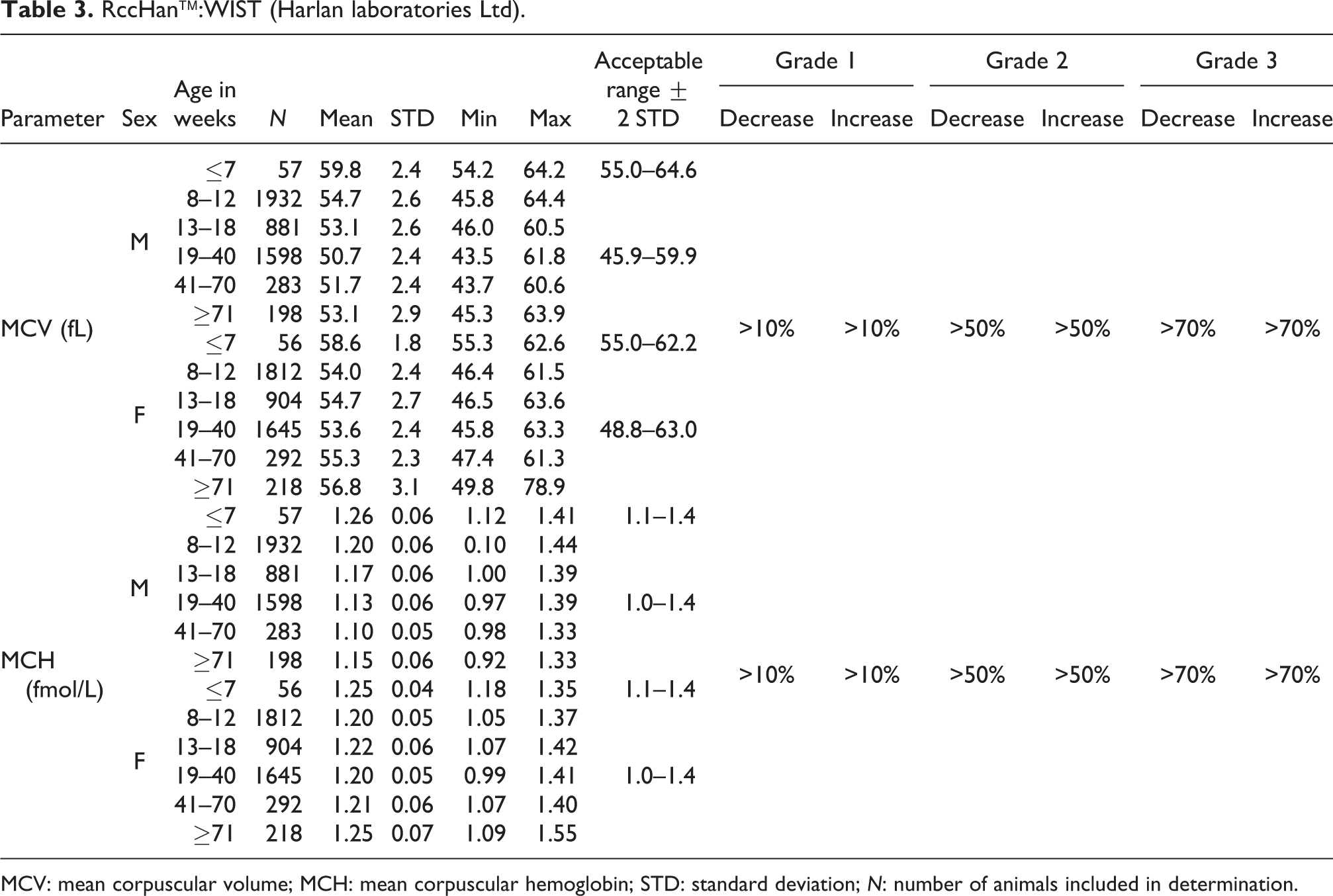
MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
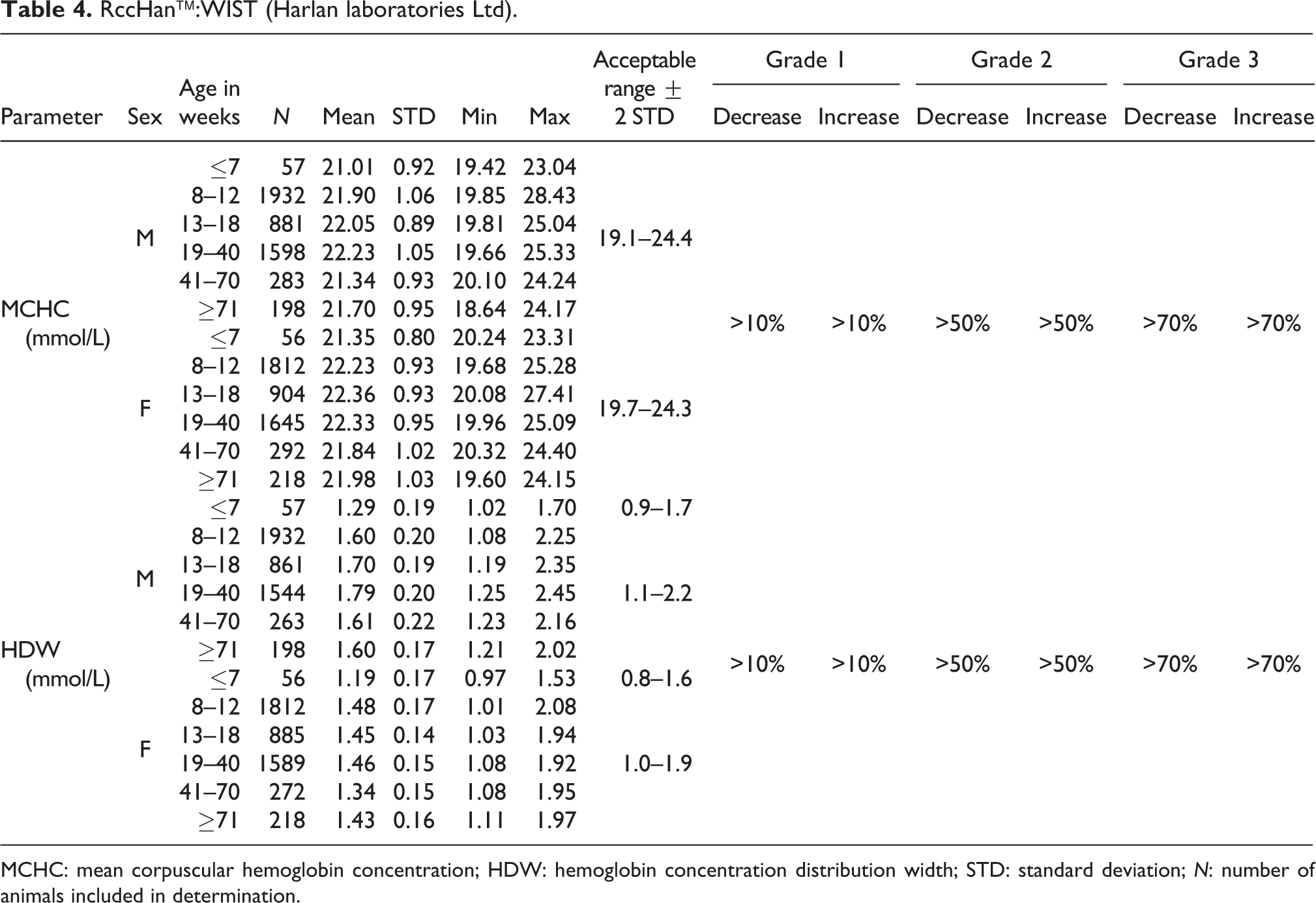
MCHC: mean corpuscular hemoglobin concentration; HDW: hemoglobin concentration distribution width; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
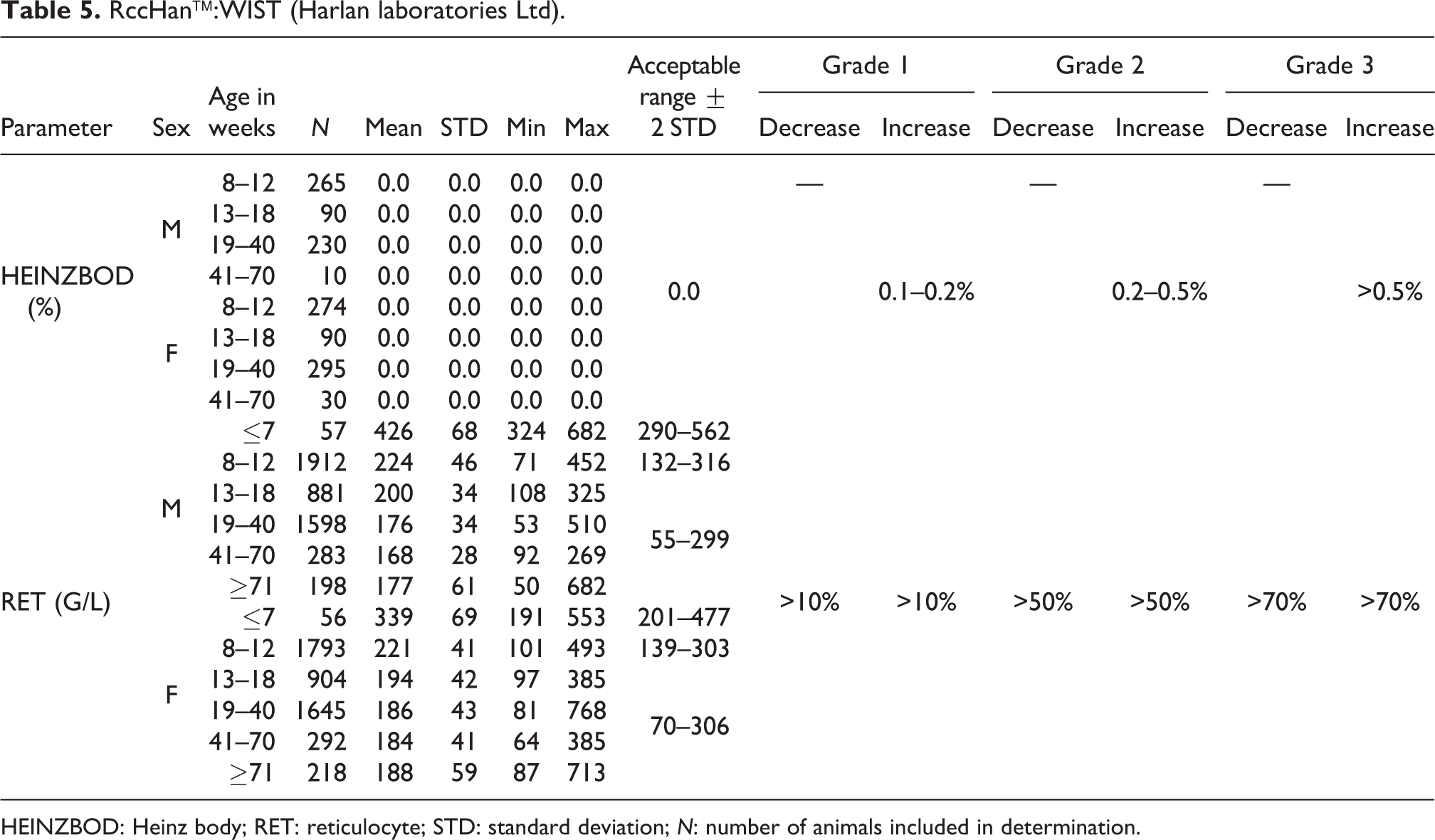
HEINZBOD: Heinz body; RET: reticulocyte; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
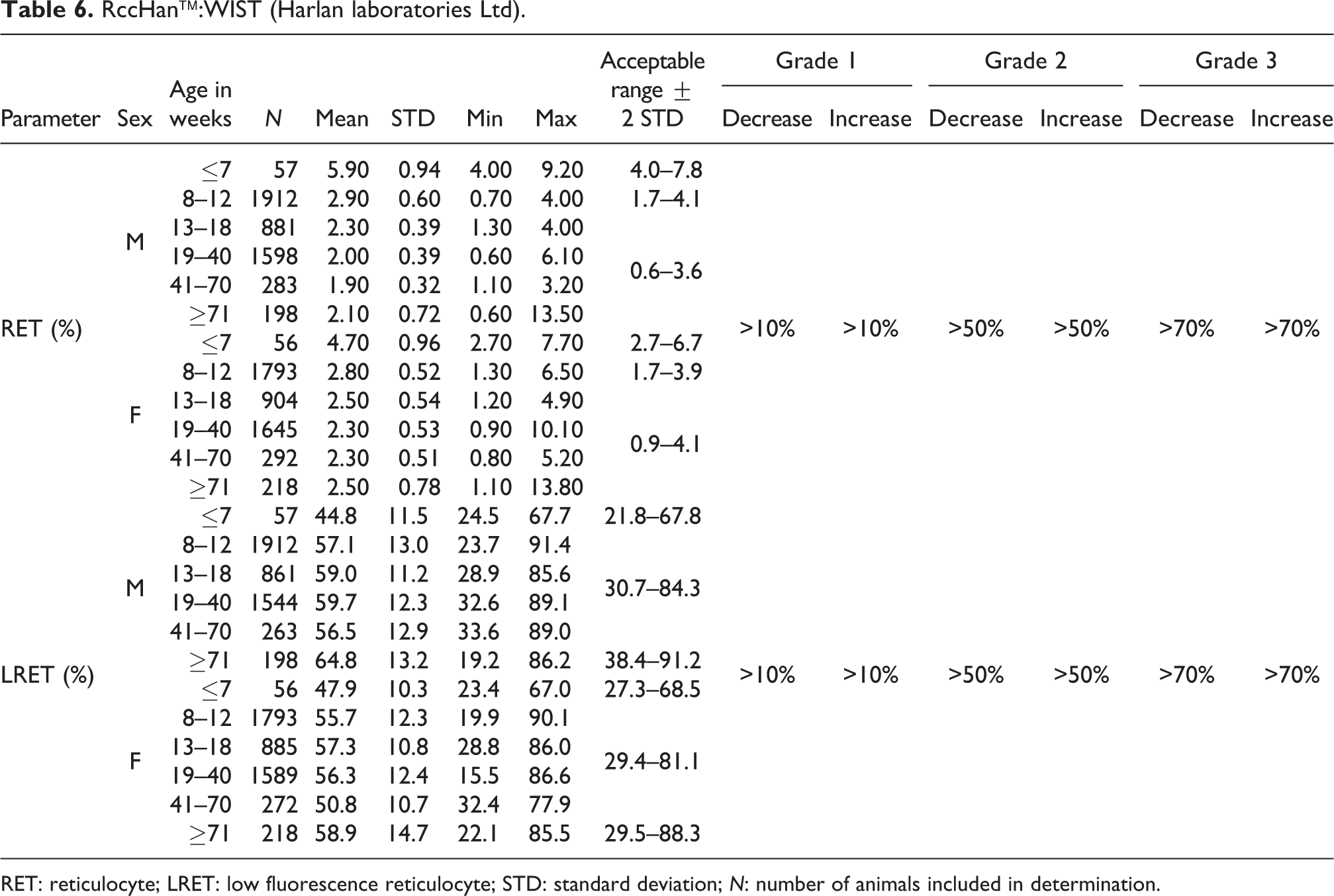
RET: reticulocyte; LRET: low fluorescence reticulocyte; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
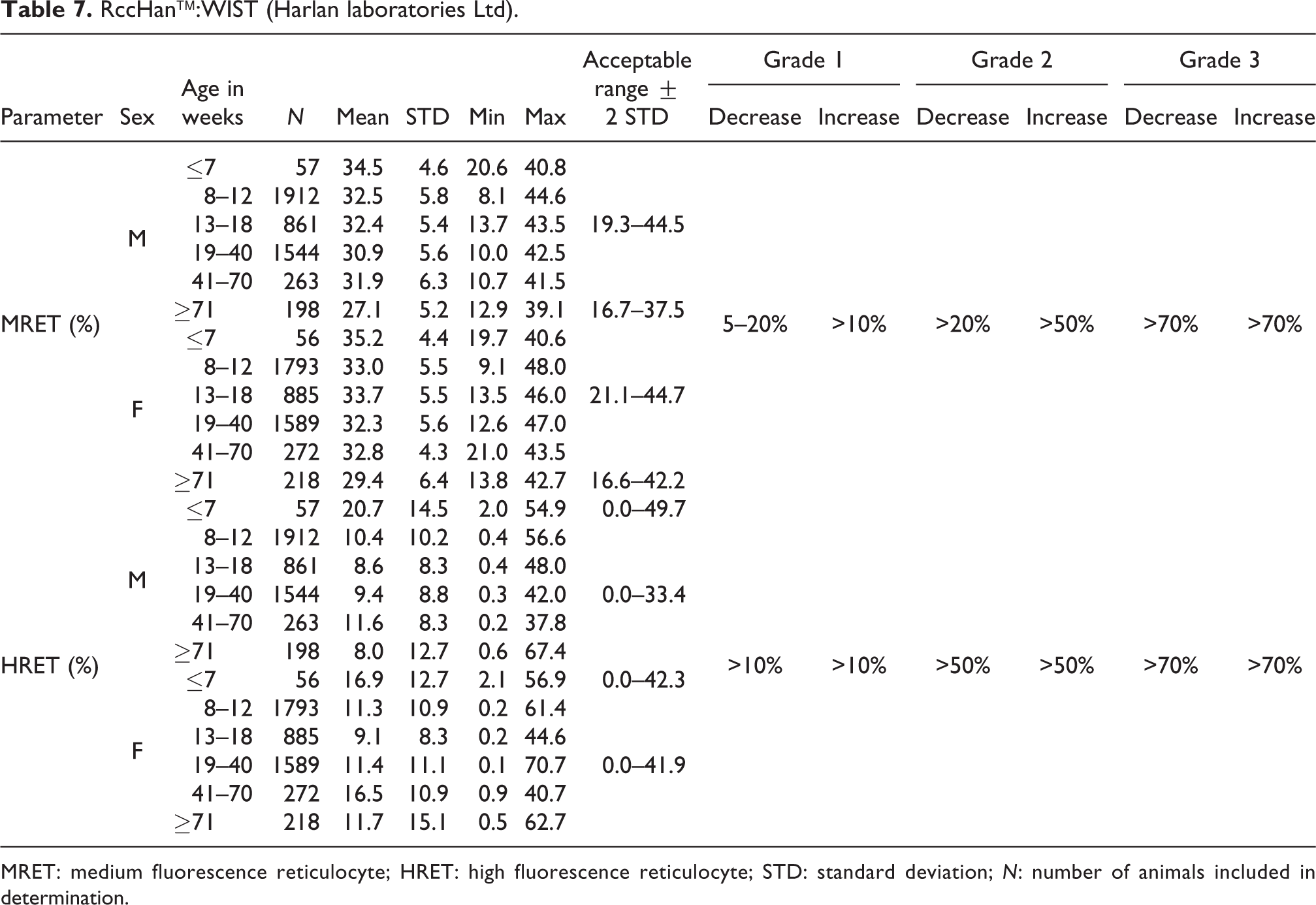
MRET: medium fluorescence reticulocyte; HRET: high fluorescence reticulocyte; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
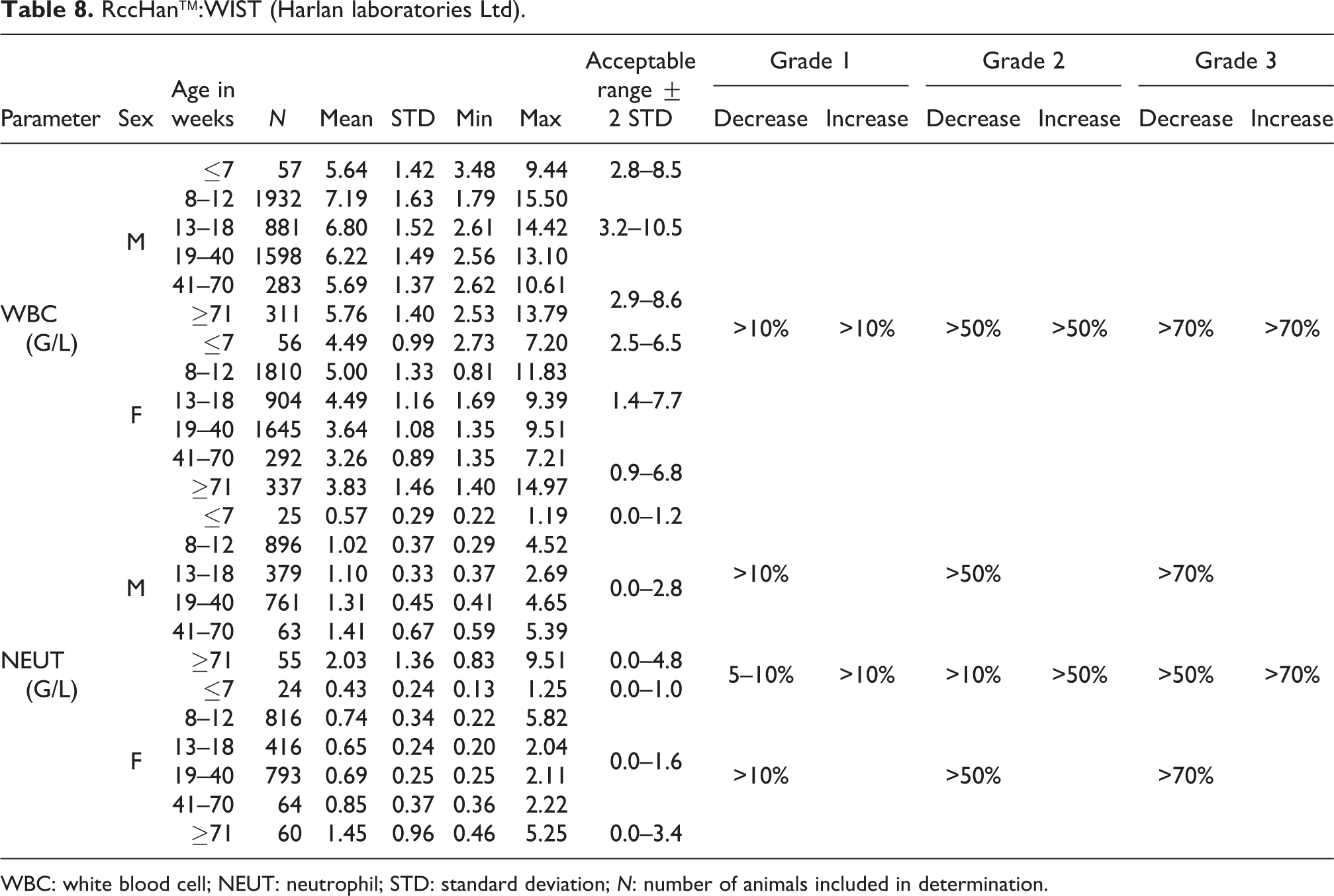
WBC: white blood cell; NEUT: neutrophil; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan Laboratories Ltd)
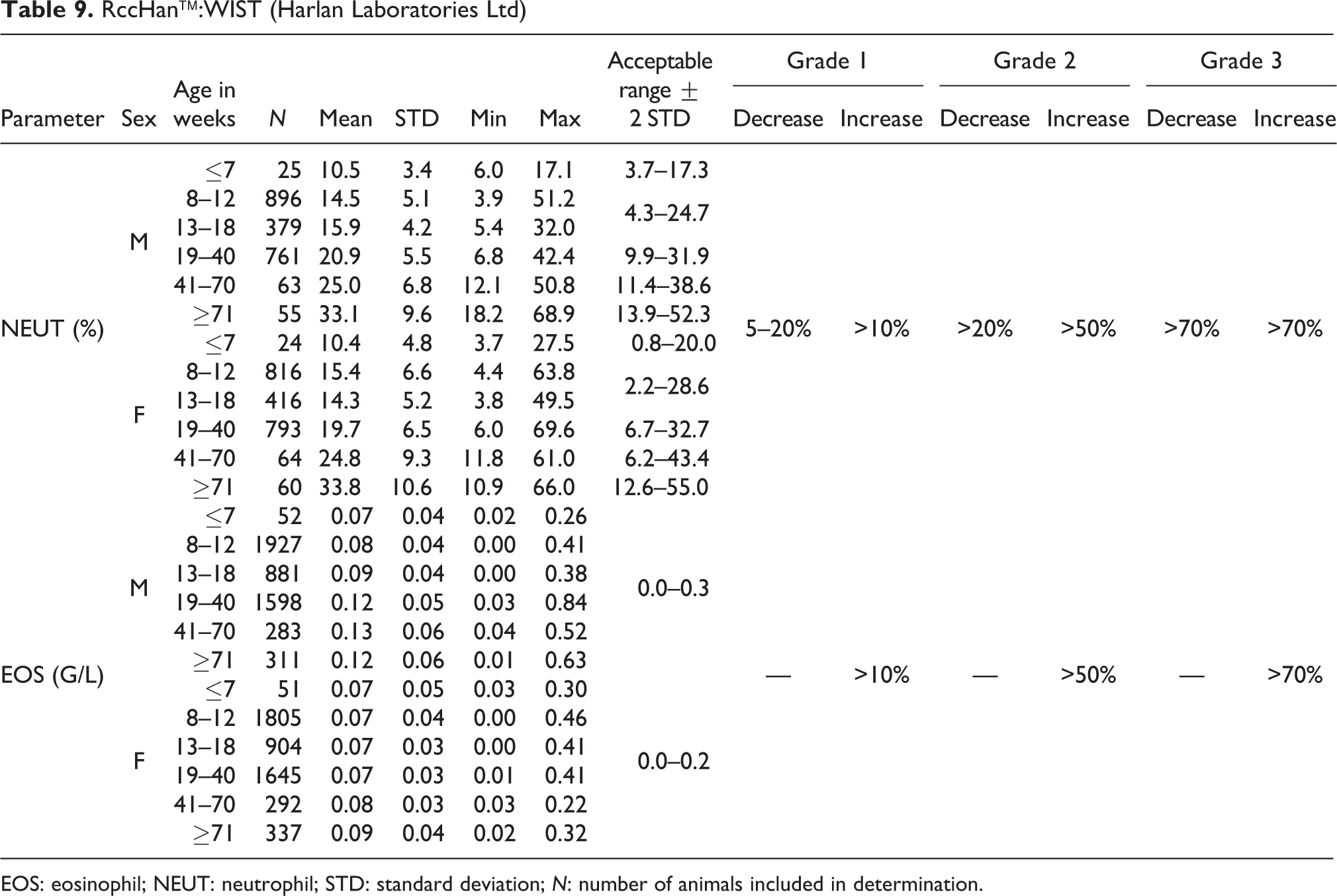
EOS: eosinophil; NEUT: neutrophil; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
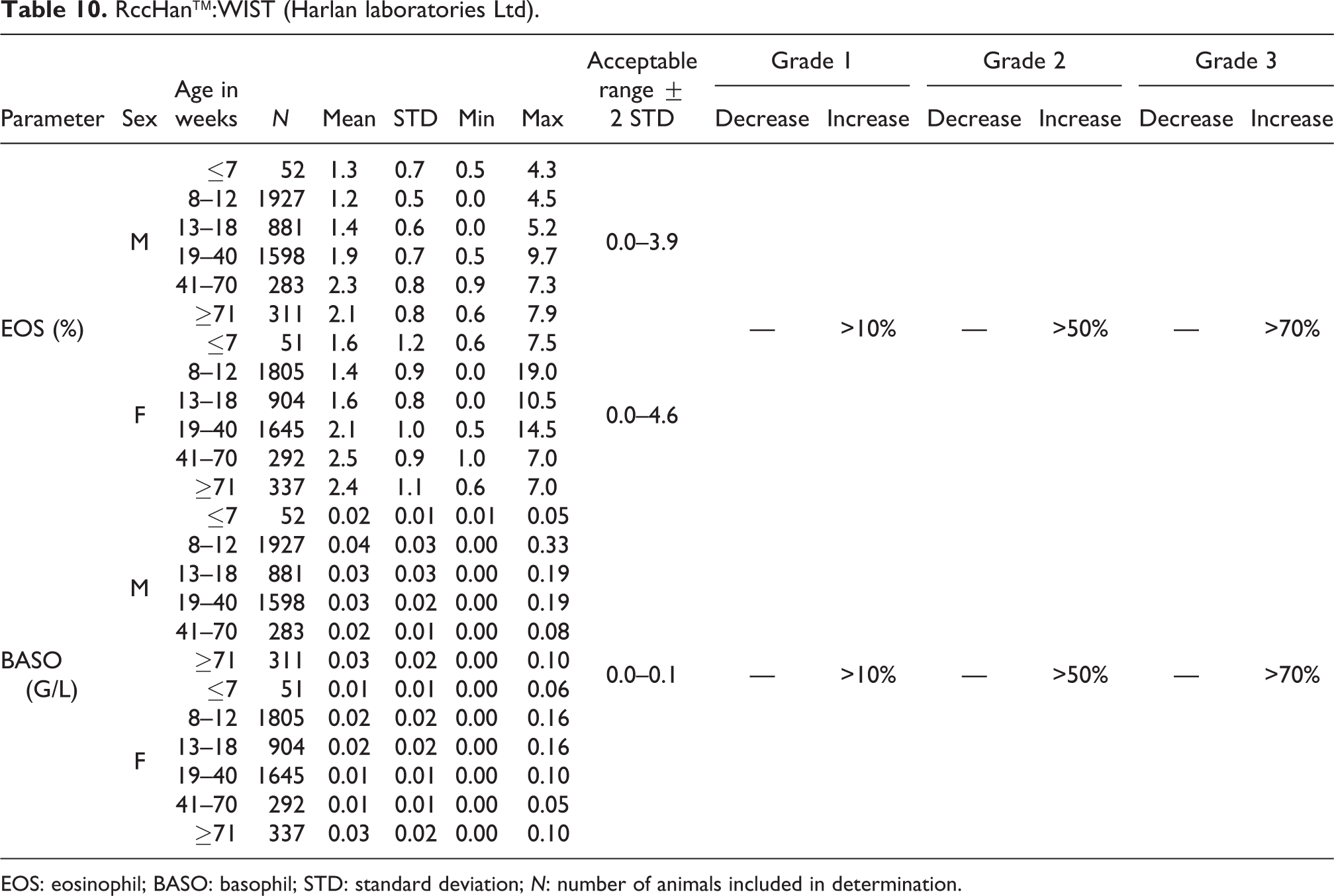
EOS: eosinophil; BASO: basophil; STD: standard deviation; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
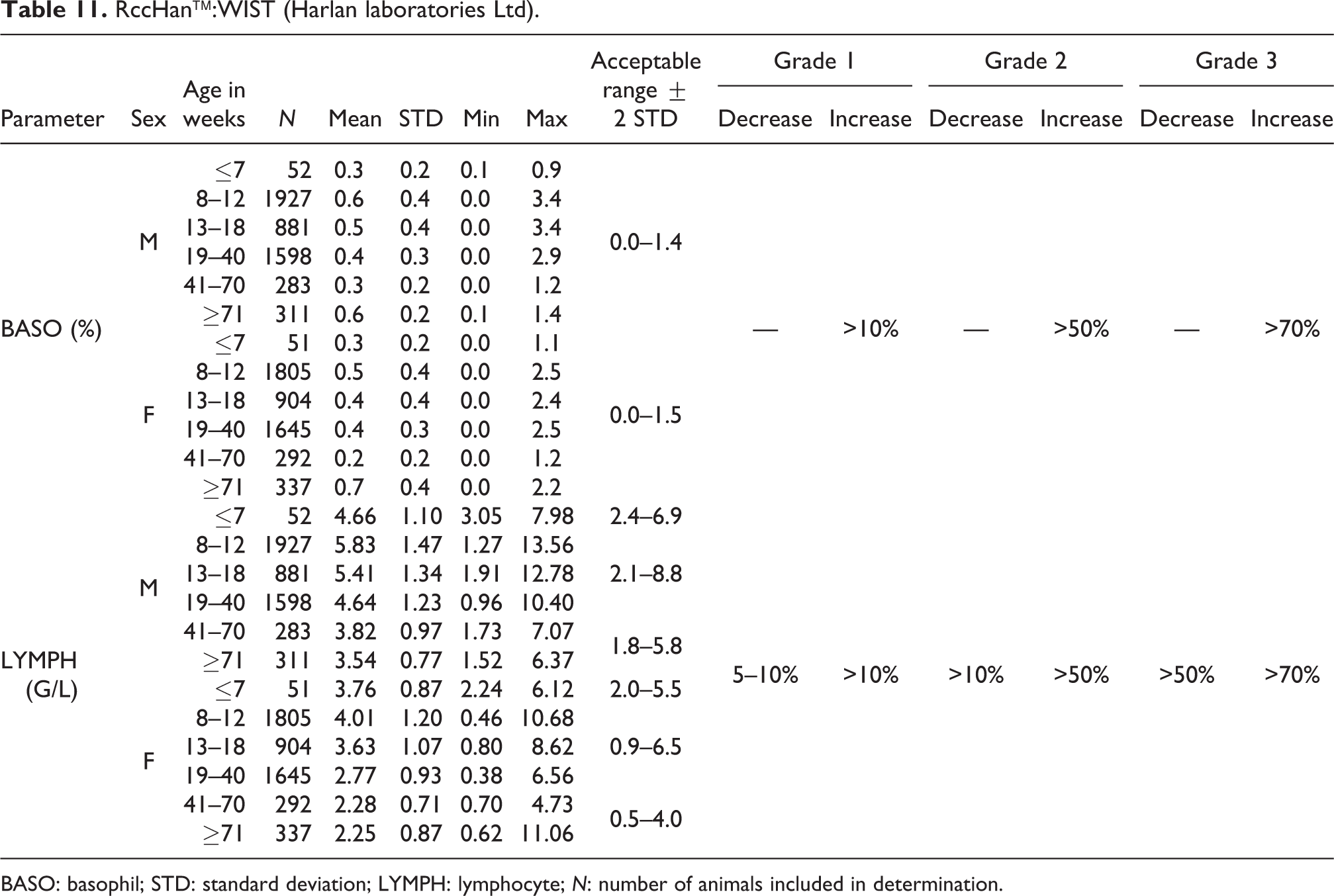
BASO: basophil; STD: standard deviation; LYMPH: lymphocyte; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
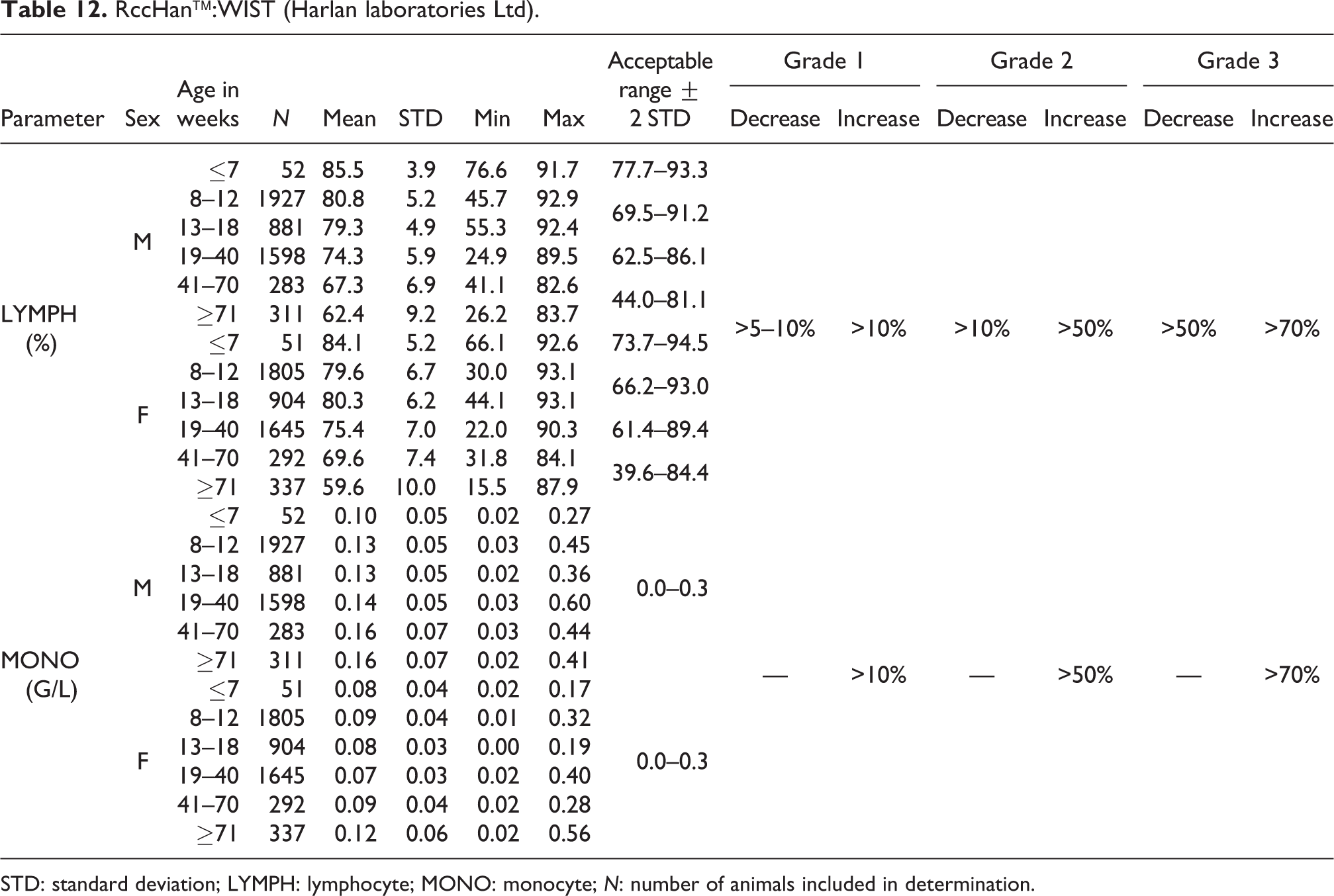
STD: standard deviation; LYMPH: lymphocyte; MONO: monocyte; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).

STD: standard deviation; MONO: monocyte; LUC: large unstained cell; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
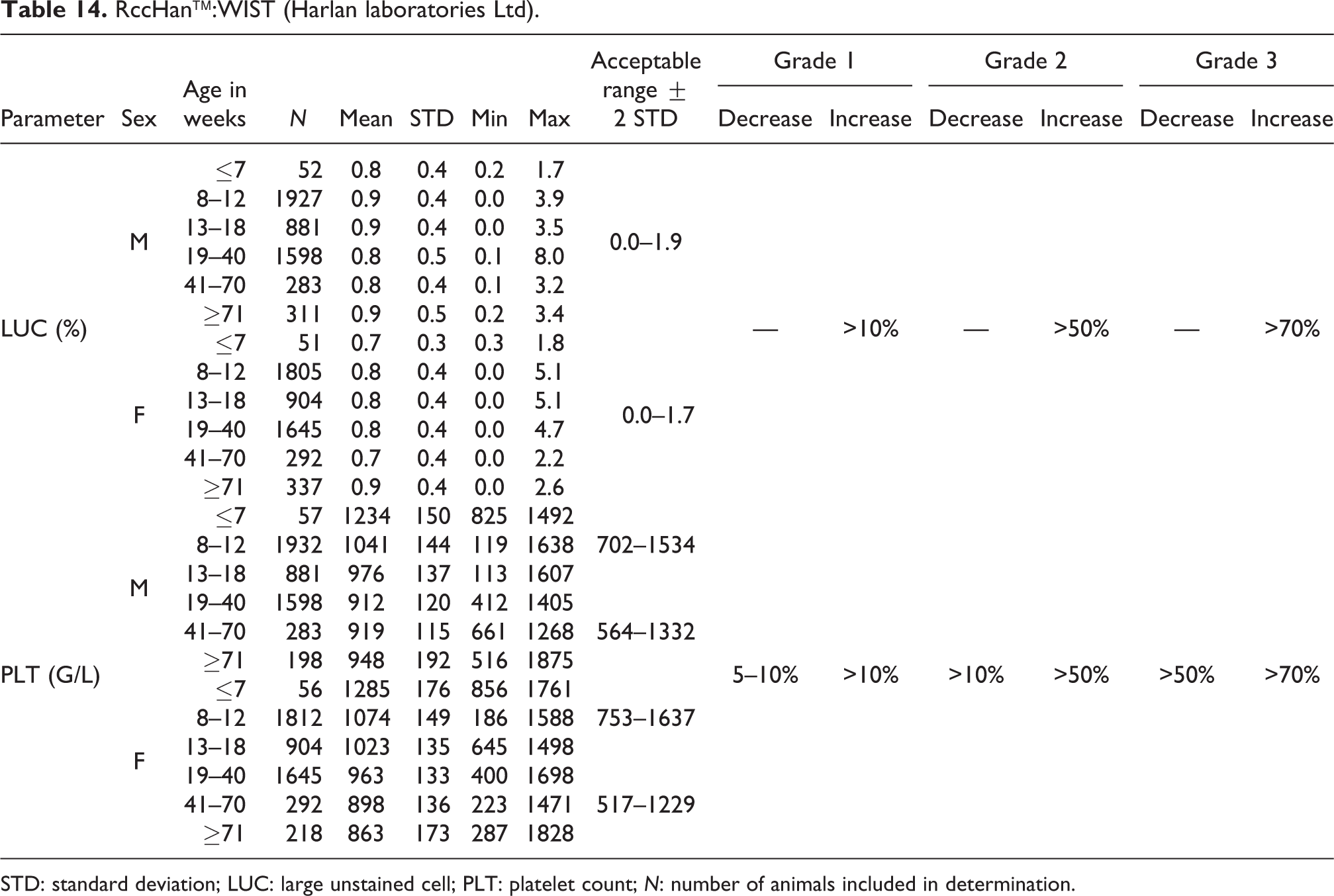
STD: standard deviation; LUC: large unstained cell; PLT: platelet count; N: number of animals included in determination.
RccHan™:WIST (Harlan laboratories Ltd).
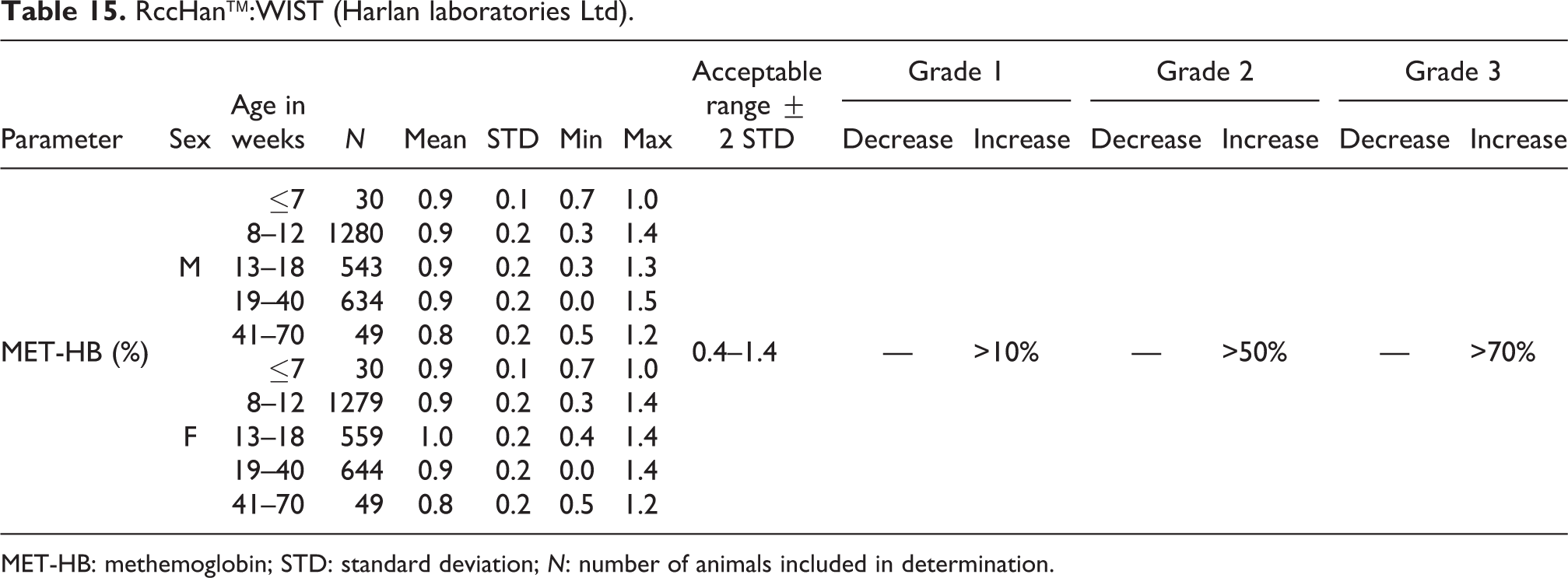
MET-HB: methemoglobin; STD: standard deviation; N: number of animals included in determination.
Crl:WI(Han) (BSL).
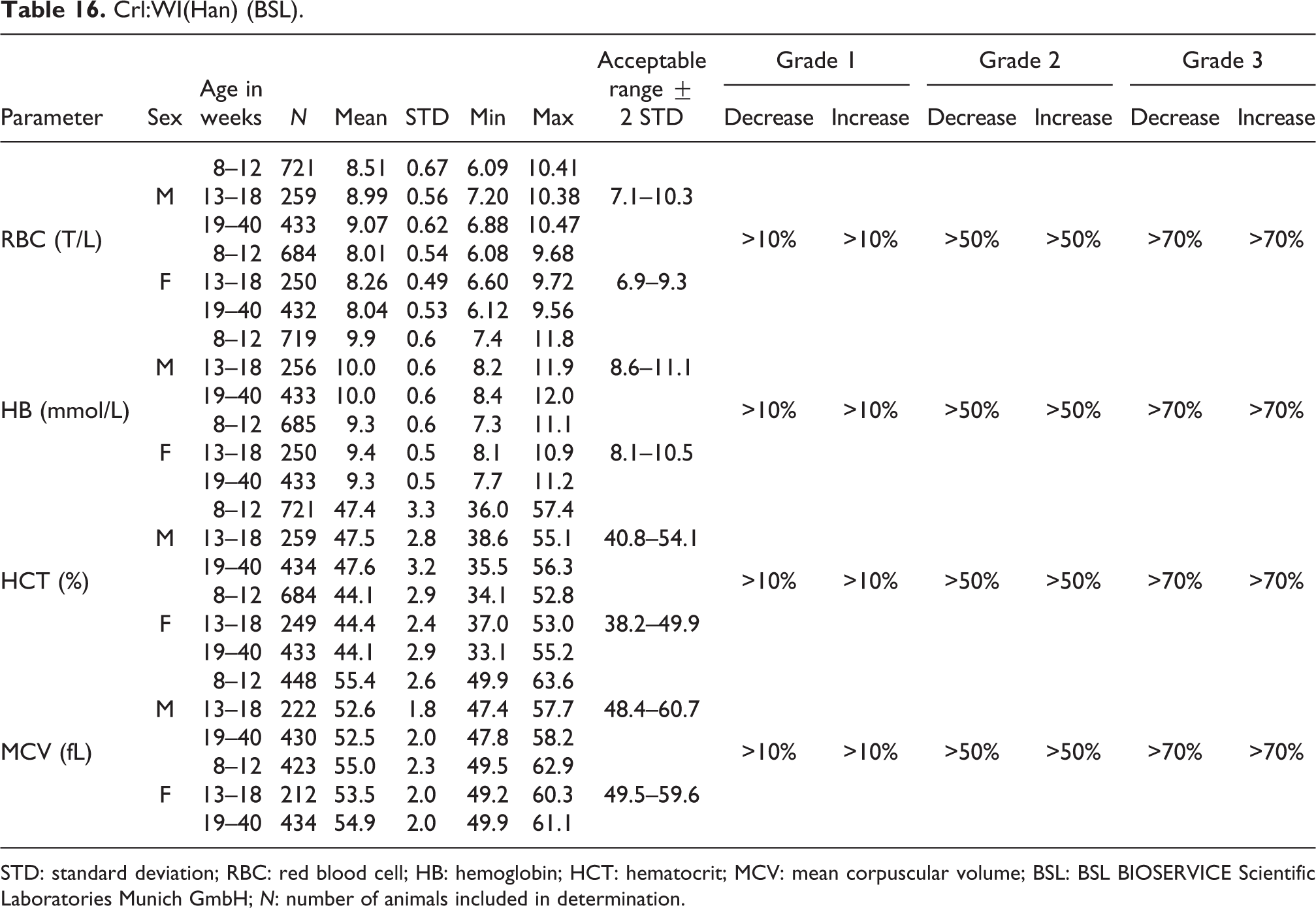
STD: standard deviation; RBC: red blood cell; HB: hemoglobin; HCT: hematocrit; MCV: mean corpuscular volume; BSL: BSL BIOSERVICE Scientific Laboratories Munich GmbH; N: number of animals included in determination.
Crl:WI(Han) (BSL).
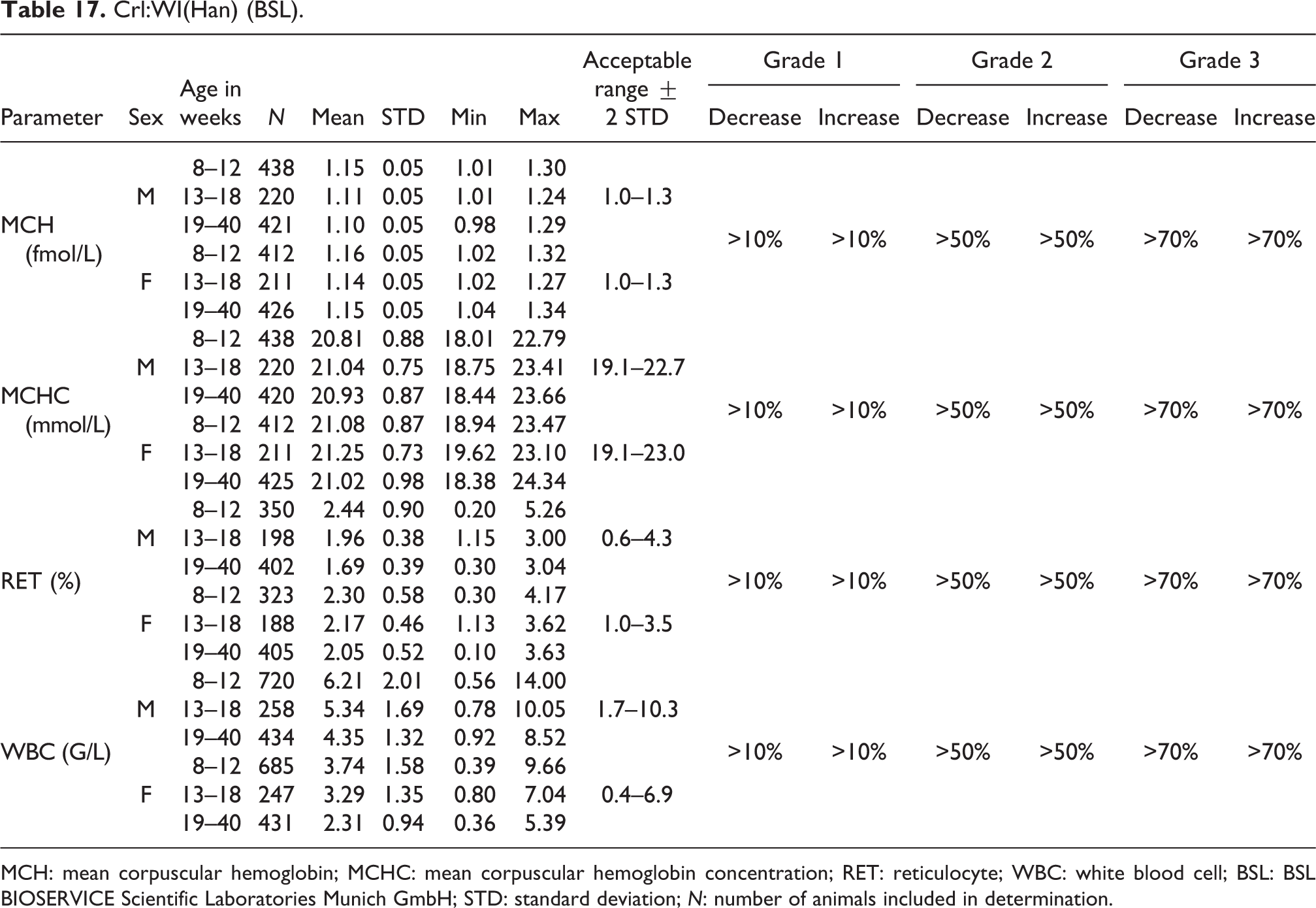
MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; RET: reticulocyte; WBC: white blood cell; BSL: BSL BIOSERVICE Scientific Laboratories Munich GmbH; STD: standard deviation; N: number of animals included in determination.
Crl:WI(Han) (BSL).
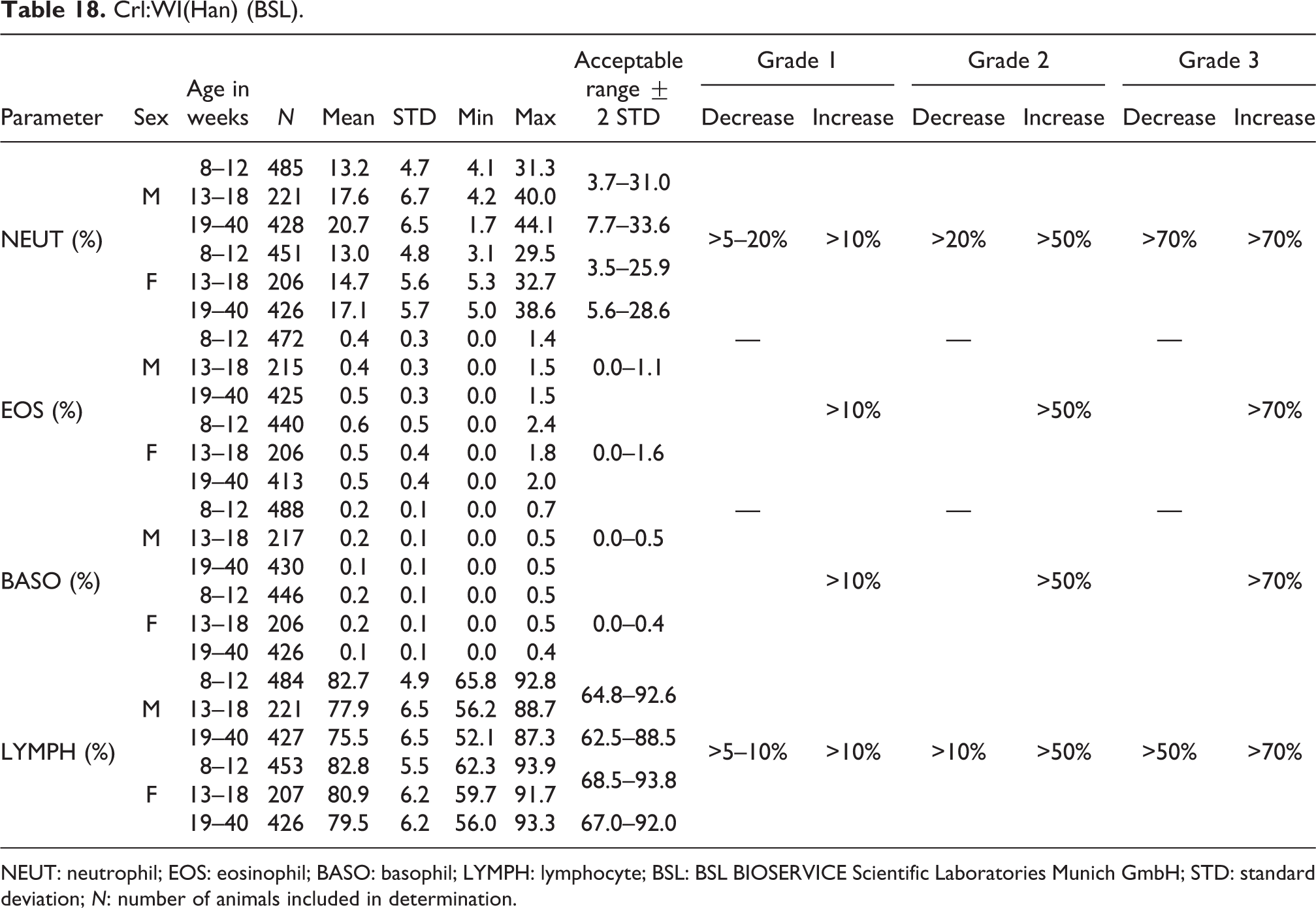
NEUT: neutrophil; EOS: eosinophil; BASO: basophil; LYMPH: lymphocyte; BSL: BSL BIOSERVICE Scientific Laboratories Munich GmbH; STD: standard deviation; N: number of animals included in determination.
Crl:WI(Han) (BSL).
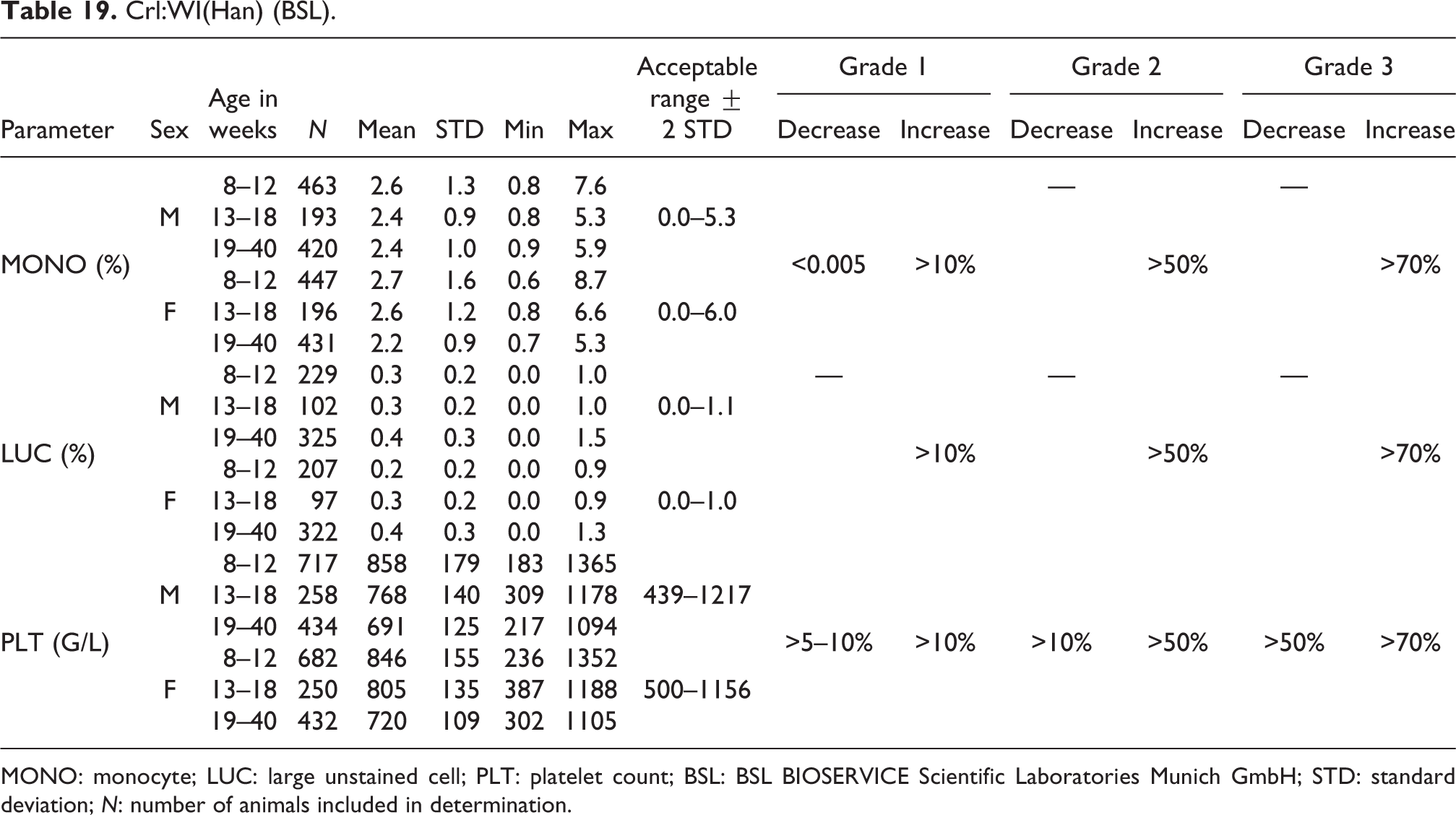
MONO: monocyte; LUC: large unstained cell; PLT: platelet count; BSL: BSL BIOSERVICE Scientific Laboratories Munich GmbH; STD: standard deviation; N: number of animals included in determination.
Values obtained from RccHan™:WIST rats
As a standard evaluation, the following parameters are included: RBC, HB, HCT, total and differential leukocyte count (WBC), MCV, MCH, MCHC, RET, large unstained cell (LUC) and PLT. 13 Additional parameters, such as erythrocyte indices MET-HB and HEINZBOD, were evaluated depending on the type of the toxicity study or test substance. Values were available from the following age-groups: ≤7 weeks, 8–12 weeks, 13–18 weeks, 19–40 weeks, 41–70 weeks, and ≥71 weeks.
RBC
RBC mean values from 8.32 to 8.88 T/L in males and 7.58 to 8.05 T/L in females were considered normal ranges for animals at ages from 8 to ≥71 weeks. In males and females of ages ≤7 weeks, the mean values were 7.27 and 7.25, respectively. Therefore, acceptable ranges were lower in young (6.4–8.1 T/L in both sexes) compared to older animals (7.0–9.8 T/L in males vs 6.5–8.9 T/L in females), whereas the ranges for females were lower than for males (see also Table 1).
HB
Mean HB values were also slightly lower in animals at ages ≤7 weeks compared to older animals (males and females: 9.1 mmol/L, with an acceptable range of 8.5–9.7 mmol/L). The acceptable data ranges were not significantly different between sexes at ages between 8 and ≥71 weeks (males, 8.3–11.1 mmol/L; females, 8.3–10.7 mmol/L) with mean values ranging from 9.7 to 10.0 mmol/L in males and from 9.5 to 9.7 mmol/L in females (see also Table 1).
HCT
Relative HCT showed mean values that were comparable and very stable between sexes and ages, whereas the mean values in males were higher compared to their female counterparts. The acceptable ranges were defined in males as 39.0–51.0%, and in females as 37.0–49.0% (see Table 2).
RDW
The mean values for the relative RDW in both sexes were comparable at different ages. Slightly lower mean values were noted in females (12.2–14.3%) than in males (13.1–15.1%). The acceptable ranges for animals ≤7 weeks of age were considered 9.1–19.1% for males and 10.1–15.3% for females. In older animals, the acceptable ranges were estimated as slightly lower for females (5.3–23.3%) than for males (6.5–23.3%) (see also Table 2).
MCV
The mean MCV value in males at ≤7 weeks of age was 59.8 fL with an acceptable range from 55.0 to 64.6 fL, which was slightly higher compared to older animals with mean values ranging from 50.7 to 54.7 fL. In females, the mean MCV value at ≤7 weeks of age was 58.6 fL with an acceptable range from 55.0 to 62.2 fL, which was slightly higher than the mean values ranging from 53.6 to 56.8 fL in older females. The acceptable ranges established for males from 8 weeks onward were from 45.9 to 59.9 fL and in their female counterparts from 48.8 to 63.0 fL (see also Table 3).
MCH
In general, mean MCH values were slightly higher in females at ages >13 weeks than in males. The highest values were noted in the youngest animals (≤7 weeks). The mean value in young males was 1.26 fmol/L compared to older animals with mean values ranging from 1.10 to 1.20 fmol/L. Although in females at ≤7 weeks of age, the mean value of 1.25 fmol/L was higher than in females between 8 and 70 weeks of age (1.20–1.22 fmol/L), the animals at ages ≥71 weeks showed a similar mean value of 1.25 fmol/L. The acceptable range was established at 1.1–1.4 fmol/L for both sexes at ≤7 weeks of age compared to older animals for which it ranged from 1.0 to 1.4 fmol/L in both sexes (see also Table 3).
MCHC
The mean MCHC values in young animals at ≤7 weeks of age were slightly lower than in older animals (males, 21.01 mmol/L vs. mean values ranging from 21.34 to 22.23 mmol/L; females, 21.35 mmol/L vs. mean values ranging from 21.84 to 22.36 mmol/L). The acceptable range was established at 19.1–24.4 for males and at 19.7–24.3 mmol/L for females at all ages (see Table 4).
HDW
HDW values were also slightly lower in young animals at ages ≤7 weeks when compared to older animals (males, 1.29 mmol/L vs. mean values ranging from 1.60 to 1.79 mmol/L; females, 1.19 mmol/L vs. mean values ranging from 1.34 to 1.48 mmol/L). In general, the values were lower in females than in males. The acceptable ranges were set at 0.9–1.7 mmol/L for males and 0.8–1.6 for females at ages ≤7 weeks, whereas for animals at ages ≥8 weeks acceptable ranges were 1.1–2.2 and 1.0–1.9 mmol/L for males and females, respectively (see also Table 4).
HEINZBOD
Several compounds can oxidize HB to MET-HB, which causes precipitation and/or formation of HEINZBODs in the erythrocytes. 20 –24 Normally in healthy rats, approximately 1% of oxyhemoglobin is metabolized into MET-HB. As no HEINZBODs were found in the RccHan™:WIST control groups in animals at ages of 8 to 70 weeks, the acceptable range is deemed to be 0.0 (see Table 5). HEINZBODs were evaluated by special staining when preparing slides only if changes in the MET-HB levels were seen in treated animals.
RET
RET values were significantly higher in young animals at ages of ≤7 weeks compared to older animals. Between 8 and 12 weeks of age, the values were still higher than in animals at ages from 13 weeks onward. At ages more than 12 weeks, lower variation was noted between animals of different ages; however, mean values in males were lower than in females.
Absolute numbers showed a mean value of 426 G/L and acceptable range of 290–562 G/L in males at ≤7 weeks of age and a mean value of 224 G/L (acceptable range of 132–316 G/L) in males at ages between 8 and 12 weeks compared to males at ages ≥13 weeks with mean values ranging from 168 to 200 G/L (acceptable range of 55–299 G/L) (see also Table 5).
For relative values, males at ≤7 weeks of age showed a mean value of 5.9% with an acceptable range of 4.0 to 7.8% and males between 8 and 12 weeks of age had a mean value of 2.9% and an acceptable range of 1.7–4.1% (compared to older animals, 13 weeks and onward, with values ranging from 1.9% to 2.3% and an acceptable range of 0.6–3.6%) (see also Table 6).
Absolute numbers in females ≤7 weeks showed a mean value of 339 G/L (acceptable range of 201–477 G/L), and a mean value of 221 G/L (acceptable range of 139–303 G/L) in females at ages between 8 and 12 weeks compared to females at ages of 13 weeks and older whose mean values ranged from 184 to 194 G/L (acceptable range of 70–306 G/L) (see also Table 5).
For relative values, females at ≤7weeks of age showed a mean value for 4.7% with an acceptable range of 2.7–6.7% and females between 8 and 12 weeks of age had a mean value of 2.8% with an acceptable range of 1.7–3.9% (compared to older animal values ranging from 2.3% to 2.5% with an acceptable range of 0.9–4.1%; see also Table 6).
Relative values for LRET (young stages) were significantly lower in animals of both sexes at ages ≤7 weeks compared to older animals. In animals at ≥71 weeks of age, the mean values tended to increase again. In young males at ≤7 weeks of age, the mean value was 44.8% (acceptable range of 21.8–67.8%) compared to animals between 8 and 70 weeks of age with mean values ranging from 56.5% to 59.7% (acceptable range of 30.7–84.3%) with slightly increased mean values in males at ages of 71 weeks and older at 64.8%. In females at ≤7 weeks of age, the mean value was 47.9% (acceptable range of 27.3–68.5%) compared to females aged 8 to 70 weeks with mean values ranging from 50.8% to 57.3% (acceptable range of 29.4–81.1%) and females older than 70 weeks with a mean value of 58.9%. In general, the female ranges, except at ≤7 weeks of age, are lower than their male counterparts (see also Table 6).
Medium fluorescence reticulocytes (MRET) showed similar values for animals from both sexes at ages ≤7 to 70 weeks (males had mean values from 30.9% to 34.5% and an acceptable range of 19.3 44.5%; females had means ranging from 32.3% to 35.2% and an acceptable range of 21.1–44.7%). In animals older than 70 weeks, the mean value was 27.1% for males and 29.4% for females, demonstrating a reduction with increasing age. Therefore, the acceptable ranges for males and females ≥71 weeks old were set at 16.7–37.5 and 16.6–42.2%, respectively (see also Table 7).
The mean values for mature, HRETs varied in wide ranges. In males, there were mean values from 8.0% to 20.7% and in females, there were mean values between 9.1% and 16.9%. Due to the wide variation, the acceptable ranges were set from 0.0% to 33.4% for males and 0.0% to 41.9% for females at ages from 8 weeks onward and from 0.0% to 49.7% for males and 0.0% to 42.3% for females at ages ≤7 weeks (see also Table 7).
WBC
The WBC values for males were slightly higher than for their female counterparts, whereas the mean values for both sexes increased significantly from 8 to 12 weeks compared to younger animals. Thereafter, mean values showed a reduction with increasing age whereas the values increased again in animals at 71 weeks and older. The mean values for males ranged from 5.64 to 7.19 G/L with an acceptable range from 2.8 to 8.6 G/L at ages of ≤7 and from 41 weeks onward, and 3.2 to 10.5 G/L at ages between 8 and 40 weeks. The mean values for females ranged from 3.26 to 5.0 G/L with an acceptable range from 0.9 to 6.8 G/L in females at ages of ≤7 and from 41 weeks onward, and 1.4 to 7.7 G/L at ages between 8 and 40 weeks (see also Table 8).
NEUT
Absolute NEUT numbers were lowest in animals aged ≤7 weeks and highest in animals at ≥71 weeks. In males, the mean value at ≤7 weeks was 0.57 G/L (acceptable range of 0.0–1.2 G/L) and at ≥71 weeks was 2.03 G/L (acceptable range of 0.0–4.8 G/L). In males from 8 to 70 weeks old, the mean value ranged from 1.02 to 1.41 G/L (acceptable range of 0.0–2.8 G/L). In females, the mean value at ≤7 weeks was 0.43 G/L (acceptable range of 0.0–1.0 G/L) and in animals ≥71 weeks was 1.45 G/L (acceptable range of 0.0–3.4 G/L). In females of ages from 8 to 70 weeks, the mean values ranged from 0.65 to 0.85 G/L (acceptable range of 0.0–1.6 G/L) (see also Table 8).
Relative NEUT values increased with age, with mean values ranging from 10.5% to 33.1% in males and from 10.4% to 33.8% in females, across the different age-groups. The acceptable ranges are different for different age-groups, starting from 0.8–20.0% in animals at ≤7 weeks of age up to 12.6–55.0% in animals at ages ≥71 weeks (see also Table 9).
EOS
Mean absolute values for EOS ranged from 0.07 to 0.13 G/L with an acceptable range of 0.0–0.3 G/L in males and from 0.07 to 0.09 G/L in females with an acceptable range of 0.0–0.2 for all ages (see Table 9).
Relative EOS values were slightly higher in animals at ages from 19 weeks onward compared to younger animals. In males from ≤7 to 18 weeks, the mean values were from 1.2% to 1.4%, and in older animals between 1.9% and 2.3%. In females from ≤7 to 18 weeks, the mean was 1.4% to 1.6%, and in older animals from 2.1% to 2.5%. The acceptable ranges were established as 0.0–3.9% for males and 0.0–4.6% for females (see Table 10).
BASO
The absolute mean number of BASO was low in both sexes at all ages with no significant difference between females and males. The acceptable range was 0.0 to 0.1 G/L for both sexes and all ages (see Table 10). Similarly, the acceptable relative value range was established at 0.0–1.4% for males and at 0.0–1.5% for females (see Table 11).
LYMPH
The absolute mean values decreased slightly at ages at and above 19 weeks in both sexes. For males ≤7 weeks old, the mean value was 4.66 G/L, from 8 to 40 weeks, between 4.64 and 5.83 G/L, and in older males, between 3.54 and 3.82 G/L. For females at ages of ≤7 weeks, the mean value was 3.76 G/L, from 8 to 40 weeks of age, between 2.77 and 4.01 G/L, and in older females, between 2.25 and 2.28 G/L. The acceptable range for males at ages of ≤7 weeks was established at 2.4–6.9 G/L, for ages between 8 and 40 weeks, at 2.1–8.8 G/L, and for older males, at 1.8–5.8 G/L. The acceptable range for females at ages of ≤7 weeks was established at 2.0–5.5 G/L, for ages from 8 to 40 weeks, at 0.9–6.5, and for older females, at 0.5–4.0 G/L (see also Table 11).
Relative lymphocyte (LYMPH) values behaved similarly. For males at ages of ≤7 weeks, the mean value was 85.5%; between 8 and 40 weeks, from 74.3% to 80.8%; and between 62.4 and 67.3% in older males. In females at ages of ≤7 weeks, the mean value was 84.1%; from 8 to 40 weeks, between 75.4 and 80.3%; and between 59.6 and 69.6% in older females (see also Table 12).
MONO
The absolute mean values were slightly higher in males (0.10–0.16 G/L) than in females (0.07–0.12 G/L). There were no differences between female and male ranges at different ages, however, an increase in both sexes can be seen for older animals. The acceptable range was set at 0.0–0.3 G/L for males and females (see Table 12).
The relative MONO values showed a minor increase in animals aged 19 weeks and older. In males aged ≤7 to 18 weeks, the mean value ranged from 1.8% to 1.9% (acceptable range of 0.4–3.2%). In older males, the mean values ranged from 2.2% to 2.8% (acceptable range of 0.8–4.6%). In females aged ≤7 to 18 weeks, the mean value ranged between 1.7% and 1.8% (acceptable range of 0.4–3.2%), and in older females, the mean values ranged from 2.0% to 3.1% (acceptable range of 0.6–5.1%) (see also Table 13).
LUC
LUC mean values ranged from 0.02 to 0.06 G/L for both sexes and all age-groups. Therefore, the acceptable range was established at 0.0 to 0.2 G/L for both sexes (see Table 13).
Relative LUC mean values ranged from 0.8% to 0.9% for males, with an acceptable range of 0.0–1.9%. For females, the relative mean values were between 0.7% and 0.9%, with an acceptable range of 0.0–1.7% (see also Table 14).
PLT
The absolute mean values were slightly higher in both sexes at ages from ≤7 weeks to 18 weeks compared to mean values from older animals. In males, at ages from ≤7 to 18 weeks, the mean values ranged from 976 to 1234 G/L (acceptable range of 702–1534 G/L), and in older males, the mean values ranged from 912 to 948 G/L (acceptable range of 564–1332 G/L). In females at ages from ≤7 to 18 weeks, the mean values ranged from 1023 to 1285 G/L (acceptable range of 753–1637 G/L), and in older females, the mean values ranged from 863 to 963 G/L (acceptable range of 517–1229 G/L) (see also Table 14).
MET-HB
Mean relative MET-HB values ranged from 0.8% to 1.0% for both sexes and all age-groups. The acceptable range was established as 0.4% to 1.4% (see Table 15).
Values obtained from Crl:WI(Han) rats
Obtained outliers were examined on available histopathology data. 25 Therefore, outliers were associated with histopathology findings like, for example, inflammatory pulmonary lesions following vehicle aspiration or spontaneous sperm granuloma. After excluding all statistically identified outliers, the following values from the standard parameter list were evaluated: RBC, HB, HCT, WBC, MCV, MCH, MCHC, RET, LUC, and PLT. Values were only available for the 8–12, 13–18, and 19–40 week age-groups.
RBC
Mean RBC values in animals at ages of 8–40 weeks ranged from 8.51 to 9.07 T/L (acceptable range of 7.1–10.3 T/L) for males and were somewhat lower for females, 8.01 to 8.26 T/L (acceptable range of 6.9–9.3 T/L; see also Table 16).
HB
Mean HB values in animals at ages of 8–40 weeks ranged from 9.9 to 10.0 mmol/L (acceptable range of 8.6–11.1 mmol/L) for males and were slightly lower for females, 9.3 to 9.4 mmol/L (acceptable range of 8.1–10.5 mmol/L; see also Table 16).
HCT
Relative HCT values were slightly higher in males (mean values ranging from 47.4% to 47.6%; acceptable range of 40.8–54.1%) than in females (mean values ranging from 44.1% to 44.4%; acceptable range of 38.2–49.9%; see also Table 16).
RBC values for all age-groups were higher in males than in females. While the female RBC values were fairly constant, values in males increased with age. In contrast, HB and HCT values were constant throughout all age-groups but were higher in males than in females (Figure 1).
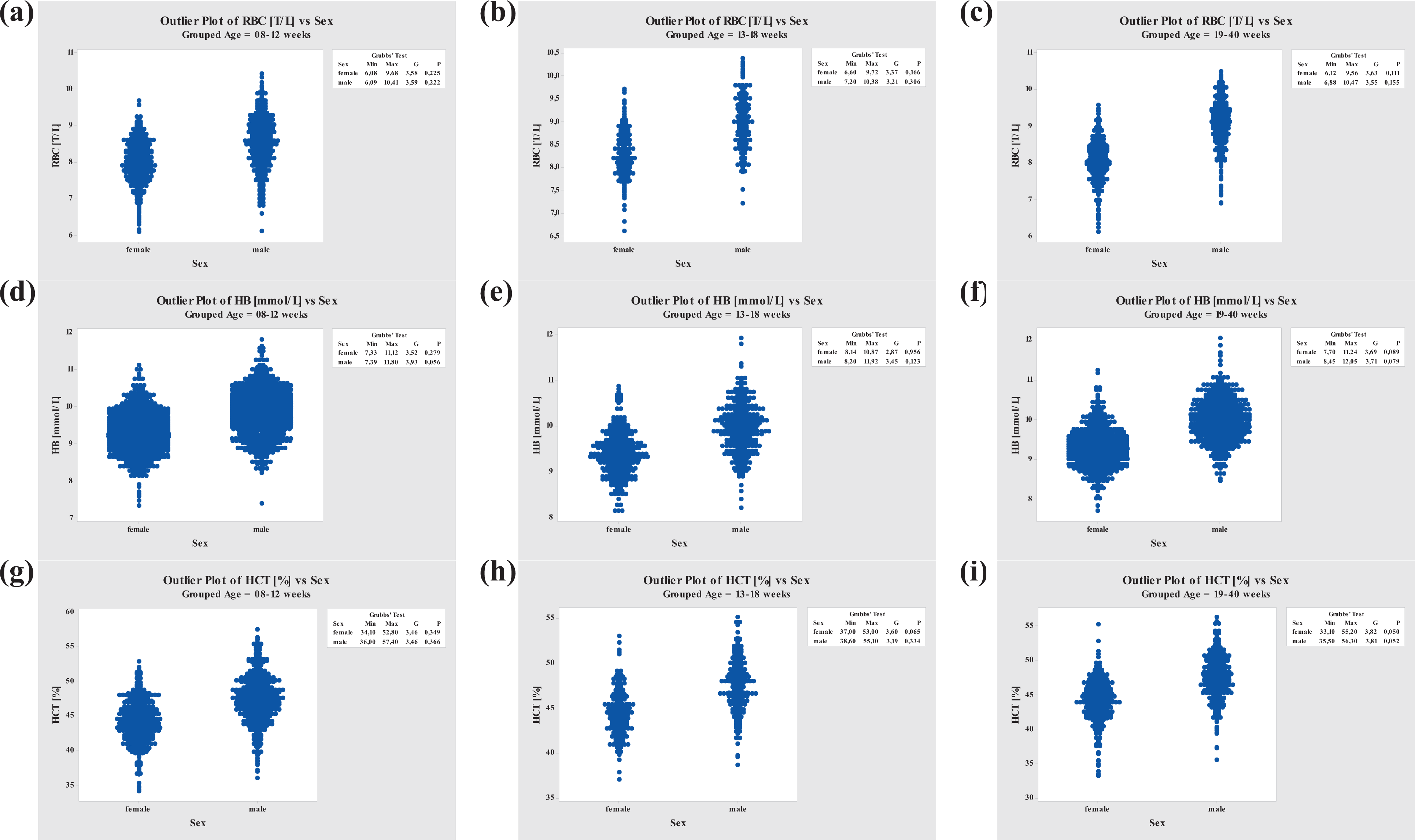
Grubbs’ Outlier Test performed for RBC, HB, and HCT. RBC: red blood cell; HB: hemoglobin; HCT: hematocrit.
MCV
The mean MCV values in animals between 8 and 40 weeks old were similar in males and females (males, 52.5 to 55.4 fL; females, 53.5 to 55.0 fL), as were the acceptable ranges for males, 48.4 to 60.7 fL, and for females, 49.5 to 59.6 fL (see Table 16).
MCH
The MCH acceptable ranges in 8- to 40-week-old animals were the same for both sexes, reaching from 1.0 to 1.3 fmol/L (see Table 17).
MCHC
Mean MCHC values were similar for both sexes (males, 20.81 to 21.04 mmol/L; females, 21.02 to 21.25 mmol/L). The acceptable ranges were slightly different between sexes, 19.1 to 22.7 mmol/L and 19.1 to 23.0 mmol/L, respectively, for males and females (see Table 17).
MCV, MCH, and MCHC values were generally slightly higher in females than in males. Females and males at ages between 8 and 12 weeks showed rather constant MCV and MCH values but varied at higher ages, whereas the MCHC values remained constant throughout all age-groups (Figure 2).
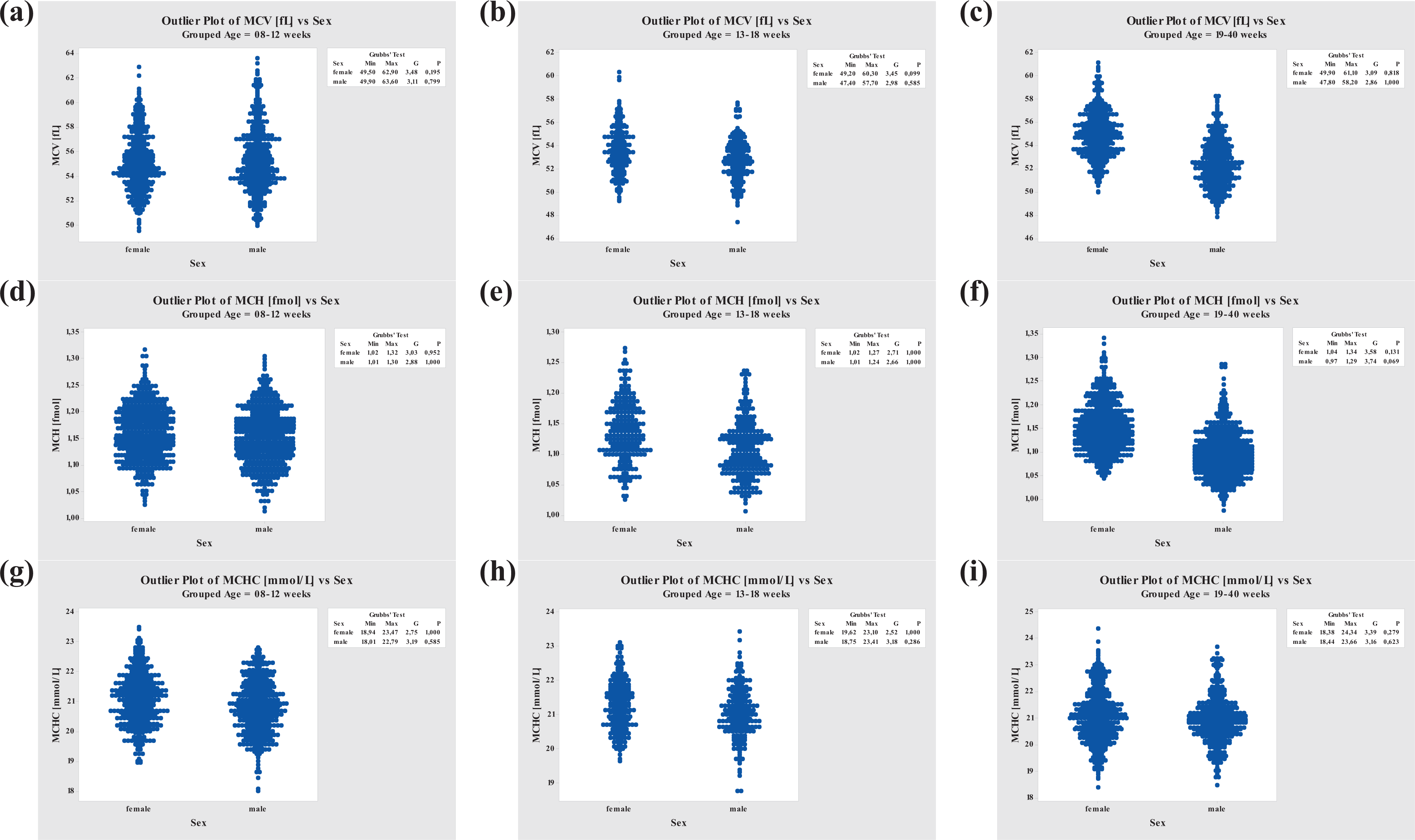
Grubbs’ Outlier Test performed for MCV, MCH, and MCHC. MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration.
RET
For relative RET values, again there were similarities between sexes and among all age-groups (males, 1.69 to 2.44%; females, 2.05 to 2.30%) with acceptable ranges of 0.6–4.3% for males and 1.0–3.5% for females. The RET count decreased with increasing age. The decrease through age-groups was more significant in males than females (see also Table 17).
WBC
The mean WBC values were higher in males (4.35–6.21 G/L, acceptable range of 1.7–10.3 G/L) than in females (2.31–3.74 G/L, acceptable range of 0.4–6.9 G/L). Similar to the RET count, the WBC values decreased in older animals, again more significantly in males than in females (see also Table 17).
NEUT
The mean relative values for NEUT were higher in males (13.2–20.7%) than in females (13.0–17.1%) with acceptable ranges at ages between 8 and 18 weeks of 3.7% to 31.0% in males and 3.5–25.9% in females. The acceptable range for animals between 19 and 40 weeks of age increased for both males (7.7–33.6%) and females (5.6–28.6%; see also Table 18).
Figure 3 demonstrates that WBC and RET decreased with increasing age in both sexes, whereas RET values were slightly higher and WBC values were significantly lower in females compared to their male counterparts. In contrast, the NEUT values were higher in males than in females and varied more the older the animals were. However, the values were very similar between animals of both sexes at ages between 8 and 12 weeks.
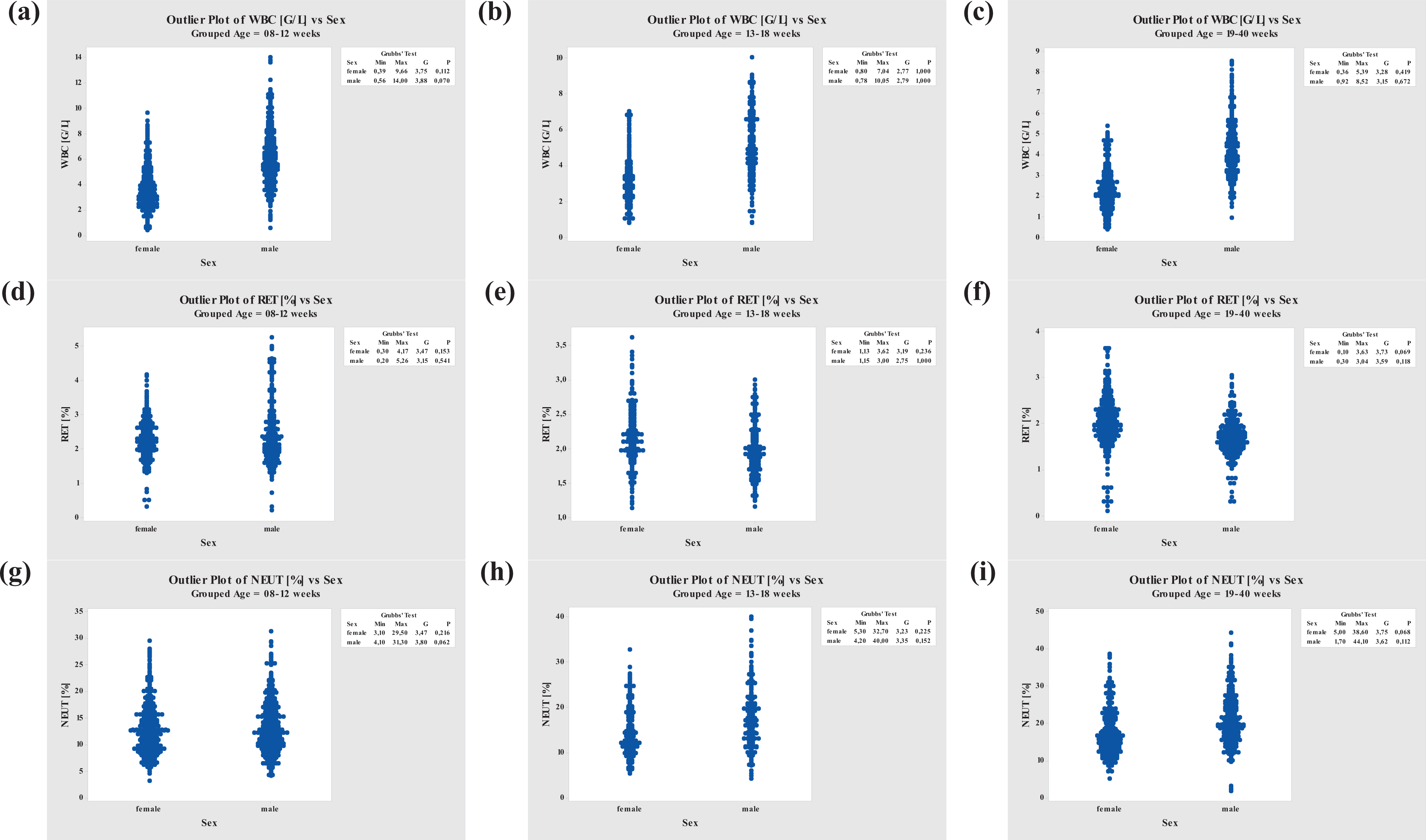
Grubbs’ Outlier Test performed for WBC, RET, and NEUT. WBC: white blood cell; RET: reticulocyte; NEUT: neutrophil.
EOS
The mean relative EOS values were similar for males (0.4–0.5%) and females (0.5 to 0.6%) with acceptable ranges of 0.0–1.1% for males and 0.0–1.6% for females (see Table 18).
BASO
The mean relative BASO values were exactly the same for males and females (0.1–0.2%) at all ages, decreasing in older individuals, with acceptable ranges of 0.0–0.5% for males and 0.0–0.4% for females (see Table 18).
LYMPH
Relative LYMPH values were higher in females than in males, reaching 75.5% to 82.7% and 79.5% to 82.8% in males and females, respectively, with acceptable ranges of 64.8–92.6% for males and 68.5–93.8% for females at ages between 8 and 18 weeks. The acceptable ranges for animals at ages between 19 and 40 weeks decreased for both males (62.5–88.5%) and females (67.0–92.0%; see also Table 18).
MONO
Mean relative MONO values were similar for males (2.4–2.6%) and females (2.2–2.7%) with respective acceptable ranges of 0.0–5.3 and 0.0–0.6% for males and females. MONO values were constant throughout all age-groups (see also Table 19).
LUC
The mean relative values for LUCs were similar in both sexes but increased minimally throughout the age-groups. The values ranged in both sexes from 0.2% to 0.4% with acceptable ranges of 0.0–1.1% for males and 0.0–1.0% for females (see Table 19).
PLT
Mean platelet values are relatively similar in males (691–858 G/L) and females (720–846 G/L) with acceptable ranges of 439–1217 G/L for males and 500–1156 G/L for females. PLT decreased with increasing age, somewhat more in males than in females (see also Table 19).
For further information, please see supplementary data Figures S1 and S2.
As already mentioned, all hematology parameters (except MET-HB) were measured with the ADVIA 120 Hematology System. The ADVIA 120 is an instrument with a well-established clinical track record and years of successful use in a large number of laboratories worldwide. It was also compared to newer systems like the ADVIA 2120 by Harris et al. 26 Overall, both systems showed a very high similarity, thus the confidence intervals of both systems are comparable. 27
Within BSL, a small stability study with 19 untreated animals was done, the results showed that the samples remain stable for 72 h at 2–8°.
Comparison of parameters from different study types
Selected parameters have been summarized and compared for Crl:WI(Han) rats in Figures 4 to 6 .
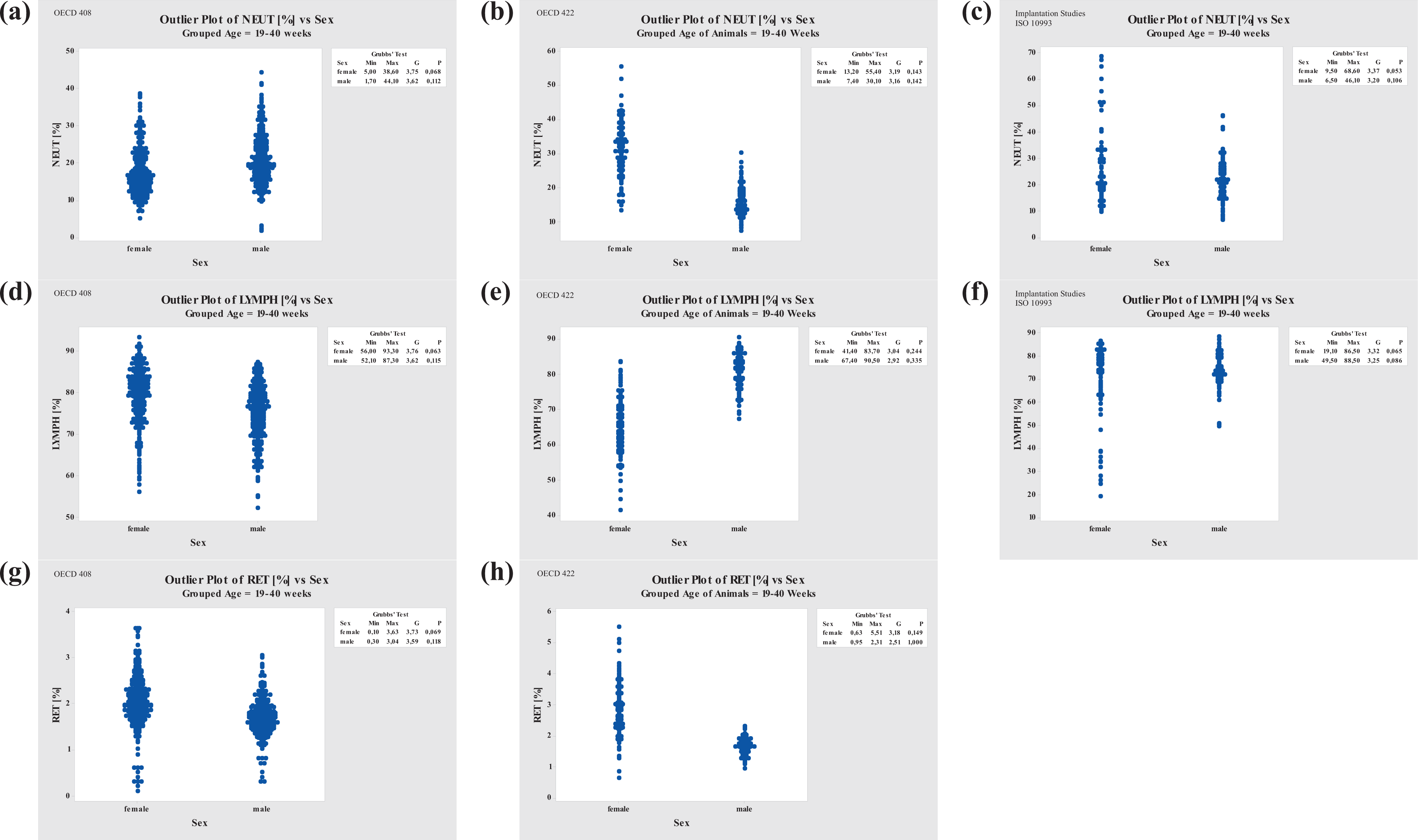
Grubbs’ Outlier Test comparison of NEUT, LYMPH, and RET for three different study types. Left column, repeated-dose studies (OECD 408); middle column, reproduction toxicity studies (OECD 422); right column, implantation studies (ISO 10993). NEUT: neutrophil; LYMPH: lymphocyte; RET: reticulocyte.
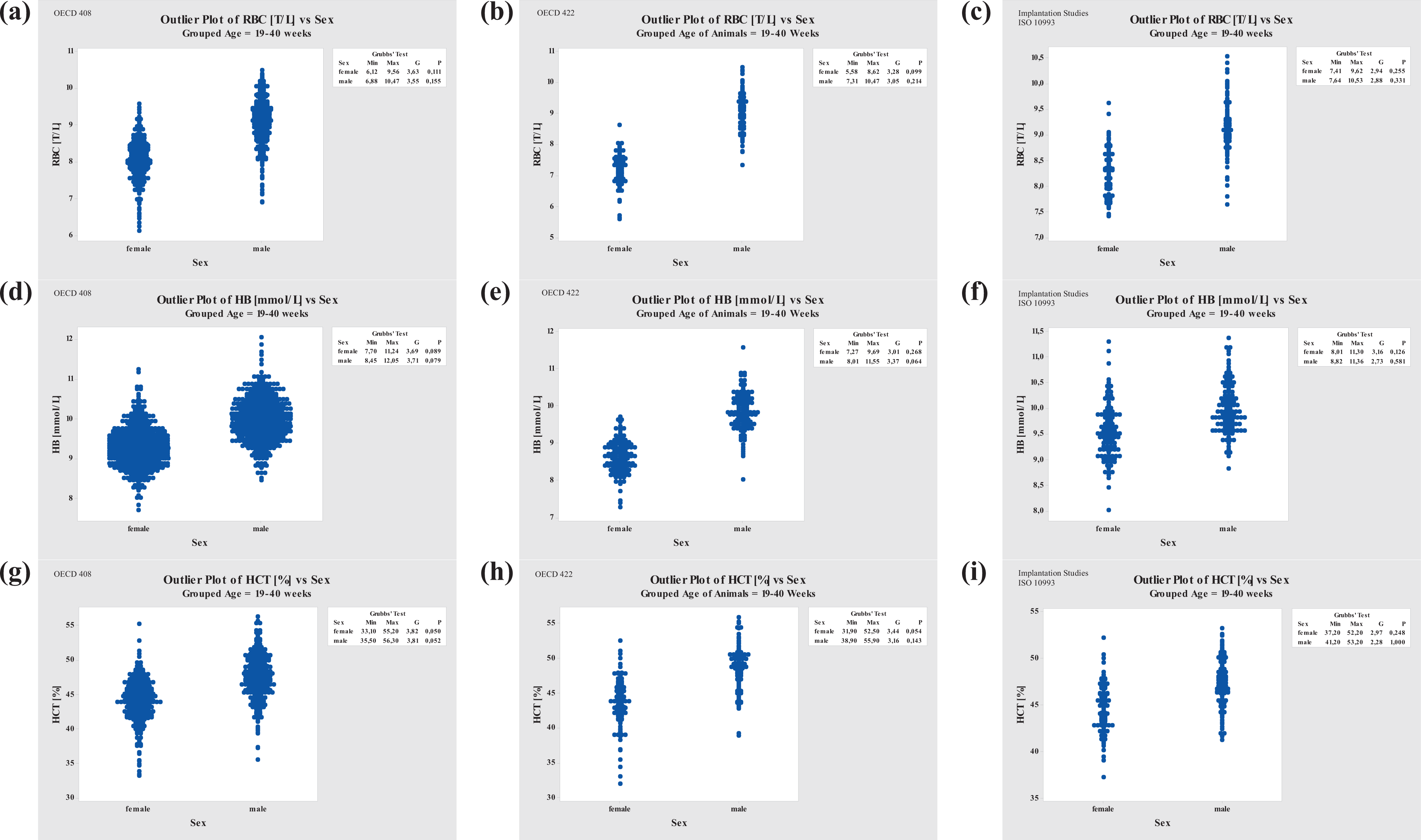
Grubbs’ Outlier Test comparison of RBC, HB, and HCT for three different study types. Left column, repeated-dose studies (OECD 408); middle column, reproduction toxicity studies (OECD 422); right column, implantation studies (ISO 10993). RBC: red blood cell; HB: hemoglobin; HCT: hematocrit.
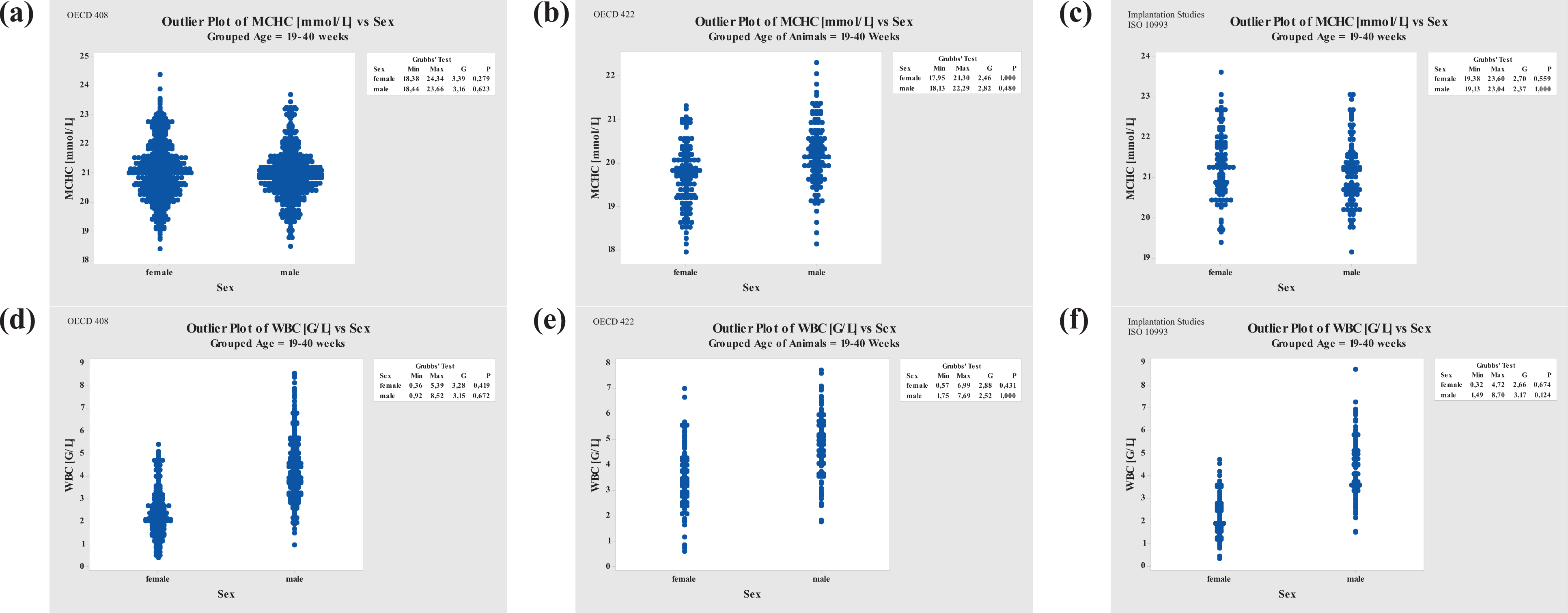
Grubbs’ Outlier Test comparison of MCHC and WBC for three different study types. Left column, repeated-dose studies (OECD 408); middle column, reproduction toxicity studies (OECD 422); right column, implantation studies (ISO 10993). MCHC: mean corpuscular hemoglobin concentration; WBC: white blood cell.
A comparison of repeated-dose toxicity studies (OECD 408) 10 with reproduction (OECD 422) 28 and implantation (ISO 10993) 29 studies revealed several differences in hematology parameters.
After variance analysis, almost every parameter among males from repeated-dose and reproduction toxicity studies resulted in unexpected high variation. Males at the end of reproduction toxicity studies (OECD 422) were about 18–19 weeks old. Animals at that age were included in age-groups 13–18 and 19–40 weeks for better comparability because those at the end of repeated-dose toxicity (OECD 408) and implantation studies were older than 19 weeks. A second variance analysis comparing reproduction study males at the age of 18–19 weeks with repeated-dose toxicity study males at the age of 13–18 weeks only showed variation in MCHC and WBC.
Reproduction study females compared to repeated-dose toxicity study females showed differences in almost every parameter except RBC, HCT, HB, and MCH, as expected.
Implantation study males were rather similar to males in repeated-dose toxicity studies, whereas females showed more variances, especially in the differential leukocyte count.
In repeated-dose studies with animals at ages from 19 to 40 weeks, NEUT values were slightly higher in males than in females. In contrast, in reproductive toxicity studies, the NEUT ranges were higher in females than in males. The male NEUT values in reprotoxicity studies were lower compared to implantation and repeated-dose studies due to their real age of 18–19 weeks, that means, these animals were at the lower limit of their age-group. When comparing the mean values of reprotoxicity study males with repeated-dose study males, no significant difference was noted. Therefore, NEUT values in males are deemed to have remained relatively constant throughout the different study types. Finally, in implantation studies, higher variation was noted for females, whereas the NEUT values in males were slightly lower than in their female counterparts (Figure 4).
In repeated-dose toxicity studies, LYMPH values shifted toward lower values with increasing age (higher shift in males than in females). In young animals (8–12 weeks), there were no differences for LYMPH in either sex. By contrast, LYMPH values decreased in females from reproduction and implantation studies compared to females from repeated-dose studies, whereas at first glance, male LYMPH values in reprotoxicology studies seemed to be higher. However, due to their real age as mentioned above, male LYMPH values can be said to be comparable for all the different study types analyzed (Figure 4).
Generally speaking, RET values varied in high ranges in all studies. Comparing RET values of reprotoxicology with repeated-dose toxicity studies, a clear indication of increased RET values was noted in females in reprotoxicology studies (Figure 4).
RBC, HB, and HCT values were somewhat invariable among the different study-type categories. To confirm this suspicion, a variance analysis was done, verifying that RBC, HB, and HCT values of all three study types are comparable (Figure 5).
MCHC values decreased slightly in females and males from reproduction studies compared to repeated-dose studies, whereas no changes were noted in implantation studies (Figure 6).
WBC values showed significant differences between sexes in reprotoxicology studies compared to repeated-dose studies and were not comparable, based on variance analysis, whereas WBC values from repeated-dose toxicity and implantation study animals were comparable (Figure 6).
Number, mean values, and standard deviations are summarized in Table 20. Other hematology parameters were not compared. For further information, see supplementary data Figures S3 to S5.
Number, mean values, and STDs of RBC, HB, HCT, MCHC, RET, WBC, NEUT, and LYMPH for three different study types: Repeated-dose studies (OECD 408), reproduction toxicity studies (OECD 422), and implantation studies (ISO 10993).
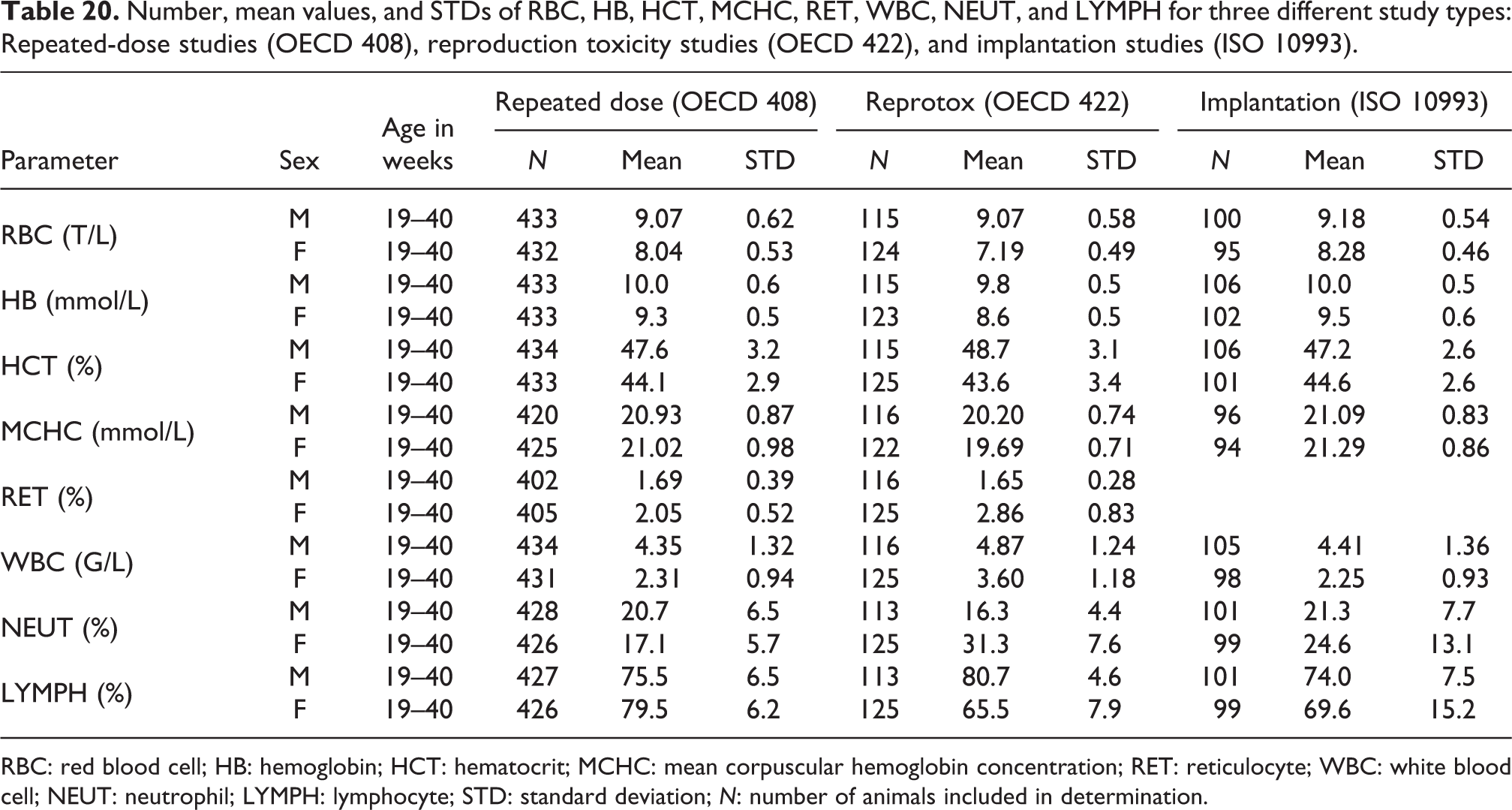
RBC: red blood cell; HB: hemoglobin; HCT: hematocrit; MCHC: mean corpuscular hemoglobin concentration; RET: reticulocyte; WBC: white blood cell; NEUT: neutrophil; LYMPH: lymphocyte; STD: standard deviation; N: number of animals included in determination.
Vehicle effects
The following vehicles were used in repeated-dose toxicity studies performed with Crl:WI(Han) rats: aqua ad injectionem, control diet, corn oil and dried corn oil, cotton seed oil, dimethyl sulfoxide (DMSO) in water, DMSO/polyethylene glycol 400 (PEG400) 1:5, hydroxyethyl cellulose, methylcellulose, NaCl 0.9%, paraffinum liquidum, polyethylene glycol 300 (PEG300), PEG400, and sesame oil.
Most data were available for studies performed with aqua ad injectionem (water) and corn oil. Therefore, vehicle effects were statistically evaluated only for these vehicles (variance analysis, Levene’s test, 30 Bonett’s test 31 ). For other vehicles, the sample size was insufficient to perform statistical analyses (Figures 7 to 14 ).
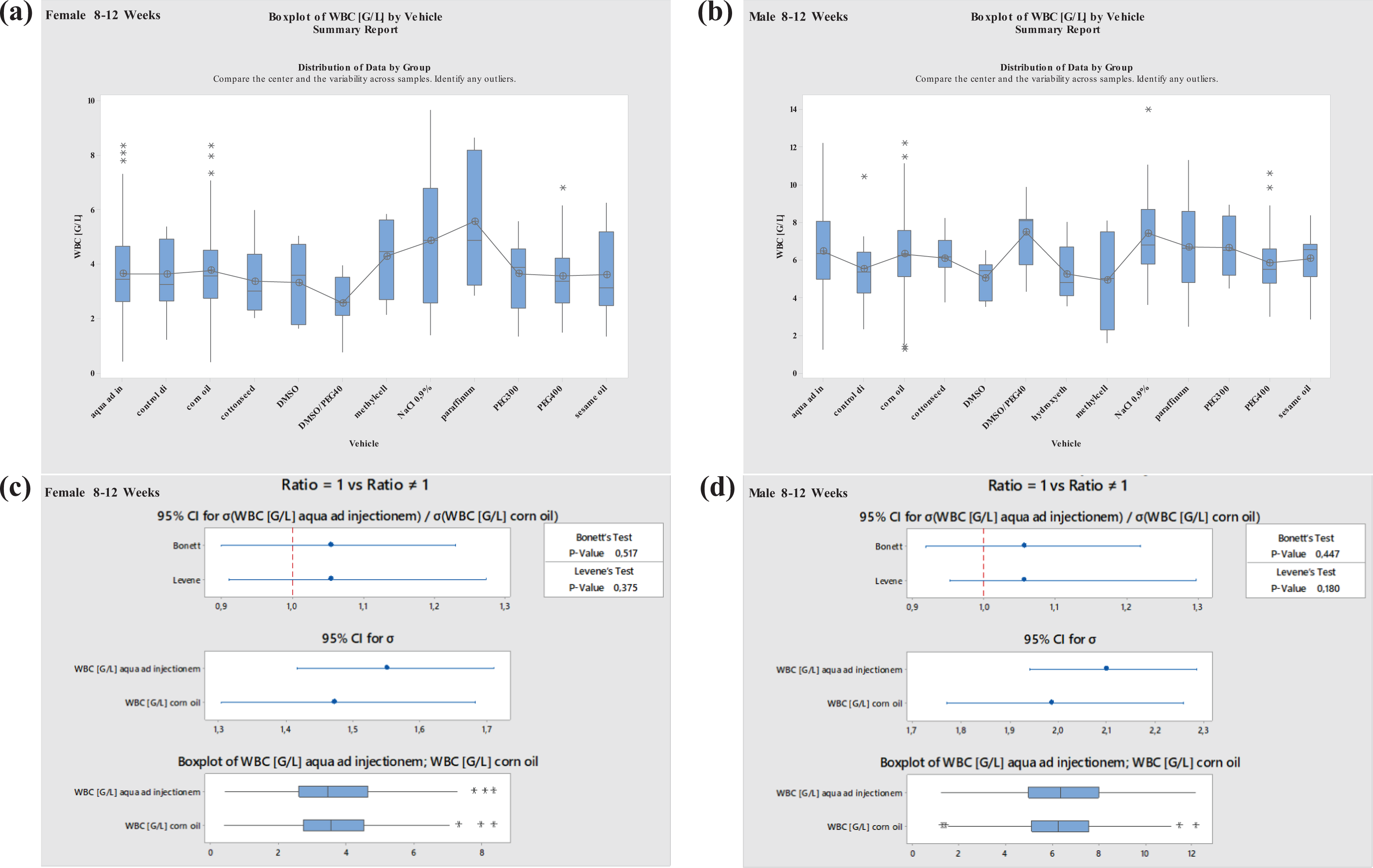
Boxplots—Comparison of different vehicles possibly impacting hematology parameter WBC in males and females at 8–12 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, cotton seed oil, DMSO in water, DMSO/PEG400 1:5, hydroxyethyl cellulose, methylcellulose, NaCl 0.9%, paraffinum liquidum, PEG300, PEG400, sesame oil. WBC: white blood cell; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400; PEG300: polyethylene glycol 300.
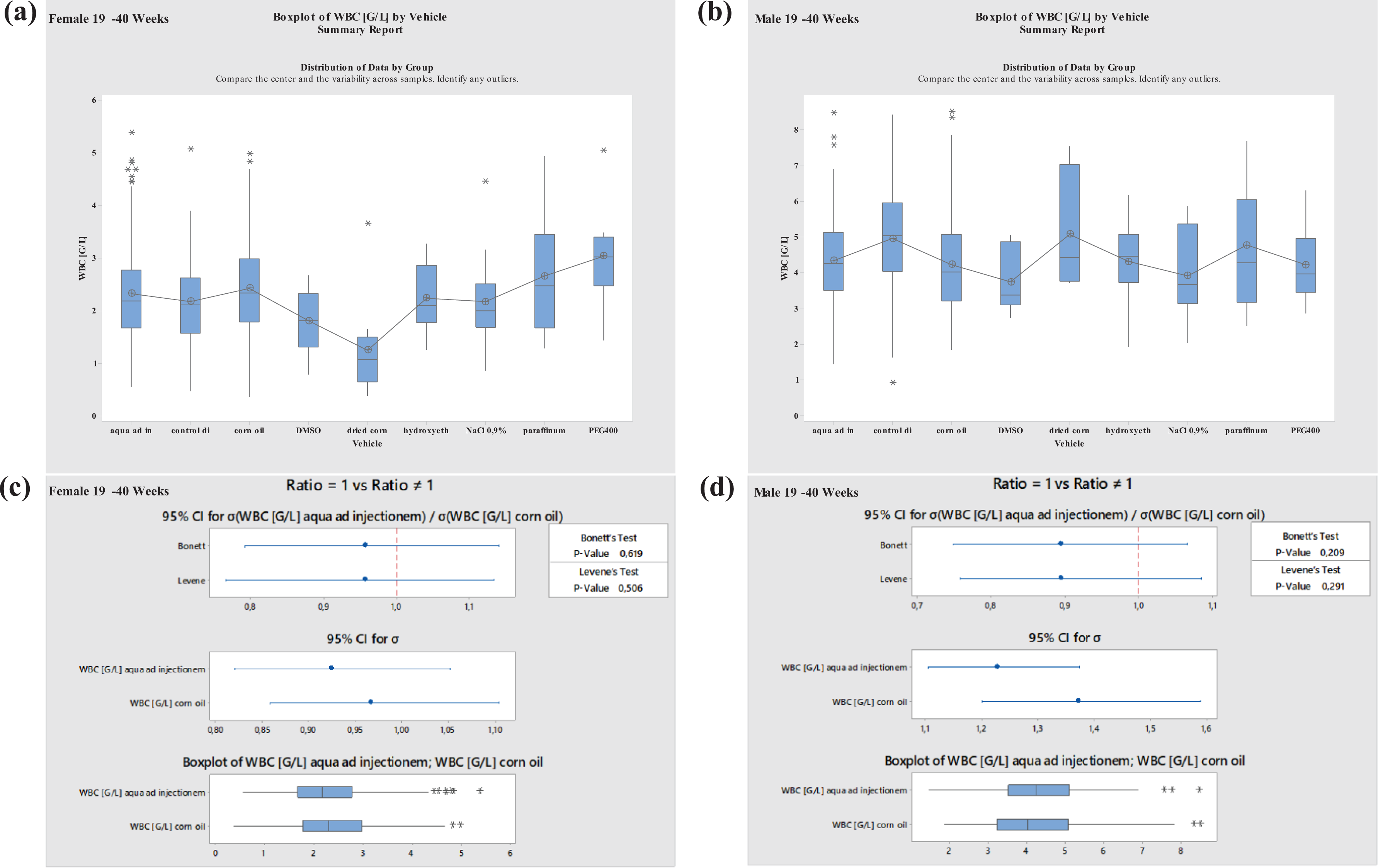
Boxplots—Comparison of different vehicles possibly impacting hematology parameter WBC in males and females at 19–40 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, DMSO in water, dried corn oil, hydroxyethyl cellulose, NaCl 0.9%, paraffinum liquidum, PEG400. WBC: white blood cell; PEG400: polyethylene glycol 400; DMSO: dimethyl sulfoxide.
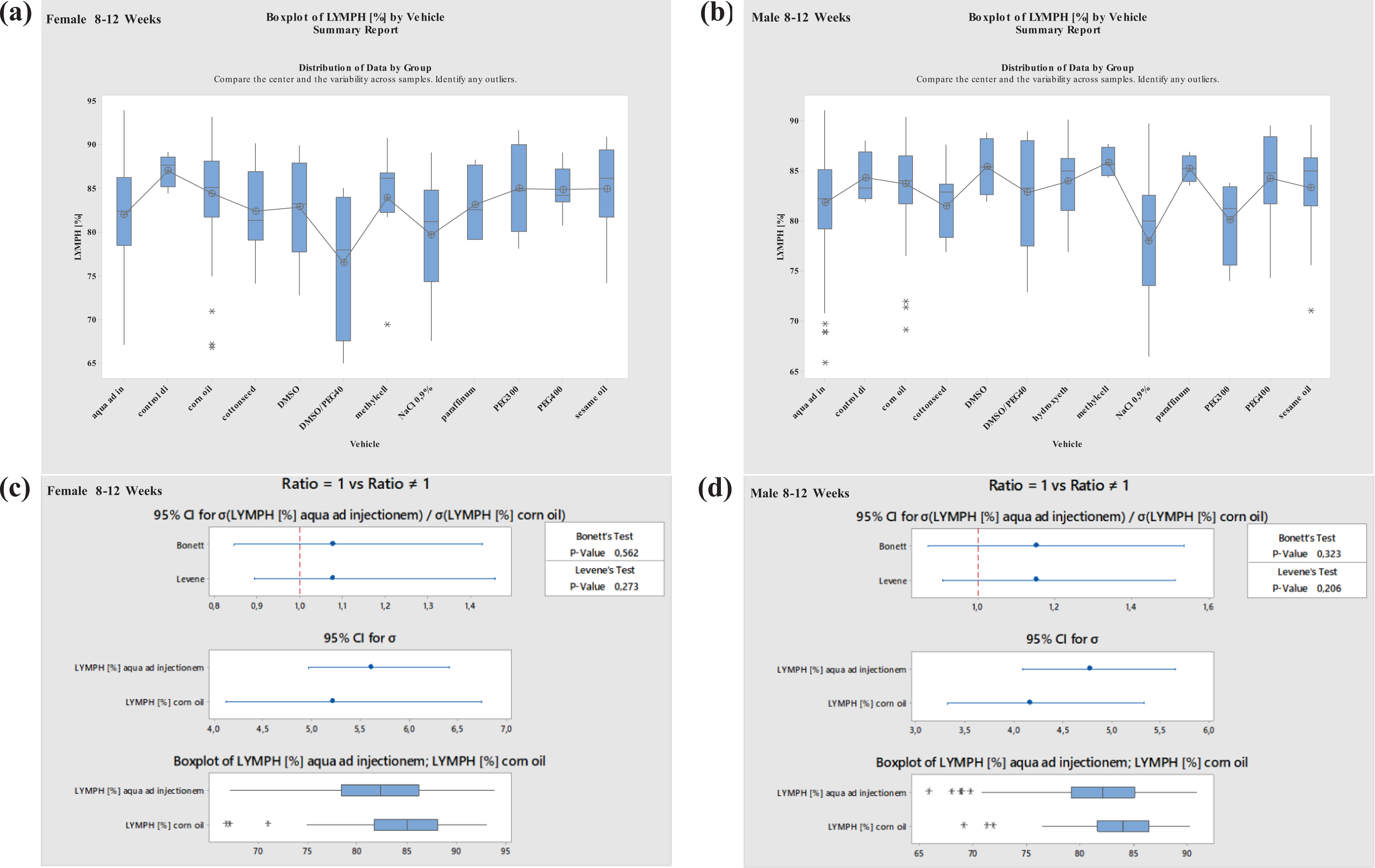
Boxplots—Comparison of different vehicles possibly impacting hematology parameter LYMPH in males and females at 8–12 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, cotton seed oil, DMSO in water, DMSO/PEG400 1:5, hydroxyethyl cellulose, methylcellulose, NaCl 0.9%, paraffinum liquidum, PEG300, PEG400, sesame oil. LYMPH: lymphocyte; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400; PEG300: polyethylene glycol 300.
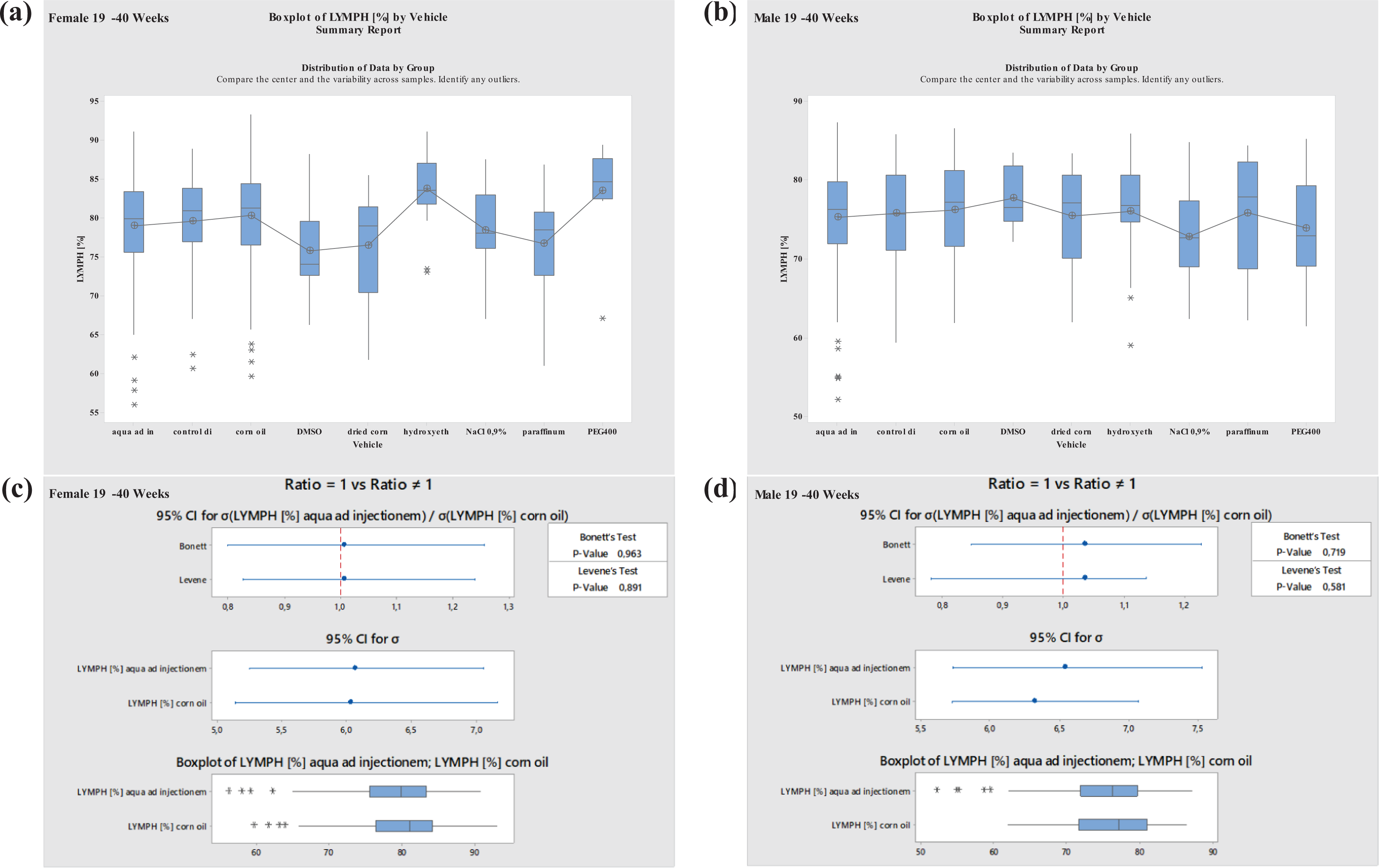
Boxplots—Comparison of different vehicles possibly impacting hematology parameter LYMPH in males and females at 19–40 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, DMSO in water, dried corn oil, hydroxyethyl cellulose, NaCl 0.9%, paraffinum liquidum, PEG400. LYMPH: lymphocyte; PEG400: polyethylene glycol 400; DMSO: dimethyl sulfoxide.
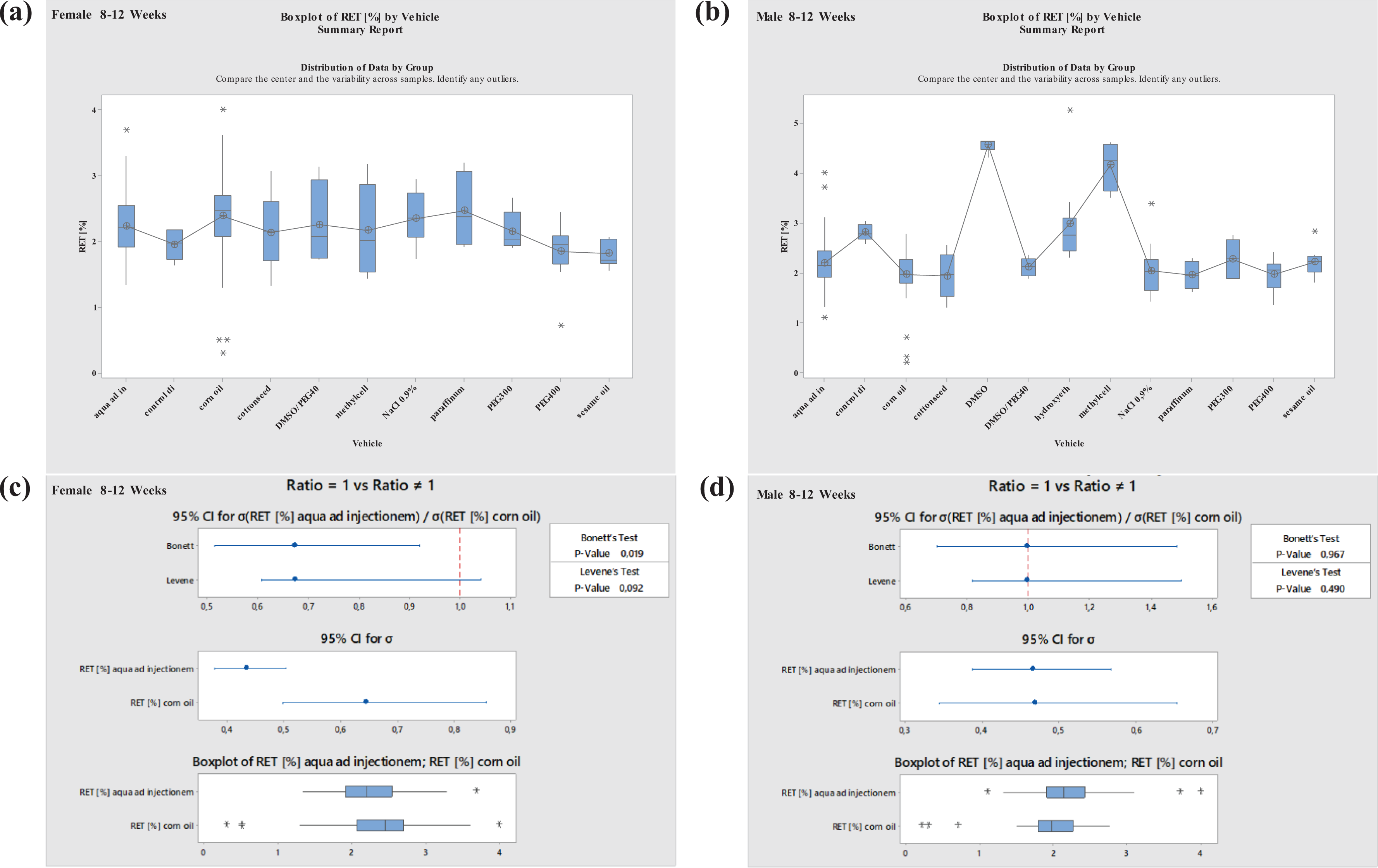
Boxplots—Comparison of different vehicles possibly impacting hematology parameter RET in males and females at 8–12 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, cotton seed oil, DMSO in water, DMSO/PEG400 1:5, hydroxyethyl cellulose, methylcellulose, NaCl 0.9%, paraffinum liquidum, PEG300, PEG400, sesame oil. RET: reticulocyte; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400; PEG300: polyethylene glycol 300.
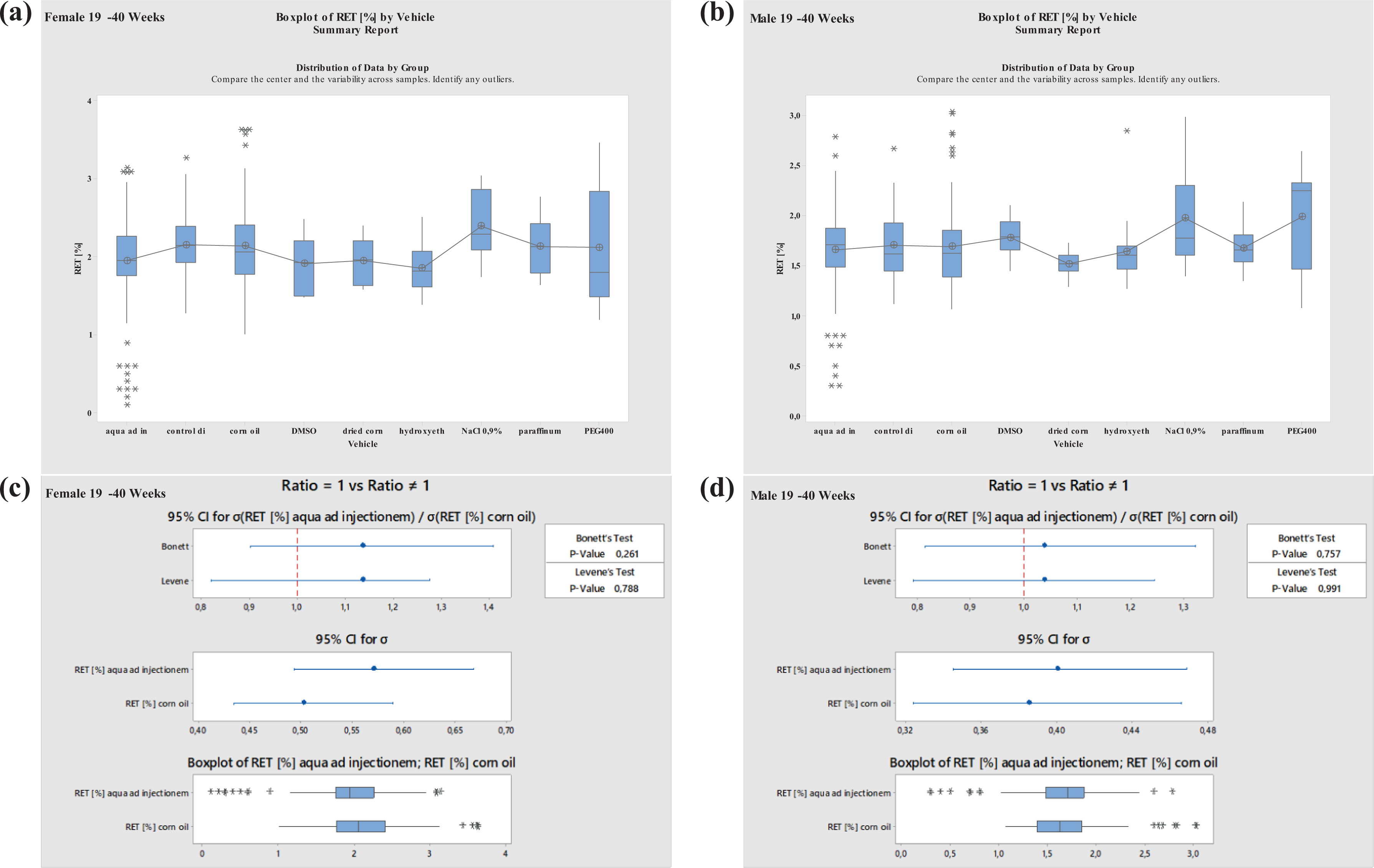
Boxplots—Comparison of different vehicles possibly impacting hematology parameter RET in males and females at 19–40 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, DMSO in water, dried corn oil, hydroxyethyl cellulose, NaCl 0.9%, paraffinum liquidum, PEG400. RET: reticulocyte; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400.
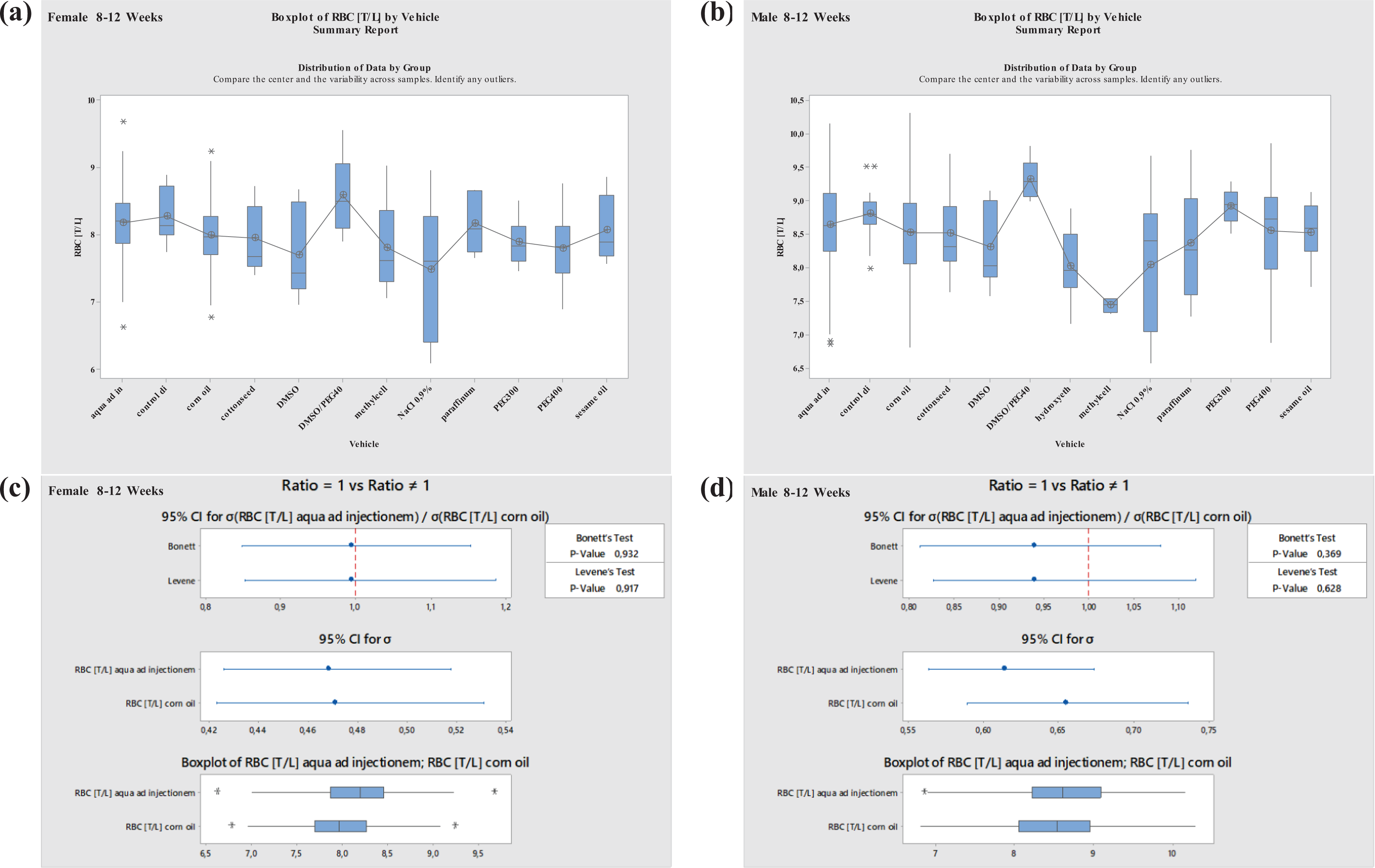
Boxplots—Comparison of different vehicles possibly impacting hematology parameter RBC in males and females at 8–12 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, cotton seed oil, DMSO in water, DMSO/PEG400 1:5, hydroxyethyl cellulose, methylcellulose, NaCl 0.9%, paraffinum liquidum, PEG300, PEG400, sesame oil. RBC: red blood cell; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400; PEG300: polyethylene glycol 300.
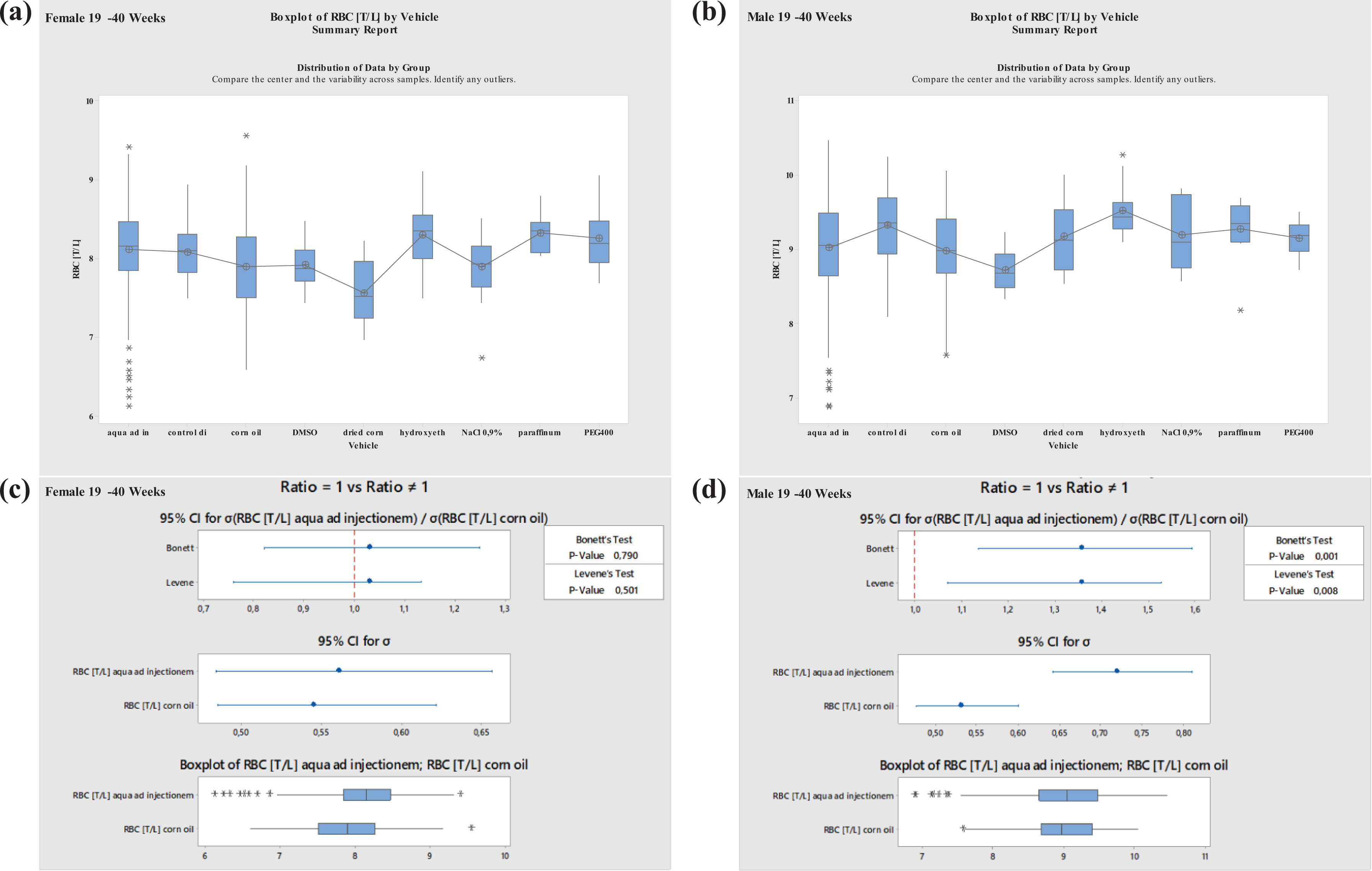
Boxplots—Comparison of different vehicles possibly impacting hematology parameter RBC in males and females at 19–40 weeks of age as well as corresponding variance analysis plots (Bonett’s and Levene’s test). Aqua ad injectionem, control diet, corn oil, DMSO in water, dried corn oil, hydroxyethyl cellulose, NaCl 0.9%, paraffinum liquidum, PEG400. RBC: red blood cell; DMSO: dimethyl sulfoxide; PEG400: polyethylene glycol 400.
No significant differences regarding WBC were established for either of the most commonly used vehicles, aqua ad injectionem or corn oil. No differences were noted for males or females at ages of 8–12 or 19–40 weeks. However, there were clear increases in males and females at ages of 8–12 and 19–40 weeks using paraffinum liquidum and decreases using DMSO (Figures 7 and 8 ). Using DMSO/PEG400 1:5, WBC increased in males at ages of 8 to 12 weeks and decreased in females at the same ages; using dried corn oil, WBC increased in males and decreased in females at ages between 19 and 40 weeks.
Further, no significant differences were noted in LYMPH values when using the most common vehicles, aqua ad injectionem and corn oil. However, values were seen to decrease when using the vehicle NaCl 0.9% in males at the ages of 8–12 or 19–40 weeks. Using PEG400, the values increased at ages between 8 and 12 weeks in males. Using PEG300, LYMPH increased in females but decreased in males at ages of 8 to 12 weeks. Variations in LYMPH values were noted using PEG400 in animals at ages between 19 and 40 weeks, the values increased in females and decreased in males (Figures 9 and 10 ).
For RET, there was a large variation. A significant increase was noted in animals at 19–40 weeks of age using NaCl 0.9% (Figures 11 and 12 ).
For RBC, the highest variation was noted for DMSO/PEG400 and methylcellulose in males and for DMSO/PEG400 and NaCl 0.9% in females at the ages of 8 to 12 weeks. Using paraffinum liquidum, an increase in RBC values was noted in animals at ages between 19 and 40 weeks in both sexes. A clear decrease in RBC was noted in 19- to 40-week-old females when using dried corn oil (Figures 13 and 14).
Other data are negligible due to limited sample sizes. For more information and interpretation of graphical representation, see supplementary data page 6 and Figures S6 to S27.
Comparison of Crl:WI(Han) data collected at BSL and RccHan™:WIST data collected at RCC Ltd/Harlan Laboratories Ltd, Switzerland
Summarized data from BSL and RCC Ltd/Harlan Laboratories Ltd, Switzerland, were compared in Minitab® using a two-sample t-test at a significance level of 95.0% and the hypothesized differences were reported as absolute values (see supplementary data Table S1 to S3).
For RBC (T/L), there was a male-specific hypothesized difference of 0.2 g/L, whereas the respective female data showed no difference (see supplementary data Table S1). For data from the 8–12-week and 19–40-week age-groups, the zero hypothesis could not be rejected. For the 13–18-week-old males and females, significantly higher values were reported than would be expected from normal statistical differences, thus the differences could be attributable to anesthesia or blood sampling methods.
Another possible explanation could be the use of different rat strains in the two laboratories, for example, the RBC values of the youngest and the oldest animals were similar, but the 13–18-week-old animal group is different between RCC Ltd/Harlan Laboratories Ltd, Switzerland, and BSL. These could possibly be explained by the BSL animals growing faster and larger and thus needing more RBCs for oxygen transport.
For HB (mmol/L), there was a female-specific difference of 0.3 mmol/L (see supplementary data Table S1). For males, no similar hypothesized difference was noted. The two-sample t-test showed that all data sets were derived from the same basic statistical population.
The male HCT (%) values had a slightly higher difference (2.5%) than the summarized female values (1.2%). The two-sample t-test showed no significant difference (see supplementary data Table S1).
Sex-nonspecific hypothesized differences were found for PLT, 215 G/L; MCHC, 1.17 mmol/L; RET, 0.37%; WBC, 1.355 G/L; MCV, 0.55 fL; and MCH, 0.055 fmol/L. The respective t-test results can be extracted from supplementary data Tables S1 to S3. The differences could be associated with methodological differences between the two laboratories.
The differences between blood samples could have different reasons: firstly, the samples were drawn from different positions, from the retro-orbital plexus in the case of RCC Ltd/Harlan Laboratories Ltd, Switzerland, and from the abdominal aorta in the case of BSL; secondly, different types of anesthesia were used (5% isoflurane/95% oxygen) in the case of RCC Ltd/Harlan Laboratories Ltd, Switzerland, and ketamine/xylazine 32 in the case of BSL. Additionally, in the RCC Ltd/Harlan Laboratories Ltd, Switzerland, studies, blood samples were collected twice, whereas in the BSL studies, a single final blood sample was collected where recovery of blood parameters was unnecessary.
The respective t-test results can be extracted from supplementary data Lists S1 to S9.
Discussion
In the present article, accumulated historical control data for hematology parameters are presented and evaluated. The data are derived from different laboratories. 13,25 The available data were compared to establish possible differences for different Wistar rat strains and to set acceptable control ranges in order to differentiate “normal” from “impaired” values. 12,14,33 Setting acceptable control ranges is necessary due to the high variation in parameter values obtained from a large number of studies. Data variability and range are discussed in the literature. 34,35
Therefore, for statistical correctness and data set comparability, the acceptable ranges were defined as “mean value ± 2 standard deviations.” However, acceptable ranges started in some cases at 0.0% or 0.0 G/L (e.g. BASO, EOS, LUC). This does not mean that 0.0 values are really acceptable, for example, in the case of relative HRET or absolute NEUT values.
The bulk of control data were available for RccHan™:WIST 13 and Crl:WI(Han) 25 rats. Both data sets are based on a full range of standard hematology parameters required by different guidelines (ICH (M3 R2 2009), 8 OECD (407/2008, 9 408/2018 10 ), EPA 2000 11 ). The following standard parameters were available for all sources: RBC, HB, HCT, total and differential leukocyte count (WBC), MCV, MCH, MCHC, RET, and PLT. Additional parameters, such as the erythrocyte indices MET-HB and HEINZBOD, have been used for several studies with RccHan™:WIST rats. For this rat strain, absolute and relative differential leukocyte count values have also been presented in contrast to the BSL 25 data for Crl:WI(Han) where only relative values are given. Moreover, for RccHan™:WIST rats, RET values were split into maturity indices using the fluorescence behavior of these cells. The latter data are deemed useful for judging recovery after blood sampling procedures, blood loss or anemia. 36 –42
As for the age ranges applied, it should be considered that Wistar rats at the start of toxicity studies are between 6 and 8 weeks of age, therefore, animals in 28-day studies are necropsied at ages of 10–12 weeks, or, calculating a 2-week recovery period, at ages of 12–14 weeks. Animals in 13-week studies are necropsied at ages of 19–21 weeks, or considering a 4-week recovery in such studies, at ages of 23–25 weeks, and so on. These increasing ages are associated with increasing incidence and severity of spontaneous background alterations. Some of these alterations 43 may influence the hematology evaluation values, for example, chronic progressive nephropathy (CPN), inflammatory changes, and so on. Occasionally, in young adult rats, even neoplasms are present. 5
Data recorded for RccHan™:WIST rats included animals at ages of ≤7 up to ≥71 weeks, from various study types with different administration routes, such as implantation, oral, intravenous, intramuscular, subcutaneous, and inhalation. 13 Data from BSL 25 for Crl:WI(Han) rats included data from animals at 8–40 weeks of age, from repeated-dose toxicity studies with oral, intravenous, intramuscular, and subcutaneous application routes. Implantation study data were excluded from the calculation of the aforementioned data set due to shifts in erythrocyte and related values and WBC values that are deemed a consequence of surgical trauma. These values were collected separately. Published data by Charles River Laboratories (Crl) 12 showed selected data only from animals at ages of 8 to 16 and >17 weeks (n < 200, 2002 to 2007), and data from Taconic Farms, Inc. are very limited (no information about single cell populations or RET) and recorded for animals at ages of 5, 10 and 19, 21, 32, and 106 weeks (n ≤ 10, August 2003 to April 2004). 33
In general, Harlan Laboratories RccHan™:WIST hematological data are comparable with Crl:WI(Han) and Global Alliance for Laboratory Animal Standardization (GALAS) Wistar rat data due to their common background (see below). Minor variations between strains were likely due to differences in blood sampling, anesthesia, 32 methods of analysis, and nutrition. The higher WBC values of GALAS rats, especially at the age of 10 weeks (males, n = 10), may be related to the low number of animals (see Supplemental Data Table S4). However, hematology parameters for all these rat strains are considered similar due to their genetical origin, this means that, similarities are deemed related to the three rat strains’ history, tracing back to a common origin. In 1892, the Wistar Institute 44 of Anatomy and Biology in Philadelphia was founded by General Isaac Wistar. 45 In 1905, Milton J Greenman 44 was appointed Director of the Institute. With Chief Scientist Henry H Donaldson 46 and several breeding pairs of albino rats on board, the Wistar Institute concentrated part of its services on establishing an animal colony, mass production, and distribution of rats bred for use in the laboratory. In 1915, Donaldson’s 46 book containing data and reference tables on the laboratory rat was published. It was central to the recognition of the rat as the first “standardized” laboratory animal. It offered “base line values” to which those of the controls used in any investigation may be referred. By the time when the book was updated in 1931, 44,46 the Wistar rats were being used in laboratories around the world. Breeding animals were transferred from the Wistar Institute to ICI Blackley 47 in 1944 and later to ICI’s pharmaceuticals 47 division at Alderley Park (1957), where, from the 1960s, the strain was referred as the “Alderley Park Strain 1.” 48 In 1964, rats were transferred to the Zentralinstitut für Versuchstierzucht 48 in Hannover, Germany (ILAR lab code: Han), 48 for breeding. In 1989, a nucleus colony was established at Biological Research Laboratories Limited (BRL), Füllinsdorf, Switzerland (formerly owned by Research and Consulting Company; later, RCC Laboratories Ltd, since 2005, Harlan Laboratories Ltd and since 2013, Envigo). 48,49 The rat strain was finally named RccHan™:WIST) 49 in 2008.
Taconic received a breeding stock from RCC in 1998. The rats were refreshed with cryopreserved embryos in 1998. Each member of GALAS received 50 breeding stock from RCC in 1998. The rats were derived by caesarean in 1999. Taconic replaced its previous Wistar Hannover stock with Wistar Hannover GALAS™ in June 2000. 33 The Crl:WI(Han) rats were originally rederived by GlaxoWellcome from Han Wistar rats supplied by BRL (Switzerland). They were transferred to Charles River UK in 1996, and transferred to Charles River in 1997, and rederived into an isolator-maintained foundation colony. 50
In contrast to the above discussed Wistar strains, the origin of SD rats (Hsd:SD) dates back to the Sprague-Dawley Company in 1925 through a series of crosses by Robert W Dawley begun with a single-hooded male and six albino females of unknown origin. The Harlan Laboratories colonies are direct descendants of this original colony. 51,52 CD® (Sprague Dawley) IGS rats went to Charles River in 1950 from Sprague Dawley, Inc. In 1991, eight colonies were selected to form the IGS Foundation Colony. It was rederived into an isolator foundation colony in 1997. 51
Comparing the Wistar data with data available for rats with Sprague-Dawley origin, a number of differences were evident. WBC values for Crl:(CD(SD)) and Hsd:CD® rats in published data were almost twofold and PLT count was approximately 20% higher than for Crl:WI(Han). LYMPH values were somewhat higher in Hsd:CD® than in Crl:WI(Han) rats, whereas NEUT values shifted toward lower values 53 (see Supplemental Data Table S4).
Wistar rats are considered sexually mature at ages of 7 to 8 weeks. 54 –56 As hematology parameters are under the influence of androgens 57 –59 and estrogens, 60 different hematology values are expected in peripubertal stage animals (age ≤a weeks) than in sexually mature animals. This was evident in data obtained for RBC, HB, RDW, MCV, MCH, HDW, RET%, LRET%, HRET%, NEUT, NEUT%, LYMPH, LYMPH%, and WBC.
Data between sexes are expected to be different considering the aforementioned endocrine influences but there are also differences in body weight, food and water consumption, which in Wistar rats are higher in males than in females at ages of 5 weeks onward. 61
Hematology value ranges might be influenced by technical issues, for example, accidental aspiration of high viscosity vehicles 62,63 or by gavage-related reflux-induced lesions. 64 Furthermore, some vehicles are considered harmful, for example, Cremophor®. Nonetheless, no changes were noted after 24 h in studies with Cremophor® at a single dose >7.5 mg/kg at a viscosity of 1.0 mPa·s. Therefore, Cremophor® EL was regarded as safe in rats at doses of 100 mg/kg daily for 1 month. 65,66 In patients, Cremophor® EL as a vehicle was linked to effects like severe anaphylactoid hypersensitivity reactions, hyperlipidemia, abnormal lipoprotein patterns, erythrocyte aggregation, and peripheral neuropathy. 67 As the sample size of animals treated with the vehicle Cremophor® was too small, data were excluded and no comparison was made with the vehicles most commonly used at BSL, aqua ad injectionem and corn oil.
The only significant differences found for the vehicles aqua ad injectionem and corn oil were for BASO and MONO in males and females at the ages of 8 to 12 weeks and for LUC at the ages of 19 to 40 weeks. These parameters were already discussed as highly variable and were therefore excluded from further discussion. In males at ages between 19 and 40 weeks, differences between the common vehicles mentioned above were seen for HCT, RBC, and HB, which may be explained by CPN starting earlier and being more pronounced in males than in females. This was evident, as no differences were seen between the commonly used vehicles, aqua ad injectionem and corn oil, in males at the ages between 8 and 12 weeks.
DMSO treatment had significant effects on RBC in vitro, affecting osmotic fragility and increasing hemolysis. This effect can also be observed in Crl:WI(Han) rats when using DMSO as vehicle in repeated-dose toxicity studies. Free HB levels increased in vitro compared with controls. The PLT aggregation induced by adenosine diphosphate, thrombin and thrombin receptor activator peptide is inhibited by DMSO treatment in vitro, 68 something also observed in males at the ages of 8–12 and 19–40 weeks when compared to animals that were treated with aqua ad injectionem. Furthermore, in humans, DMSO inhibited platelet activation at low doses. 69
An additional vehicle effect can be seen using paraffinum liquidum in oral studies, which is associated with lung affections following aspiration. Hence, blood parameter changes could be related to induced lesions. NaCl-related differences, however, remain unclear.
The ranges might also be influenced by outliers which are not due to technical errors but rather to background alterations. The older the animals are, the higher the incidence and severity of background lesions. Hence, CPN may be considered to influence hematology parameters. Although hematology data are not considered in publications on CPN, 70 chronic nephropathy is deemed a risk factor for erythropoiesis by reducing erythropoietin in humans. 71,72 Furthermore, the older the animals are, the greater the influence serious age-related lesions may have on hematology ranges. Therefore, for several parameters (NEUT, NEUT%, LYMPH, LYMPH%, MONO, MONO%, and PLT), data for animals at ages 19 up to ≥71 weeks were considered separately. Finally, for some values (HCT, MCHC, MRET%, and WBC), there were no obvious differences between ages ≤7 to ≥71 weeks.
A comparison of 90-day repeated-dose studies with reproduction and implantation studies revealed differences in a few hematology parameters. While in repeated-dose studies with animals at ages from 19 to 40 weeks, NEUT values were slightly higher in males than in females, in reproductive toxicity studies, the NEUT ranges were higher in females than in males. This finding was associated with a decreased LYMPH count in females in reproduction toxicity studies. Similar data have been shown for bone marrow differential counts in which the number of lymphoid cells in pregnant females dropped massively. 73,74 Furthermore, similar data have been reported for humans, like, increases in granulocytes during pregnancy whereby no changes in LYMPH have been noted by some authors. 75 Other authors described a decreasing LYMPH count during pregnancy. 76 A higher variation for NEUT and LYMPH is expected for implantation studies as was noted in the control data presented. These are either post-surgical/post-traumatic effects or may even be stress-related (e.g. implant bearing animals, wound healing).
In 90-day repeated-dose toxicity studies, LYMPH values shifted toward lower values with increasing ages (higher values in females than in males). Also, in humans, one of the most prominent signs of an aging immune system is a significant reduction in naïve LYMPH in blood. These age-related changes have been considered as changes in circulating LYMPH cytokines or cytokine receptors in serum that might influence age-associated changes of LYMPH in circulation. 77
In young animals, there were no differences for LYMPH in either sex. In rats, age-related changes in LYMPH populations have been described in detail, for example, CD4+ LYMPH in Peyer’s patches and spleen. 78
RET values varied strongly in all study groups. However, a clear trend toward increased RET values was noted in females in reprotoxicology studies. Similarly, changes in erythropoiesis have been noted in women whereby mature RET increased during the second and third trimesters and which was associated with increased serum transferrin receptors. The authors concluded that erythropoiesis increases with gestational age during pregnancy and returns to normal 5 weeks after delivery. 79 On the other hand, anemia is a common pregnancy-related complication in humans due to effects on the HB concentrations whereby the main cause might be considered due to iron deficiency followed by folate deficiency-related megaloblastic anemia. 80 In contrast, data have been provided for rats necropsied at day 20 of gestation (after Cesarean section) with significantly increased erythropoiesis in bone marrow 73,74 that might be considered related to increasing RET values in the periphery due to increased demand for oxygen supply. Changes in hematology parameters in rats during pregnancy have been summarized by de Rijk et al. 81 and include drastically decreased RBC count values, HB concentration, and HCT, suggesting that hemodilution (and consequently anemia) occurs, and increased RET count. It has been suggested that progesterone is involved in initiating enhanced erythropoiesis. These changes are not deemed fully visible in rats that have been necropsied 12 days postpartum (OECD 422), therefore, RBC, HB, and HCT values were not affected in females from reproduction studies. However, MCHC values decreased in female rats from reproduction studies which is indicative of changes in HB synthesis. The latter is reflected by an increased RET count. Determining values for LRET, MRET, and HRET and not only for the total amount of RET should be considered important in order to obtain more information about the erythropoiesis of untreated animals. A number of toxic compounds or biological products, like erythropoietin, exert an influence on erythrocyte development.
Conclusion
This study showed that it is very important to note that historical control data of different studies can vary and that some parameters are more age dependent as others. It can also be seen that the differential blood count varied stronger through the different ages while red blood count values were more constant except in animals younger than 7 weeks.
It was a big challenge to define acceptable ranges and in practical trials there will be deviations according to discussed circumstances, for example, used vehicle, blood sampling and anesthesia method as well as strain and origin. However, there will be always unexplainable outliers too, for example, outliers without histopathological background or outliers reasoned by methodical issues, which always have to be excluded from the considered control data set, via outlier test, first. As the historical control data set contains a huge sample size over several years, originated variances can be neglected and the set can be used to evaluate control data sets of single studies with smaller sample sizes. Values within the acceptable ranges of the historical control data should not have undergone critical methodological effects.
As the respective true value for most samples remains unknown, the summarized data could help investigators to interpret differences in observed data sets. In the ideal case only minor differences, which are from normal random deviation were seen and values of control animals should be within the defined acceptable ranges. If the values show larger differences, this could be part of yet unknown effects from, for example, treatment or handling. Major differences could also help to find not yet evaluated methodological differences or to improve the measurement setup.
Future investigators should take all these into consideration by analyzing their study-based control parameters before comparing them to groups dosed with test substances.
Supplemental material
Supplementary_Data - Historical control data for hematology parameters obtained from toxicity studies performed on different Wistar rat strains: Acceptable value ranges, definition of severity degrees, and vehicle effects
Supplementary_Data for Historical control data for hematology parameters obtained from toxicity studies performed on different Wistar rat strains: Acceptable value ranges, definition of severity degrees, and vehicle effects by Marlies de Kort, Klaus Weber, Björn Wimmer, Katharina Wilutzky, Patricia Neuenhahn, Philip Allingham and Anne-Laure Leoni in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
