Abstract
Meldonium is the active molecule from Mildronate® with similar chemical structure to an amino acid, and it is known as (3-(2,2,2-trimethylhydrazine) propionate) (CAS 76144-81-5). This pharmaceutical substance is approved in Eastern Europe for cerebral and myocardial ischemia and has been on the World Doping Association’s banned substances list since January 2016. The goal of this review is to relate the use of meldonium as a doping agent, considering its pharmacological, toxicological, and analytical aspects. This review is based on the scientific literature from digital platforms. The main mechanism of action of meldonium is based on a decrease in
Introduction
The World Anti-Doping Agency (WADA) is an independent organization created by the International Olympic Committee to protect the rights of athletes to participate in doping-free competitions and to ensure athlete health, fairness, and equality among participants. According to WADA, 1 doping is defined as the use of substances and/or methods that improve athletic performance, and it can be harmful to the athlete himself/herself as well as to other competitors. Therefore, anti-doping programs seek to preserve “the spirit of sport,” the pursuit of human excellence through the dedicated perfection of an individual’s natural talents 1
Many reasons lead to doping such as status, fame, money, pressure from sponsors, and/or club or even the state or country which athletes represent. On some occasions, the technical team or state officials are responsible for the athlete’s doping, as occurred with the Soviet Union during the Cold War, 2 Finland in 2001, 3 and recently with Russia. 4 These issues make doping a complex subject.
According to WADA, 1 the list of monitored substances and methods is classified as (1) substances and methods prohibited at all time, (2) prohibited substances in competition, and (3) prohibited in particular sports. Despite the efforts of WADA and other sectors of society, there are new substances and/or methods that can be used for doping purposes, such as meldonium. 1
Meldonium, also known as MET-88, is a substance present in the medicinal product Mildronate®, which is marketed in Eastern Europe, specifically in Azerbaijan, Belarus, Georgia, Kazakhstan, Kyrgyzstan, Moldova, Ukraine, Uzbekistan, and Russia. It is used for the treatment of cerebral and cardiac ischemic disorders. 5,6
Since January 2016, meldonium has been classified as a WADA type I substance. Indeed, this substance is included in “Metabolic and Hormonal Modulators,” which also includes insulin and its mimetics, activators of adenosine monophosphate-activated protein kinase (AMPK), peroxisome proliferator-activated receptor agonist gamma (PPAR
Since its prohibition from sporting events, there have been several doping cases that involve meldonium, especially for Eastern European athletes. 7,8 According to the WADA, 9 meldonium was added (to the Prohibited List) because of evidence of its use by athletes with the intention of enhancing performance. 9 Therefore, there are studies that may indicate its ergogenic effects. 10 –13 However, according to Schobersberger et al., 6 there is insufficient scientific evidence on performance enhancement in athletes caused by meldonium. 6
Given this relatively new meldonium status, the goal of this review is to relate the use of meldonium as a doping agent, considering its pharmacological, toxicological, and analytical aspects.
Methods
Study design
The review was based on the following platforms: MEDLINE (Pubmed), LILACS (Scielo), EMBASE (Elsevier), Scopus (Science Direct), and Academic Google. The keywords employed for this review were Meldonium; Doping; Review; Pharmacology; Pharmacokinetics; Pharmacodynamics; Toxicology; Sports; Risk; Determination; Analyze; Biological Matrices; Urine; Blood; Chromatography; Mass spectrometry; Physicochemical properties and their Portuguese, Spanish and English versions.
Criteria for inclusion and exclusion
All published studies in the scientific literature on meldonium are according to the digital platforms. Absence of ISSN indexing, references without authors, journalistic notes without scientific nature, and impossibility of accessing the full article were used as criteria for exclusion.
Meldonium
Physicochemical properties
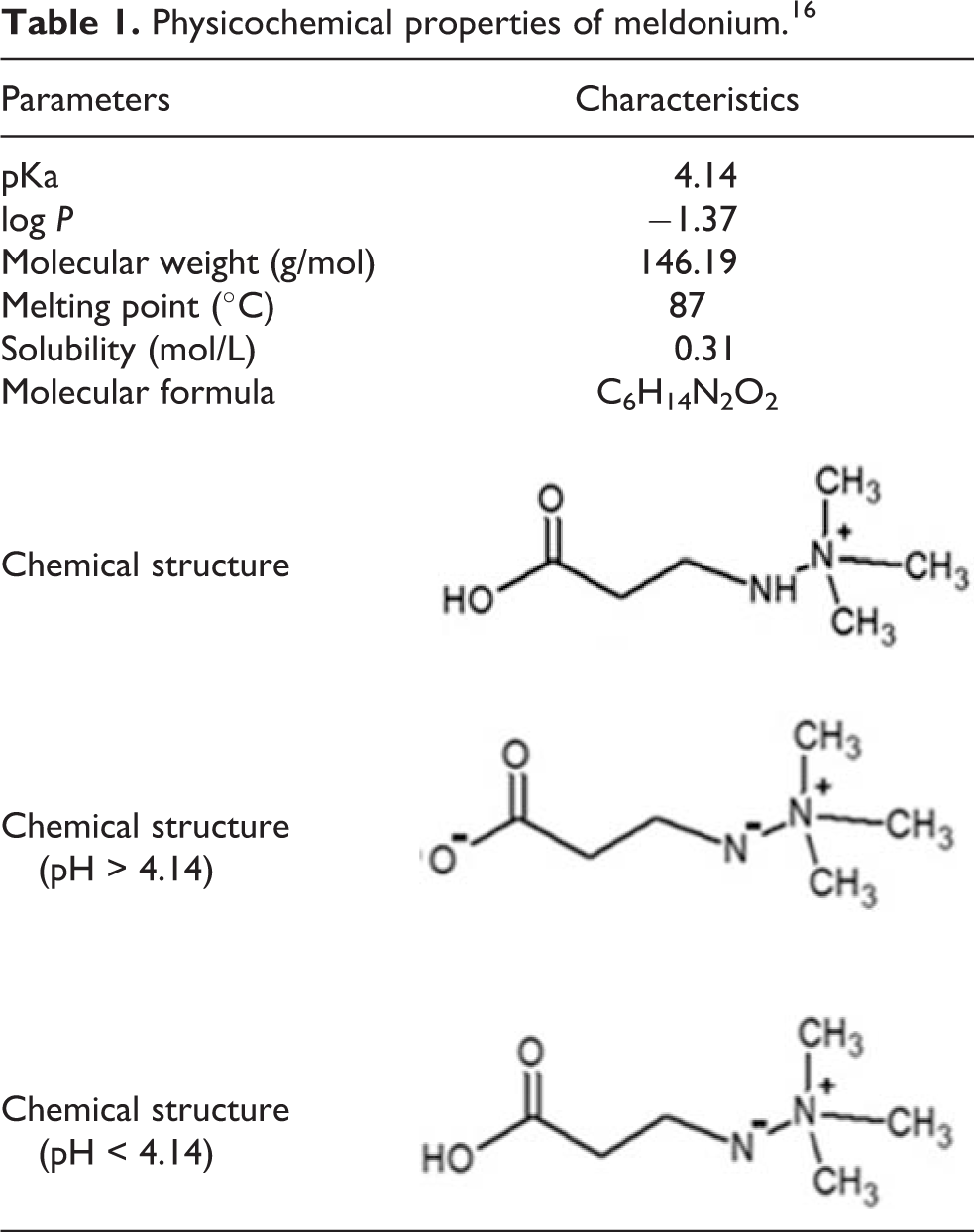
Meldonium is the active molecule from Mildronate, and it is known as (3-(2,2,2-trimethylhydrazine) propionate) (CAS 76144-81-5). Originally synthesized by the Latvian Institute for Organic Synthesis in the 1970s with anti-ischemic action, Mildronate is not approved by the Food and Drug Administration (FDA). This substance is used only in Eastern Europe and Russia. 5,6 Meldonium has a chemical structure similar to an amino acid. This molecule exhibits ionized and non-ionized fraction and its physicochemical properties are described in Table 1. 14 –17
Physicochemical properties of meldonium. 16
Pharmacokinetics
Meldonium is a relatively small, highly hydrophilic molecule that has no affinity for plasma proteins.
14–15
The maximum plasma concentration occurs at approximately 1 h, and its half-life varies depending on the posology, treatment time, and pharmaceutical form.
6
For the ingestion of meldonium capsules,
Meldonium excretion and maximum urine concentrations vary according to the type of administration; the single dose injectable form has a linear excretion profile, whereas administration of multiple doses shows nonlinear elimination. 20 Single or multiple oral dose administration also exhibits nonlinear excretion. 18 Physical exercise may also influence meldonium excretion. 21 However, Forsdahl et al. 22 reported that the maximum urine meldonium concentration is observed shortly after injection of the substance. 22
In the study by Rabin et al., 21 32 volunteers received meldonium treatment for 21 days, who were divided into two groups. The first group of 14 volunteers (7 men and 7 women) received 1.0 g of orally daily meldonium, while the second group of 18 volunteers (9 men and 9 women) received 2.0 g of meldonium orally daily. Six volunteers were selected on the basis of the lowest meldonium urinary concentration to perform single functional load testing (physical exercise). The volunteers underwent the Wingate test on a veloergometer performed by the Ramp-30 protocol, which caused a decrease of meldonium urinary concentration in the six volunteers (3 men and 3 women). The changes in meldonium urinary concentrations were 0.38–0.13; 0.12–0; 0.04–0.09; 0.19–0.08; 0.085–0.068; and 0.015–0 µg/mL. 21
Meldonium can be metabolized mainly in the liver by gamma-butyrobetaine hydroxylase (BBOX) to form dimethylamine, 2-hydroxymethyl-2(hydroxymethylamino)-propane-1,3-diol, 3-amino-4-(hydroxymethyl-methyl-amino)-butyric acid, and 3-hydroxypropionic acid. 14,20,23 This last metabolite can be converted into succinic acid, the main metabolite found in plasma, as illustrated in Figure 1. 14,20 The main meldonium excretion route is in urine; approximately 34–60% of this substance is eliminated unchanged 15,20 as described in Figure 1.
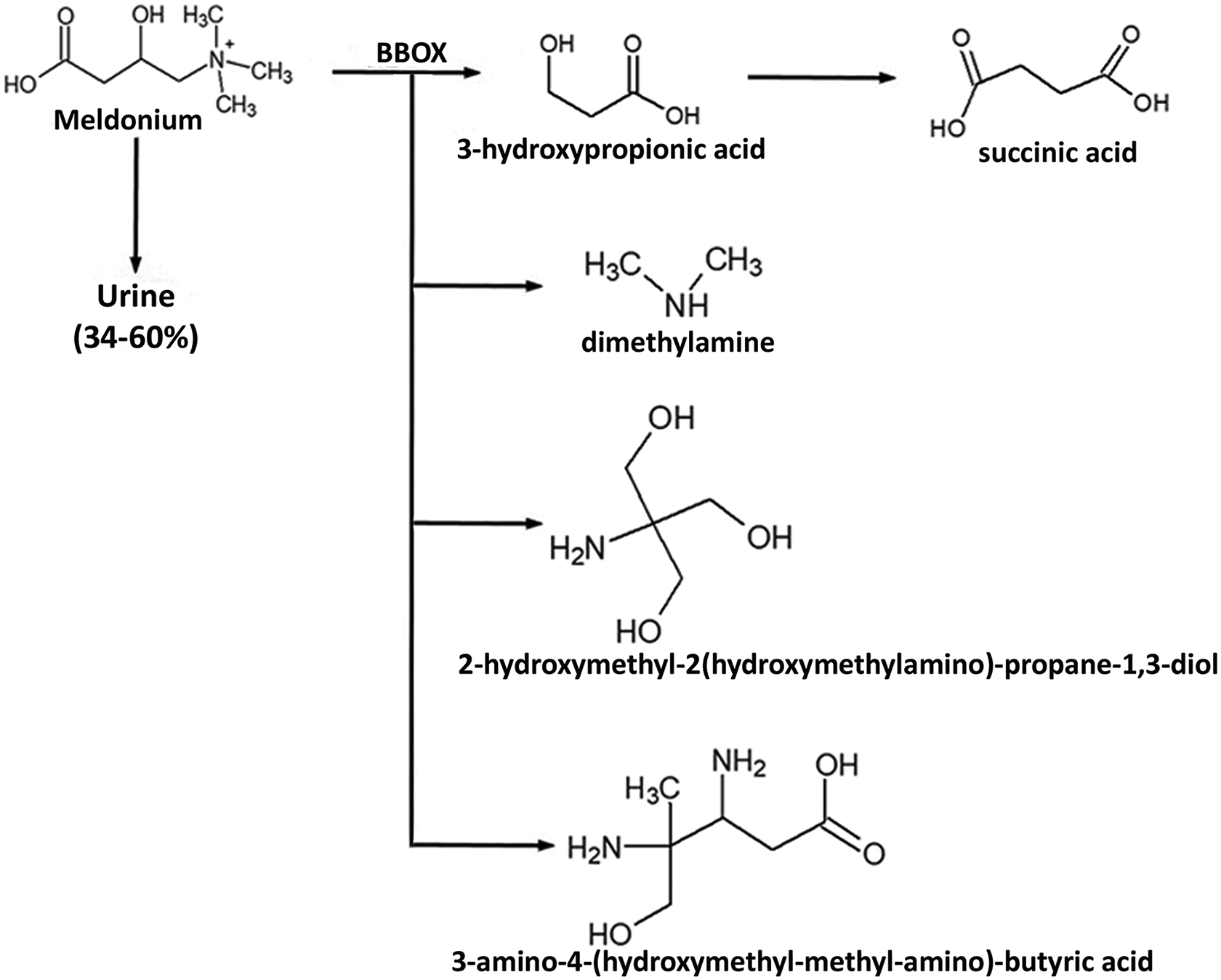
Main route of elimination and its route of metabolization by BBOX. BBOX: gamma butyrobetaine hydroxylase.
Meldonium accumulates in various tissues that express organic cation transporter 2 (OCTN2), such as the heart, kidney, brain, and skeletal muscles. 14,24 In fact, the meldonium transport system into the cells includes capture of ionic compounds, association with macromolecules, and distribution in lipids. This substance is a polar-ionized molecule with low solubility in the double lipid layer of cell membranes and its ability to travel across cell membranes by OCTN2. An excess of meldonium is localized outside of the double lipid layer as ion pairs. Probably due their amphoteric characteristic, endogenous substances can carry out the transfer across biological membranes that transport organic anions or cations in vivo. However, this effectiveness depends on the pH and pKa values, and it can reach the fullness limit. So, saturation limit of meldonium and inhibition of other molecules can be involved in the carnitine cycle, as described by Rabin et al. 21
Blood circulation of meldonium forms hydrophobic ion pairs with plasma lipids based on non-covalent complexes with polar molecules and with neutral weakly hydrophobic molecules. This particular ability of meldonium allows its easy penetration into membrane cells, including the blood–brain barrier. 21
Excretion is reduced approximately by 70% when multiple doses are administrated. These nonlinear excretion parameters may be due to BBOX, a key enzyme of meldonium metabolization.
18,20,25
Based on the study of Forsdahl et al.,
22
meldonium has an excretion profile by a three-compartment model: alpha (
According to Zhao et al., 20 meldonium BBOX saturation occurred in healthy Chinese volunteers after a single dose (750–1500 mg) or multiple daily doses (500 mg) for 5 days. 20 Another explanation for the nonlinear kinetics is the absorption phase. Since meldonium is highly soluble in water, active transport in the gastrointestinal tract becomes very important, and saturation of these transporters could lead to this kinetics. 24
Tretzel et al. 26 orally administered a single dose (500 mg) or multiple doses (3 × 500 mg/day for 6 consecutive days) of meldonium and evaluated the substance concentration by dried blood spot (DBS). DBS method is a form of biosampling where blood samples are blotted and dried on a specific filter paper, with positive results after 16 days for a single dose or 28 days for multiple doses. In urine samples, this substance was detected after 49 days from oral administration of a single dose (limit of detection: 20 ng/mL). Considering these results, the authors suggest that meldonium has the ability to bind erythrocytes, a phenomenon that would also explain the lack of linearity for urinary excretion. 26
Pharmacodynamics
Meldonium has the capacity to interfere in
The main meldonium mechanism of action is inhibition of
In the intestine,
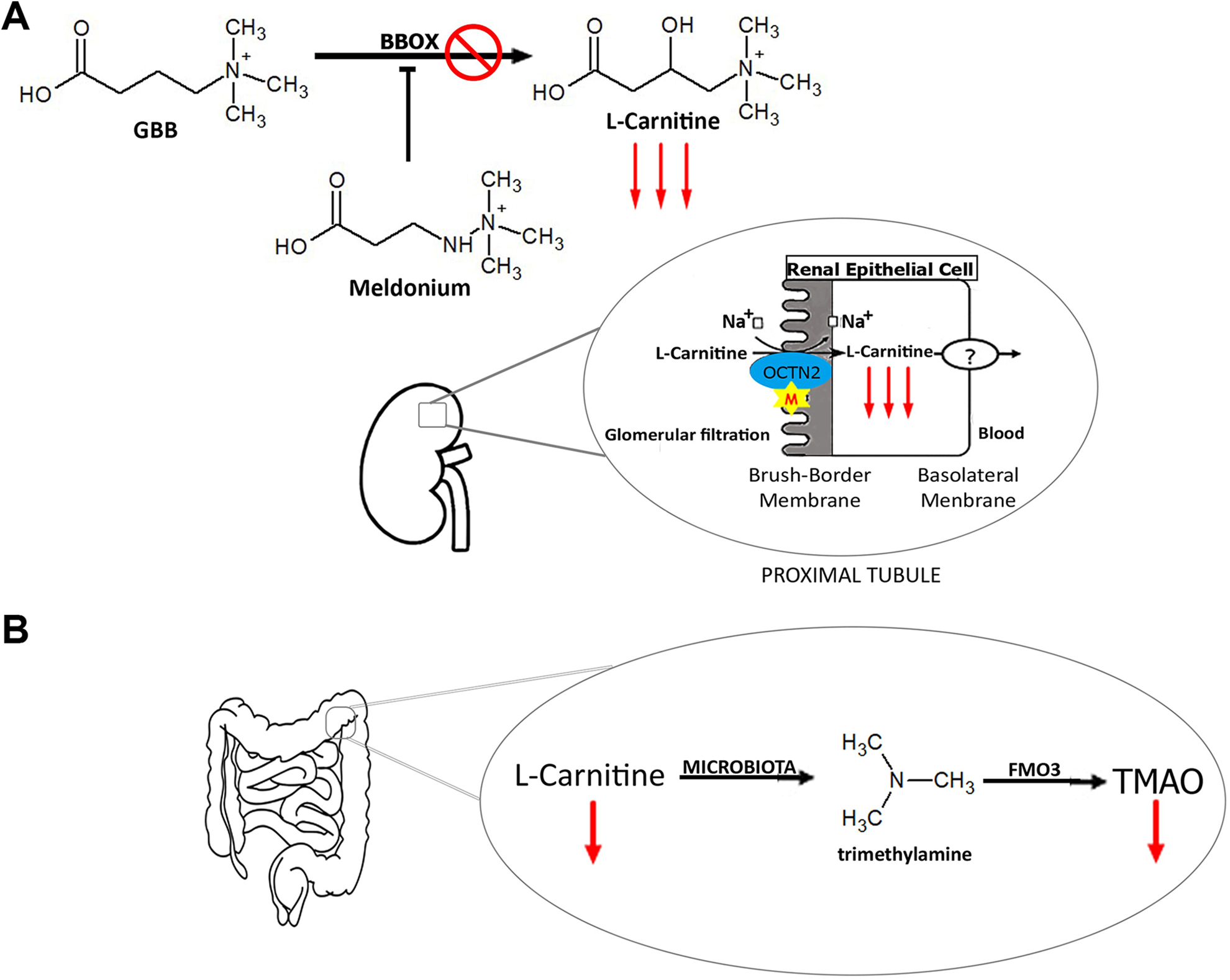
Meldonium pharmacodynamics in (a) kidneys and (b) intestines. M: meldonium; BBOX: gamma butyrobetaine hydroxylase; GBB: gamma butyrobetaine; OCTN2: organic cation transporter 2; FMO3: hepatic flavin monoxigenases 3; TMAO: N-oxide-trimethylamine; ?: unknown carrier; XXX: inhibition.
Another meldonium mechanism of action includes the inhibition of carnitine palmitoyltransferase-1 (CPT1). This enzyme uses
Meldonium inhibition of CPT1 leads to an accumulation of acyl-CoA and fatty acids in the cytosol. This behavior enhances the gene expression of peroxisome proliferator-activated receptor alpha (
In diabetes, there are high acylcarnitine concentrations that induce insulin resistance. 14,38,39 Dambrova et al. 14 showed that meldonium treatment induces GLUT4 gene expression and insulin receptor proteins in rat hearts. This meldonium-mediated regulation decreases lactate levels, an action that indicates an increase in aerobic glucose oxidation. These authors indicated changes in glucose metabolism caused by meldonium through the inhibition of CPT1 and decreased acylcarnitines. 14,40
Type 2 diabetic rats treated with meldonium exhibited decreased glycated hemoglobin and normalized GLUT1 gene expression levels in the heart, liver, kidney, and skeletal muscle. In normal rats treated with meldonium, there was a small increase in GLUT1 expression in these tissues. 41 The most probable hypothesis proposed by the authors was increased glucose metabolism in tissues and consequently reduced blood glucose. 41 Similar findings were described in studies by Liepinsh et al. 40,42 in previous years. These mechanisms are shown in Figure 3.
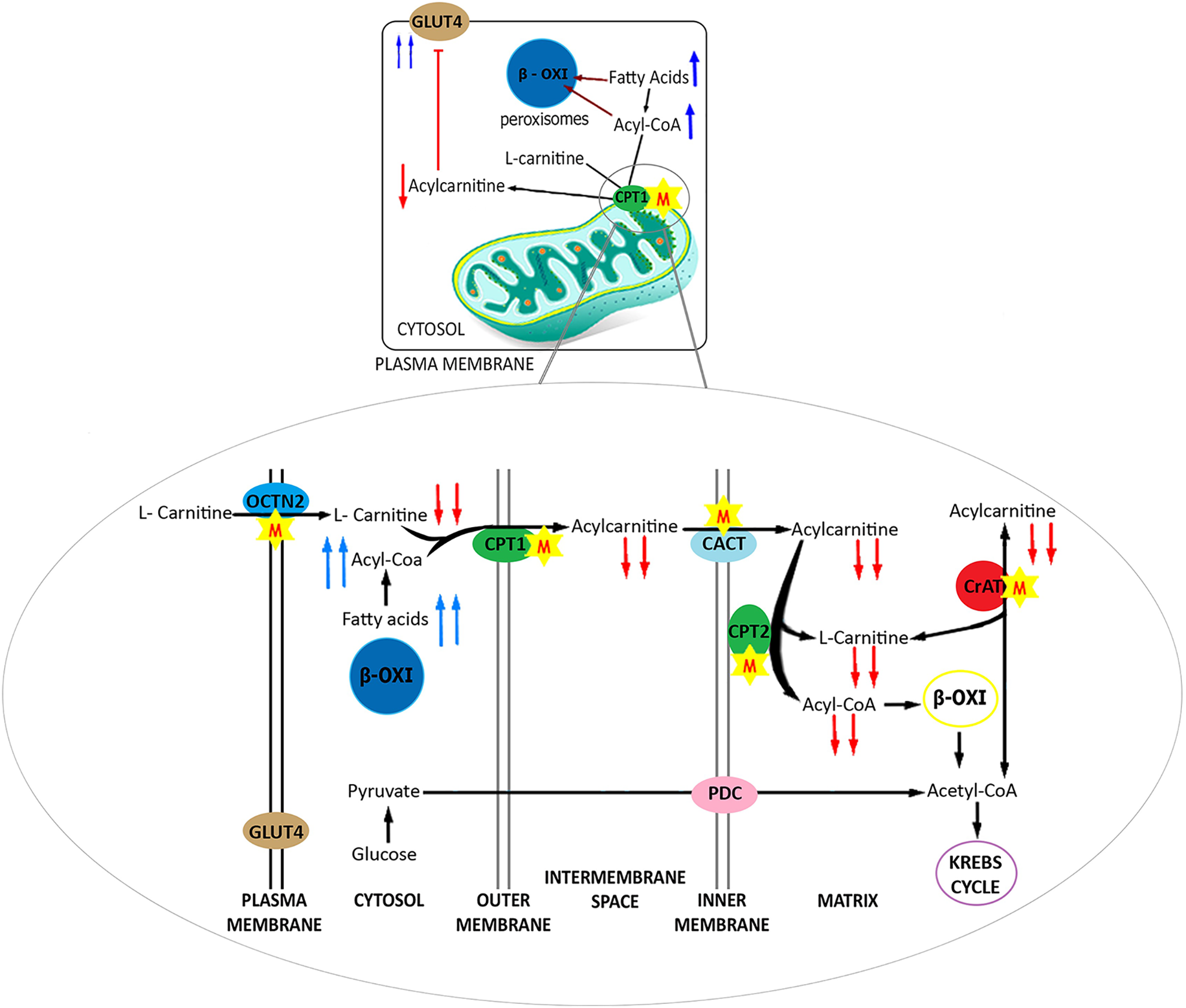
Mitochondrial effect of meldonium. M: meldonium; OCTN2: organic cation transporter 2; CPT1: carnitine palmitoyl transferase 1; GLUT4: glucose transporters 4; CACT: carnitine/acylcarnitine transferase; CPT2: palmitoyl transferase 2; CrAT: carnitine acetyltransferase; IR: insulin receptor.
Carnitine transport in cardiomyocytes occurs via Na+-dependent channels. Pathological acylcarnitine dysregulation in myocardial cells is bypassed with meldonium treatment. 29 A reduction in acylcarnitine affects the endoplasmic reticulum and sarcoma, which regulates the activity of Ca2+ ATPase and Na+-K+ ATPase, respectively. Thus, meldonium regulates myocardial contractility and increases hexokinase I activity. 23,29,41,43 Peroxisome-mediated fatty acid metabolism increases the AMP/ATP ratio, which activates AMPK, an enzyme responsible for restoring ATP levels, and increases cellular glucose oxidation. 44
In the blood–brain barrier, OCTN2 is apparently the main mechanism that allows meldonium entry into the central nervous system (CNS). 29,45 Meldonium in the brain improves hemodynamics, electrolyte balance, and oxygenation in cerebrovascular disorders. 29 Meldonium normalizes caspase 3 and cellular apoptosis susceptibility protein (CAS) levels 45,46 and induces nitric oxide synthase (iNOS) expression that is activated under ischemic conditions. 29,45 –47 It also promotes improved glucose absorption in the CNS, 29 probably due to increased GLUT4 expression. However, these mechanisms have not yet been fully elucidated. 29,47
In the brain, BBOX inhibition displaces GBB into an alternative route of esterification by an unknown enzyme, and this process generates GBB methyl ester (at low levels) and GBB ethyl ester. The GBB ethyl ester structure is similar to acetylcholine, and it displays cholinergic activity, 29 as demonstrated by Dambrova et al. 48 based on decreases in diastolic and systolic pressure. 48 GBB esters are hydrolyzed by GBB esterase (acting as an acetylcholinesterase). This hydrolysis product acts as a secondary signal to activate NO synthesis. This reaction may occur in other tissues, including the cerebral cortex, cerebellum, liver, heart, and kidneys, considering that endothelial nitric oxide synthase (eNOS) is activated by GBB esters in the endothelium via muscarinic receptors 29 (Figure 4).
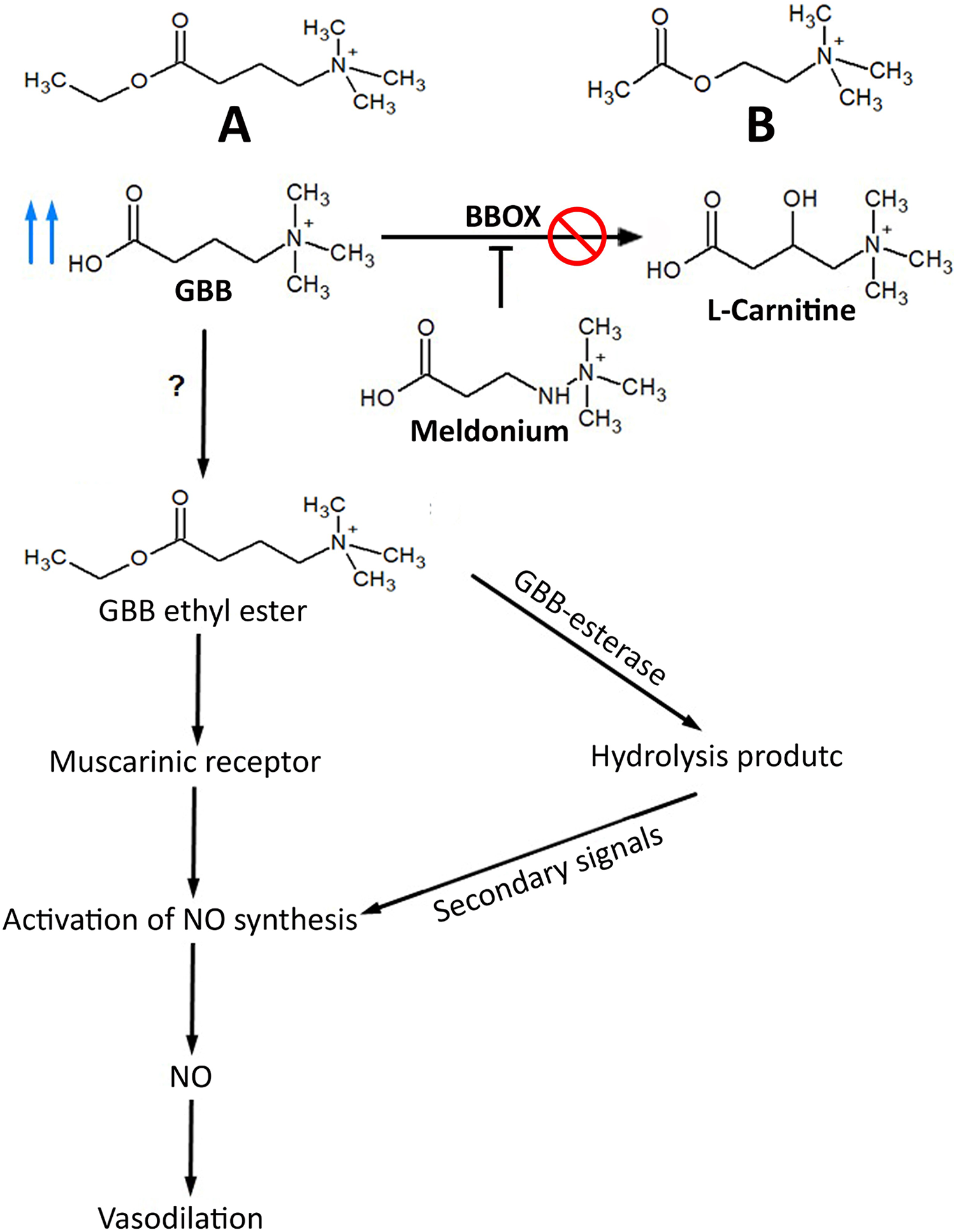
Indirect effect of meldonium on cholinergic system: (a) GBB ethyl ester and (b) acetylcholine, GBB, unknown enzyme (?), NO, and inhibition. GBB: gamma butyrobetaine; NO: nitric oxide; XXX: inhibition.
In addition to all the above-mentioned effects, meldonium acts as an immunomodulator, since it supports humoral immunity. It also acts as a bronchodilator in asthmatics; however, this mechanism has yet to be elucidated. 49
Therapeutic use and acute and chronic clinical manifestations
Mildronate GX, which contains meldonium as the active substance, is marketed in capsule form with a therapeutic dose of 500 mg orally twice daily, mainly for the treatment of cardiac and neurological disorders such as ischemia. The maximum administrated dose in humans is 2000 mg by oral form per day. 6 However, with appropriate posology, this substance can be applied for other clinical disorders such as asthma, bronchoconstriction, gastric ulcers, cocleovestibular dysfunction, glaucoma, diabetes, and vaccine adjuncts. 14,29,49
There may be differences in meldonium dosage and treatment time according to the symptoms, but the usual regimen is 500 mg twice daily for 4–6 weeks. Glucose levels and cardiac parameters change after 4 weeks of treatment (100 or 200 mg/kg orally daily).
41,42
Two weeks of meldonium treatment reduced
Another study conducted by Liepinsh et al. 40 administered 200 mg/kg meldonium intraperitoneally in mice once a day for 20 days. Ten days of treatment decreased blood glucose levels and increased glycemic metabolism related to gene expression in the heart. 40 Kūka 51 administered 100 mg/kg meldonium daily for 4 weeks to Wistar rats. This treatment reduced arrhythmia duration and ventricular fibrillation frequency after 14 days of treatment compared to the control. After 4 weeks, blood pressure was not significantly different in the animals. However, after 8 weeks of treatment, there was a decreased heart rate in treated Dahl rats compared to rats given a high salt diet. 51
Other studies showed several positive effects from meldonium treatment, mainly against chronic heart failure, systolic function disturbances, 10 and left ventricle dysfunctions. 43,44 Thus, it reduces acute myocardial infarction Casilde 52 and prevents atherosclerosis. 14 This substance also combats peripheral vascular resistance. 11 In the CNS, concomitant intraperitoneal meldonium (120 mg/kg) and GBB (30 mg/kg) treatment increased brain NO levels within 15 min of administration. 29
Preliminary meldonium administration (10 mg/kg orally) reduced hemodynamic disturbance and improved oxygenation.
29
Considering meldonium effects in the brain, daily intraperitoneal administration of this substance (250 mg/kg) during 26 days was able to significantly reduce intracerebral tumor growth in orthotropic glioblastoma (GBM) mouse model. GBM is related to OCTN2 overexpression and
In the study of Di Cristo et al. (2019),
54
transgenic Huntington’s disease (HD)
Based on the literature findings (Table 2), beta-oxidation inhibition, lipid metabolism modulation, and consequently the beneficial cardiac effects require at least 10 days of meldonium administration in ICR mice. 29,40 However, cerebral and vasodilatory effects are pronounced less than 1 h after intraperitoneal administration in Wistar rats. 29,56
A resume of the studies of meldonium treatment and its impact.
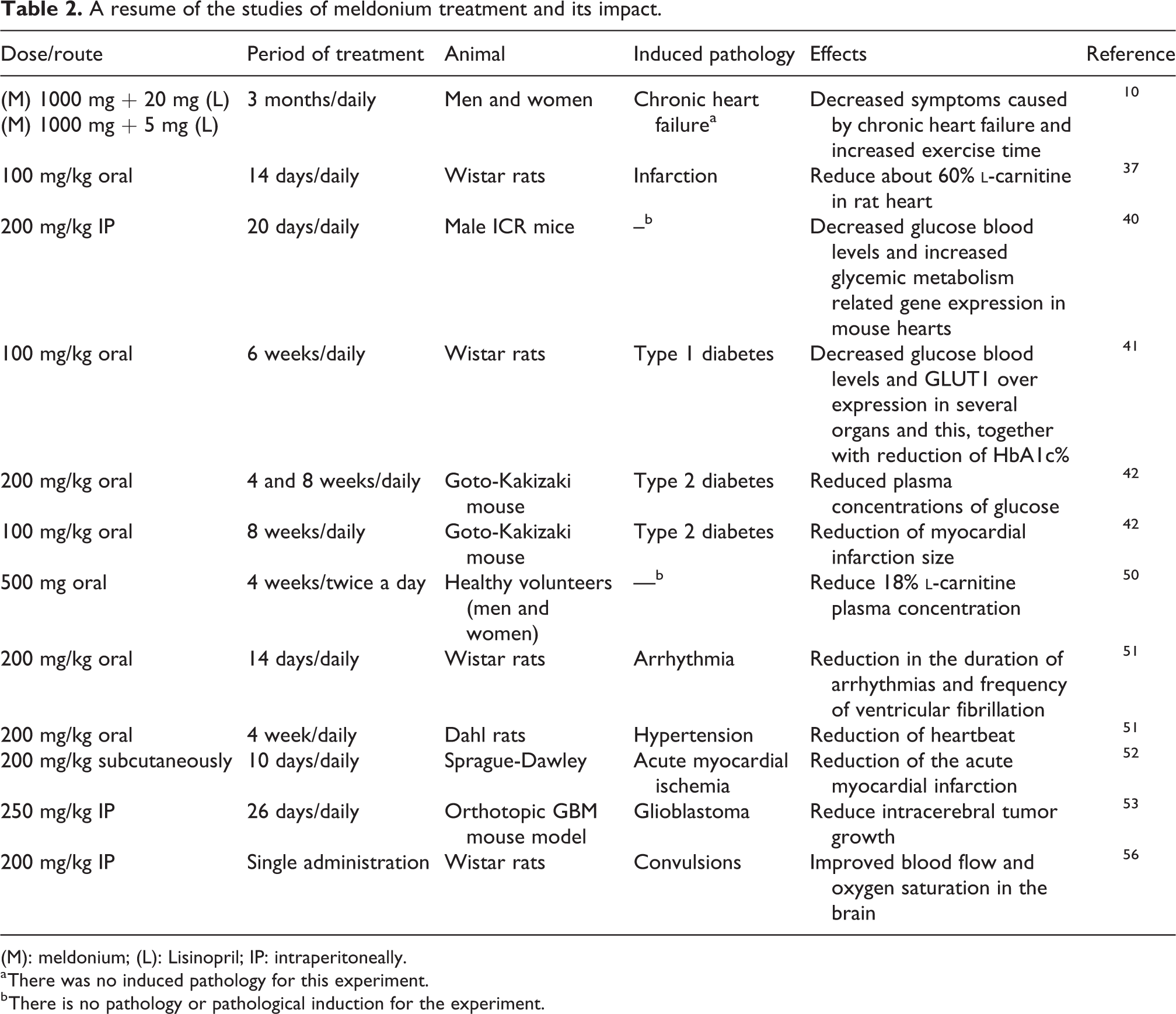
(M): meldonium; (L): Lisinopril; IP: intraperitoneally.
a There was no induced pathology for this experiment.
b There is no pathology or pathological induction for the experiment.
Adverse effects and toxicity
According to the manufacturer of Mildronate GX, the commercial pharmaceutical product of meldonium, there are some rare adverse effects, including allergic reactions (redness and itchy skin, urticaria, rash, and/or angioedema), dyspepsia, tachycardia, and change (increase or decrease) in blood pressure. There are no reported toxicological symptoms in healthy patients in human clinical studies, and the overall substance toxicity is low. 6,56
A study of 40 Chinese volunteers that consumed Mildronate GX (500 mg/day three times daily for 13 days) revealed no toxicity, and the only adverse effects, insomnia, allergic reactions, dizziness, and nausea, disappeared after treatment cessation; these results are similar to those described by the Mildronate GX manufacturer. 18 Daily oral administration over 6 months to rats did not alter any tissues. 29
Another study conducted in male Sprague-Dawley rats (200 mg/kg orally for 3 or 6 weeks) demonstrated hepatic steatosis, increased acyl-CoA long chain and elevated very low-density lipoproteins (VLDL) for both treatment periods. 57 Oral administration of 400 mg/kg daily for 60 days increased triglyceride levels in the liver for male Sprague-Dawley rats; however, the liver functions and heart triglyceride levels remained unchanged. 58
A 13-week daily oral administration of 1600 mg/kg experiment was performed on Sprague-Dawley rats to obtain nontoxic data (unpublished observations). The nontoxic meldonium dose was estimated for female rats at 25 mg/kg/day, while it was 100 mg/kg/day for male rats (
Analytical methods for biological matrices in vivo
Biological matrices in vivo
Urine, blood, and plasma are the traditional biological matrices to determine meldonium concentrations. Urine is the biological matrix of choice for anti-doping analysis in athletes because of noninvasive collection, amount of sample, and a reasonable window of detection. 6,59,60 Total blood, plasma, and serum samples for meldonium determination were used for other approaches, mainly pharmacokinetics, pharmacodynamics, bioequivalence, and biochemical studies. 19,20,24,32,61 –64 There are no scientific papers that have used alternative biological matrices, such as hair, nails, sweat, or oral fluid, despite the viability of these matrices, especially oral fluid (because meldonium lacks affinity for plasma proteins).
Tretzel et al. 26 investigated the possibility of using DBS for analysis; it is considered an alternative matrix. Recently, a study published by Thevis et al. 61 determined meldonium in exhaled breath using breath sampling units from SensAbues® (Huddinge, Sweden). This methodology allows the determination of this substance without invasive approach and great facility to collect the biological sample. 61
Sample treatment
Sample treatment varies according to the biological matrix and available equipment. Most studies use urine samples to determine meldonium concentration. For this specific biological matrix, dilutions with different solvents and combinations were evaluated in specific proportions, including acetonitrile:ammonium acetate, 60,65 acetonitrile:ammonium formate, 24,61 deionized water 58,60 or deionized water followed by an acetonitrile:methanol mixture. 32,51 Lv et al. 63 did not dilute urine samples, but rather mixed them with an internal standard solution and centrifuged the mixture to remove large particles. Subsequently, the supernatant was injected into a liquid chromatography–mass spectrometry (LC-MS) system. 63
For serum and plasma samples, deproteinization followed by centrifugation is necessary to obtain an appropriate supernatant. Some studies added acetonitrile:methanol,
32,50
acetonitrile:acetic acid,
24,61
or methanol.
19,62
–64
There are variations among the proportion of the solvent(s) and its combinations as well as the amount of sample. Liepinsh et al.
66
used solid phase extraction (SPE) with a SAX cartridge (silica gel with
DBS samples require different steps for adequate analysis. Ethylenediaminetetraacetic acid (EDTA)-stabilized whole blood is spotted on DBS cards followed by centrifugation. Plasma is separated from the erythrocyte fraction, and red blood cells retained on DBS cards are washed twice with phosphate-buffered saline (pH 7.4). Plasma and washed erythrocytes are spotted onto DBS cards and stored at 4°C in a plastic bag with desiccant until analysis. An online DBS sample preparation is performed using a DBS card autosampler directly coupled to an automated solid-phase extraction that employed an exchange module cartridge. The spots are extracted by flow-through desorption technology using acetonitrile:water (70:30, v/v). 26
Thevis et al.
61
developed a method to determine meldonium in exhaled breaths. The samples were collected from volunteers according to the SensAbues guidelines in an approximate volume of 20 L. Methanol was incorporated into the filter membrane that contained a cartridge. This device was gently agitated for 5 min, followed by elution of the solvent into a glass tube. Subsequently, the cartridge outlet was placed into the ground orifice of the tube and centrifuged at 660 ×
Equipment
Due to its structural and physicochemical characteristics, LC-MS is the most viable option for meldonium determination. 6,18,19,24,26,32,61,63,64 It is also possible to analyze this substance with liquid chromatography with a refractive index detector (LC-RID) 62 and capillary electrophoresis with capacitive detection of coupled conductivity (EC-C4D) using urine samples as the biological matrix. 59
There are no chromophore groups in meldonium; however, it is possible to identify this substance after a derivatization step. This procedure was employed for liquid chromatography with an ultraviolet detector (
A description of meldonium determination in biological matrices in vivo is presented in Table 3.
A resume of meldonium determination in biological matrices
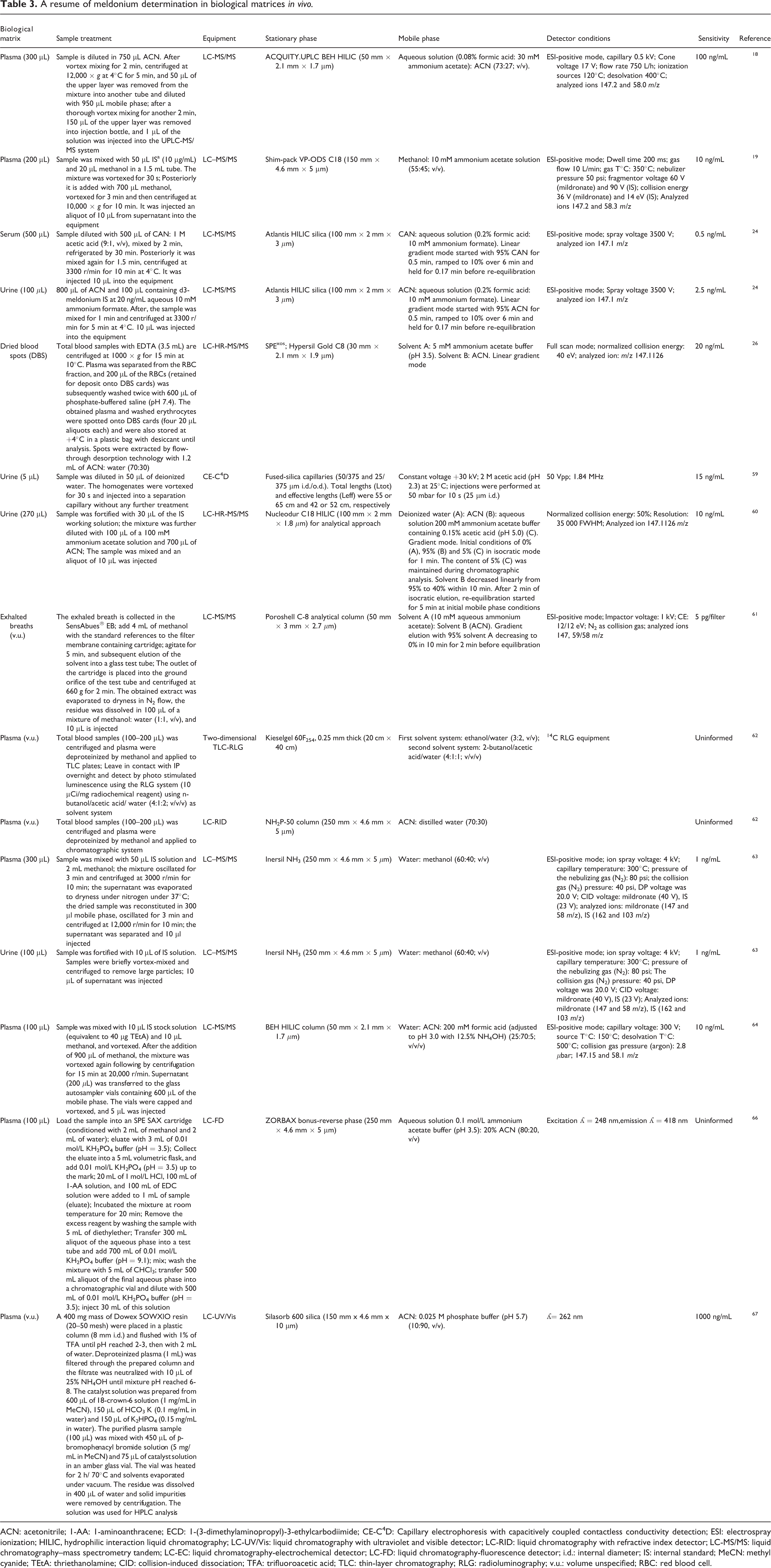
ACN: acetonitrile; 1-AA: 1-aminoanthracene; ECD: 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide; CE-C4D: Capillary electrophoresis with capacitively coupled contactless conductivity detection; ESI: electrospray ionization; HILIC, hydrophilic interaction liquid chromatography; LC-UV/Vis: liquid chromatography with ultraviolet and visible detector; LC-RID: liquid chromatography with refractive index detector; LC-MS/MS: liquid chromatography–mass spectrometry tandem; LC-EC: liquid chromatography-electrochemical detector; LC-FD: liquid chromatography-fluorescence detector; i.d.: internal diameter; IS: internal standard; MeCN: methyl cyanide; TEtA: thriethanolamine; CID: collision-induced dissociation; TFA: trifluoroacetic acid; TLC: thin-layer chromatography; RLG: radioluminography; v.u.: volume unspecified; RBC: red blood cell.
Discussion
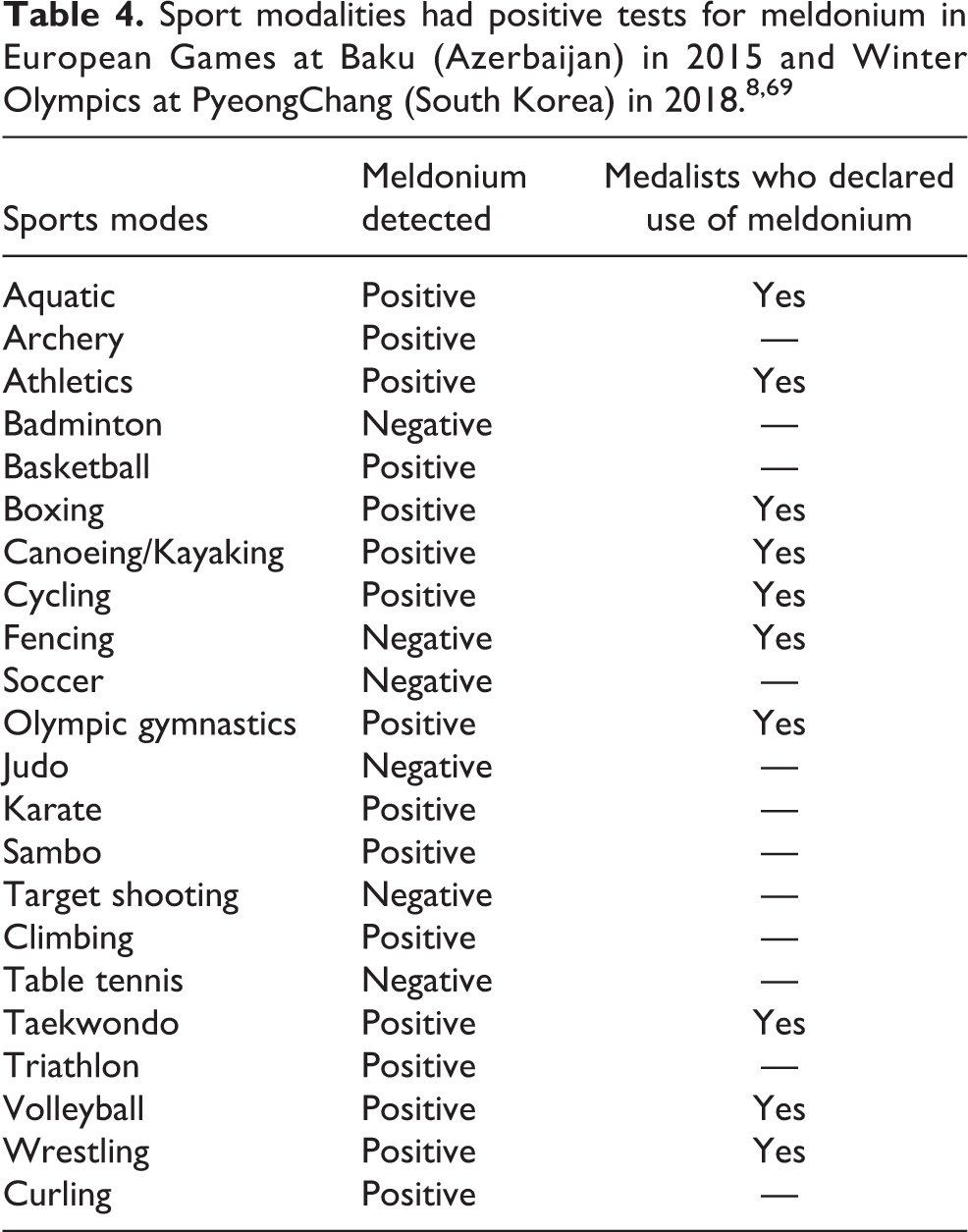
Recently, Russia has been involved in doping, a reality that makes the doping issue very complex. 68 Substances such as meldonium were being used indiscriminately by athletes, a large majority of whom were from Eastern Europe, and this situation came to a head at the 2015 European Games in Baku, Azerbaijan. In this competition, 66 athletes tested positive for meldonium (0.7–273 mg/mL); 23 athletes declared their meldonium use during the games and 13 athletes who used the substance won medals. Notably, of the 21 sports, meldonium use was detected in 15. 69 In 2016, athletes were caught and banned for using meldonium, 7 and most recently at the 2018 Winter Olympics in PyeongChang, South Korea. 8 Table 4 presents the sports for which athletes tested positive for meldonium at the last European Games and Winter Olympics. 8,69
Following the addition of meldonium to the Prohibited List, WADA stated that meldonium was banned for evidence that athletes would be using for the purpose of increasing the sporting performance. 9 In the current literature, there are few studies that link meldonium to sports performance in humans, although studies indicate its ergogenic effect. 6,10 –12,69
A study conducted with seven judokas from the Georgia National team who used 250 mg meldonium orally four times daily for 20 days concluded that meldonium could increase physical labor capacity. However, the methodology was of a relatively poor quality, due to the small sample size, the utilized dose, and other factors. 6
Male and female patients with chronic coronary disease and decreased physical tolerance (
Another study evaluated 119 male and female patients aged 30–80 years with heart failure and coronary atherosclerosis. They were separated into three groups: ML20 received meldonium (M) 1000 mg and lisinopril (L) 20 mg daily; ML5 received M 1000 mg and L 5 mg, and L20 (control) received L 20 mg daily. The groups were referred for the ergonomic bicycle test following the standard protocol: the initial load was 25 W, which was increased by 25 W within 3 min. Before treatment, the final load was approximately 70 W, and at the end of the treatment, it was approximately 80 W. The 6-min walk test was also performed, which evaluates the distance traveled over 6 min. At the end of treatment, the ML20 group covered approximately 25 m, an increase compared to the 14 m walked by the control group. 10
In animals, Baulin et al. 12 evaluated 10 adult albino rats in the forced swimming test that simulated a physical load of 7% of the rat body weight in a cylindrical pool. The test protocol was initial swimming, 5 min of rest followed by the second swimming session until exhaustion, and finally 10.7 mg/kg of meldonium (via oral gavage) followed by 40 min rest and the last swimming session. Meldonium improved recovery and increased performance by 65.02 and 49.4%, respectively, compared to the control. 12
Another approach to determine the ergogenic effects from meldonium could be by examining AMPK activation, as described by Liepinsh et al.. 37 Although this study did not directly test the relationship between physical exercise and meldonium, the authors reported increased AMPK activity. 37 This enzyme plays a key metabolic role in exercise, since it acts directly as a metabolic fuel gauge in skeletal muscle as the AMP/ATP and creatine/phosphocreatine ratios increase. Consequently, there is an inhibition of ATP-consuming pathways and activation of other pathways from carbohydrate and fatty acid metabolism to reestablish ATP levels. Thus, meldonium may increase skeletal muscle response to exercise training due to an ability to alter muscle fuel reserves and exercise-responsive gene expression based on AMPK activity. 13,70,71
Meldonium could also enhance physical performance by elevating testosterone levels. Bruveris et al. 72 administrated 2 g of meldonium (diluted with 2–3 mL of water) with food to wild boars. This study revealed a significant increase in testosterone levels compared to before treatment, namely, an increase from 2.0 ng/mL (±0.6) to 3.4 ng/mL (± 0.7). However, this mechanism remains unclear. Although the authors used wild boars, testosterone level increase is a very important factor for improving physical performance. If these conditions could be repeated in humans, it may explain the ergogenic effects of meldonium. Therefore, further studies should be performed. 72
In addition to effects related to exercise, a study demonstrated the beneficial effects of meldonium on the vasodilatory capacity of atrial conduction vessels and resistance vessels and during static muscle loading with the static load tolerance test. This study reported that the combination of meldonium and lisinopril improves quality of life, exercise capacity, and peripheral circulation mechanisms compared to lisinopril. 10 Therefore, as demonstrated by meldonium pharmacodynamics, it benefits sports performance and we must relate its use in doping to its effects.
Among the meldonium benefits (Table 5), its cardioprotective action is considered as the main positive effect. The increase of PPARα and PGC1α activity shifts beta oxidation from the mitochondrial matrix to peroxisomes in the cell cytosol. Therefore, PPARα activity is also related with rise of pyruvate dehydrogenase kinase 4 (PDK4) enzyme mRNA expression in all tissues, however, it was not shown to increase PDK4 protein expression. 73 Probably the increase of PDK4 enzyme mRNA expression may be related in an attempt to regulate glucose metabolism.
Possible ergogenic effects of meldonium.

CNS: central nervous system.
Studies have shown that meldonium increases glucose metabolism, decreases lactate production, and increases liver glycogen. 14,40,50 Therefore, the meldonium metabolic effect that decreases insulin resistance and increases glucose receptor expression (e.g. GLUT1 and GLUT4) provide higher glucose concentrations in tissues without hypoglycemia. 14,40,41 These effects decrease lactic acid production during exercise, which consequently reduces fatigue and improves physical recovery. 14,28,69,73
Activation of eNOS causes vasodilatation and increases blood flow in several organs, increasing the availability of substrates necessary for exercise. Since meldonium increases cerebral oxidation and hemodynamics, during long physical exercise there are several benefits. With optimal brain oxygenation during long periods of physical exercise, movement control, decision-making, and cognitive function are preserved. 74
BBOX inhibition by meldonium decreases
Meldonium decreases methemoglobin concentrations and consequently allows greater O2 transport by erythrocytes.
49
The structural similarity of meldonium to
Another important point that must be considered is posology (Table 2). The dose and administration time should correlate with the desired effect. For metabolic effects, changes in plasma and cardiac glucose levels and heart failure symptom reduction, at least 2 weeks of meldonium treatment is necessary. 40 For CNS effects and eNOS activation, a single administration promotes biological actions. 29,56 However, Tretzel et al. 26 showed ergogenic performance improvement in athletes based on oral doses of meldonium of up to 2.0 g per day over 2–3 weeks during pre-competitions period. 26
With regard to meldonium toxicity, Kirimoto et al. 44 demonstrated non-lethal meldonium doses of 25 and 100 mg/kg for female and male Sprague-Dawley rats, respectively. Thus, the non-lethal dose in female rats may be four times lower compared to male rats, a result that suggests females may have greater susceptibility to toxicity. 44 A hypothesis for this particular behavior may be the excretion rate. Forsdahl et al. 22 demonstrated differences between women and men volunteers, although it is not the purpose of the study. 22 It was verified by Rabin et al., 21 showing differences on meldonium excretion in male versus female human volunteers, where male volunteers showed a higher excretion ratio of this substance than female volunteers. In contrast, plasma concentrations in female volunteers were higher compared to male volunteers, 21 demonstrating greater susceptibility of female volunteers to meldonium toxicity.
This fact makes the use of meldonium even more dangerous, since athletes often use ergogenic substances without adequate knowledge about them. However, Zhang et al. 18 administered meldonium orally in 40 Chinese volunteers (20 men and 20 women), specifically a single dose of 250, 500, 1000, 1250, or 1500 mg, and volunteers who received the 500 mg dose continued treatment for 13 days three times daily. The study revealed no meldonium sex difference; 18 however, it is possible that this dose does not reveal potential differences. Furthermore, the administration route utilized which was not reported by Kirimoto et al. 44 may influence sex differences.
Considering the collateral effects of meldonium, one point that should be explored is the use of this substance in the medium and long periods for athletes and/or healthy people and their respective negative effects. In fact, at this moment, it was not finding any data addressing chronic exposure to meldonium in these populations.
Meldonium determination is another important point (Table 2); a medalist detailed the use of meldonium as a doping agent, but this substance was not detected in the athlete’s sample. There is no official method for analysis by anti-doping agencies; however, high-resolution MS-MS or MS equipment is necessary for confirmatory purposes. For WADA, this agency establishes the minimum concentration to detect the Reliable Substance called Minimum Required Performance Levels (MRPL) at 200 ng/mL in urine samples. 6,65,77 There are many methods to identify meldonium in biological matrices, and most of them are based on dilute-and-shoot after sample preparation (blood and/or urine) and LC-MS determination. Given meldonium toxicokinetics, it is possible to determine this molecule in the body for several days, even after use cessation.
According to Knych et al., 24 meldonium accumulates in tissues due to its affinity for OCTN2, which leads to reabsorption in the renal tubules and uptake in various tissues that express this transporter. 24 However, Peng et al. 19 suggest that meldonium accumulates in plasma while Rabin et al. 21 showed higher levels of plasmatic meldonium in female volunteers compared to male volunteers based on the excretion capacity of this substance. 21 Forsdahl el al. 22 noted that meldonium has a three-compartment model excretion profile, which these authors verified as meldonium detection in urine in a period between 94 days and 162 days. 22 Another explanation for meldonium accumulation is the ability of this molecule to bind to erythrocytes (16–28 days), even after substance consumption ends. 26 In this sense, DBS samples seem to have great potential for examining meldonium accumulation, as demonstrated by Tretzel et al. 26
For screening tests, other approaches could be used to determine meldonium use. One possibility is assessing urine and/or blood
Conclusion
The use of meldonium for sports competitions is considered to violate sports ethics according to WADA. In this review, it was verified that meldonium showed ergogenic effect in animals and humans volunteers. However, this drug has therapeutic applications in patients with cardiovascular diseases. Also, meldonium can be used for the treatment of other pathologies, and it has no pronounced adverse effects, demonstrating a certain safety and effectiveness. In fact, this molecule shows high lethal dose and its positive effects are pronounced at doses much lower than its lethal dose, which decreases problems due to overdose. Therefore, there are few studies that demonstrate the ergogenic effect of meldonium and its mechanisms of action, mainly in athletes. The same aspect occurs with toxicological effects of meldonium, for which there are limited toxicological data. Furthermore, studies with meldonium are required mainly in healthy individuals, and new analytical methodologies should be developed for the detection of this molecule in anti-doping tests.
Footnotes
Acknowledgement
The authors thank CAPES for the Master’s scholarship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.