Abstract
An increase in copper concentration in body may lead to hepatolenticular degeneration which is considered as one clinical feature of Wilson’s disease. Chelation therapy using
Introduction
Copper, the third most abundant essential transition metal in human tissues, is absorbed from diet through intestinal enterocytes and reaches to the hepatic circulation. 1 The fate of absorbed copper involves sequestration by glutathione (GSH), mobilization to targets like superoxide dismutase 1 (SOD1), cytochrome c oxidase and copper-transporting Adenosine triphosphate (ATP). Specific copper-transporting ATPase has two subtypes ATP7A and ATP7B which mobilizes copper in to trans-Golgi network in response to the peripheral demand of copper. More specifically, ATP7A appears to be responsible for distribution of copper in the blood stream (for their incorporation in copper-dependent enzymes) while ATP7B facilitates the biliary excretion of copper. 2
Copper homeostasis ensures the proper distribution of copper to essential proteins without causing any adverse conditions to cellular defence system. Copper toxicity cases have been observed primarily due to accident/occupational hazards, environmental contamination and ATP7B gene mutation, which lead to accumulation of copper in hepatic and central nervous system. 3 The elevated copper level has been reported to be associated with several diseases including Wilson’s, Alzheimer’s and Parkinson disease. 4,5 Copper overload has been reported to cause mitochondrial dysfunction and to reduce antioxidant factors which may lead to oxidative damage. 6
Current management of copper toxicity involves use of chelating agents (
Monoisoamyl-2, 3-dimercaptosuccinic acid (MiADMSA), a C5-branched-chain alkyl monoester of dimercaptosuccinic acid, contains two vicinal sulfhydryl groups responsible for its chelating property. MiADMSA has potential chelating property against arsenic toxicity 12,13 and is under clinical trials. Copper depletion from different organs was reported as a potential adverse effect of MiADMSA, 12 which advocates its possible use in copper toxicity.
The present study was thus planned to (i) compare the therapeutic efficacy of MiADMSA and
Materials and methods
Drugs and chemicals
Copper sulphate (CuSO4.5H2O) and
Animals
Female Sprague Dawley rats (220–250 g) obtained from the Animal House Facility of CSIR-Central Drug Research Institute, Lucknow, India, were used in the study. Animals were kept in standard cages and had access to water and food (standard chao pallet from ATNT, Germany) ad libitum. They were maintained on ad libitum pellet diet and water in an air-conditioned room (room temperature of 25 ± 2°C) with regular alternate cycles of 12 h light and darkness. The metal contents of the animal feed (in ppm dry wt.) were Cu 10, Mn 55, Co 5, Zn 45 and Fe 70. Before the start of the experiment, the animals were acclimatized to the laboratory conditions for a week and experimental works were carried out from 08:00 h to 16:00 h. All the experimental protocols included in this study were duly approved by the Institutional Animal Ethics Committee of National Institute of Pharmaceutical Education and Research, Raebareli, and the care of the animals was taken according to the guidelines provided by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forest, Government of India.
Experimental protocol
This study included two major groups. Group I (naïve group, n = 5) consisted of untreated normal animals, while group II was exposed to copper exposure (CuSO4.5H2O, 100 mg/kg/day, p.o.; n = 15) for 6 weeks.
14
After 6 weeks, copper-exposed animals were subdivided into three groups. Subgroup IIa received vehicle treatment (normal saline; 4 ml/kg; p.o.); subgroup IIb received
Different treatments in the subgroups were given for 5 days. All the animals were euthanized after 48 h of the last treatment.
Behaviour evaluations
The effect of different treatments on behaviour was evaluated during and after exposure.
Assessment of spontaneous locomotor activity
The effect of different treatments on spontaneous locomotors activity was evaluated using Optovarimex-4 (Columbus Instruments, Columbus, Ohio, USA) following the method described by Bernal-Morales and colleagues with slight modification. 16 The cages of this instrument are equipped with horizontal and vertical sets of infrared photocells, which send continuous unseen light beams. The number of light beam interruptions due to the movement of animal inside the cage was automatically recorded. Each rat was placed individually in the activity cage floor, and distance travelled and resting time of each animal were recorded within 10 min duration as an index of locomotor activity. To remove any confounding olfactory stimulus, the apparatus was cleaned with alcohol–water solution after each rat. The experiment was repeated on each animal on 4th and 6th week of exposure and after the treatment with various drugs.
Rotarod test
The muscle strength, coordination and balance of rats were assessed through the rotarod test, as determined by Shiotsuki et al. 17 The apparatus consisted of a horizontal cylinder (diameter 6 cm), elevated 20 cm above the switch floor. The cylinder was divided into four separate parts by circular plates, so that four rats could be tested simultaneously. Each rat was placed on the rotating cylindrical rod at a constant speed of 25 r/min, and the fall-off time was recorded. The experiment was repeated on each animal on 4th and 6th week of exposure and after the treatment with various drugs.
Forced swim test
The forced swim test mainly evaluates depression like phenotypes. It consisted of cylindrical tank containing water at 24 ± 1°C and animals were placed individually. The immobility period was noted in 6 min duration. 18 Immobility was defined as the total absence of movement in the whole body, that is, when the animal stopped struggling and kept motionless, floating on water, or when the animal was only doing the necessary movements to keep its head above the water. Subsequently, the animals were removed from the water and dried with towel and returned to their home cages.
Passive shock avoidance test
For the passive avoidance test, all the rats were introduced into the light compartment, over 3 consecutive days following the method described by Lee and Noh 19 with slight modifications. Briefly, the passive shock avoidance test apparatus (PACS-30; Columbus Instruments, USA) consisted of two compartments (23 × 24 × 24 cm3), a light and a dark compartment divided by a guillotine door. The instrument was set to run in passive mode with conditioned stimulus light intensity of 10 lux and unconditioned stimulus of 0.3 mA current with door closure upon transfer and inescapable shock mode. The rats were placed one at a time, in the lighted compartment, following which the door separating the two chambers was automatically lifted up. As soon as the rat entered the dark compartment, the separating door was closed and inescapable shock was delivered. After the shock, animals were returned to their home cages This process was repeated for 5 trials a day for 3 days. Thereafter acquisition of the learned task was measured by placing the animal again in light compartment. During acquisition trial, the shock was not delivered. The transfer latency was recorded as the index of memory.
Biochemical estimations
After behavioural assessments (after 48 h of last treatment), all the rats were euthanized by cervical dislocation and blood was collected in heparinized vials by cardiac puncture. After blood collection, brain, liver and kidneys were isolated for biochemical estimation. The blood samples were immediately analysed for changes in δ-aminolevulinic acid dehydratase (δ-ALAD) level. On the other hand, the brain samples were analysed for the changes in acetylcholinesterase (AChE) activity. Rest all samples (blood, brain, liver and kidney) were analysed for the changes in pro/antioxidant markers. Blood haemoglobin was analysed using Sahli’s apparatus, haematocrit count was estimated using Wintrobe’s tube, and plasma aspartate transaminase level was monitored using Automatic Clinical Biochemistry Analyser (EM200, Transasia). Additionally, metal content analyses were done in tissue samples (brain, liver and kidney) to determine the changes in copper and zinc content.
Blood δ-ALAD
The activity of δ-ALAD was determined by the method of Berlin and Schaller. 20 Briefly, heparinized blood was haemolysed at 37°C for 10 min followed by addition of aminolevulinic acid (ALA) solution (0.083 g ALA in 50 ml of phosphate buffer, pH 7.4) in the experimental tube while trichloroacetic acid (TCA) was added to the control tubes. Tubes were mixed well and incubated for 60 min at 37°C. The reaction was stopped with TCA in experimental tube and ALA in controls. Supernatant was collected following centrifugation and equal volumes of Ehrlich reagent were added. Absorbance at 555 nm was noted, and activity was expressed as nanomoles per minute per millilitre.
Glutathione
The analysis of blood GSH concentration was performed by the method described by Ellman 21 and modified by Jollow et al. 22 In brief, 0.2 ml of whole blood was added to 1.8 ml of distilled water and incubated for 10 min at 37°C, for complete hemolysis. After adding 3 ml of 4% sulphosalicylic acid, tubes were centrifuged at 2500 r/min for 15 min. To the supernatant, 0.2 ml of 10 mM solution of 5,5-dithiobis-(2-nitrobenzoic acid) (DTNB) was added in the presence of phosphate buffer (0.1 M, pH 7.4). Absorbance recorded at 412 nm was used for the calculation of GSH concentration.
Reduced GSH and oxidized glutathione contents
Reduced GSH and oxidized glutathione (GSSG) levels were measured fluorometrically using the method of Hissin and Hilf. 23 The tissue homogenate (10%) was prepared in 3.75 ml of 0.1 M sodium phosphate, 0.005 M EDTA buffer, pH 8.0 and 1.0 ml 25% HPO3. The homogenate was centrifuged at 10,000 × g (Centrifuge 5810 R, Eppendorf, Germany) for 30 min at 4°C. Supernatant obtained was used for the assay. For GSSG assay, 0.5 ml supernatant was incubated at room temperature with 200 µl of 0.04 M N-ethyl maleimide for 30 min to interact with GSH present in the tissue. To this mixture, 4.3 ml of 0.1mol NaOH was added. A 100 µl sample of this mixture was taken for the measurement of GSSG using the procedure described above for the GSH assay, except that 0.1 M NaOH was used as the diluent instead of phosphate buffer. After dilution, 0.2 ml O-phthaldehyde (200 µg/ml in absolute methanol) was added. The mixture was thoroughly mixed, incubated for 15 min at room temperature. The fluorescence is measured at excitation wavelength (λ Ex) 350 nm and emission wavelength (λ Em) 420 nm. Standard stock solutions of GSH and GSSG were prepared and calibration curve was prepared using similar assay conditions as used for experimental tubes. The concentration of reduced GSH and oxidized GSSG was expressed as micrograms per gram of tissue.
Reactive oxygen species
The amount of reactive oxygen species (ROS) in blood was measured using 2,7-dichlrofluoresceindiacetate (DCF-DA) that gets converted into highly fluorescent DCF by cellular peroxides (including hydrogen peroxide). The assay was performed as described by Socci et al. 24 For the estimation of ROS, 5% RBC haemolysate/10% tissue homogenate was prepared and diluted to 1.5% with ice-cold 40 mM Tris-HCl buffer (pH 7.4). The tissue was homogenized (10 mg) in 1 ml of ice-cold 40 mM Tris-HCl buffer (pH 7.4), further diluted to 0.25% with the same buffer and placed on ice. Then, 40 ul of 1.25 mM DCF-DA in methanol was added for ROS estimation. All samples were incubated for 15 min in a 37°C water bath. Fluorescence was determined at 488 nm excitation and 525 nm emission using a fluorescence plate reader.
Thiobarbituric acid reactive substances
The measurement of lipid peroxidation was done by the method described by Ohkawa et al. 25 Tissue lipid peroxidation was measured in tissue homogenate (5% homogenate (w/v) in 150 Mm KCl for brain and 10% homogenate (w/v) in 150 mM KCl for liver and kidneys) for 30 min at 37°C. The incubation was interrupted by adding 1 ml of 10% TCA. After centrifugation, 1 ml of the supernatant was mixed with 1 ml of 0.65% thiobarbituric acid. The mixture was then kept in a boiling water bath for 15 min to get the red colour of thiobarbituric acid–malondialdehyde (MDA) complex, the absorbance of which was recorded at 535 nm. The amount of thiobarbituric acid reactive substances (TBARS) was calculated using a molar extinction coefficient of 1.56 × 105/M/cm.
In case of blood, 0.1 ml of 5% RBC haemolysate was added to 0.2 ml of 8.1% Sodium Dodecyl Sulfate (SDS) (w/v) and incubated for 10 min; 1.5 ml of 20% acetic acid (pH 3.5) was added followed by the addition of 1.5 ml of 0.8% thiobarbituric acid (w/v) and 0.7 ml distilled water. The mixture was then incubated for 1 h in boiling water bath. One millilitre of distilled water was added to the solution after cooling and centrifuged at 6000 r/min for 15 min. The absorbance of supernatant was read at 532 nm, and the values were expressed as moles of MDA per millilitre.
Superoxide dismutase
The SOD activity in whole brain, liver, kidneys and blood was assayed spectrophotometrically as described by Kakkar et al. 26 Reaction mixture contained 1.2 ml (0.052 mM, pH 7.0) sodium pyrophosphate buffer, 0.1 ml (186 µM) phenazinemethosulphate and 0.3 ml (300 µM) nitro blue tetrazolium; 0.2 ml of the supernatant obtained after centrifugation (1500 × g, 10 min followed by 10,000 × g, 15 min) of 5% haemolysate/homogenate (prepared in phosphate buffer, pH 7.4, 100 mM) was added to the reaction mixture. Reaction was initiated by adding 0.2 ml NADH (780 µM) and stopped by adding 1 ml glacial acetic acid. Colour intensity of the chromogen was measured at 560 nm, and the activity was expressed as units per minute per milligram of protein.
Catalase activity
The catalase activity in tissue was assayed following the procedure of Sinha, 27 at room temperature; 0.1 ml of 5% RBC haemolysate/tissue homogenate was incubated with 0.5 ml of H2O2 (0.2 M) at 37°C for 90 s precisely, in the presence of 0.01 M phosphate buffer (pH 7.4). Reaction was stopped by adding 5% dichromate solution. Further samples were incubated at 100°C for 15 min in boiling water bath. Amount of H2O2 consumed was determined by recording absorbance at 570 nm.
Glutathione peroxidase
The glutathione peroxidase (GPx) activity was measured by the procedure of Flohé and Günzler. 28 Supernatant obtained after centrifuging 5% tissue homogenate for 10 min at 1500 × g, followed by 10,000 × g for 30 min at 4°C, was used for GPx assay. One millilitre of reaction mixture was prepared, which contained 0.3 ml of phosphate buffer (0.1 M, pH 7.4), 0.2 ml of GSH (2 mM), 0.1 ml of sodium azide (10 mM), 0.1 ml of H2O2 (1 mM) and 0.3 ml of tissue supernatant. After incubation at 37°C for 15 min, reaction was terminated by the addition of 0.5 ml of 10% TCA. Tubes were centrifuged at 1500 × g for 5 min and supernatant was collected; 0.2 ml of phosphate buffer (0.1 M, pH 7.4) and 0.7 ml of DTNB (0.4 mg/ml) were added to 0.1 ml of the reaction supernatant. After mixing well, absorbance was recorded at 420 nm.
Glutathione-S-transferase
The glutathione-S-transferase (GST) activity was determined following the procedure of Habig et al. 29 Supernatant was obtained after centrifuging 5% tissue homogenate at 1500 × g for 10 min followed by 10,000 × g for 30 min at 4°C. Reaction mixture contained 0.02 ml of 1-chloro-2,4-dinitrobenzene (1 mM), 2.9 ml of GSH (0.3 mg GSH/ml in 0.2 M phosphate buffer, pH 7.4) and 30 ml of tissue supernatant. Change in colour was monitored by recording absorbance (340 nm) at 30 s intervals for 3 min.
Nitrite estimation
The nitrite content was measured in tissue homogenates using Griess reagent method, with slight modification suggested by Mishra and Goel. 30 Fifty microlitre of the filtered homogenate (10% w/v in 50 mM, phosphate buffer pH 7.4; centrifuged at 6000 × g for 20 min at 4°C)/standard (NaNO2) was mixed with 50 µl of the Griess reagent using 96 well plates. The plate was shaken at 150 shakes/min for 1 min to ensure proper mixing of samples/standard with Griess reagent. The plate was incubated for 30 min at room temperature. The absorbance of nitrite containing samples was measured at 540 nm using Microplate Reader (Multiscan FC, Thermo Scientific, Mumbai, India) against the photometric reference (blank: 50 µl HPLC grade water + 50 µl Griess reagent). The concentration of total nitrite was estimated using the straight line equation of nitrite (y = 0.0054x + 0.0915; R 2 = 0.9902; where x is the concentration in ng/ml and y is the area under peak) drawn against standard solution of sodium nitrite at 10, 20, 40, 60, 80 and 100 ng/ml. The results were expressed as nanograms per gram of wet tissue.
AChE activity
For the estimation of brain AChE activity, Ellman method was followed with slight modification suggested by Mishra and Goel. 30 Briefly, 40 µl of filtered brain homogenate (10% w/v in 50 mM, phosphate buffer pH 7.4; centrifuged at 6000 × g for 20 min at 4°C) was mixed with 160 µl of Ellman’s reagent (39.6 mg DTNB and 15 mg NaHCO3 dissolved in 10 ml 50 mM phosphate buffer pH 7.4) and 200 µl of 50 mM phosphate buffer (pH 8) in 96 well plate and shaken properly. The absorbance of the reaction mixture was recorded prior to the addition of substrate at 412 nm. The reaction was initiated by adding 10 µl of the enzyme substrate (10 mM acetylthiocholine iodide) to each well and was allowed to incubate for 15 min. Yellow colour developed, and the solution was read at 412 nm using Microplate Reader (Multiscan FC, Thermo Scientific). A molar extinction coefficient of 14,150/M/cm was used to calculate enzyme activity. Enzyme activity was expressed as micromolar of acetylcholine hydrolyzed per milligram of wet tissue.
Metal estimations
Copper concentrations in different tissues (brain, liver and kidney) were measured after wet acid digestion using nitric acid and perchloric acid (5:1 ratio). Samples were brought to a constant volume and determination of tissue copper content was performed using an ICP-OES Spectrometer (Optima 8000, Perkin Elmer, Mumbai, India). 31
Statistical analysis
Statistical analysis was performed using Student’s t test (observations during copper exposure at 4th and 6th week), one-way analysis of variance followed by Tukey’s test to compare means among the different treatment groups. Statistical significance was considered at p < 0.05, and all values were expressed as mean ± standard error of mean (SEM).
Results
Exposure to CuSO4.5H2O was continued for 6 weeks and after a gap of 7 days copper pre-exposed animals were treated with
Effect on spontaneous locomotor activity
Oral administration of copper for 4 weeks have not shown significant changes in distance travelled and resting time as compared to naïve animals. However, 6 weeks of copper treatment significantly (p < 0.05) decreased distance travelled and increased resting time as compared to naïve animals. After discontinuation of copper exposure and simultaneous administration of vehicle,
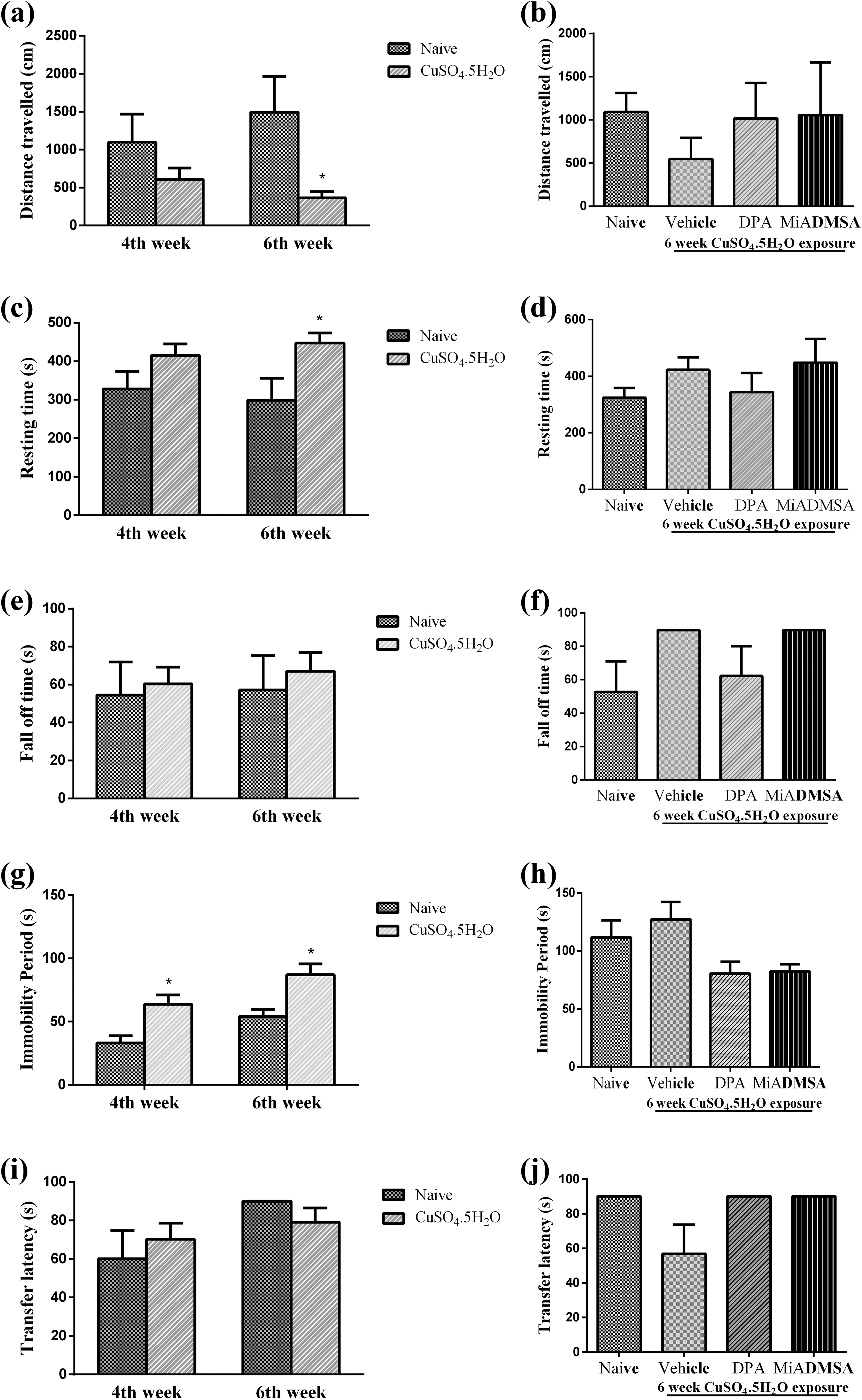
Effect of different interventions on neurobehavioural functions. (a to d) Effect of different interventions on distance travelled and resting time in spontaneous locomotor activity test, (e) effect of copper exposure on fall-off time in rotarod test, (f) effect of
Effect on fall-off time in rotarod test
The treatment with copper did not alter the fall-off time in rotarod test when compared with that of naïve animals. Similarly, no significant change in fall-off time was recorded on day 5 of treatment in different treatment groups (F (3,14) = 1.567; p = 0.25; Figure 1e and f).
Effect on immobility period in forced swim test
The immobility period was significantly increased (p < 0.05) after 4 and 6 weeks of copper treatment as compared to that of naïve animals. After discontinuation of copper treatment and simultaneous administration of vehicle,
Effect on transfer latency in passive shock avoidance test
There was no significant change in transfer latency observed in copper-treated group after 4 and 6 week of treatment as compared to that of naïve animals. Similarly, no significant change in transfer latency was recorded on day 5 of treatment in different treatment groups (Figure 1i and j).
Effects on haematopoietic variables and blood oxidative stress
Six weeks treatment with CuSO4.5H2O was not found to alter haemoglobin level, percent haematocrit level and AST level (Figure 2). Further, there were no significant changes in haemoglobin level, percent haematocrit level and AST level with D-Pencillamine and MiADMSA treatments.
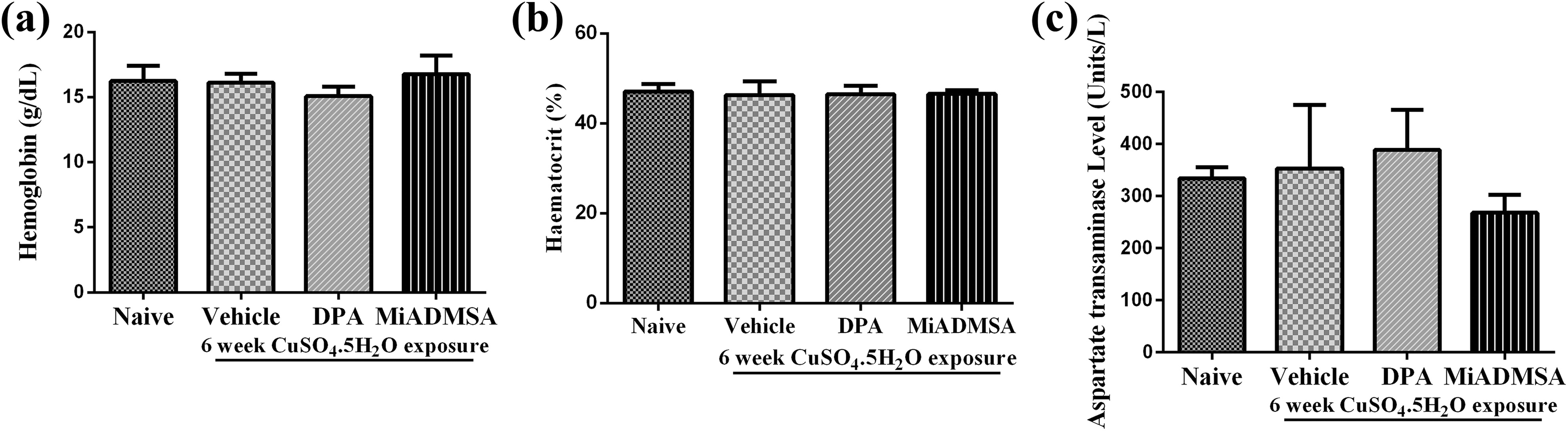
Effect of
Effect of different treatment in different oxidative stress markers is shown in Figure 3. The treatment with copper for 6 weeks did not alter blood ALAD level (Figure 3a), GSH:GSSG ratio (Figure 3d) and ROS level (Figure 3g), as compared to naïve animals. However, with copper exposure, activities of catalase (Figure 3b), GPx (Figure 3c), GST (Figure 3f), SOD (Figure 3h), and TBARS levels (Figure 3i) increased significantly (p < 0.05) compared to that of naïve animals. The blood GSH level was significantly reduced (p < 0.05) in copper exposure group as compared to that of naïve group (Figure 3e). The treatment with
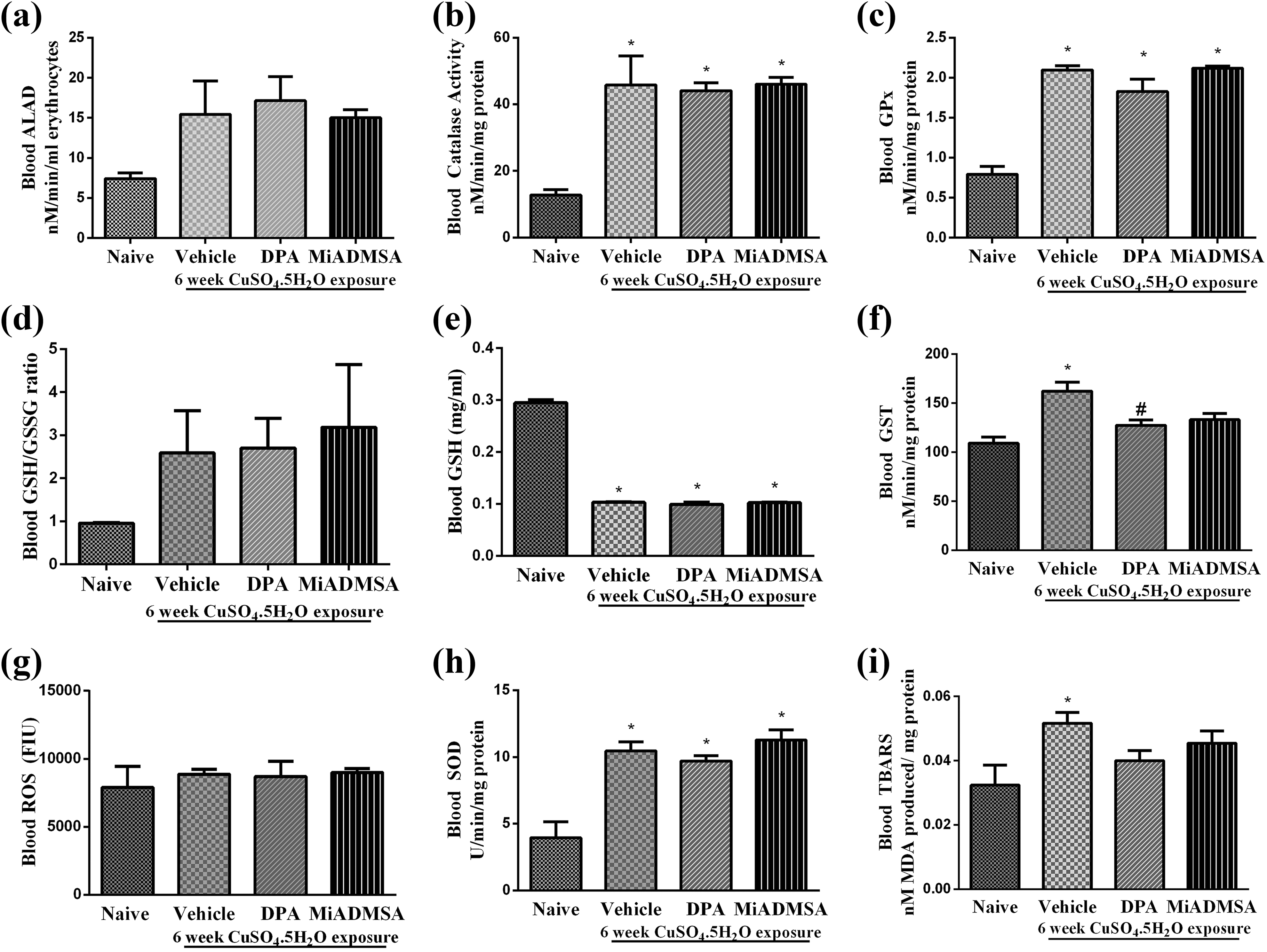
Effect of
Effect on brain oxidative stress
Effect of different treatments on brain oxidative stress and AChE activity has been shown in Figure 4. Treatment with copper for 6 weeks duration significantly increased (p < 0.05) TBARS levels (Figure 4h) in brain and significantly reduced (p < 0.05) the GPx levels (Figure 4b) in brain as compared to that of naïve animals. However, the levels of ROS (Figure 4f), SOD (Figure 4g), nitrite (Figure 4e), GSH:GSSG ratio (Figure 4c), GST level (Figure 4d), catalase activity (Figure 4a) and AChE activity (Figure 4i) remained unchanged with copper treatment as compared to that of naïve animals. Treatment with
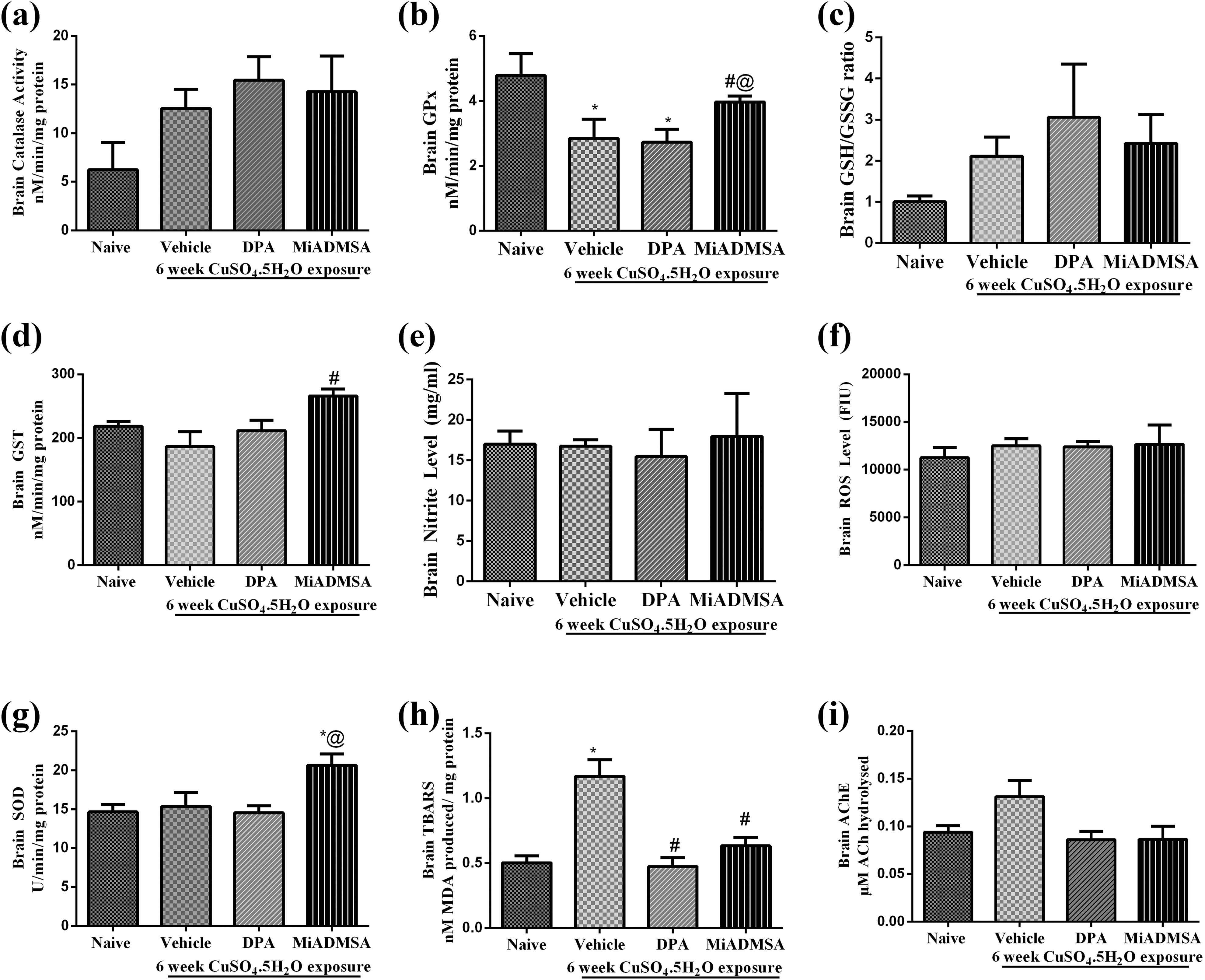
Effect of
Effect on hepatic oxidative stress
Effect of different treatments on hepatic oxidative stress has been mentioned in Figure 5. Treatment with copper for 6 weeks significantly decreased (p < 0.05) the GST (Figure 5d) and GPx levels (Figure 5b) in liver as compared to that of naïve animals. However, the levels of ROS (Figure 5f), SOD (Figure 5g), nitrite (Figure 5e), GSH:GSSG ratio (Figure 5c), catalase activity (Figure 5a) and TBARS (Figure 5h) in liver remained unchanged with copper treatment as compared to that of naïve animals. Treatment with
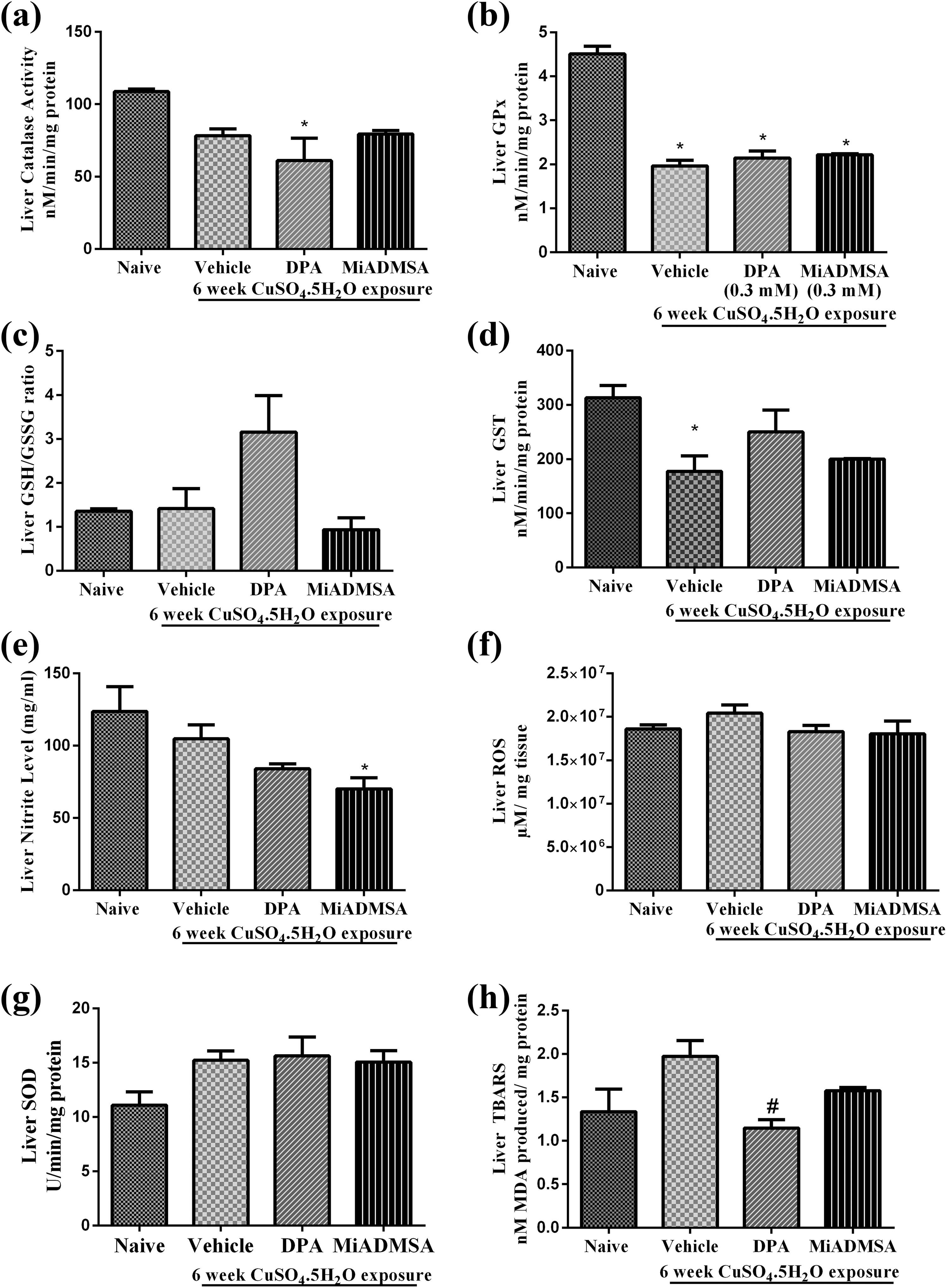
Effect of
Effect on renal oxidative stress
Effect of different treatments on renal oxidative stress has been illustrated in Figure 6. Treatment with copper for 6 weeks significantly increased (p < 0.05) the nitrite level (Figure 6e), GSH:GSSG ratio (Figure 6c) and TBARS level (Figure 6h) and significantly reduced catalase activity (Figure 6a) and GST level (Figure 6d) in kidney as compared to that of naïve animals. However, the levels of ROS (Figure 6f), SOD (Figure 6g) and GPx (Figure 6b) in kidney remained unchanged with copper treatment as compared to that of naïve animals. The treatment with
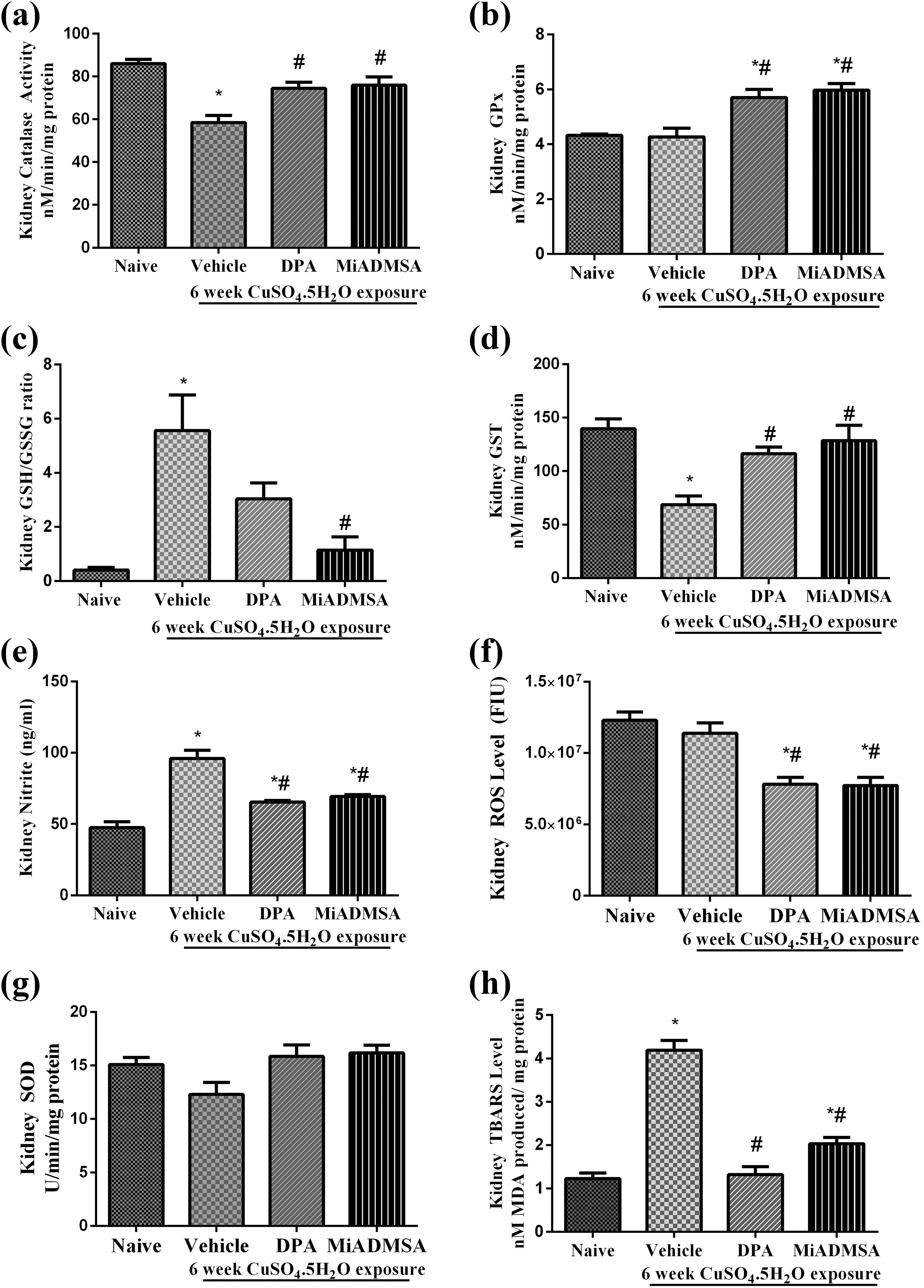
Effect of
Effect on copper concentration
Copper concentrations in different tissues are shown in Figure 7a to c. Copper exposure for 6 weeks significantly raised (p < 0.01) copper load in liver compared to animals from naïve group. Treatment with
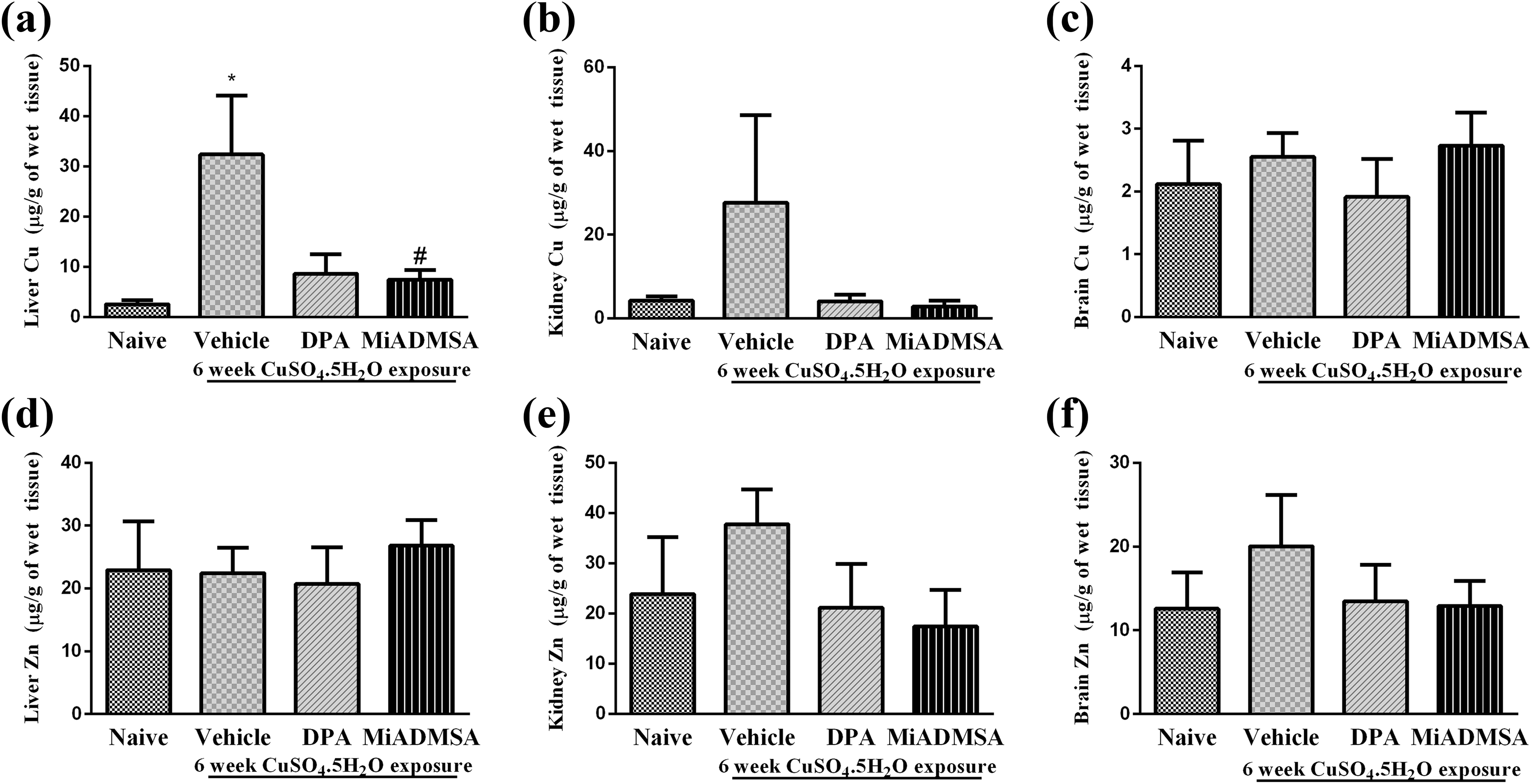
Effect of
Effects of different treatments on zinc concentration in different tissues are mentioned in Figure 7c to e. Neither copper exposure nor subsequent treatment with
Discussion
We compared the efficacy of MiADMSA and
Orally administered copper gets absorbed through intestine and reaches liver. Being a transition metal copper undergoes redox cycling to promote formation of ROS. In the presence of GSH and superoxides, Cu++ gets converted to Cu+ (Haber–Weiss reaction) which further facilitates the formation of hydrogen peroxides and hydroxyl radicals.
34
Copper interacts with GSH and forms Cu(I)-[GSH](2) complex, which reduces molecular oxygen into superoxide,
34,35
and further gets converted to H2O2 by the action of SOD. This might be the one possible reason for the reduced GSH level and increased SOD activity in the blood of copper-exposed animals. Further, copper exposure significantly raised blood catalase, GPx and GST activities which might suggest the activation of defensive mechanisms to protect against copper-associated free radical generation. In oxidative stress conditions, it is well known that there is an accompanied increase in catalase, GPx and GST levels.
36
The peroxides generated due to copper exposure appear to be detoxified by catalase and GPx. Elevated GST activity suggests the neutralization of strong electrophile (free radicals and metal ions) by their interaction with GSH. Elevated TBARS levels on copper exposure suggest copper-mediated oxidative damage. Treatment with
Around 90% of total SOD activity in eukaryotic cells is attributed by CuZnSOD. It is abundant in cytosol, however, minor fractions have also been noticed in cellular organelles. The SOD activity is essential in terms of protection against ROS and prevents their leakage from mitochondria. 37 In our study, 6 weeks of copper exposure remarkably increased total SOD activity in blood. The elevation in SOD activity suggests the activation of antioxidant defence mechanism in response to mild oxidative stress.
Liver has been suggested as the primary storage site and a possible target for copper.
38
Data for metal analysis suggested significantly higher amount of copper in liver in copper-exposed animals (naïve animals). Pronounced oxidative damage in liver might be associated with higher copper concentration in liver. Treatment with
Renal copper concentration remained unchanged in different groups, however, we noted moderate (non-significant) rise in copper contents in kidney in the copper-exposed animals and a small depletion (non-significant) by
Free copper has been reported to cross blood–brain barrier
37
and may show potential toxic effects via free radical-mediated oxidative damage. In brain, rapid sequestration of copper occurs with GSH which might have led to the reduction in GSH level in copper-exposed animals.
2
However, in our study, we noted no significant change in brain copper content in different groups which support the behavioural findings; however, a significant depletion of GPx activity and elevation in TBARS levels were recorded. The treatment with
In addition to excess copper, zinc deficiency has been linked to Wilson’s disease. 14 Apart from this, deficiency of essential metal ions has been reported as the side effect of chelation treatment. 43,44 Therefore, in this study, tissue zinc levels have also been monitored and noted that zinc levels in different tissues were found to be unaffected.
Conclusion
A comparable therapeutic efficacy of both
Footnotes
Acknowledgements
The authors would like to acknowledge the Department of Pharmaceuticals, Ministry of Chemical and Fertilizers, Govt. of India, and National Institute of Pharmaceutical Education and Research (NIPER) Raebareli for necessary infrastructure and facility. The authors would also like to acknowledge Prof AA Mahdi, Head, Department of Biochemistry, King George Medical University, Lucknow, India, for extending facility for the metal estimations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
