Abstract
The term oxidative stress was introduced by the National Toxicology Program (NTP) in 1985. It is now invoked as a contributing mechanism to the induction of tumors in chronic rodent cancer bioassays following administration of a chemical agent, especially via inhalation. The term oxidative stress is confusing as there are two different types of oxidative stress. Direct oxidative stress derives from chemical damage to mitochondrial energy-producing structures with concomitantly altered mitochondrial function. Indirect oxidative stress is frequently quantitatively greater than direct oxidative stress in terms of release of activated oxygen or nitrogen species secreted by one or more of 12 potential in situ or recruited inflammatory cell types. Evidence supporting a role for direct oxidative stress in chemical carcinogenesis comes from cell culture studies wherein the cells have been removed from their natural environment and denuded of defense systems, for example, removal of particles by cilia, macrophage engulfment of particles, and suspension and dilution of chemicals or particles in pulmonary surfactant. In contrast, extensive direct evidence from bronchoalveolar lung washes conducted on humans shows elevations in pulmonary macrophage and neutrophil counts, release of intravascular proteins into the extracellular space, and activation of pro-inflammatory cytokines that correlate with observed pulmonary pathology. Higher rates of lung cancer in smokers with chronic obstructive pulmonary disease (COPD) than in smokers at the same cigarette consumption level but without COPD demonstrate a positive contribution by pulmonary inflammation to pulmonary carcinogenesis. Indirect oxidative stress from pulmonary inflammation should be considered before allocating a primary carcinogenic role for direct oxidative stress.
Introduction
The toxicology and pathology literature commonly use the term oxidative stress. This terminology is confusing because there are two types of oxidative stress. Direct oxidative stress derives from chemical damage to mitochondrial energy-producing structures with concomitantly altered mitochondrial function. Indirect oxidative stress is frequently quantitatively greater than direct oxidative stress in terms of release of activated oxygen or nitrogen species secreted by one or more of 12 potential in situ or recruited inflammatory cell types. In this review, the literature on the two different types of oxidative stress will be critically evaluated to demonstrate that in inhalation exposures of chemicals or complex chemical mixtures that it is the indirect variety of oxidative stress resulting from pulmonary inflammation that predominates.
Between 1978 and 2018, National Toxicology Program (NTP) conducted 596 studies and issued 480 reports. The term oxidative stress was introduced in 1985. 1,2 Three hundred of the 596 studies had been completed prior to the use of the term oxidative stress. Since the introduction of the term in 1985, NTP has proffered oxidative stress as a potential contributor to the formation of rodent tumors on 58 different occasions. 3 The 58 different cases result from an amalgamation of attributions. First, in several reports written after 1985, the authors retrospectively note that some chemicals tested before 1985 would have invoked oxidative stress as a possible carcinogenic mechanism. Second, the majority of NTP reports do not speculate about mechanisms underlying the observation of rodent tumors. Third, 146/480 (30.4%) of NTP reports show no evidence of neoplasms. Fourth, many of the chemicals tested by NTP have also been tested by non-NTP scientists. Several of these non-NTP studies invoked oxidative stress as a possible mechanism for carcinogenicity even when the NTP report authors did not. 4,5 For example, since 2014 there have been 832 articles written on the carcinogenicity of benzene. Of these, 121 (14.5%) have indicated that oxidative stress was the primary cause of cancer by benzene. 6
An imbalance between the production of reactive oxygen species (ROS) such as superoxide anion, hydroxyl radical, hydrogen peroxide, and singlet oxygen and their detoxification results in oxidative stress, thereby leading to cellular damage (Table 1). When a chemical is inhaled at a dose sufficient to induce cellular damage, oxidative stress can be induced by two different although not completely independent sources—(a) direct toxicity to cells lining the airways and (b) toxicity to cells lining the airways from ROS, nitrogen reactive species, and enzymes released via inflammatory processes.
Reactive oxygen and nitrogen species.a

Direct oxidative stress to mitochondria
The process of direct oxygen-related toxicity to cells can be best illustrated by cell culture studies where a single parenchymal cell type is cultured in the absence of the 12 cell types that can participate in inflammatory processes listed in Table 2. The mitochondrial respiratory chain generates the majority of ROS. The electron flow rate through respiratory chain complexes is the primary modulator of mitochondrial ROS production. Approximately, 1–4% of mitochondrial oxygen consumption is diverted to the formation of ROS under physiological conditions. 39 Based on the aforementioned ROS conversion rate, and an average rate of utilization of oxygen in each human cell of approximately 2.5 × 10–18 mol/s (i.e. 2.2 × 1010 molecules/day), approximately 1 billion molecules of ROS are being produced by each cell every day in vivo. 39
Inflammatory cells and their secreted reactive oxygen and nitrogen species.
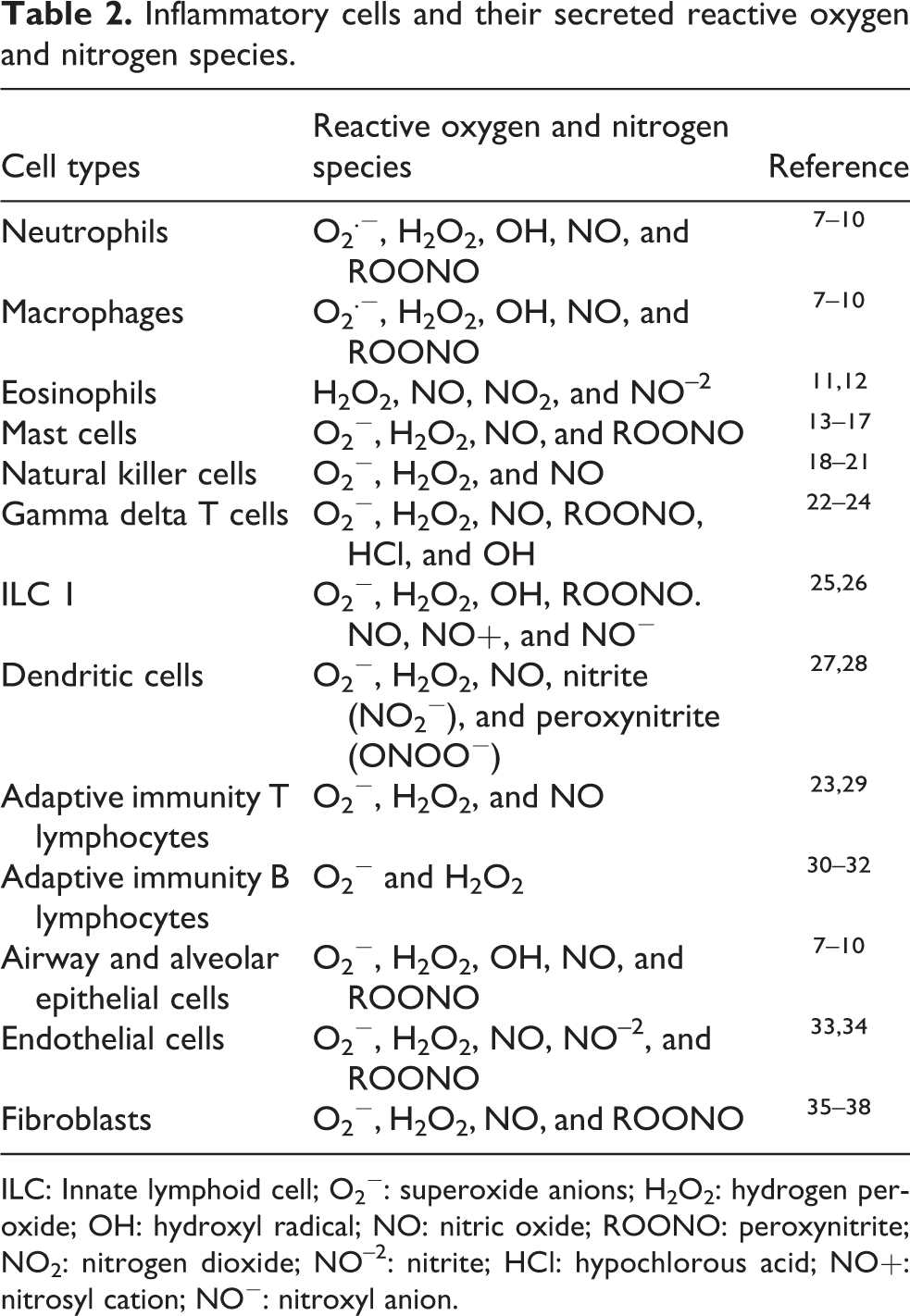
ILC: Innate lymphoid cell; O2 −: superoxide anions; H2O2: hydrogen peroxide; OH: hydroxyl radical; NO: nitric oxide; ROONO: peroxynitrite; NO2: nitrogen dioxide; NO–2: nitrite; HCl: hypochlorous acid; NO+: nitrosyl cation; NO−: nitroxyl anion.
Mechanistic studies are sometimes conducted in cultured cells in an attempt to elucidate the mode of action of chemicals. The partial pressure (pO2) of ambient atmospheric oxygen is 150 mm/Hg, which is equivalent to 21% O2. Inhaled O2 levels progressively decrease in humans as the gas reaches various internal organs and tissues. 40 The level of O2 and its tissue distribution depends on the balance between capillary blood flow and oxygen utilization. In healthy humans, arterial pO2 averages approximately 100 mm/Hg or 14% O2. As blood flow reaches the highly vascularized parenchymal organs such as lungs, liver, and kidneys, O2 levels range from 4% to 14%. 40 In less vascularized organs and tissues including the brain, eye, and bone marrow, the O2 concentration ranges between 0.5% and 7%. 40 In contrast to the relatively low O2 concentrations seen in human organs, in vitro cultures of immortalized cells use a higher O2 concentration of 21%. Under these higher O2 concentrations, ROS production can be increased several fold. Cultured primary cells are cultured at lower O2 levels than 21% due to their inability to grow at higher O2. Although excess ROS can lead to oxidative stress, moderate to low levels of ROS function in cellular signaling pathways. 41 Oxidative stress occurs in all cell culture media. It is especially important in serum-free and protein-free media because many of the anti-oxidation properties of serum are missing.
Cells in culture may behave differently from cells in vivo in many ways. Immortalized cells cultured under high oxygen tension can demonstrate a higher inflammatory response and greater redox imbalance. 40 Halliwell 42,43 has argued that cells that survive and grow in culture might use ROS-dependent signal transduction pathways that rarely or never operate in vivo. In addition, cell culture media can catalyze the oxidation of certain test chemicals, resulting in apparent cellular effects due in fact to oxidation products such as ROS. Such artifacts may have affected many studies on the effects of ascorbate, thiols, flavonoids, and other polyphenolic compounds on cells in culture. 42
Indirect oxidative stress from inflammatory processes
When a rodent inhales a chemical, it is difficult to disentangle contributions to oxidative stress from the following scenarios: (1) direct damage to mitochondria of cells lining the distal airways that does not induce inflammation (significant cellular damage occurring in vivo and not eliciting any degree of inflammation appears unlikely.); (2) direct damage to mitochondria of cells lining the distal airways, exposing either antigenic or immunogenic intracellular contents or extracellular membrane components that then elicit additional oxidative stress from activation of one or more of the 12 different cell types described in Table 2; (3) metabolic conversion of the inhaled chemical to a cytotoxic metabolite that then either damages the mitochondria or exposes antigenic or immunogenic cellular components that stimulate the inflammatory process; and (4) without damaging cellular mitochondria lining the distal airways, a chemical, particle, or dust elicits an inflammatory response that secretes large amounts of ROS and reactive nitrogen species. In the human lung, the inflammatory response can make a profound contribution to the levels of oxidative stress as seen in the clinical sequelae of a number of diseases or conditions including silicosis, 63,64 black lung (pneumoconiosis) in coal miners, 65,66 idiopathic pulmonary fibrosis, 67,68 adult respiratory distress syndrome, 69,70 bird fanciers lung, 71,72 and other fibrotic lung conditions.
Genotoxicity and increased cellular proliferation interact to increase neoplasia
Kiraly et al. 73 provided an elegant demonstration of inflammation-induced cell proliferation greatly potentiating exposure-induced mutations. For many years, it has been recognized that cancer risk was raised in certain chronic inflammatory diseases, for example, phagedenic ulcer of the skin, reflux esophagitis with Barrett’s esophagus, chronic atrophic gastritis, chronic ulcerative colitis, cirrhosis of the liver, cholelithiasis of the gallbladder, and Paget’s disease of the bone. 74 Ulcerative colitis may represent the most dramatic example of a strong association between chronic inflammation and cancer. In patients experiencing inflammation of the entire colon (pancolitis) for 10 or more years, the relative risk for developing colon cancer as compared with normal controls is 20- to 30-fold. 75 Thirty-five years following diagnosis, these relative risks are equivalent to an absolute risk of 30%. 76 (Fortunately, if initial pathologic examination does not reveal dysplasia, the rate of progression to dysplasia and carcinoma is much lower than the 30% figure noted in the aforementioned worst-case scenario. 77,78 ) With an understanding of the relationship between cancer risk and chronic inflammatory disease and pathogen-induced inflammation in hand, Kiraly et al. 73 designed two experiments.
Fluorescent yellow direct repeat (FYDR) refers to “fluorescent yellow direct repeat (FYDR) mice harboring a reporter that detects misalignments during homologous recombination (HR).” In the first experiment, FYDR mice were exposed to a potent inducer of pancreatic inflammation called cerulein. The cerulein-induced pancreatic inflammation causes double-stranded breaks (γH2AX foci). These double-stranded breaks are associated with an increase in cell proliferation. An acute inflammation by itself did not increase HR. In contrast, HR was increased significantly when inflammation-induced DNA damage and inflammation-induced cell proliferation overlapped.
In the second experiment, Kiraly et al.
73
exposed mice to the alkylating agent methylnitrosourea. While alkylation alone induced some measure of HR, when paired with inflammation-induced cell proliferation, HR was produced in a synergistic fashion. These authors summarized their findings as: Taken together, these results show that, during an acute bout of inflammation, there is a kinetic barrier separating DNA damage from cell proliferation that protects against mutations, and that inflammation-induced cell proliferation greatly potentiates exposure-induced mutations.
Pulmonary inflammation in cigarette smokers
In a bronchoalveolar lavage (BAL) procedure, a bronchoscope connected to a syringe containing 100 ml of a saline buffer solution is wedged into the second bifurcation of the large airways of the human lung. After slow injection of five separate infusions of 20 ml, approximately 70 ml can be drawn back into the syringe representing a washout of that section of the lung. 79 The 70 ml of lavage fluid are then centrifuged with the pellet separated from the supernatant. In healthy normal cigarette smokers, for example, the cellular composition of the pellet is approximately 90% pulmonary macrophages and 10% neutrophils. 80 In addition, the supernatant contains proteins from intravascular leakage and a number of inflammatory cytokines. The opacity of the BAL fluid and relatively large number of macrophages and neutrophils collected from but a single section of the lung from a healthy asymptomatic smoker as compared with the clear BAL supernatant from a healthy nonsmoker demonstrates the large amount of material in the lungs of a patient experiencing pulmonary inflammation. 80
Table 3 lists some of the many pro-inflammatory changes reported in the lungs of cigarette smokers. As compared with nonsmokers, smokers can experience four- to five-fold increases in the number of neutrophils and macrophages recovered in BAL fluid. In addition, inflammatory mediators, pro-inflammatory cytokines, and connective tissue matrix damaging enzymes are also increased. Visual bronchoscopic examination of smoker’s lungs frequently displays areas of redness, swelling, and mucus secretion. 80 Rennard and Daughton 80 have demonstrated a dose–response relationship between the amount of smoking and the degree of pulmonary inflammation in smokers without chronic bronchitis. In their study, 15 two-pack-per-day smokers with normal pulmonary function were bronchoscopically examined and bronchoalveolar lavaged and then switched down to one-pack-per-day smoking, followed for 2 months and reexamined with BAL. A reduction in smoking of approximately 50% for 2 months resulted in a significant reduction in visible, subclinical airway inflammation using a quantitative visual index. Airway neutrophilia decreased in the proximal bronchial lavage fluids. Distal “alveolar” fluids showed a reduction in total inflammatory cells, in absolute number of recovered neutrophils, and in recovered neutrophil elastase. Thus, smoking reduction lessened all measures of lower respiratory tract inflammation.
Pro-inflammatory changes in smokers’ lungs.
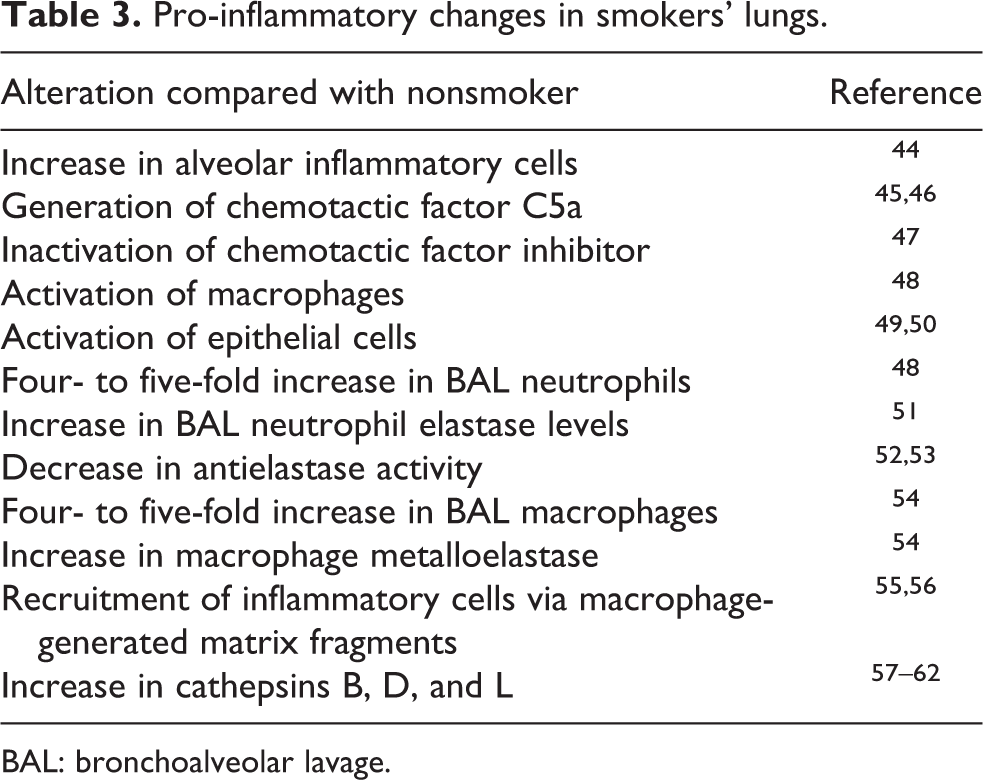
BAL: bronchoalveolar lavage.
Lung cancer is greatly increased in COPD
Increased lung cancer risk in smoking-associated chronic obstructive pulmonary disease (COPD) is an exemplar of the synergistic role played by exposure to chemical carcinogens in the presence of increased cell proliferation as demonstrated by Kiraly et al. 73 The two most common inflammatory conditions contributing to obstructed airflow in COPD are chronic bronchitis and emphysema. 81 Figures 1 to 3 illustrate the interaction of inflammatory cells with epithelial cells in human airways. In the lower left of Figure 1, the lumen of the bronchial branch shows acute inflammation by neutrophils, while the upper right of Figure 1 shows chronic inflammation in the lung wall. The upper part of Figure 2 shows respiratory epithelial cells with cilia and a few inflammatory cells including an eosinophil infiltrating the tissue layer. The bottom of Figure 2 shows neutrophils in the lumen of the bronchial branch. The bronchial branch with ciliated respiratory epithelial cells is seen in Figure 3. The lumen in Figure 3 is to the lower left. Chronic inflammation and a few eosinophils are observed in the wall. Some inflammatory cells are also seen in the respiratory epithelial cell layer.
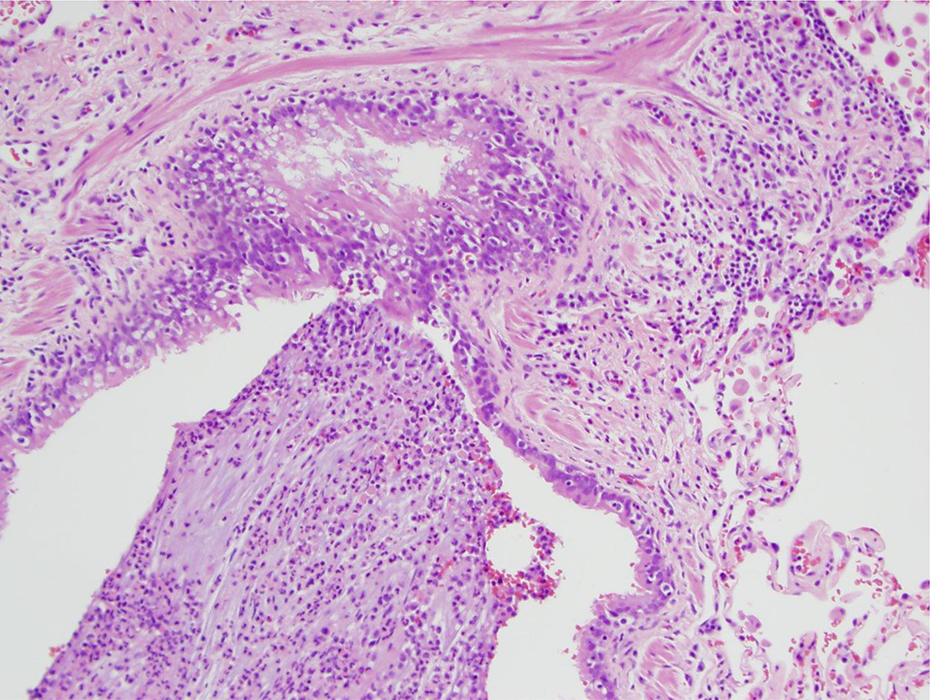
Acute inflammation (neutrophils) in bronchial branch (lumen; lower left) and chronic inflammation in the wall (upper right). hematoxylin and eosin (H&E) stain ×10 objective (×100).
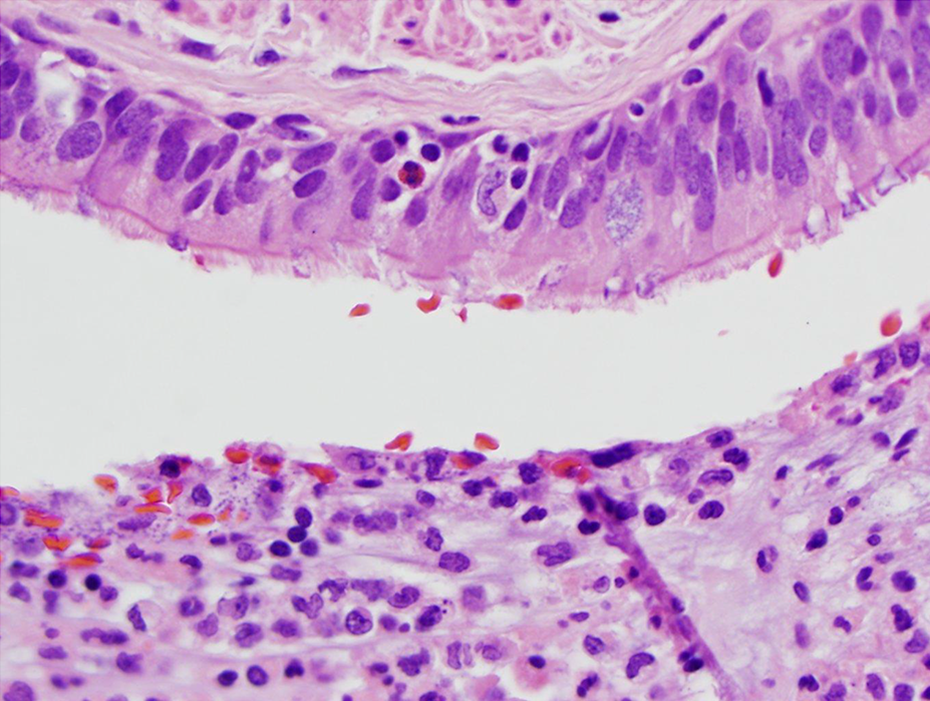
Respiratory epithelial cells with cilia (upper part of photo) with few inflammatory cells in the layer (including an eosinophil). Bottom of photo shows neutrophils in the lumen of the bronchial branch. H&E stain ×40 objective (×400).
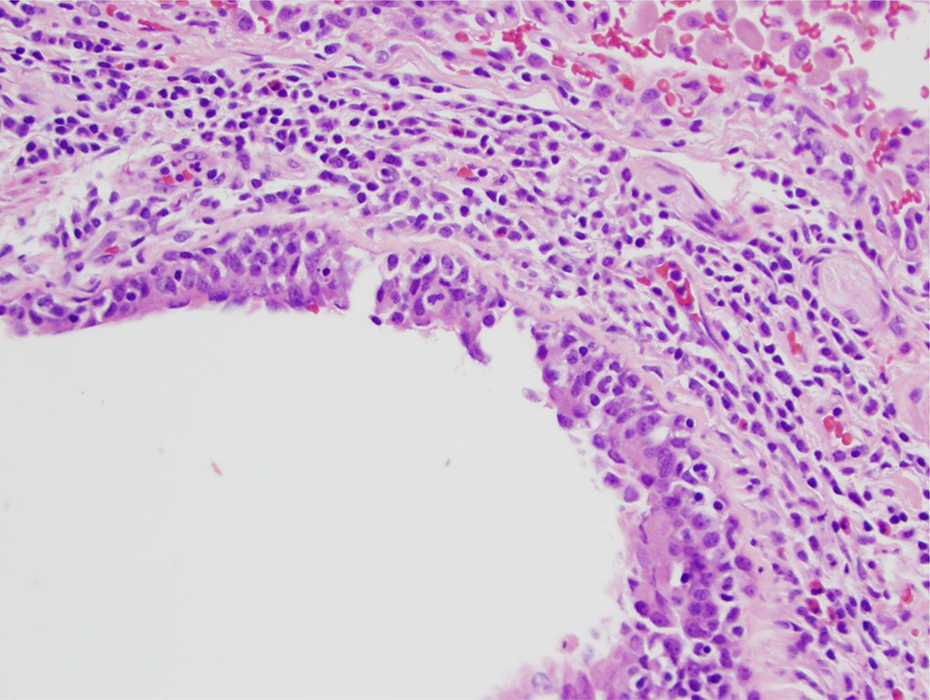
Bronchial branch with ciliated respiratory epithelial cells. Lumen to the lower left. Chronic inflammation (and few eosinophils) in the wall. Some inflammatory cells in the respiratory epithelial cell layer. H&E stain ×20 objective (×200).
A number of different cell types are stimulated to release activated oxygen and nitrogen species (Table 1) in the lungs of COPD patients including cells involved in innate immunity (neutrophils, macrophages, eosinophils, mast cells, natural killer cells, γδ T cells, innate lymphoid cells, and dendritic cells), adaptive immunity (T and B lymphocytes), and structural cells (airway and alveolar epithelial cells, endothelial cells, and fibroblasts; Table 2). 7 –38
Recent screening studies have reaffirmed the results from many previous studies 82 –89 that smokers with COPD are particularly susceptible to lung cancer. 90 The chronic inflammation associated with COPD is postulated to both cause cellular damage and promote cellular proliferation. 44,48,50,55,91 –98 In addition, the chronic inflammation in emphysema appears capable of conferring some level of lung cancer risk even in the absence of the genotoxic exposure from smoking. 99,100
Inflammatory states are associated with hypoxia
Pulmonary inflammation can result from a wide variety of agents and actions causing injury to the cells lining the airways, including inhalation of a wide variety of chemicals including irritants, cytotoxic compounds, and certain metals and metallic complexes. 101 The vast majority of neutrophils and macrophages found in pulmonary inflammation are not normally resident in the lung, but rather are recruited to inflammatory lesions. 102 One of the metabolic changes associated with active inflammation is development of hypoxia with concomitant accumulation of lactic acid and sometimes metabolic acidosis depending on the amount of lactic acid and the degree of buffering in the tissue environment. 103 –105 A number of factors can contribute to inflammation-induced tissue hypoxia including increased metabolic demands of cells and reductions in metabolic substrates caused by thrombosis (blood clots), trauma, compression (interstitial hypertension), or atelectasis (airway plugging). 106
Hypoxia due to inflammation is a stressor that can preferentially select for expansion of clonal populations of cells with particular mutations, for example, preferential survival of epidermal growth factor receptor (EGFR) positive clones. 107 In hypoxic mice with lung tumors induced by urethane, significant overexpression of EGFR, fibroblast growth factor receptor 2, and platelet-derived growth factor receptor were seen. 107 Similarly, using the HCC827 NSCLC cell line, Lu et al. 108 provided evidence that hypoxia/HIF2α activation mediates upregulation of EGFR protein levels. This observation provides a potential non-mutational explanation for the EGFR overexpression often seen in human adenocarcinomas. The authors hypothesized that, “The data presented in this contribution also introduce the intriguing possibility that the tumor microenvironment may act as a universal oncogenic trigger that drives the autonomous growth of tumor cells.”
Human EGF is a 6-kDa protein and cytokine 109,110 comprised of 53 amino acid residues and containing three intramolecular disulfide bonds. 111 EGFR is a protein found on the surface of some cells and to which EGF binds. Cell division is stimulated following the binding of EGF to EGFR. ECGR is found at abnormally high levels on the surface of many types of cancer cells, so these cells may divide excessively in the presence of EGF. Alternate names for EGFR are ErbB1 and HER1. 112
Non-small cell lung cancers (NSCLs) are a group of lung cancers so named for either the normal cell of origin or the microscopic appearance of the tumor cells. The three main types of NSCL are squamous cell carcinoma, large cell carcinoma, and adenocarcinoma, respectively. NSCL is the most common kind of lung cancer. 113 Approximately, 10% of patients with NSCLC in the United States and 35% in East Asia have a mutation in the EGFR gene in the DNA of their lung tumor. 114 –116 In both the United States and East Asia, EGFR mutations are more common in tumors from female never smokers (<100 cigarettes in patient’s lifetime) with adenocarcinoma histology. 114 –116 The percentage of EGFR mutation frequency rises to 60–65% in female East Asian never-smoker adenocarcinoma patients. 117 However, EGFR mutations are sometimes seen in squamous cell carcinoma and large cell carcinoma in both former and current smokers. 117
The EGFR mutations occur with exons 18–21 (Both the DNA sequence within a gene and the corresponding sequence in RNA transcripts are termed exons. 118 ) EGFR exons 18–21 encode a portion of the EGFR kinase domain. EGFR mutations are usually heterozygous and display gene amplification, that is, increase in number of copies. 119 The overwhelming majority, that is, approximately 90% of EGFR mutations, are deletions in exon 19 or point mutations in exon 21 L858 R. 120 These mutations increase the kinase activity of EGFR. Kinase-induced phosphorylation activates signaling pathways that block apoptosis. 121 In the vast majority of cases, EGFR mutations seen in NSCLC do not overlap with KRAS mutations observed in the same tumors, 117 although KRAS mutations are found in 25–35% of newly diagnosed non-small cell, non-squamous cell patients. 116
Conclusions
Many different chemical 122,123 or mechanical exposures (e.g. pressure overload), 124 pathogens, 125 and clinical conditions can induce pulmonary inflammation. At least 12 distinct cell types can be activated in situ or recruited to sites of lung injury and generate an inflammatory response. The quantity of inflammatory cells, proteins, and cytokines from BAL washouts of inflamed human lungs testifies to the robustness of the pulmonary inflammatory response. Higher rates of lung cancer in smokers with COPD than in smokers at the same cigarette consumption level but without COPD demonstrate a positive contribution by pulmonary inflammation to pulmonary carcinogenesis. Most of the evidence supporting a role for direct oxidative stress in chemical carcinogenesis comes from cell culture studies wherein the cells have been removed from their natural environment and denuded of defense systems, for example, cilia removal, macrophage engulfment, and suspension and dilution in pulmonary surfactant.
In real-world exposures to inhaled chemicals or complex chemical mixtures, the indirect oxidative stress burden from pulmonary inflammation is likely to greatly exceed the contribution to oxidative stress from direct chemical damage to mitochondrial structures. Before invoking a role for direct oxidative stress in the carcinogenesis of a particular chemical, the role of indirect oxidative stress from pulmonary inflammation should first be considered.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
