Abstract
Background:
Intravenous iloprost has been widely used for the treatment of systemic sclerosis peripheral vasculopathy. No agreement has been found on the regimen and the dosage of intravenous iloprost in different scleroderma subset conditions. This study aimed to evaluate the modalities of intravenous iloprost administration within a large cohort of systemic sclerosis patients from the SPRING Registry and to identify any associated clinical-demographic, instrumental or therapeutic data.
Patients and Methods:
Data of systemic sclerosis patients treated with intravenous iloprost for at least 1 year (case group) were retrospectively analyzed, including different timing and duration of intravenous iloprost session, and compared with those of untreated patients (control group).
Results:
Out of 1895 analyzed patients, 937 (49%) received intravenous iloprost treatment, while 958 (51%) were assigned to the control group. Among cases, about 70% were treated every 4 weeks, 24% with an interval of more than 4 weeks, and only 6% of less than 4 weeks. Most patients receiving the treatment every 4 weeks, or less, underwent infusion cycle for 1 day only, while if it was scheduled with an interval of more than 4 weeks, a total number of 5 consecutive days of infusions was the preferred regimen. The comparison between the two groups revealed that patients treated with intravenous iloprost had a higher frequency of DUs (p < 0.001), pitting scars (p < 0.001), diffuse cutaneous involvement (p < 0.001), interstitial lung disease (p < 0.002), as well as higher rates of anti-topoisomerase I, “late” scleroderma pattern at nailfold videocapillaroscopy. These findings were confirmed by multivariate analysis.
Conclusion:
Our data provide a picture on the Italian use of intravenous iloprost among systemic sclerosis patients and showed that it was usually employed in patients with a more aggressive spectrum of the disease. The disparity of intravenous iloprost treatment strategies in the different centers suggests the need of a rational therapeutical approach based on the clinical characteristics of different patients’ subsets.
Background
Systemic sclerosis (SSc) is a severe autoimmune disease characterized by a prominent vasculopathy with a wide range of clinical features, such as Raynaud’s phenomenon (RP) and digital ulcers (DUs). 1
Intravenous (IV) iloprost (ILO) is a stable synthetic analogue of prostacyclin used for the treatment of RP and ischemic complications in SSc. In the clinical practice, ILO in infusion cycles has obtained efficient and safe results.2–5
According to the European League Against Rheumatism (EULAR) recommendation on SSc, IV ILO is employed for severe RP after failure of oral vasoactive drugs and, as first-line therapy, for the treatment of DUs. 6 These endorsements are supported by meta-analyses and randomized clinical trials (RCTs) demonstrating that IV ILO reduces the frequency and severity of RP attacks,4,7,8 and may prevent the occurrence and boost the healing of DUs. 9 Moreover, ILO has been registered for the treatment of severe pulmonary arterial hypertension (PAH) associated with SSc, although it has a strength of recommendation “B,” since data are obtained from one RCT including patients with SSc and other connective tissue diseases. 6
A systematic review of the literature on IV ILO in SSc, enriched by a Delphi consensus exercise, confirmed its efficacy, without identifying accurately the most appropriate regimens, as for dosage, duration, and/or frequency. It should be also considered that all the existing published studies have been conducted on limited numbers of patients. 10
Indeed, there is a great variability on its use in daily clinical practice and therapeutic indications differ among countries: overall, the recommended dosage varies between 0.5 and 2.0 ng/kg/min for an infusion of 6 h/per day, depending on patient’s tolerance (as reported in the technical data sheet). 11 In some countries, IV ILO is available with the approved indication for RP secondary to SSc for 3–5 days and in Italy also for Buerger’s disease.10,11 Data derived from expert opinion suggested a 1- to 3-day monthly regimen for RP and DUs healing, and 1 day monthly for DUs prevention. 10 Therefore, the lack of uniformity on the type of regimen, dosage, frequency, and duration prompts in practice the use of IV ILO mainly based on personal experience and convenience.
Thus, the aim of our study was to evaluate how IV ILO therapy is used and administered by rheumatologists within a large national cohort of SSc patients, included in the Italian “SPRING” (Systemic Sclerosis Progression InvestiGation) Registry, to investigate the association between clinical-demographic, instrumental, and therapeutic data, and to understand whether there were features that could drive its specific timing and dosage.
Patients and methods
In this case–control study, we retrospectively evaluated clinical-demographical, instrumental, and therapeutical data from patients affected by definite SSc, classified according to the 2013 EULAR/American College of Rheumatology (ACR) criteria, 12 enrolled in the SPRING Registry.
SPRING project is a prospective cohort study, with a consecutive recruitment of SSc-spectrum cases, promoted by the Italian Society for Rheumatology-SIR in 2015, as a strategic no-profit project involving 37 Italian centers (the reference number of the Coordinating Centre is OSS 15.010, AOU Careggi-Firenze). All patients gave their written informed consent to participate. Study data were collected and managed using Research Electronic Data Capture (REDCap), a web-based application to support data collection. As previously described, 13 the cohorts were categorized as RP (primary and suspected secondary), Very Early Diagnosis of SSc (VEDOSS), 14 and definite SSc. 12
At baseline and at yearly follow-up visit, demographic, clinical, instrumental, and laboratory features of each patient, aged >18, were collected, together with the disease history, lifestyles, and comorbidities. Information included age, sex, age of disease onset, as well as the following clinical variables: skin signs (sclerodactyly, puffy fingers, calcinosis, and telangiectasia), peripheral vascular signs (digital pitting scars, DUs, gangrene), presence of comorbidities (smoking habit, arterial hypertension, dyslipidemia, diabetes).
Among instrumental features, non-invasive cardiac diagnostic testing was performed by electrocardiogram (ECG) and trans-thoracic echocardiography (including pulmonary arterial pressure—PAPs estimation). Investigations for lung involvement consisted of pulmonary function tests (total lung capacity—TLC, forced vital capacity—FVC), with diffusion capacity for carbon monoxide (DLCO) and high-resolution computed tomography—HRCT (to detect interstitial lung disease—ILD). Nailfold videocapillaroscopic (NVC) data were collected, using the classification proposed by Cutolo et al. 15
Previous and current treatments were also reported, including both vasodilators/vasoactive drugs (calcium-channel blockers—CCBs, prostanoids, endothelin receptor antagonists—ERAs, phosphodiesterase-5 inhibitors—PDE5i, angiotensin-converting enzyme inhibitors—ACEi, anti-platelets).
For the study, only patients classified as definite SSc were evaluated, while VEDOSS and RP patients were excluded. The sample selection process is illustrated in Figure 1.
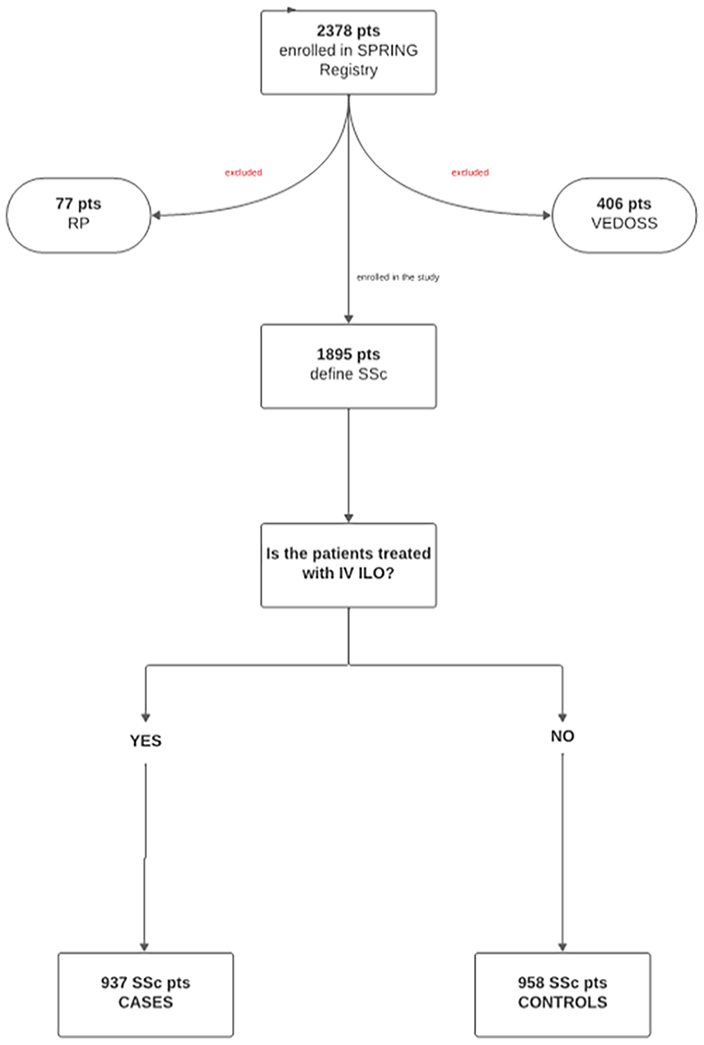
Selection process of the sample: from the SPRING Registry to the definition of case and control cohorts.
From the cohort of definite SSc, those treated with IV ILO were selected, evaluating the different timing of ILO infusions and in detail the frequency and duration of infusion itself. The second step was to collect and stratify patients based on the type of IV ILO regimens.
In addition, clinical, demographic, and instrumental features, as well as therapies, were compared between SSc patients treated with IV ILO (case group) and those without (control group). Besides, we evaluated whether there was any difference among patients treated with different frequency of IV ILO infusion, and among their characteristics, such as the presence of DUs and/or pitting scars, SSc-specific autoantibodies (anti-topoisomerase 1/Topo 1, anti-centromere/ACA, anti-RNA polymerase), organ involvement, severity of RP, NVC patterns, or presence of limited (lcSSc)/diffuse (dcSSc)/sine SSc (ssSSc) subsets of the disease.13,16
Statistical analysis
Descriptive analyses were reported as absolute and relative frequencies for categorical variables, mean and standard deviation (SD) for continuous ones. Median (interquartile range (IQR)) has been provided in place of mean (SD) when significant asymmetry of distributions was present.
The chi-square test was used to compare categorical variables, while quantitative variables were compared using Student’s t test or Mann–Whitney U test depending on their distribution, as appropriate.
Multivariable logistic regression analysis was also performed to examine the strength of the association between demographic and clinical variables and the use of IV ILO. The regression model was adjusted for the covariates with a p < 0.05 in univariate models. Odds ratio (OR) values were reported with their 95% confidence intervals (95% CIs).
The level of significance was set at <0.05. Data were analyzed using Stata v.14.
Results
The analysis of SPRING database showed that 1895 out of 2378 patients were classified as definite SSc. Of them, 937/1895 (49.45%) were treated (cases) and 958/1895 (50.55%) were not treated (control group) with IV ILO.
The case group was analyzed from a geographical perspective by sorting the overall number of SSc patients enrolled in the entire database, based on their Italian macro-area of origin, which included 911 patients from the North, 339 patients from the Center, and 565 from the South. The IV ILO treatment was found to be more frequently used in Central Italy (189/339—55.7%) compared to the Northern (397/911—43.6%) and Southern macro-areas (269/565—47.6%) (p = 0.006). A subgroup analysis was conducted to assess differences among patients undergoing IV ILO therapy across the Northern, Central, and Southern Italy. It revealed that patients receiving IV ILO in Central Italy exhibited a higher prevalence of pitting scars (141/189—74.6% vs Northern: 244/397—61.5% and Southern: 155/269—57.6%; p = 0.0001), of dcSSc subset of disease (70/189—37% vs Northern: 95/397—23.9% and Southern: 67/269—27.9%; p = 0.002) and of a scleroderma late pattern at NVC (76/189—40.2% vs Northern: 103/397—25.9% and Southern: 71/269—26.4%; p = 0.0008) than patients from Northern and Southern regions. No statistically significant differences were observed in other clinical manifestations, except for ILD at HRCT, that was more frequently encountered among patients in Northern Italy (117/397—29.5% vs Central: 36/189—19% and Southern 67/269—27.9%; p = 0.0008).
The main clinical and demographic data of all patients at baseline, including laboratory, and instrumental findings, are shown in Table 1.
Comparison of clinical, demographic, and instrumental characteristics between SSc patients treated (cases) and not treated (controls) with IV ILO.
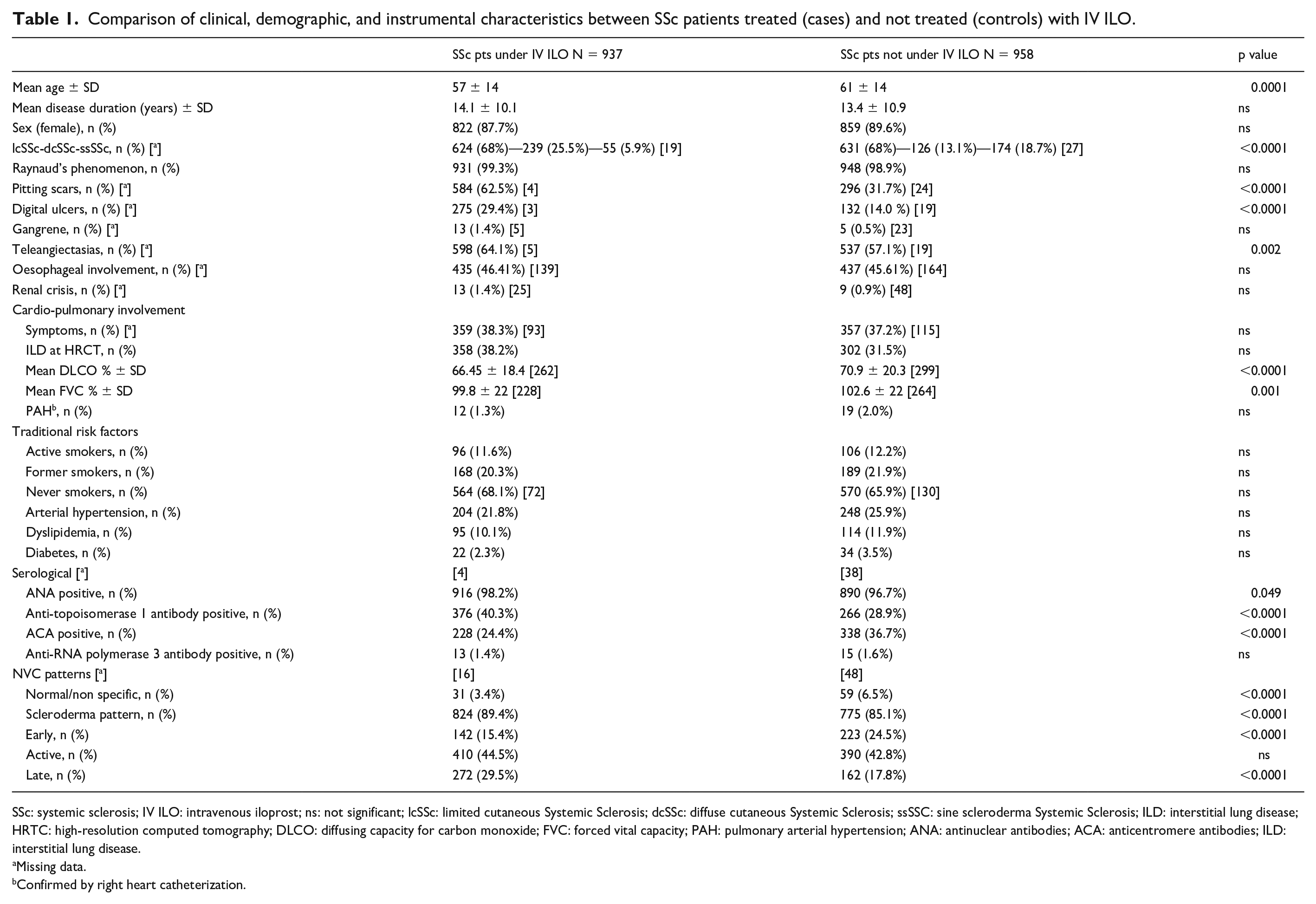
SSc: systemic sclerosis; IV ILO: intravenous iloprost; ns: not significant; lcSSc: limited cutaneous Systemic Sclerosis; dcSSc: diffuse cutaneous Systemic Sclerosis; ssSSC: sine scleroderma Systemic Sclerosis; ILD: interstitial lung disease; HRTC: high-resolution computed tomography; DLCO: diffusing capacity for carbon monoxide; FVC: forced vital capacity; PAH: pulmonary arterial hypertension; ANA: antinuclear antibodies; ACA: anticentromere antibodies; ILD: interstitial lung disease.
Missing data.
Confirmed by right heart catheterization.
The comparison between the two groups revealed that the median age of the controls was significantly higher than that of the cases (61±14 vs 57±14 years ±SD; p-value = 0.0001). However, the two groups were well-matched in terms of gender and disease duration.
Almost all patients (99%) in both groups had RP. Regarding other clinical signs of peripheral vasculopathy, patients treated with IV ILO showed a higher frequency of DUs (cases vs controls: 275/934—29.4% vs 132/939—14.0%; p < 0.001) and pitting scars (cases vs controls: 584/933—62.5% vs 296/934—31.7%; p < 0.001). Baseline NVC showed a normal or non-specific pattern in 31/921 (3.4%) cases and 59/910 (6.5%) controls, while a NVC scleroderma pattern was significantly more frequent among cases (824/921—89.4%) than controls (775/910—85.1%; p < 0.0001). In addition, cases more frequently presented a “late” scleroderma pattern than controls (cases vs controls: 272/921—29.5% vs 162/910—17.8%; p < 0.0001).
In all the Italian SPRING centers, IV ILO was administered between 0.5 and 2.0 ng/kg/min for 6 h, according to manufacturer indication and patient tolerability.
A detailed description of the differently available regimens of IV ILO treatment in SSc patients, including frequency (<, >, or =4 weeks) and number of days of infusion (from 1 to 6 days) for each cycle, is shown in Table 2. Most of the patients (602/861—69.9 %) were on IV ILO every 4 weeks, 49/861 (5.7 %) with an interval less than 4 weeks, and 210/861 (24.4%) with an interval of more than 4 weeks. Most patients (311/602—51.6%) on treatment every 4 weeks underwent IV ILO infusion for only 1 day. The single-day cycle was also preferred for patients receiving IV ILO for less than 4 weeks (35/49, 71.4%). When IV ILO was scheduled with an interval of more than 4 weeks, most of the patients received a total number of 5 consecutive days of infusions (125/210, 59.5%).
Different regimens of IV ILO treatment in the cases group: frequency (<, >, or =4 weeks) and number of infusions (from 1 to 6 days) for each cycle.
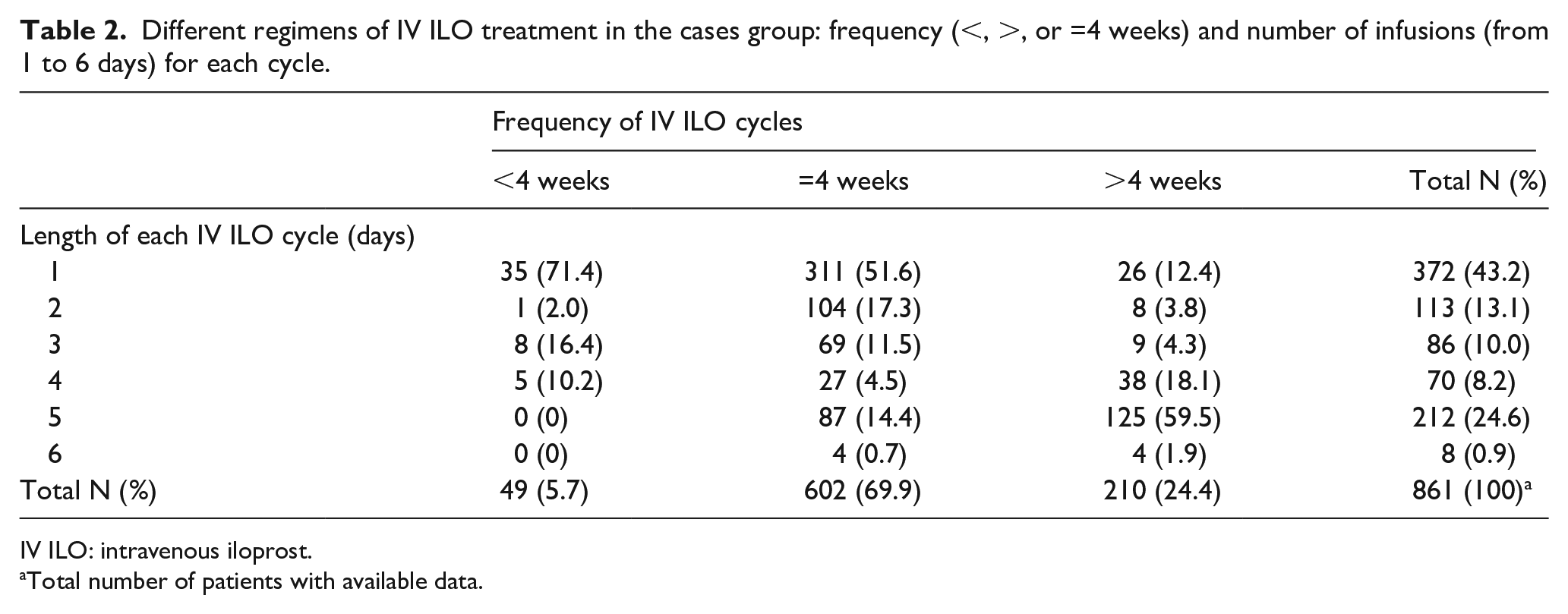
IV ILO: intravenous iloprost.
Total number of patients with available data.
Patients who received an IV ILO infusion with an interval of less than every 4 weeks had significantly more DUs (27/49—55.1% of cases) than patients treated every 4 weeks (178/602—29.5%) or with an interval of more than 4 weeks (63/210—30%) (p = 0.002). Similarly, patients on IV ILO infusion more often over 4 weeks reported more severe RP than subjects treated with other infusion schedules (IV ILO < 4 weeks, N = 22/49—44.8%; IV ILO every 4 weeks, N = 136/602—22.5%; IV ILO > 4 weeks, N = 42/210—20%; p < 0.002).
No difference was found for other clinical features, NVC patterns, or other concomitant vascular therapies based on the different IV ILO regimens.
It should be noted that 129 controls were previously treated with IV ILO. The reasons for withdrawal included toxicity (36%), recovery of symptoms (21%), presence of comorbidities (8%), and inefficacy (7%).
Besides, patients receiving IV ILO therapy showed a more aggressive disease (Table 1): a significantly higher proportion of cases were dcSSc (25.5% vs 13.1%, p < 0.0001), showed ILD on HRCT (38.2% vs 31.5%, p = 0.002), DUs and pitting scars (62.5% vs 31.7%, p < 0.0001, for both). This observation is also consistent with the serological findings (Table 1), as patients on IV ILO therapy were more frequently anti-Topo (40.3% vs 28.9%, p < 0.0001), while controls were more frequently ACA-positive (24.4% vs 36.7%, p < 0.0001). In contrast, controls showed a higher percentage of patients with ssSSc (18.7% vs 5.9%, p < 0.0001).
A detailed description of previous or ongoing treatments in 937 patients on IV ILO therapy and in 958 controls is reported in Table 3; as expected ERA (290/937—30.9% vs 110/958—11.5%; p < 0.0001) and anti-platelet agents (446/937—47.6% vs 385/958—40.2%; p = 0.001) were prescribed more frequently in cases, while there was no significant difference in the use of CCBs and PDE5 inhibitors between the two groups.
Concomitant vascular therapies carried out by cases and controls.
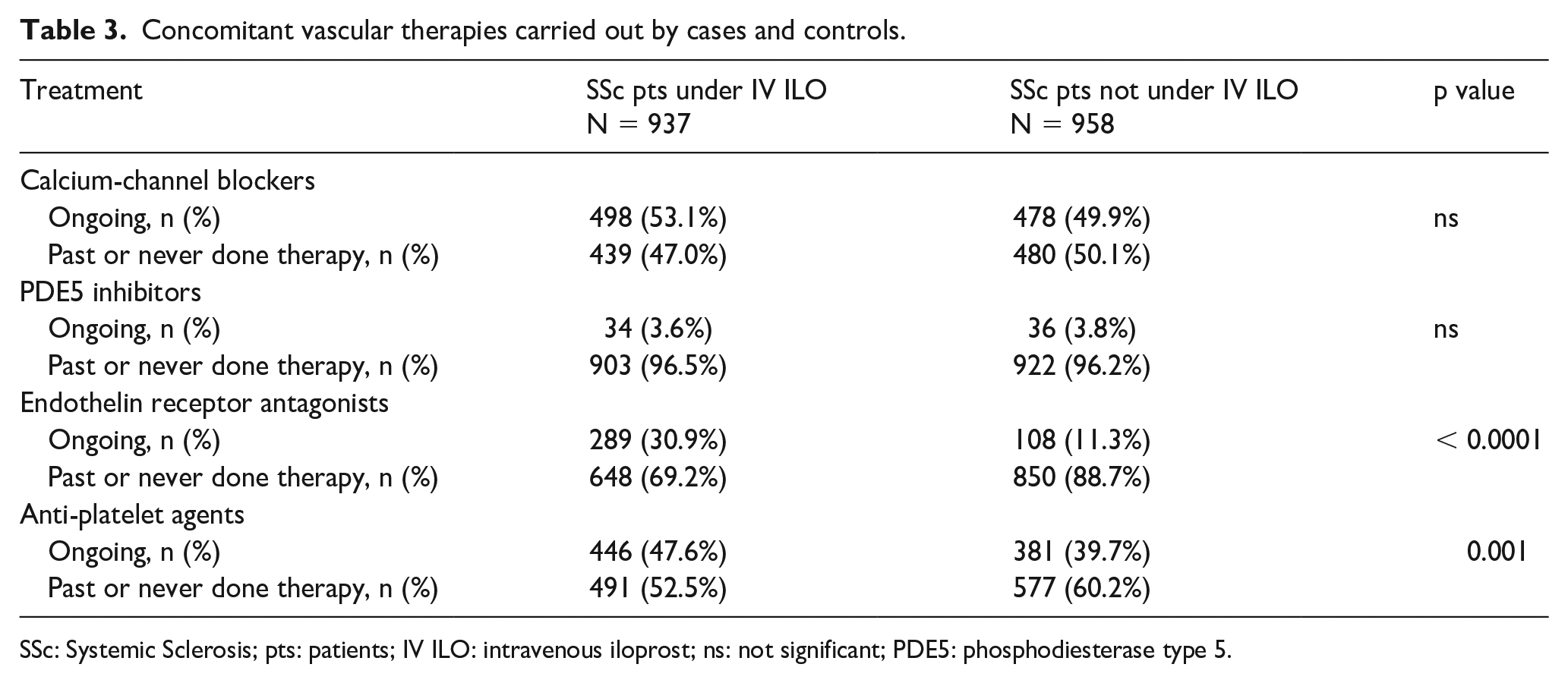
SSc: Systemic Sclerosis; pts: patients; IV ILO: intravenous iloprost; ns: not significant; PDE5: phosphodiesterase type 5.
The multivariate analysis revealed that patients’ age (p < 0.0001), presence of pitting scars (p < 0.0001), and therapy with ERAs (p < 0.0001), and/or anti-platelet agents (p = 0.049) were significantly associated with the IV ILO use (Table 4).
Univariate and multivariate analysis for variables associated with IV ILO treatment.
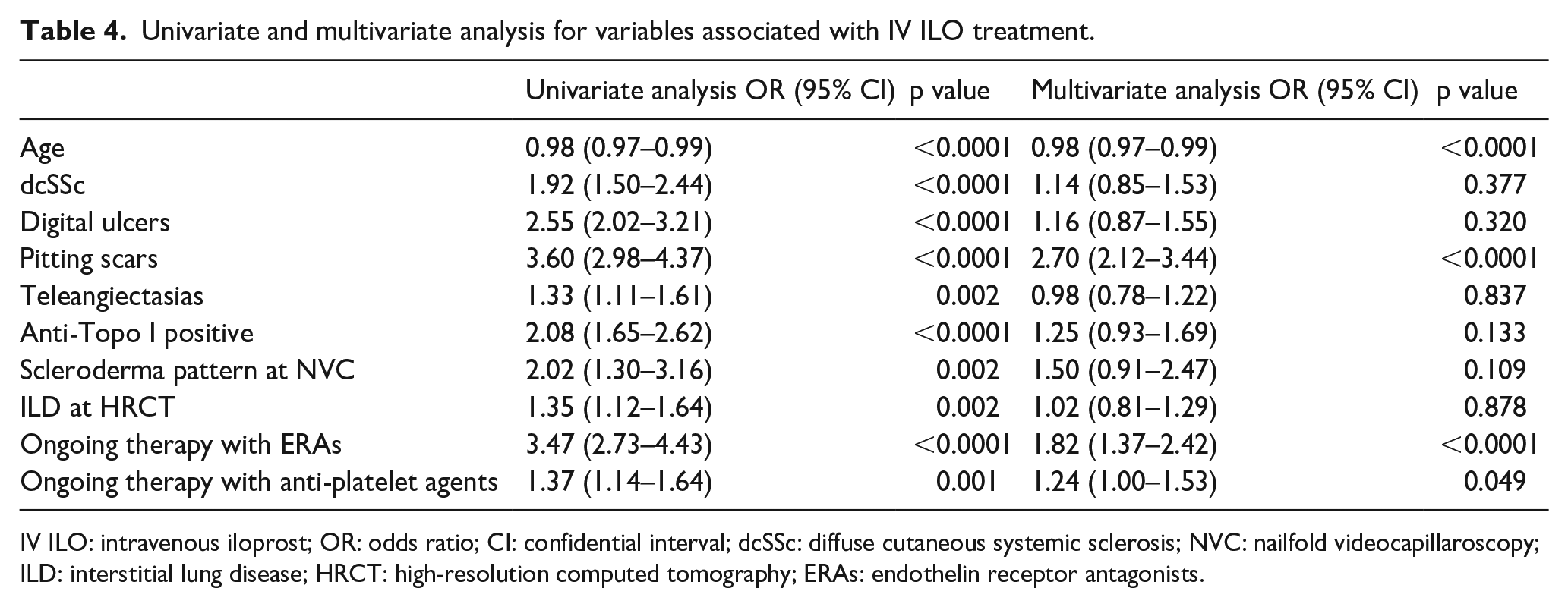
IV ILO: intravenous iloprost; OR: odds ratio; CI: confidential interval; dcSSc: diffuse cutaneous systemic sclerosis; NVC: nailfold videocapillaroscopy; ILD: interstitial lung disease; HRCT: high-resolution computed tomography; ERAs: endothelin receptor antagonists.
An overall overview of the IV ILO regimens as detected from our study is given in Table 5.
Most frequent IV ILO regimens as detected from our study.

IV ILO: intravenous iloprost; RP: Raynaud’s phenomenon.
Discussion
Our data show that, in Italian centers, IV ILO is employed in patients with a more aggressive spectrum of the disease, namely those patients with clinical features defined by previous studies 17 as risk factors for disease worsening (i.e. DUs, ILD, diffuse cutaneous involvement). Thus, it is partly in agreement with the recent EULAR recommendations. 6 Usually, IV ILO is employed for 3–5 days of infusion, but our study found that different treatment regimens were employed in a large SSc Italian national cohort. The clinical-demographic, laboratory, and instrumental features, as well as other vascular therapies, were investigated to identify whether there was a preferential regimen, given the absence of well-defined guidelines on the use of IV ILO in SSc.
Almost half of all the SSc cases, amounting to 1895, were on IV ILO, and up to date no study on such a large population has been reported in the literature.
A different geographical distribution of the IV ILO was recorded among the main Italian macro-areas, as higher percentages of SSc patients treated with IV ILO were resident in Central and Southern Italy, rather than Northern Italy. This finding may result quite paradoxical because Northern regions have a colder average annual climate and therefore patients should be affected with a more severe RP and DUs. 5 Indeed, our analysis showed that patients from Central Italy more frequently have some features indicating a more severe form of disease, especially regarding peripheral vascular microangiopathy (pitting scars, scleroderma late pattern) similar to what was found in a previous clinical-demographical analysis of SPRING Registry that has shown as patients from Southern Italy were characterized by a more aggressive disease, accounting for a greater need of IV ILO treatment. 18 The different geographical distribution of SSc subsets has been previously emphasized, and may probably be related to referral bias as well to different environmental and/or genetic factors. 18
According to the 2017 EULAR recommendations for the treatment of SSc, IV ILO is indicated for RP management after failure of oral vascular therapies such as CCB and PDE5i or as first choice for DU healing. 6 Almost all cases (99%) complained about RP, while only one-third presented DUs. However, as this study is a cross-sectional analysis, it was not possible to clearly identify the reason for prescribing IV ILO, although we can hypothesize that the presence of RP was the main indication, in agreement with the results of expert consensus. 10 Moreover, the comparison of the clinical characteristics of cases and controls showed that IV ILO is prescribed to those SSc patients presenting a more severe vascular involvement, as cases were more frequently affected by DUs and pitting scars and exhibited a higher incidence of a “late” scleroderma pattern at NVC. In addition, ERA and anti-platelet treatments were prescribed more frequently in cases than controls. In our SSc cases, it is clear that the manifestations of SSc vasculopathy seem to drive the prescription of IV ILO, in line with EULAR recommendations. 6
In addition, cases treated with IV ILO were more frequently dcSSc, anti-Topo I positive and affected by ILD with respect to controls. This observation highlights that in real life the prescription of IV ILO is also guided by the whole SSc severity. A similar finding was observed in previous studies.19,20 In our IV ILO–treated patients, the higher ILD prevalence is not surprising, as DUs and anti-Topo I are present in more severe patients, including those with ILD.21,22 It is interesting to note that, despite the lack of RCTs, ILO seemed able to improve skin thickness and pulmonary arterial systolic pressure in observational studies,23–25 again suggesting its use in the more aggressive subsets of the disease.
The very recent 2023 update of EULAR recommendations for the treatment of SSc still does not specify the dose or the therapeutic regimen for IV ILO. 26 Currently, no trials are available providing guidance on the regimen. In some countries, IV ILO is available and approved for RP secondary to SSc, for 3–5 consecutive days cycle, with no indication on the infusion frequency. Thus, according to patients characteristics 10 and the organization of the hospital center, the physician may consequently choose the best regimen, which includes dosage, duration, and frequency. 10 In the future, portable infusion pumps might be applied to selected subjects with a remote monitoring system, managed by expert physician or nurse, thus sparing costs for the patients and the centers. 27
As regards concomitant vascular therapies, a combination strategy with IV ILO is considered the best therapeutic option for RP refractory to oral therapies as well as for DUs. 28 Anti-platelet drugs, used by nearly 50% of our cases, are possibly prescribed with IV ILO in preventing DUs, as recommended in the PROSIT study. 28 The combination of ILO + ERAs is believed to be aimed to increase the rate of healing for DUs 18 and prevent the development of new DU. 29 In fact, in a long-term follow-up, ILO + ERAs has proven to increase fingertip blood perfusion and the absolute nailfold capillary number/mm, reducing of 80% the incidence of new DU. 30
One of the greatest concerns for the use of IV ILO is represented by the choice of its administration regimen. 10 Neither in the EULAR recommendations nor in the manufacturer datasheet, a specific dosage, duration, or frequency of infusion is indicated, the latter only suggesting that the drug should be administered at a dose of 0.5–2 ng/kg of body weight/min. This was also the most frequent dosage employed in our cohort. In a prospective RCT on 46 SSc subjects, an 8-h IV ILO infusion was used as a daily dose of 2 ng/kg/min for 5 days. 8 Another placebo-controlled double-blind study on 131 SSc patients showed IV ILO efficacy in reducing severity, frequency, and duration of RP at a dosage of 2 ng/kg/min over 6 h a day for 5 consecutive days. 4 In 28 SSc patients, Auriemma et al. 31 showed an amelioration of RP severity and number of RP attacks reduction using a median lower dosage (0.5–2 ng/kg of body weight/min) for 1–3 days every 30 days. However, similar results were detected also with different approaches including higher or lower dosages of ILO. 32
In most of our patients, the treatment regimen was 1-day IV ILO every 4 weeks. This result is in agreement with the report suggesting that IV ILO could be administered 1–3 days monthly to treat RP and DUs healing and 1 day per month for DU prevention. 10
Thus, in our study, the reason for driving the choice of a more frequent infusion may mainly be due to a more severe vascular disease characterized by RP, DUs, and pitting scars of the extremities.
Attention was also focused on the number of infusions per cycle ranging from a single-day dose or cycles of 2–5 consecutive days. A single-day infusion was used for treatment regimens every 4 weeks or less, while when IV ILO was scheduled for infusions with an interval of more than 4 weeks, 5 consecutive days of infusions were the most frequently used regimen.
The strength of this study is represented by the extensive data obtained from a nationwide registry, which provide insights into the real-life IV ILO regimens of tertiary-rheumatology referral centers. At the same time, this type of data collection may have some limitations, including the heterogeneity of the involved centers from different areas of the country with potential geographical referral bias. 18
In conclusion, the observed data indicate that the choice of the IV ILO dosage and duration of a single infusion are generally made according to the main recommendations suggested in the datasheet. In particular, the following regimens have been most frequently detected in the Italian centers:
Dosage range = 0.5–2 ng/kg/min (tapered according to patient’s needs);
Infusion duration = six consecutive hours for each cycle (as reported in the manufacturer datasheet);
Infusion frequency = more often than 4 weeks in the presence of severe vascular features; every 4 weeks or more in stable RP;
Cycle frequency = single-day infusion, if repeated within 4 weeks; from 2 to 5 consecutive days, for intervals longer than 4 weeks.
Overall, the frequency and dosage of IV ILO administration depend on the severity of both peripheral vascular involvement (i.e. RP and DUs) and SSc variants. For a shared therapeutical approach, appropriate RCTs should be planned, allowing to elaborate the most effective and well-tailored IV ILO treatment modalities for different SSc patients’ subgroups.
Footnotes
Acknowledgements
This study was supported by the Italian Society for Rheumatology (SIR)
Convenors
Clodoveo Ferri, University of Modena & Reggio Emilia, Italy;
Marco Matucci-Cerinic, University of Florence, Italy;
Investigators (in alphabetical order)
Abignano Giuseppina, AOR San Carlo di Potenza;
Agnes Cecilia, Ospedale San Lorenzo, Carmagnola (TO), ASL-TO5
Amato Giorgio, AOU Policlinico—Vittorio Emanuele, Catania;
Ariani Alarico, AOU Parma; dott.
Bagnato Gianluca, Università degli Studi di Messina;
Bajocchi Gianluigi, Arcispedale S. Maria Nuova, Reggio Emilia;
Barsotti Simone, AOU Santa Chiara, Pisa;
Bellando-Randone Silvia, University of Florence;
Benenati Alessia, AOU ‘Policlinico—Vittorio Emanuele, Catania;
Beretta Lorenzo, Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Milano;
Bianchi Gerolamo, ASL3 Genova; gerolamo.
Bosello Silvia, Policlinico “A. Gemelli” –IRCCS—UOC di Reumatologia; Roma;
Cacciapaglia Fabio, UO Reumatologia—DETO, Università di Bari;
Calabrese Francesca, SSD Reumatologia, Reggio Calabria;
Caminiti Maurizio, Ospedale Bianchi-Melacrino-Morelli, SSD Reumatologia, Reggio Calabria;
Campochiaro Corrado, Ospedale S. Raffaele, Milano;
Carignola Renato, AOU San Luigi Gonzaga, Orbassano (TO);
Cavazzana Ilaria, Spedali Civili di Brescia;
Ciano Giovanni, Ospedale Ariano Irpino, ASL Avellino;
Cipolletta Edoardo, Clinica Reumatologica, Università Politecnica delle Marche, Ancona;
Codullo Veronica, Policlinico San Matteo, Pavia;
Cozzi Franco, Villa Salus, Mestre;
Cuomo Giovanna, Università degli Studi della Campania—Luigi Vanvitelli, Napoli;
D’Angelo Salvatore, AOR San Carlo di Potenza;
Dagna Lorenzo, Ospedale S. Raffaele, Milano;
Dall’Ara Francesca, UO Medicina Interna-Ambulatorio Reumatologia, Ospedale di Lodi;
De Andres Ilenia, AO ARNAS Garibaldi, Catania;
De Angelis Rossella, Clinica Reumatologica, Università Politecnica delle Marche, Ancona;
De Cata Angelo, Ospedale Casa Sollievo della Sofferenza, San Giovanni Rotondo (FG);
De Luca Giacomo, Ospedale S. Raffaele, Milano;
De Santis Maria, Istituto Clinico Humanitas, Rozzano, Milano;
Della Rossa Alessandra, AOU Santa Chiara, Pisa;
Di Vico Claudio, Università degli Studi della Campania “Luigi Vanvitelli”;
Doria Andrea, Università degli Studi di Padova;
Doveri Marica, ASL3 Genova;
Foti Rosario, AOU Policlinico San Marco, Catania;
Furini Federica, Department of Medical Sciences, University of Ferrara;
Fusaro Enrico, AOU Città della Salute e della Scienza di Torino;
Generali Elena, Istituto Clinico Humanitas, Rozzano, Milano;
Gigante Antonietta, Università degli Studi “La Sapienza,” Roma;
Giollo Alessandro, AOUI Verona;
Girelli Francesco, Ospedale GB Morgagni, Forlì;
Giuggioli Dilia, University of Modena/Reggio Emilia;
Govoni Marcello, AOU S. Anna, Ferrara;
Guiducci Serena, University of Florence;
Iannone Florenzo, UO Reumatologia– DETO, Università di Bari;
Ingegnoli Francesca, Università degli Studi di Milano;
Iuliano Anna Maria, AO San Camillo Forlanini, Roma;
Lazzaroni Maria Grazia, Spedali Civili and University of Brescia;
Lepri Gemma, University of Florence;
Lubrano Ennio, Università del Molise, Campobasso;
Lumetti Federica, University of Modena & Reggio Emilia;
Magnani Luca, Arcispedale S. Maria Nuova, Reggio Emilia;
Mennillo Gianna, AOR San Carlo di Potenza;
Murdaca Giuseppe, Department of Internal Medicine, University of Genoa, IRCCS Ospedale Policlinico San Martino, Genoa, Italy;
Pagano Mariano Giuseppa, Ospedale Bianchi-Melacrino-Morelli, Reggio Calabria;
Parisi Simone, AOU Città della Salute e della Scienza, Torino;
Pellegrino Greta, Sapienza, Università di Roma;
Peroni Clara Lisa, AOU Città della Salute e della Scienza, Torino;
Pigatto Erika, UOC Medicina Interna, Ospedale San Bassiano, Bassano del Grappa, Vicenza;
Riccieri Valeria, Sapienza Università di Roma;
Romeo Nicoletta Rheumatology Unit ASO Santa Croce e Carle, Cuneo;
Rosato Edoardo, Università degli Studi di Roma “La Sapienza” Policlinico Umberto I;
Sambataro Gianluca, Azienda Ospedaliera Cannizzaro, Catania
Saracco Marta, Ospedale Mauriziano, Torino;
Sebastiani Giandomenico, AO San Camillo Forlanini, Roma;
Spinella Amelia, University of Modena & Reggio Emilia;
Talotta Rossella, L. Sacco Hospital, Milan;
Visalli Elisa, AOU Policlinico San Marco, Catania;
Vultaggio Licia, AOU S. Anna, Ferrara,
Zanatta Elisabetta, Università degli Studi di Padova;
Zanframundo Giovanni, Policlinico San Matteo, Pavia;
Study Center of the Italian Society of Rheumatology (SIR)
Carlo Scirè, Università degli Studi, Milano-Bicocca, Milan;
Greta Carrara, Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy;
Gianpiero Landolfi, Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy;
Davide Rozza, Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy;
Anna Zanetti, Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy;
Author’s note
The Editor/Editorial Board Member of JSRD is an author of this paper; therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Board member had no involvement in the decision-making process.
Author contributions
V.R. and G.P. conceived the idea for the study, contributed to the study design, supervised data analysis, interpreted the results, reviewed the literature, co-wrote the first draft of the manuscript, and critically reviewed the manuscript. R.D.A., M.M.C., and C.F. contributed to the study design, supervised data analysis, interpreted the results, and critically reviewed the manuscript. E.C. performed data analysis, interpreted the results, and critically reviewed the manuscript. D.G., G.B., S.B.R., L.D., G.Z., R.F., F.C., G.C., A.A., E.R., G.L., F.G., E.Z., S.L.B., I.C., F.I., M.D.S., G.M., G.A., N.R., A.D.R., M.C., A.I., G.C., L.B., G.B., E.L., I.D.A., A.G., M.S., C.A., F.L., A.S., L.M., C.C., G.D.L., V.C., E.V., C.D.V., A.G., F.S., M.G.L., F.F., E.G., G.M., S.B., G.P.M., F.C., F.F., L.V., S.P., C.L.P., G.B., F.C., F.C., S.D.A., A.D., E.F., M.G., S.G., F.I., C.S., G.D.S. collected clinical data and critically reviewed the manuscript. All the author approved the submitted manuscript. V.R. is responsible for the overall content as guarantor and attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study involves human participants and was approved by reference number OSS 15.10 Azienda Ospedaliera Universitaria Careggi-Firenze. Participants gave informed consent to participate in the study before taking part.
