Abstract
This article reports the diagnosis and treatment of a 50-year-old female patient with systemic sclerosis complicated by a nonhealing wound on her left hand. Following comprehensive treatments including debridement, vacuum sealing drainage, ozone therapy, skin grafting, and phalangeal fusion, the wound healed successfully. This article highlights the potentially contradictory role of interleukin-6 inhibitors in the treatment of patients with systemic sclerosis and ulcers as well as emphasizes the application value of ozone therapy in improving tissue hypoxia and promoting healing.
Introduction
Systemic sclerosis (SSc) is a relatively rare systemic autoimmune disease characterized by skin thickening, fibrosis, and involvement of internal organs. It is marked by microvascular lesions, immune dysregulation, and progressive fibrosis that typically affects the skin and is often accompanied by varying degrees of internal organ involvement. The pathogenesis of SSc remains unclear,1,2 potentially associated with fibrotic responses following early inflammatory infiltration and endothelial injury. 3 The main symptoms include Raynaud’s phenomenon (RP), digital ulcers (DU), fatigue, and musculoskeletal pain. Patients with severe microvascular lesions in SSc often develop ulcers on their fingertips or toes. Clinically, local debridement or even digital amputation is frequently performed; however, due to impaired blood circulation in the extremities, the wounds often fail to heal.4,5 In light of this, our hospital adopted a treatment approach combining surgical debridement, negative pressure wound therapy, and ozone autohemotherapy as an adjunct, which resulted in successful wound healing. 6
Clinical data
Basic information
A 50-year-old female patient was admitted with a 2-year history of skin induration along with a nonhealing, infected wound on the left hand, which developed following a trauma approximately 3 months ago. Two years ago, the patient developed progressive skin hardening and swelling on both hands, with the skin turning reddish-purple. She experienced tenderness at the fingertips of the second, third, and fourth fingers on both hands as well as tenderness in both knee joints. The condition progressed to tightening of the skin all over the body, with a waxy luster and a “mask-like” facial expression. Stiffness was observed in the limbs, accompanied by pain and limited mobility in joints including the metacarpophalangeal and knee joints. Autoantibodies and rheumatoid factor were negative, and the immunoglobulin G (IgG) level was 7.33 g/L. Skin biopsy revealed “collagenization of the dermal fibrous tissue,” leading to a diagnosis of SSc. Chest X-ray revealed pulmonary fibrosis. The patient was treated with oral methylprednisolone (48 mg once daily) and intravenous tocilizumab (200 mg/10 mL once monthly), resulting in a gradual improvement in her condition. Three months ago, a blister appeared on the patient’s left middle finger at home without obvious cause. The patient attempted self-disinfection, but after the blister ruptured, the wound further expanded. Subsequently, wound dressing treatment was performed every 2 days at a community clinic, without any adjustment to the dosage of hormones or monoclonal antibodies. The wound continued to expand, extending to the back of the hand. Clearance and skin grafting surgery were performed at another hospital; however, the skin graft failed, and the wound further expanded to the back of the hand, accompanied by worsening infection of the extensor tendons of the second, third, and fourth fingers.
Admission examination
The patient’s vital signs were normal. The skin over the entire body was tight with a waxy luster, and the facial expression was stiff, presenting a “mask-like” appearance (Figure 1). Spinal mobility was limited, whereas joint mobility in the limbs was acceptable. Muscle strength in both lower limbs was grade 4. The fingers were flexed in a claw-like shape. A wound measuring approximately 4 cm × 6 cm was visible on the back of the left hand, exposing the middle phalanx joint of the left middle finger and the extensor tendons of the second, third, and fourth fingers (Figure 2). Pulmonary auscultation revealed coarse breathing sounds, with Velcro rales heard in both lungs. Cardiac examination showed no obvious abnormalities. The abdomen was flat, with no tenderness or rebound tenderness, and the liver and spleen were not palpable below the costal margins.

Conditions upon admission. (a) The patient exhibited a pained facial expression, with tight, waxy, lustrous skin and restricted flexion of the elbow joints and (b) condition of the wound upon admission, showing necrotic tendons and exposed phalangeal joints. We have obtained the necessary permissions from the copyright holders.

Pathological image of hand skin biopsy (H&E staining, ×100). The image shows epidermal and dermal tissues, with epidermal atrophy observed. The dermal collagen fibers appear thickened, and a small amount of lymphocyte infiltration is present around the small blood vessels. We have obtained the necessary permissions from the copyright holders. H&E staining: hematoxylin and eosin staining.
Right hand skin pathology diagnosis
Epidermal and dermal tissues were examined, revealing epidermal atrophy, thickened dermal collagen fibers, and a small amount of lymphocyte infiltration around small blood vessels.
Treatment course
Treatment of underlying disease
Hormone therapy for the primary disease was continued with oral methylprednisolone (20 mg once daily). Tocilizumab injection therapy was discontinued.
Treatment of skin ulcer
Systemic treatment. Antibiotic therapy with cefotiam was administered for infection control, along with treatments to improve microcirculation and provide nutritional support. Ozone water irrigation therapy. Prior to skin grafting surgery, 1000 mL of ozone water containing 45 μg/L ozone was flushed into the wound once daily through a negative pressure drainage device at a controlled speed. Surgical treatment. The first surgery involved debridement combined with vacuum sealing drainage (VSD). During the operation, necrotic tissues of the middle phalanx and the metacarpophalangeal joint capsule of the middle finger were observed, with an open joint cavity. After removing necrotic tendons and some necrotic tissue, a negative pressure drainage device was applied to the wound. Postoperatively, the wound was slowly irrigated with a saline solution containing growth factors (500 mL/24 h). On the 6th postoperative day, upon removal of the negative pressure device, the wound area had slightly decreased compared with that before surgery, with fresh granulation tissue at the base of the wound. The residual extensor tendon of the index finger was visible near the proximal wound, and no necrotic tissue was observed at the wound edges (Figure 3).
Conditions of the first debridement surgery. (a) Wound condition before surgery. (b) Wound condition during surgery and (c) wound condition after surgery. We have obtained the necessary permissions from the copyright holders.
The second surgery involved debridement with skin grafting, phalangeal fusion, and VSD. Following intraoperative debridement of edematous granulation tissue and interstitial tissue, K-wire fixation was performed for fusion of the second and third phalanges of the middle finger. Using an electric skin harvesting device, a 1% autologous split-thickness skin graft (approximately 0.3 mm thick) was harvested from the medial aspect of the patient’s left upper arm. This harvested skin was prepared as postage stamp-sized grafts and transplanted onto the left hand wound, followed by placement of a negative pressure device. Postoperatively, the wound was continued to be irrigated with a saline solution containing growth factors (500 mL/24 h). On the 4th postoperative day, upon removal of the negative pressure device, the grafted skin in the surgical area was found to be viable. Wound dressing treatment was continued. The wound was alternately irrigated and disinfected with hydrogen peroxide and saline solution, covered with an antimicrobial dressing, and secured with a dry gauze bandage (Figure 4). One month postoperatively, during an outpatient follow-up, the wound was completely healed with mild scar hyperplasia (Figure 5).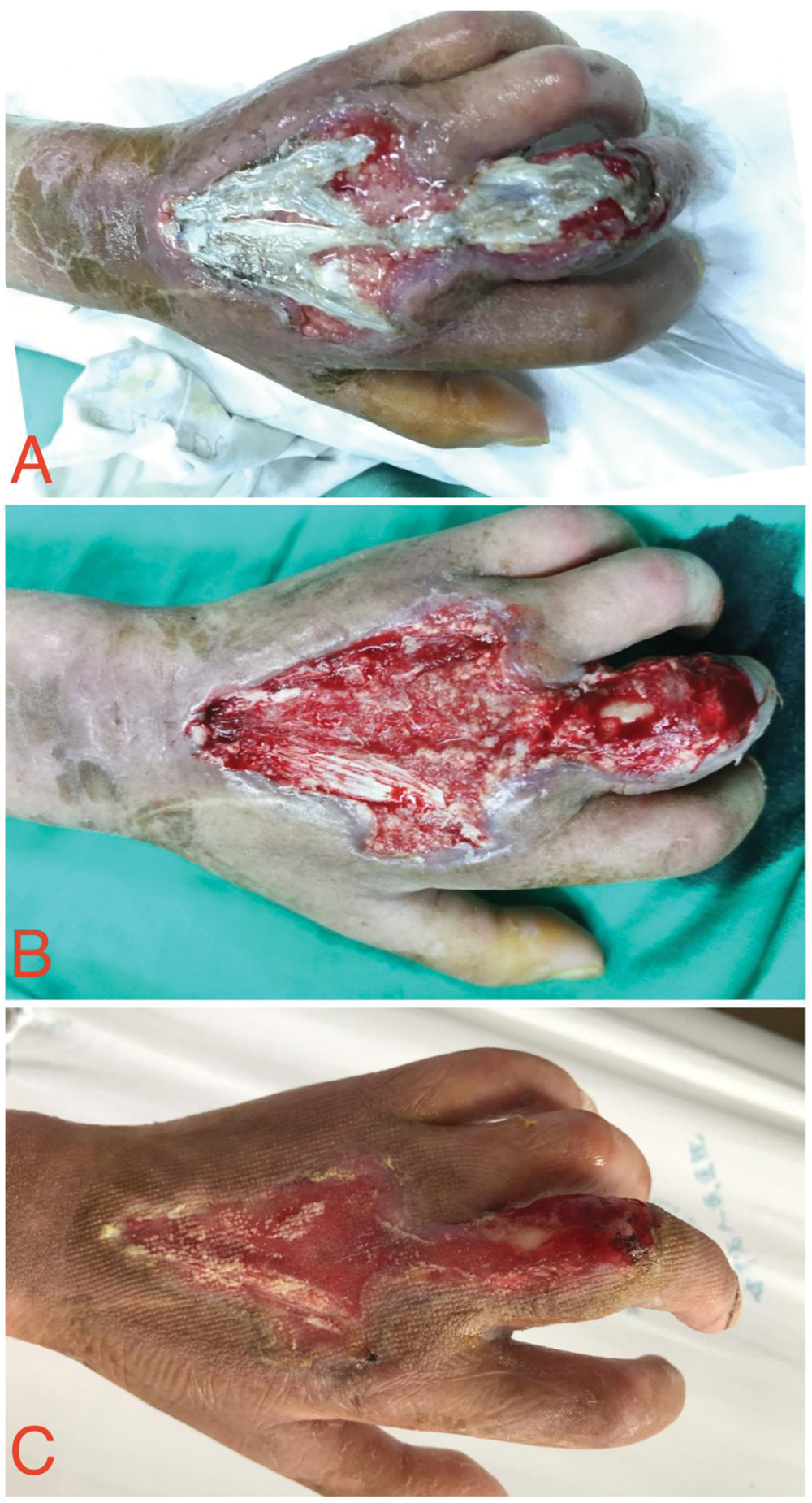

Wound condition after the second surgery. We have obtained the necessary permissions from the copyright holders.
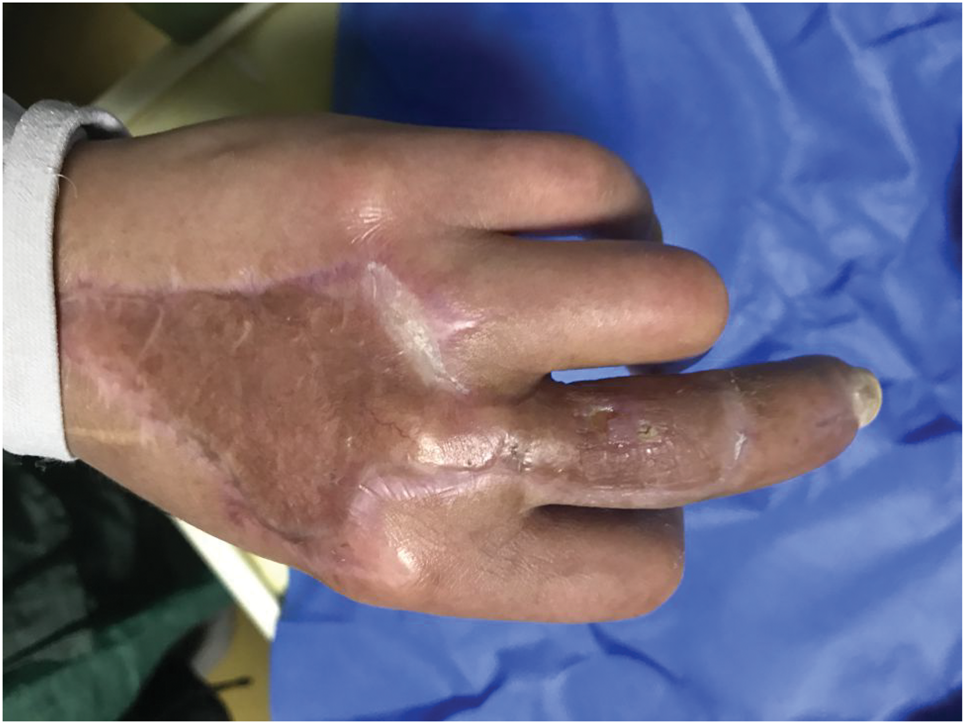
Wound condition 1 month after discharge. We have obtained the necessary permissions from the copyright holders.
Discussion
SSc is a relatively rare, multistage disease characterized by vasculopathy, immune dysregulation, and progressive fibrosis of the skin and internal organs. Despite its low prevalence (50–300 cases per million people), SSc poses a major health concern due to its high mortality rate, which is associated with organ failure, particularly of the lungs and heart.
7
Diagnosis is primarily based on the classification criteria established by the American College of Rheumatology (ACR),
8
with definitive diagnosis requiring a combination of clinical manifestations, immunological testing, skin pathology, and nailfold capillaroscopy. The primary clinical manifestations of this disease include RP, thickening and hardening of the skin, and musculoskeletal lesions, with skin thickening and hardening being characteristic features and often initially appearing on the fingers.
9
Pathological examination of the affected skin reveals an increase in dense collagen fibers in the reticular dermis, thinning of the epidermis, disappearance of dermal papillae, and atrophy of skin appendages. The pathogenesis of SSc is complex; however, the fibrotic pathological mechanism may be associated with increased collagen synthesis and reduced collagen breakdown by fibroblasts. Excessive deposition of collagen fibers in tissues and organs, including the skin, lungs, and digestive tract, leads to their hardening and subsequent dysfunction.
10
The formation of skin ulcers in SSc results from the combined effects of multiple factors, which can be broadly categorized into the following two types:
Vasculopathy. This is the primary pathogenic mechanism in SSc. Characteristic vascular lesions in SSc include intimal hyperplasia, luminal narrowing, or even occlusion of small arteries and microvessels.
Tissue ischemia. Vascular narrowing reduces blood flow, causing the skin tissue to remain in a state of chronic ischemia and hypoxia, which renders it abnormally fragile.
RP. Nearly all patients with SSc experience severe RP. Arteries in the fingers or toes undergo intense spasms in response to cold or emotional stress, causing complete interruption of blood flow. Repeated severe ischemic episodes can ultimately lead to tissue infarction and ulceration at the fingertips.
Skin fibrosis. Excessive deposition of collagen causes the skin to harden, tighten, and lose elasticity. This tightened skin further compresses already narrowed microvessels, worsening ischemia. Skin hardening over joints restricts movement, increasing the risk of skin tearing during joint flexion and extension.
The common sites of skin ulcers are as follows:
DU. This is the most common manifestation and is typically caused by ischemic infarction secondary to severe RP.
Knuckles. Skin tightness can result in laceration ulcers that commonly develop at bony prominences during joint flexion and extension.
Elbows, knees, and heels. Bony prominences such as the elbows, knees, and heels are particularly susceptible because of skin fibrosis, loss of elasticity, and localized pressure.
Calcinosis. Approximately one-quarter of patients with SSc develop subcutaneous calcium salt deposits (calcinosis). These hard, calcified nodules can compress the skin, rupturing and releasing chalky material, which results in the development of difficult-to-heal ulcers.11–13
The clinical characteristics of skin ulcers are as follows:
Intense pain. Ulcers involve areas rich in nerve endings and therefore are typically accompanied by significant pain.
Poor healing. Impaired local blood supply and persistent disease activity often result in chronic ulcers that may persist for weeks, months, or even years.
High infection risk. Ischemic tissue exhibits poor resistance to infection, making it highly susceptible to bacterial infections (e.g. cellulitis and osteomyelitis), which may lead to gangrene and the need for amputation. 7
This case represents a relatively rare instance of SSc complicated by a nonhealing wound, with the rapid progression of distal skin ulceration on the fingers being particularly uncommon. Apart from the approximately 1% of normal-appearing skin on the medial aspect of the left upper arm, the patient’s entire skin surface exhibited a waxy sheen and a coarse, rigid texture. Fortunately, this small area of normal skin was sufficient to repair the hand wound through skin grafting. Despite early detection and timely treatment, the ulcer development could not be controlled. Following a detailed review of the patient’s medication history upon admission, we decided to discontinue immunosuppressive therapy. During preoperative conservative dressing changes, we observed that the wound infection was largely controlled and showed signs of gradual improvement. This clinical course prompted us to raise an important question: Do interleukin-6 (IL-6) signaling pathway inhibitors interfere with wound healing?
The patient had been treated with the immunosuppressive agent tocilizumab, a recombinant humanized IL-6R inhibitor that competitively binds to IL-6 and IL-6R, thereby reducing collagen synthesis and improving tissue fibrosis. Literature has indicated that IL-6 is a proinflammatory cytokine produced by various cells including endothelial cells, dendritic cells, macrophages, and lymphocytes, playing a crucial role in the pathogenesis of SSc. Studies have shown that IL-6 contributes to vascular endothelial activation and apoptosis during the early stages of SSc, resulting in cardiac microvascular dysfunction and vascular lesions that subsequently impair myocardial blood supply. 14 Furthermore, elevated IL-6 levels have been found to correlate positively with the modified Rodnan skin score (mRSS), 15 supporting its critical role in the pathological process of SSc. Mechanistically, IL-6 promotes fibroblast proliferation and migration by activating these cells, thereby increasing collagen synthesis and leading to fibrosis in skin and visceral organs. 16 Conversely, activated fibroblasts can upregulate the IL-6 gene, further increasing IL-6 production and creating a self-perpetuating cycle that amplifies the fibrotic process. 17 IL-6 mediates signal transduction through three mechanisms: classic signaling, trans-signaling, and trans-presentation. The IL-6/soluble IL-6 receptor (sIL-6R) complex binds to the glycoprotein 130 (gp130) receptor, activating multiple downstream signaling pathways, including JAK-STAT3, JAK-SHP2-MEK-MAPK, and JAK-PI3K-AKT. IL-6 activates fibroblasts via the JAK-STAT3 pathway, promoting their differentiation into myofibroblasts and enhancing the synthesis and excessive deposition of extracellular matrix (ECM) components including collagen types I/III and fibronectin, in synergy with TGF-β1 to amplify fibrosis. However, IL-6 is also required for wound healing. It activates neutrophil infiltration to accelerate pathogen clearance; stimulates hepatocytes to produce acute-phase proteins such as C-reactive protein (CRP) and fibrinogen to enhance systemic anti-infective capacity; and at moderate expression levels, initiates repair by stimulating endothelial cells to produce vascular endothelial growth factor (VEGF) receptor and upregulating basic fibroblast growth factor (bFGF) expression to support granulation tissue growth. Therefore, excessive inhibition of IL-6 signaling with immunosuppressive agents in patients with SSc and skin ulcers is not conducive to wound healing and may exacerbate wound infection. However, the impact of IL-6 inhibition on wound healing in humans, particularly in patients with SSc, remains inconclusive.
Additionally, apart from a small area of normal skin (<10 cm2) on the medial aspect of the left upper arm, the patient had no other available donor sites for skin grafting. Therefore, during the interval between the first debridement and the second skin grafting procedure, we used a negative pressure drainage device combined with ozone water irrigation to prevent wound infection and promote granulation tissue growth, achieving favorable outcomes and laying the foundation for improving the success rate of subsequent skin grafting. We believe that low-concentration ozone improved local tissue oxygenation and reduced tissue fibrosis. Previous studies investigating skin oxygen partial pressure and fibroblast proliferation in patients with SSc have shown that skin oxygen partial pressure is lower in these patients than in healthy individuals, and that hypoxia can aggravate skin hardening and promote fibrosis.
6
Once skin ulcers develop, fibrosis can inhibit wound healing. Medical ozone is a mixture of ozone and oxygen generated from medical-grade pure oxygen and has been used clinically for more than 100 years. In 2001, Matsumoto et al. at the Department of Surgery, Chiba Tokushukai Hospital, Japan, demonstrated the significant efficacy of ozone oil in treating postoperative refractory fistulas and wounds. In 2003, Bialoszewski and Kowalewski provided technical evidence showing that this approach can effectively control wound infections and accelerate the healing of postoperative and traumatic wounds, supporting its value as an adjunctive treatment. Research has demonstrated that ozone plays the following roles in wound healing:
It improves hemodynamics by enhancing the plasticity and fluidity of red blood cell membranes and reducing plasma viscosity.
18
It increases intracellular adenosine triphosphate (ATP) and 2,3-diphosphoglycerate (2,3-DPG) levels, shifts the oxygen–hemoglobin dissociation curve to the right, and reduces oxygen affinity for hemoglobin, thereby significantly improving tissue oxygen supply efficiency.
19
It effectively activates and modulates the immune system. It stimulates production of various cytokines including interferons (γ and β), interleukins (IL-2, IL-6, and IL-8), and tumor necrosis factor (TNF-α), thereby enhancing immune defense.
18
Through the NF-κB pathway, it activates immune cells, enhances phagocytic function in granulocytes and cytotoxic activity in monocytes/macrophages, and improves antiviral capacity.
20
It activates superoxide dismutase (SOD) and other enzymes, boosting the body’s antioxidant capacity. It stimulates platelets to release platelet-derived growth factor (PDGF) and transforming growth factor (TGF-β1), which promote tissue repair.
21
It reacts with platelets to produce peroxides, altering their aggregation patterns and thereby facilitating the dissolution of existing thrombi.
22
Clinical studies have indicated that ozone therapy has been widely applied in the treatment of various autoimmune diseases, including gout. However, its clinical applications has certain contraindications: 1. Ozone gas must not directly enter pulmonary bronchial tissues, as inhalation causes destruction of alveolar epithelial cells; 2. Patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency cannot undergo high-dose autologous ozone blood reinfusion therapy; 3. Ozone therapy may induce skin allergic reactions; 4. Excessively high concentrations or doses during treatment can trigger systemic toxicity reactions. 23 In future clinical applications and research, overcoming the limitations associated with ozone administration to wounds and developing more efficient delivery methods will be key priorities to expand its therapeutic role in wound healing.
Conclusion
Based on this case, in patients with SSc complicated by skin ulcers, the use of immunosuppressive agents targeting inflammatory receptors should be carefully minimized. Instead, adjuvant therapies such as ozone gas baths, ozone water baths, and autologous blood transfusion, combined with surgical and other comprehensive treatments, should be employed to improve wound hypoxia and microcirculation, facilitating early wound repair and healing.
Footnotes
Acknowledgments
We are grateful to the constructive feedback provided by anonymous reviewers, which significantly improved the manuscript. We also extend our deepest gratitude to our families and colleagues for their unwavering encouragement throughout this project.
Author contributions
Ruonan Lu drafted the manuscript, Hui Wang analyzed the data, Lijuan Zhang collected and analyzed clinical data, Jian Wu obtained ethics approval and patient consent, and Jun Liu reviewed and revised the manuscript. All authors agree to be accountable for all aspects of the work.
Compliance with reporting guidelines
The reporting of this study conforms to the Case Report (CARE) guidelines. 24
Data availability statement
All clinical data relevant to this case are included in the manuscript and its supplementary files. Raw data (e.g. daily wound measurements and serum cytokine levels) are stored in the electronic health record system of Gansu Provincial Hospital and can be shared for noncommercial research purposes under a data use agreement. Requests should be directed to the corresponding author’s email.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethical approval statement
This case report was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki (2013 revision) and received approval from the Medical Ethics Committee of Gansu Provincial Hospital (Approval Number: 2024-093; issued on 29 February 2024). The study protocol, including the use of innovative therapies (e.g. ozone therapy and IL-6 inhibitor administration) and surgical interventions (e.g. skin grafting and phalangeal fusion), was reviewed and deemed compliant with institutional ethical standards for clinical case reporting.
Funding
This work was financially supported by the Lanzhou Science and Technology Bureau (No. 2023-2-99) and Clinical Medical Research Center for Burns and Wound Repair in Gansu Province (No. 21JR7RA674).
Patient consent statement
Written informed consent was obtained from the 50-year-old female patient (or her legally authorized representative, if applicable) prior to treatment initiation and publication. We have de-identified all patient details.
