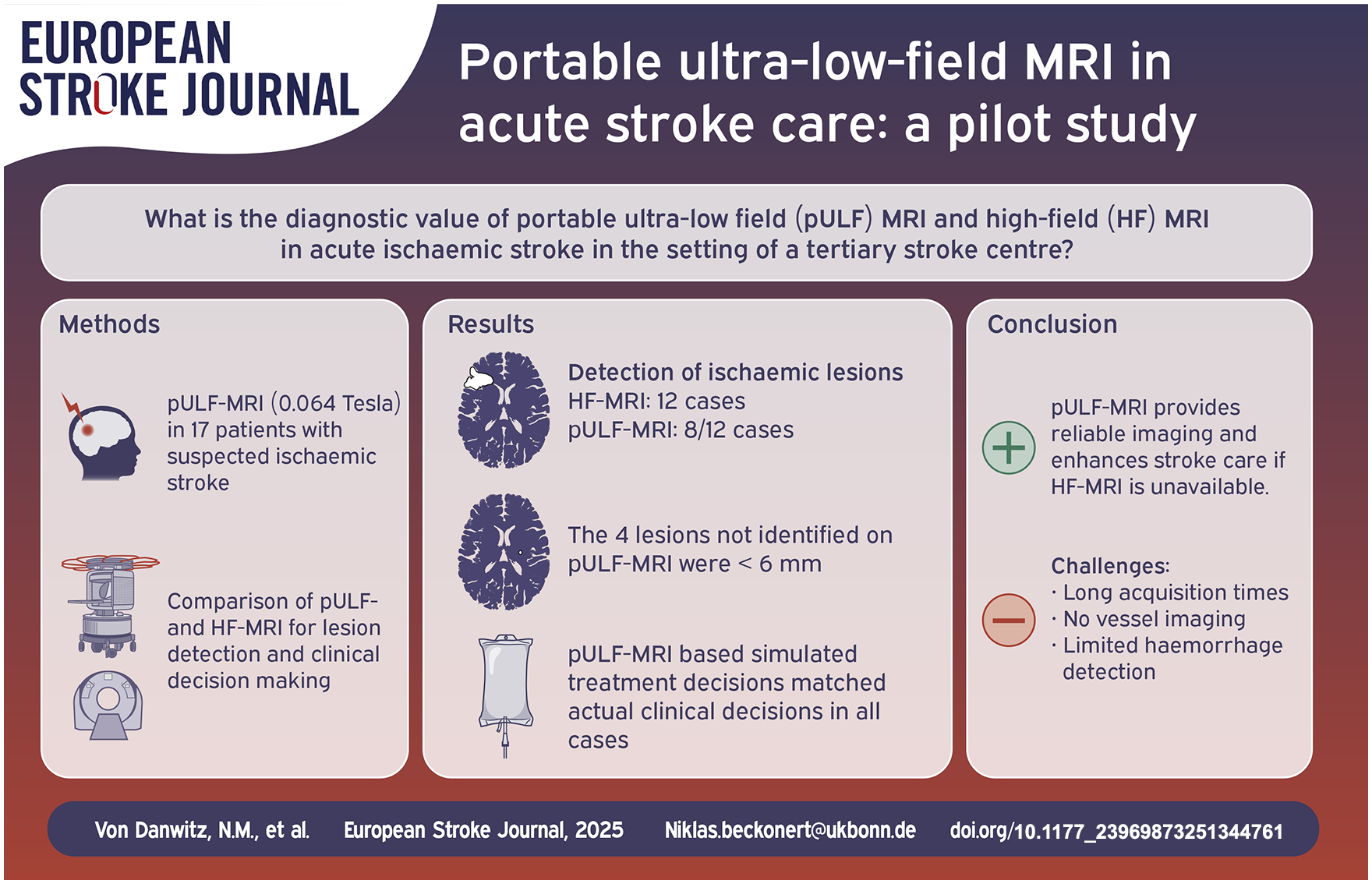
Abstract
Introduction:
Neuroimaging is a prerequisite for treatment of stroke patients, but it is not available all over the globe. Portable ultra-low field (pULF) MRI has the potential to improve access to neuroimaging and thus stroke care worldwide. In a pilot study, we were the first to utilise pULF-MRI in a European tertiary stroke centre and to evaluate its diagnostic value compared to high-field (HF) MRI.
Patients and methods:
Consecutive patients admitted for suspected ischaemic stroke underwent pULF-MRI using the 0.064 Tesla Swoop® portable MR imaging system in addition to standard imaging. HF-MRI and pULF-MRI scans were blindly assessed to compare the diagnostic accuracy and imaging-based therapeutic decisions based on pULF-MRI to HF-MRI.
Results:
Seventeen patients underwent pULF-MRI, 12 of whom had ischaemic lesions on HF-MRI. Ischaemic lesions were detected on pULF-MRI in 8/12 cases. The four infarcts not identified on pULF-MRI were all smaller than 6 mm in diameter. In all cases, a virtual treatment decision based on pULF-MRI by a blinded team matched the actual clinical decisions.
Conclusion:
This single-centre study demonstrates that pULF-MRI is a promising tool in acute stroke care, providing reliable imaging for treatment decision and follow-up monitoring. pULF-MRI may support acute stroke care if HF-MRI is unavailable and may be particularly helpful in resource-limited settings. Limitations of pULF-MRI include long acquisition times and the lack of vessel imaging and haemorrhage-sensitive sequences.
Keywords
Introduction
Stroke is the second leading non-communicable cause of death worldwide and the third leading cause of death and disability combined. 1 The majority of this burden – 86% of stroke-related deaths and 89% of disability-adjusted life-years (DALYs) – occurs in countries in the Global South. 2 In the most recent Global Burden of Disease study, stroke accounted for 160 million DALYs. 1 While the incidence of stroke has already increased by 70% in the last 30 years, the expected further increase, especially in younger people (<55 years), poses a global challenge. The World Health Organisation expects a further 50% increase in mortality by 2050.2,3
Reperfusion therapy has revolutionised stroke care, but according to a recent global analysis, only 2.9% of low-income countries could provide intravenous thrombolysis and endovascular treatment was not available in low-income countries investigated in the study. 4 Timely neuroimaging with computer tomography (CT) or magnetic resonance imaging (MRI) plays a crucial role in stroke subtype identification, lesion localisation, prognosis and therapy guidance, but global imaging availability varies widely between the global North and South. 5
In recent years, portable ultra-low field MRI (pULF-MRI) has emerged as a promising imaging alternative. Operating at fields below 0.1 T, these systems are smaller, do not require high power, sophisticated helium cooling systems, intensive electromagnetic or ferromagnetic shielding and require less staff and space, and could therefore be a cost-effective option.6,7 In recent US studies, pULF-MRI has shown high sensitivity and specificity for detecting stroke lesions 8 and DWI-FLAIR mismatch compared with HF-MRI. 9 The portable application also allows for a wide range of use cases, especially in critically ill patients in the intensive care unit, 10 and even in children and neonates. 11 The lack of radiation exposure and cost-effectiveness are also potential advantages over CT. In addition, MRI generally offers diagnostic superiority over CT in brain imaging. These two points make pULF-MRI an exciting innovation, not only in resource-limited settings. We therefore intended to pilot the use of pULF-MRI in stroke care at our European tertiary stroke centre.
In this pilot study, patients received pULF-MRI in addition to guideline-directed care. The diagnostic accuracy of pULF-MRI was compared to HF-MRI by a blinded assessment. Neuroradiologists and vascular neurologists, blinded to HF-MRI, provided a virtual therapy decision based on clinical history and pULF-MRI, which was compared with actual clinical decisions.
Methods
Study design
This pilot study was conducted in a cohort of patients with suspected ischaemic stroke who were admitted to a tertiary university stroke centre. After initial evaluation in the emergency department, patients received pULF-MRI and HF-MRI scans. The diagnostic accuracy of pULF-MRI to detect ischaemic lesions was compared to HF-MRI by blinded raters. The blinded raters also made a simulated treatment decision based on pULF-MRI and clinical information, which was compared to actual clinical decision.
Patient cohort
Over a period of 16 days (between 12/09/24 and 27/09/24), all consecutive patients of a tertiary university stroke centre with a suspected diagnosis of ischaemic stroke after initial evaluation in the emergency department were screened. The initial evaluation included cranial CT and CT angiography as standard imaging in most cases. Patients aged over 18 years within 72 h of symptom onset were included. Patients had to be able to give written informed consent. The study was approved by the Ethics Committee of the University of Bonn (number 167/22). Consent from a proxy or relative was not allowed. Patients with agitation, severe aphasia and impaired consciousness were therefore excluded. In the case of expressive aphasia, capacity to consent was assessed independently by two stroke physicians. Exclusion criteria included contraindications to MRI (e.g. implanted devices, severe claustrophobia). Patients could only be enrolled in this prospective pilot study once standard imaging was completed and acute stroke therapy had been initiated. Electronic medical records of enrolled patients were retrospectively analysed: stroke severity was assessed by vascular neurologists using the National Institutes of Health Stroke Scale (NIHSS), and premorbid disability was assessed using the modified Rankin Scale (mRS).
Clinical HF MRI standard protocol
Clinical HF MRI scans were performed using a 3 T MRI scanner (Achieva, Philips Healthcare, Best, The Netherlands) or a 1.5 T MRI scanner (Achieva, Philips Healthcare, Best, The Netherlands). The minimum protocol included diffusion-weighted imaging (DWI, slice thickness 5 mm, gap 6 mm, b-values 0, 500, 1000), axial fluid-attenuated inversion recovery (FLAIR, slice thickness 5 mm, gap 6 mm, TE 140 ms, TR 12,000 ms, TI 2850 ms), and gradient echo imaging (T2*, slice thickness 5 mm, gap 6 mm, TE 20 ms, TR 700 ms) and was performed for patients who had already undergone CT-imaging, including non-contrast enhanced CT of the brain and CT angiography, as primary imaging modality. The acquisition time of the described sequences was approximately 9 min. Sequence parameters could be varied at the discretion of the MRI technician when necessary. Additional MR sequences were selected at the discretion of the responsible neuroradiologist and included time of flight MRA, susceptibility weighted imaging or gadolinium administration for contrast enhanced MRA or contrast enhanced T1 weighted imaging.
pULF-MRI protocol
The 0.064 T Swoop® Portable MR Imaging System (Hyperfine, Inc., Guilford, CT, USA; hardware version 1.7; software version 8.7commercial) was used to acquire pULF-MRI scans. The scanner weighs 620 kg has a height of 140 cm and a diameter of 86 cm (Figure 1). For operation, the scanner is plugged into a standard electrical outlet. The scanner was operated by a neurology resident using a tablet. AG and HS trained NVD, JNM, OSS, HA, CT, TE and SS with a training duration of approximately 1 h including all safety instructions and operating procedures. The protocol utilised the standard product axial FLAIR (3D inversion-recovery turbo spin echo, acquisition time = 9 min 12 s, resolution = 1.7 × 1.7 × 5 mm, TE/TR/TI = 175/3500/1290 ms, receiver bandwidth = 64 kHz, echo train length = 68, excitation/refocusing/inversion flip angles = 90°/180°/180°) and axial diffusion-weighted imaging (3D turbo spin echo, acquisition time = 9 min 50 s, resolution = 2.4 × 2.4 × 6 mm, TE/TR = 62/1000 ms, receiver bandwidth = 52 kHz, echo train length = 40, excitation/refocusing flip angles = 90°/180°). In the case of a DWI-FLAIR mismatch, scans were repeated serially every hour for a maximum of four scans until ischaemic lesions were FLAIR-positive.
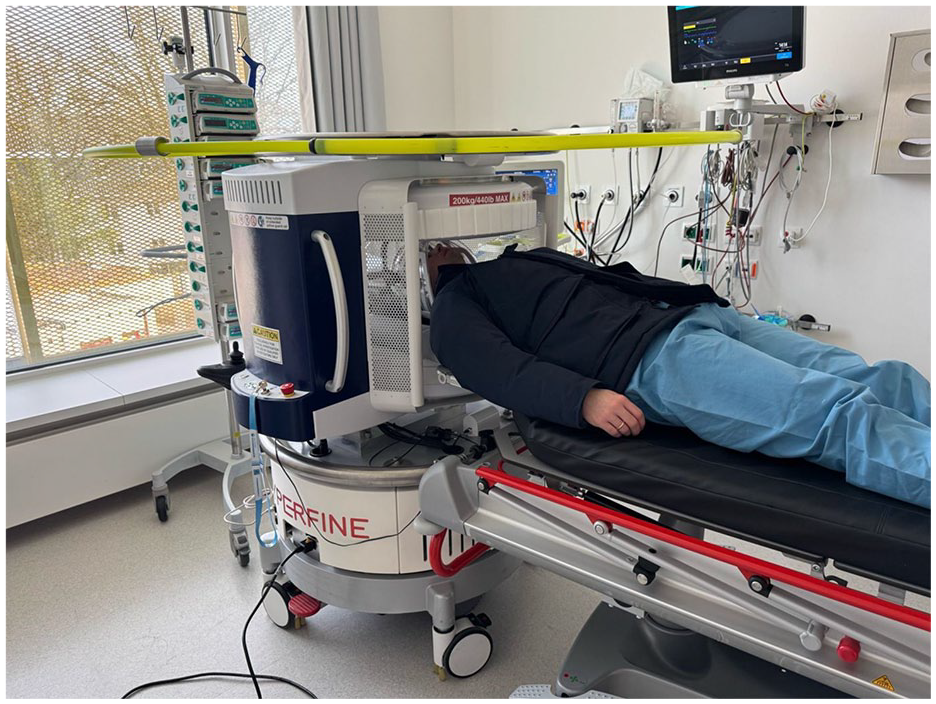
A photo of the 0.064 T Swoop® Portable MR Imaging System used in this study. The person being examined is lying on a stretcher that had been moved next to the device, but is not part of it.
MRI assessment, accuracy of treatment planning
The pULF-MRI scans were assessed by an experienced neuroradiology attending (NL) and an experienced vascular neurology attending (JMW). The readers were provided with information about the clinical presentation, but blinded to CT and HF-MRI results and also to the time of stroke onset, simulating a treatment decision for a stroke with unknown onset. A subsequent virtual treatment decision (diagnosis of ischaemic stroke, performing intravenous thrombolysis) was generated based on the clinical information and the imaging. The performance of pULF-MRI in acute stroke assessment was evaluated in the following categories:
(1) Detection and localisation of ischaemic lesions. The gold standard for diagnosis was HF-MRI, and pULF-MRI results were considered diagnostic if they demonstrated concordance with HF-MRI regarding detection and localisation of ischaemic lesions. Lesion size was measured on DWI on the axial plane with the largest diameter.
(2) Assessment of mismatch between DWI and FLAIR. In the case of a mismatch, serial scans were performed to assess the dynamics.
(3) Relevant concomitant imaging findings – this means that all relevant information from the clinical HF-MRI reading was also present in the pULF-MRI report (e.g. mass effect, presence of tumour). Intracranial haemorrhage was assessed in this category as no susceptibility weighted sequence was available for pULF-MRI.
(4) Clinical decision making (diagnosis of ischaemic stroke, intravenous thrombolysis) based on the clinical presentation and pULF-MRI.
Outcome measures
Primary outcomes included measures of diagnostic accuracy for the detection of ischaemic lesions on pULF-MRI compared to HF-MRI. Secondary outcomes included the concordance of a virtual treatment decision based on pULF-MRI compared to the actual treatment decision, and the detection of relevant concomitant findings on pULF-MRI compared to HF-MRI.
Statistical analysis and software used
No sample size calculation was performed for this pilot study, and missing data were not imputed for this analysis. Standard descriptive statistics were provided, the Wilson method was used to provide 95% CI around confidence intervals. Statistical calculations were performed using Microsoft Excel (2016 version, Microsoft) and IBM SPSS Statistics (version 22.0, IBM). Draw.io (version 26.1.1, https://app.diagrams.net/) was used to create the flowchart and InkScape (Inkscape 0.92.2, https://www.inkscape.org) and Gimp (version 2.10.30, https://www.gimp.org/) were used to create the visual abstract.
Results
Patient cohort
Eighty-six patients admitted to our emergency department with suspected ischaemic stroke over a period of 16 days were screened for inclusion in the study and 19 patients agreed to participate. The main reasons for screening failure were inability to consent due to stroke-related or premorbid functional deficits, onset of stroke symptoms longer than 72 h before admission and unwillingness to participate. Median age was 76 years and 41.2% were female (Table 1). Seventeen of the 19 patients (89.5%) included underwent at least one pULF-MRI scan. Fifteen received a HF-MRI scan (11 patients with 3T, four patients with 1.5T HF-MRI), while two patients were examined by CT and pULF-MRI only. The median time from symptom onset to first imaging was 4.75 h (interquartile range [IQR]: 1.75–14.5), the median time interval between pULF- and HF-MRI was 2 h (IQR: 0.7–9.4).
Patient cohort demographics and clinical characteristics.

HF: high-field; IQR: interquartile range; y: years; no.: number; NIHSS: National Institutes of Health Stroke Scale; mRS: Modified Rankin Scale; pULF: portable ultra-low-field; SO: symptom onset (if available, otherwise interval to last known normal).
Seven patients who presented with stroke onset < 4.5 h or with unknown symptom onset < 10 h were enrolled. Three of these patients received reperfusion therapy (intravenous thrombolysis and endovascular therapy) before the pULF-MRI scan. 10 patients gave consent later than 10 h after stroke onset after >10 h of stroke symptoms, two of whom had received endovascular therapy (Figure 2).

Flow diagram for patient inclusion.
pULF-MRI scans were well tolerated by most patients. Some pULF-MRI scans were confounded by motion artefacts and had to be repeated to ensure diagnostic quality. The relatively hard headrest and table of the pULF-MRI were uncomfortable for some patients, which we tried to alleviate with pillows and blankets. Two patients with claustrophobia found the pULF-MRI easier to tolerate than the HF-MRI due to its smaller size, quieter noise and the fact that their partner was able to stay next to them.
Detection of ischaemic lesions and clinical decision-making
Ischaemic lesions were detected on HF-MRI in 12/15 patients. On HF-MRI, the median of the maximum lesion diameter was 33 mm (IQR: 5.25–60.5). Ischaemic lesions ⩽ 10 mm were observed in 5/15 patients (33.0%), and ischaemic lesion ⩽ 5 mm in 4/15 patients (26.7%). Both readers correctly identified corresponding lesions in 8/12 cases on pULF-MRI, giving a sensitivity of 67% (95% confidence Interval (CI) [0.39, 0.86]). The four infarcts not detected on pULF-MRI were all smaller than 6 mm in diameter, resulting in sensitivity of 0% for infarcts smaller than 6 mm. Infarcts with a diameter of 6 mm or larger were all detected on HF-MRI (8/8), resulting in a sensitivity of 100%. The smallest lesion correctly identified by pULF-MRI was 6 mm in diameter (6 mm × 5 mm in axial extent). Upon inclusion of two cases who had evidence of infarction on CT, but were not examined by HF-MRI, both readers identified ischaemic lesions in 10/14 cases compared to standard imaging, resulting in a sensitivity of 71% (95% CI [0.45, 0.88]).
Three patients were found to have a stroke mimic or no evidence of DWI or FLAIR lesions on HF-MRI (diagnosed as a transient ischaemic attack (TIA) or MRI-negative stroke) after diagnostic work up, which were correctly identified in 3/3 (neuroradiologist) and 2/3 (neurologist) cases, corresponding to a specificity of 100% and 67%, respectively.
Both readers agreed on all simulated treatment decisions (17/17) based on clinical information and pULF-MRI. In summary, the simulated treatment decisions were consistent with the actual clinical decision in all cases.
Supplemental Table 1 provides a detailed description of the clinical and MRI characteristics of all 17 cases. Exemplary HF- and pULF-MRI imaging are shown in Supplemental Figures 1 and 2. A contingency table comparing the blinded assessments is provided in Supplemental Table 2.
Incidental findings: Secondary haemorrhage and space-occupying effects
In some cases, relevant findings were incidentally identified on pULF-MRI. These cases are described in brief.
An 84-year-old patient (case number 4 in Supplemental Table 1) was referred by a partner hospital for endovascular therapy for left M1 occlusion after intravenous thrombolysis. Angiography showed recanalisation of the left middle cerebral artery and mechanical thrombectomy was no longer required. Clinically, the hemiplegia had improved to moderate hemiparesis, but moderate expressive aphasia remained. The patient consented to the study and even signed with his previously paretic hand. pULF-MRI 3 h after initial presentation showed a subacute middle cerebral artery infarction, another 3 h later a space-occupying effect due to haemorrhage was observed, a CT confirmed haemorrhagic transformation (volume of 41.9 ml, estimated using the ABC/2 method) of the infarct suspected on pULF-MRI (Figure 3).
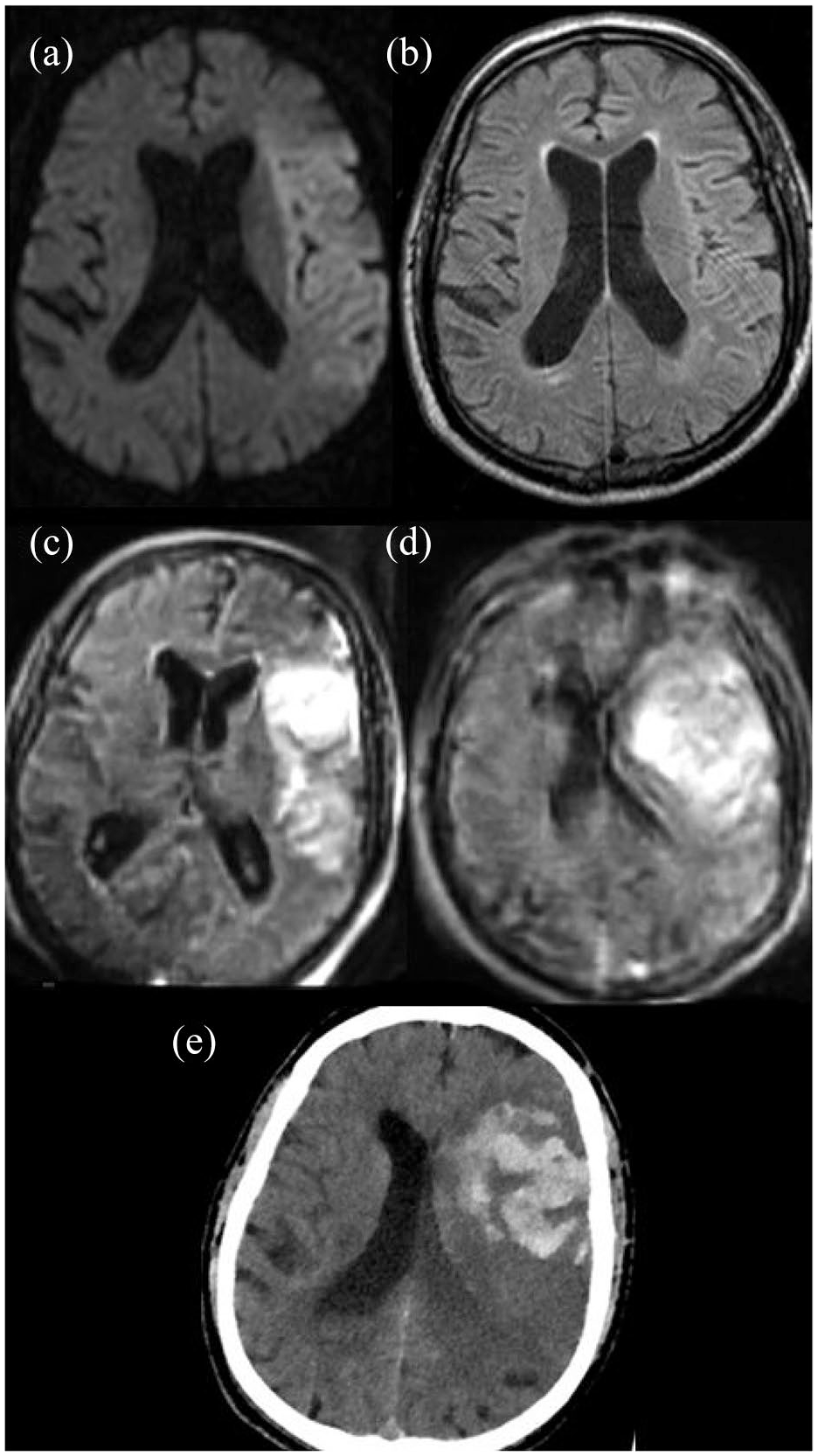
Eighty-four-year-old patient with left middle cerebral artery occlusion (not shown, case number 4 in Supplemental Table 1). (a and b) initial external MRI with DWI (a) and FLAIR (b) showing early ischaemic changes in the left middle cerebral artery territory. (c) FLAIR on pULF-MRI 3 h after initial presentation (c) with established left middle cerebral infarction and initial signs of haemorrhage in the left frontal lobe. (d) FLAIR image on pULF MRI another 3 h later with progressive haematoma in the left frontal lobe with mass effect on the left side ventricle. (e) Immediate CT scan confirmed space occupying haemorrhage in the left frontal lobe.
A 72-year-old patient (case number 8 in Supplemental Table 1) was admitted with acute onset vertigo, spontaneous nystagmus, nausea and vomiting beginning 1 h before admission. CT angiography showed extracranial occlusion of the left vertebral artery and occlusion of the left posterior inferior cerebellar artery (PICA). HF-MRI showed a FLAIR-positive infarct in the left PICA territory. Intravenous thrombolysis was not administered due to improvement in symptoms resulting in a NIHSS score of 0. Bedside pULF-MRI 36 h after admission showed incipient space-occupying left PICA infarction. Clinical monitoring was intensified and, following clinical deterioration, follow-up CT scans confirmed malignant cerebellar oedema (volume of 80.9 ml, estimated using the ABC/2 method) and the patient underwent suboccipital decompression (Figure 4).

Seventy-two-year-old patient with left posterior inferior cerebellar artery infarction (case number 8 in Supplemental Table 1). (a–c), HF-MR with DWI (a), FLAIR (b) and ADC map showing early ischaemic changes with no mass effect. (d–f), pULF-MRI 36 h later with DWI (d), FLAIR (e) and ADC map (f), showing established infarction in the left posterior inferior cerebellar artery territory with progressive mass effect. (g) Subsequent CT scan confirming space occupying left cerebellar infarction.
In one patient (case number 5 in Supplemental Table 1), a prepontine mass measuring 8 × 9 mm, most likely corresponding to an epidermoid, was discovered incidentally on HF-MRI. This abnormality was also detected on pULF-MRI by the neuroradiology reader.
Discussion
In this pilot study, the sensitivity for detection of ischaemic lesions was 67% and 72% including patients without HF-MRI, respectively, which is a decent result, but lower than previously reported rates ranging from 86% to 90%. 9 This difference may be explained by the relatively mild stroke severity in our cohort: The median NIHSS score on admission was 2 (IQR: 1–9) versus 5 (IQR: 3–8) in the aforementioned cohort. Furthermore, the frequency of infarcts with a diameter ⩽ 10 mm in that cohort was 10%, whereas the frequency of infarcts with a diameter ⩽ 10 mm was 33% in our cohort.
Feasibility studies have recently tested various applications of pULF MRI in stroke care, demonstrating relatively good diagnostic accuracy for the detection of ischaemic and haemorrhagic lesions and for the detection of a DWI-FLAIR mismatch.8,12 –14 In addition, other case series demonstrated further potential applications in neurology, broadening the scope to include bedside monitoring in intensive care units, detection of potentially epileptogenic lesions and imaging of neurodegenerative diseases.10,11,13,15 Not surprisingly, pULF-MRI images were more susceptible to motion artefacts and had lower spatial resolution than HF-MRI, especially for small ischaemic lesions: A diameter of 6 mm represented the threshold for detectable lesions on pULF MRI in our study. After the blinded review of pULF-MRI scans, the vendor introduced a research software to improve postprocessing of the MR images. To demonstrate possible improvements of image quality in the future, we included an example of the newly processed images in Supplemental Figure 1. The diagnostic limitations of pULF-MRI compared to HF-MRI did not impact the actual and virtual treatment decisions made.
We identified four major advantages of the pULF-MRI over HF-MRI in our pilot study: First, the compact size and portability of the pULF-MRI may be used directly in the emergency department without the need to transfer the patient to the MRI suite. In addition, nurses and physicians can work on the patient during the scan (e.g. precise intravenous blood pressure management), allowing closer monitoring and more sophisticated therapy without interrupting the scan. Second, in two cases, the patients’ partners were able to be present during the scan, alleviating claustrophobia symptoms and increasing patient comfort. Third, 1 h of training was sufficient to enable the stroke neurologist to operate the scanner, and no other person or special requirements were needed other than a WIFI connection and regular power supply. Forth, we presented two cases in which pULF-MRI identified early complications and thus allowed timely therapeutic interventions in these patients. This illustrates the applicability of pULF-MRI for bedside monitoring of acute stroke patients. In addition, a hand knob infarct not detected on CT was identified on pULF-MRI. In line with this, pULF-MRI has recently proven to be diagnostically superior to CT in detecting ischaemia in selected cases, 10 making pULF-MRI a potentially interesting alternative due to the lack of radiation exposure. The longer acquisition time of pULF MRI is a disadvantage compared to CT, but also HF-MRI. Furthermore, the lack of high-resolution anatomical imaging and functional sequences, such as perfusion imaging and angiography, limits the full diagnostic potential of pULF-MRI in stroke subtyping and comprehensive assessment, especially identifying candidates for endovascular therapy. Another disadvantage is the lack of susceptibility weighted sequences to detect haemorrhage. However, the only case of secondary haemorrhagic transformation was detected on pULF-MRI on DWI imaging, suggesting that haemorrhage can be detected on pULF-MRI even without haemorrhagic-sensitive sequences. A recent feasibility study indeed showed high sensitivity and specificity for detecting different types of intracranial haemorrhage on pULF-MRI.12,14 The limitations of pULF-MRI as a stand-alone imaging method compared to HF-MRI must be balanced against the cost-effective and potentially wider availability in the future. In its current state, pULF-MRI is not sufficient as stand-alone imaging for acute stroke patients due to the long acquisition time and the lack of vessel imaging and haemorrhage-sensitive sequences. It may support acute stroke care in special situations, such as wake-up stroke or for the differentiation of stroke mimics, if HF-MRI is not available or not feasible, for example, in patients requiring constant intensive medical attention.
In the Global South, where stroke care is often hampered by a lack of imaging resources, pULF-MRI offers an opportunity to bridge the diagnostic gap. The portability, lower cost (approximately US$ 260,000), reduced staff, energy and resource requirements and likely smaller environmental footprint make pULF-MRI a promising tool for improving stroke care in these settings, where the burden of stroke is steadily increasing.2,4 However, challenges remain in terms of staff training, initial implementation of stroke workflows or integration of pULF-MRI into established stroke protocols, and establishing and ensuring quality control. From a global perspective, pULF-MRI can be considered as part of a broader diagnostic strategy with potential for pre-hospital use, for example, in mobile stroke units, possibly complemented by other imaging modalities and, most importantly, education and prevention.
Our study has the following limitations: pULF-MRI presently is of limited availability in Europe, which has resulted in the study being conducted over a relatively short period of time. Consequently, only a limited number of patients could be recruited, resulting in a highly heterogeneous patient collective that is not representative and therefore limits the generalisability of the findings. In addition, our study has a strong selection bias: Due to the need for written informed consent, we had to exclude patients with severe neurological deficits such as agitation and severe aphasia, who often have large infarcts, in our pilot study, and therefore small infarcts (diameter ⩽ 5 mm) are overrepresented in our collective. Further, haemorrhagic stroke and stroke mimics are underrepresented in our cohort compared to a general cohort of patients admitted for suspected stroke due to prescreening which included CT/CTA in the majority of cases, therefore our results should be interpreted with caution as they are most likely not representative for general cohorts of patients admitted for suspected stroke. The median time of 2 h between pULF and HF MRI hinders comparable imaging, and dynamic changes in DWI/FLAIR lesions may be missed. Last, reference standard (HR-MRI) was not exactly the same for all patients, as some of them underwent 3T MRI while others underwent 1.5 T MRI.
Further studies are therefore needed. The recent CE mark for EU countries will facilitate clinical use in Europe (Certificate Number: 28620144059). An Italian multicentre study (NCT05816213) to assess diagnostic accuracy and validate imaging and the implementation of pULF-MRI in the emergency department and in a mobile stroke unit are driving developments in the field and will hopefully begin recruitment soon. 16
Conclusion
In conclusion, our pilot study shows that pULF-MRI may be a feasible and valuable tool for stroke management in the future, offering important advantages such as portability and accurate lesion detection in infarcts larger than 6 mm in diameter. Although it demonstrated reasonable sensitivity, the lower spatial resolution and longer acquisition times of pULF-MRI were limitations. The lack of iron-weighted sequences and vascular imaging hinders its use as a stand-alone imaging for deciding about intravenous thrombolysis therapy or endovascular therapy. Despite these drawbacks, pULF-MRI holds potential for use in emergency departments and resource-limited settings, where HF-MRI may not be available: The lack of radiation exposure and its potential diagnostic advantages over CT in certain cases make it a promising adjunct. However, further studies and integration into clinical protocols are needed to address its limitations and optimise its use. With ongoing research and the recent CE mark in the EU, pULF-MRI holds great promise for improving stroke diagnosis and treatment worldwide.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251344761 – Supplemental material for Portable ultra-low-field MRI in acute stroke care: A pilot study
Supplemental material, sj-docx-1-eso-10.1177_23969873251344761 for Portable ultra-low-field MRI in acute stroke care: A pilot study by Niklas M von Danwitz, Nils C Lehnen, Julius N Meißner, Omid Shirvani Samani, Hannah Asperger, Christian Thielscher, Taraneh Ebrahimi, Julia Layer, Louisa Nitsch, Franziska Dorn, Alexander Radbruch, Felix J Bode, Johannes M Weller, Anne Groteklaes, Gabor C Petzold, Hemmen Sabir and Sebastian Stösser in European Stroke Journal
Footnotes
Acknowledgements
We thank all the patients who participated in this pilot study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NVD received travel grants from Sanofi and Viatris. AR serves on the scientific advisory boards for GE Healthcare, Bracco, Bayer, Guerbet and AbbVie; has received speaker honoraria from Bayer, Guerbet, Siemens and Medscape; and is a consultant for, and has received institutional study support from, Guerbet and Bayer. All other authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open Access funding facilitated and organised by the DEAL project. The acquisition of the 0.064 T Swoop® Portable MR Imaging System was supported by the Bill and Melinda Gates Foundation (Ultralow field Neuroimaging In The Young: INV-005798). No other funding was received for this study.
Informed consent
All patients gave written informed consent for the study procedure.
Ethical approval
The study was approved by the Ethics Committee of the University of Bonn (number 167/22).
Guarantor
SS.
Contributorship
This study was conceptualised by HS, GCP and SS. The study protocol was designed by NVD, JNM, JMW, GCP and SS and carefully reviewed and edited by OSS, HA, CT, TE, JL, NL and FJB. AG and HS designed the pULF-MRI protocol, facilitated the pULF-MRI device training and were always available to answer technical questions. Patient recruitment and pULF-MRI image acquisition were performed by NVD, JNM, OSS, HA, TE, CT. Coordination of HF-MRI was organised by NL. Image interpretation and virtual decision making was performed by NL and JMW. NVD, JNM and NL analysed the data. NL designed the radiological figures including figure legends; the other figures were designed by NVD and JNM. The first draft of the manuscript was written by NVD, JNM and SS were the other major contributors, all authors revised and edited the paper before submission and approved the final version.
Availability of data and materials
Data will be provided by the corresponding author upon reasonable request.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
