Abstract
Background:
Distal embolization (DE) is a concerning complication of endovascular treatment (EVT) that is linked to adverse clinical outcomes in acute ischemic stroke (AIS). The thrombus enhancement sign (TES) has been identified as a potential biomarker of occlusion; however, its predictive value for DE remains to be fully clarified.
Methods:
We retrospectively evaluated data of consecutive patients with large vessel occlusion (LVO) in anterior circulation who underwent EVT between January 2020 and December 2023. Each included patient was underwent baseline non-contrast computed tomography (CT) and CT angiography (CTA). We assessed the incidence of DE and its association with clinical and technical outcomes. The association of DE with patient characteristics, thrombus imaging, and composition features was assessed using univariable and multivariable logistic regression.
Results:
A total of 308 patients were included in the analysis. Distal embolization occurred in 21.1% (65/308) of patients. Patients with DE had longer recanalization time (58 min vs 46 min; p = 0.002), lower proportion achieving successful reperfusion (86% vs 95%; p = 0.022) and decreased frequency of functional independence (31% vs 45%; p = 0.041). TES was significantly more frequent in the DE group compared to the non-DE group (89% vs 70%, p = 0.001). Histopathological examination revealed that TES-positive thrombi contained a higher fibrin/platelet proportion (50.9% vs 48.2% in TES-negative thrombi, p = 0.009) and fewer erythrocytes (41.7% vs 43.5% in TES-negative thrombi, p = 0.043). In multivariable analysis, TES (OR, 2.76; 95% CI, 1.23–7.10; p = 0.022) was the only independent predictor of DE.
Discussion and Conclusion:
Our study demonstrates that distal embolization is a prevalent complication of EVT that hinders full recanalization and favorable outcomes. The presence of TES on baseline CT imaging could serve as a reliable radiologic predictor of distal embolization after EVT.
Introduction
Endovascular thrombectomy (EVT) has evolved as the standard of care for patients suffering from acute ischemic stroke (AIS) due to large vessel occlusion (LVO) in the anterior circulation.1,2 Although EVT has significantly improved recanalization rates, a subset of AIS patients still fails to achieve complete recanalization due to partial removal or distal embolization (DE). DE refers to the migration of fragmented thrombi to new or more distant territories, which were not previously ischemic but become affected after being dislodged below the initial target occlusion. 3 These emboli can travel to more distant branch vessels, leading to persistent or worsened ischemia in brain regions already compromised by the initial occlusion.
Thrombectomy-related DE has been observed in approximately 40% of AIS cases and is one of the major complications closely associated with impairment of successful reperfusion rates and worse neurological improvement.4,5 However, the factors linked to DE remained poorly understood. Recent histologic studies of retrieved clots have provided insights into erythrocyte-rich thrombi, which are thought to have an unstable composition and are associated with a high likelihood of pre-interventional migration.6,7 These studies also revealed that migrated thrombi are linked to lower complete recanalization rates after thrombectomy and correlate with unfavorable outcomes. Nevertheless, some studies have found no direct relationship between thrombus composition and the risk of DE, 8 suggesting that thrombus composition may be one of several important determinants of DE. Due to the challenges in obtaining pathological samples, research on this topic is limited, and a consensus has yet to be reached.
Cranial CT-angiogram (CTA) is routinely used to identify arterial occlusion and assess thrombus origin and stability. 9 Imaging features such as thrombus length, size and collateral status have been suggested as potential indicators of DE, 10 but these measurements are difficult to obtain prior to thrombectomy and have relatively limited diagnostic accuracy. Recently, we have reported thrombus enhancement sign (TES) on thin slab maximum intensity projection (TS-MIP) image reconstruction of CT angiograms as a new imaging biomarker for predicting stroke etiology and thrombectomy efficiency.11,12 We observed that enhanced thrombi have a high prevalence in embolic sources and may be associated with subsequent migration after thrombectomy attempts. The enhanced clarity of positive TES aids in assessing the morphological characteristics of occlusions, such as thrombus length and vascular contour. Additionally, positive TES imaging even improve the visualization of distal branch vessels. Consequently, it provides valuable insights into the likelihood of thrombus migration during the thrombectomy process.
Therefore, the purpose of this study was to investigate whether TES, along with other thrombus characteristics, could be used to characterize intracranial thrombi and predict distal embolization before thrombectomy, as well as to confirm its relation to clot histological composition.
Methods
Patient selection
This single-center retrospective study was approved by institutional review board and informed consent was provided by patient or legal representative before the procedure. Patients who presented within 16 h of AIS symptom onset and underwent thrombectomy between January 2021 and December 2023 were consecutively included. Only patients with occlusion of the distal internal carotid artery (ICA) or proximal middle cerebral artery (MCA of M1 and M2 segments) and complete diagnostic and histologic workup were included for further analysis: patients with insufficient imaging quality for the required analysis were excluded.
Patients were treated with intravenous thrombolysis if they qualified for this procedure, and thrombectomy decisions were made by experienced neurointerventional radiologists, based on clinical and imaging evaluations. The demographic, clinical, and procedural data of patients were collected and compared. Functional independence was defined as a modified Rankin Scale score of ⩽2 at 90 days. Distal embolization was defined as embolization occurring in the downstream region of the initial target occlusion or in territories unrelated to the originally occluded artery.3,10 A systematic pretreatment angiography and an angiography after stent retriever deployed confirmed the initial patency of the distal middle cerebral arteries (MCAs) or anterior cerebral arteries (ACAs).
Imaging protocols
NCCT and CTA were performed using a 64-section CT scanner (Brilliance 64; Philips Healthcare) with the following parameters: tube voltage120 kV, tube current 333 mA, and rotation speed 0.75 s; or using a 640-section CT scanner (United Imaging Healthcare, Shanghai, China) with the following parameters: tube voltage 120 kVp, tube current 300 mAs, rotation speed 0.5 s. The intravenous contrast agent (60–100 mL of iopromide, 370 mg iodine per milliliter; Ultravist 370; Bayer) was injected at a flow rate of 4 mL/s. Scanning was started when the CT attenuation in the ascending aorta reached 120 HU. Volume rendering and full-slab MIP image processing were performed at a multimodality workstation (Philips Medical Systems; Philips Healthcare) using the 0.67 mm axial sections and were reconstructed at an increment of 0.8 mm. The acquired axial slice CT angiograms were transferred to a dedicated workstation for TS-MIP construction (SyngoXWP; Siemens Healthcare). After the 0.67 mm axial images were loaded into the InSpace section at the workstation, the DSA Layout application (Siemens Healthcare) was used, and coronal MIP was applied for image reconstruction with a 10-mm-thick slab.
Endovascular procedure
Angiography and thrombectomy were performed with a digital angiography unit (Artis zee; Siemens, Healthineers, Erlangen, Germany). The SWIM technique (Solitaire stent retriever in combination with the intracranial support catheter aspiration for mechanical thrombectomy) was used as the first line technique. 13 After a 90 cm sheath (Terumo, Tokyo, Japan) was introduced into the cervical segment of the internal carotid artery (ICA) on the affected side, an aspiration catheter (Penumbra, Alameda, CA, USA) was subsequently advanced into the distal ICA with a microcatheter over a microwire. Next, the microcatheter was placed distal to the thrombus, and the stent retriever (Solitaire, ev3 Neurovascular, Covidien, Irvine, CA, USA) was deployed through the thrombus according to the diameter of the occluded artery (usually 6 × 30 mm for the ICA terminus or proximal M1 and 4 × 20 mm for the distal M1 or M2); 3–5 min were allowed for clot integration. With use of stent retriever as an anchor, aspiration catheter was maneuvered up to a position just proximal to the stent to simultaneously increase the effect of aspiration. The stent retriever was withdrawn into the aspiration catheter and completely removed from the entire system. Subsequently, the aspiration catheter was removed under continuous aspiration and proximal flow arrest to ensure aspiration of potential microfragments of the thrombus. The retrieval attempt was repeated up to three times per target artery. In the case of SWIM technique failure, single catheter aspiration or stent thrombectomy were performed. Moreover, if ICAS induced stenosis was confirmed during thrombectomy and re-occlusion occurred, salvage balloon dilation or stent insertion was performed. Antegrade reperfusion was assessed using the modified Thrombolysis in Cerebral Infarction (mTICI) grade. Successful reperfusion was defined as mTICI 2b-3 after endovascular treatment.
Imaging analysis
We assessed the collateral score, thrombus enhancement grade, clot burden score (CBS), thrombus density, thrombus perviousness, the distance from terminus of intracranial carotid artery to the thrombus (DT) and thrombus length. These characteristics were assessed on pre-IVT NCCT and/or CTA imaging.
The collateral circulation was evaluated on baseline CTA raw data and MIP imaging on a scale of 0–3 according to Tan et al. 14 TES was defined as the contrast agent penetrating partially or completely into the occlude thrombus or the space between the thrombus and arterial wall to make the thrombus itself visible or discriminable on TS-MIP reconstruction of CTA imaging according to our previous experience. 11 The CBS is a 10-point score that assesses the thrombus extension in the anterior circulation according to Puetz et al. 15 Thrombus density was computed as the mean Hounsfield Units (HU) value of the manually placed three spherical regions of interests of 1 mm radius within the thrombus (proximal, middle, and distal) on NCCT. Thrombus perviousness was calculated by subtracting the mean HU value of the regions of interest on NCCT from CTA. The DT was defined as the length from the ICA-T to the most proximal thrombus border and the DT was set to zero if the thrombus proximal border was located more proximal than the ICA-T. Thrombus length was defined as the largest extension of lack of contrast filling shown on CTA, aided by the hyperdense artery sign if observed on NCCT. Two perpendicular planes on multiplanar reformatted CTA images were used to determine the ICA-T, the point of contrast truncation and the distal point of vascular refilling. In the case of bifurcating thrombi, the occluded branch with the longest thrombus was recorded.
All measurements were performed by two trained observers: GCH and YG who were blinded to the clinical information and angiographic results of the patients. Disagreements of TES and CBS were settled by joint discussion, whereas in the measurement of thrombus density, thrombus perviousness, DT, and thrombus length, the average value of the two readers was recorded. The inter-reader agreement was assessed.
Histological analysis
The retrieved thrombus was formalin-fixed, paraffin-embedded, and subsequently sectioned into 4-μm-thick slices that were stained with hematoxylin and eosin. The tissue sections were scanned with a high-definition scanner (Panoramic MIDI; 3DHISTECH). Three areas in each image were randomly selected and photographed at 340× magnification using image review software (Case Viewer; 3DHISTECH). Quantification was manually performed using Orbit Image Analysis Software (Orbit Image Analysis, Idorsia Ltd). The proportions of the red blood cell (RBC), fibrin/platelet (F/P), and white blood cell (WBC) were expressed as a percentage of the respective area. Mean values of the quantitative parameters measured in three areas of each sample were used for analysis.
Statistical analysis
Data were presented as a mean ± standard deviation, median and interquartile range (IQR), or number and proportion (%), as appropriate. Comparison between groups was done using the Mann-Whitney U test for continuous variables and χ2and Fisher’s exact test for categorical variables. The reproducibility of measurements between two observers was quantitatively measured by the intraclass correlation coefficient (ICC). Univariate and multivariate logistic regression analysis was performed to identify the independent factors associated with the incidence of DE. Variables with p values < 0.1 in univariate analysis were included in a final multivariate logistic regression model. All analyses were conducted using open-source R software (version 4.0.3; R Foundation). A two-sided p < 0.05 was considered statistically significant.
Results
Patients and occurrence of distal embolization
Between January 2020 to December 2023, 379 consecutive patients presented with acute neurological symptoms attributable to LVO and underwent thrombectomy at our comprehensive stroke center. We excluded patients lacking pre-procedure CTA or with suboptimal scans (n = 24), those whose clots were inadequately retrieved or improperly processed (n = 38), and patients who experienced no or minimal reperfusion after the final thrombectomy. The latter group was excluded because the occurrence of DE in downstream territory cannot be reliably assessed in the presence of an occluded and non-revascularized artery on DSA (n = 9). Consequently, 308 patients qualified for this study (Figure 1). Of these, 91 patients (29.5%) overlapped with cohorts from previous studies.

Flow diagram of the current study. CT: CT angiography; EVT: endovascular thrombectomy; NCCT: non-contrast CT; TES: thrombus enhancement sign.
Among the qualified patients, 21.1% (65/308) experienced DE. Patients with DE had significantly higher puncture to recanalization time (median 58 min IQR [44–80 min] vs 46 min IQR [33–55 min], p = 0.002), required a higher number of thrombectomy passes (median 2 [2–3] vs 2 [1–3], p = 0.040), had a lower frequency of successful reperfusion (86% [58/65] vs 95% [231/243], p = 0.022; Table 1). Additionally, they were less likely to achieve functional independence (31% [20/65] vs 45% [109/243]; p = 0.041; Figure 2). No significant differences were found between the two groups in terms of other demographics, cerebrovascular risk factors, stroke etiology, or use of intravenous thrombolysis.
Comparison between patients with and without distal embolization.

Data are expressed as n (%) or median and interquartile range as appropriate. ASPECTS: Alberta Stroke Program Early CT Score; DE: distal embolization; ICA: internal carotid artery; IVT: intravenous thrombolysis; MCA: middle cerebral artery; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction; NIHSS: National Institutes of Health Stroke Scale; sICH: symptomatic intracerebral haemorrhage; TES: thrombus enhancement sign.
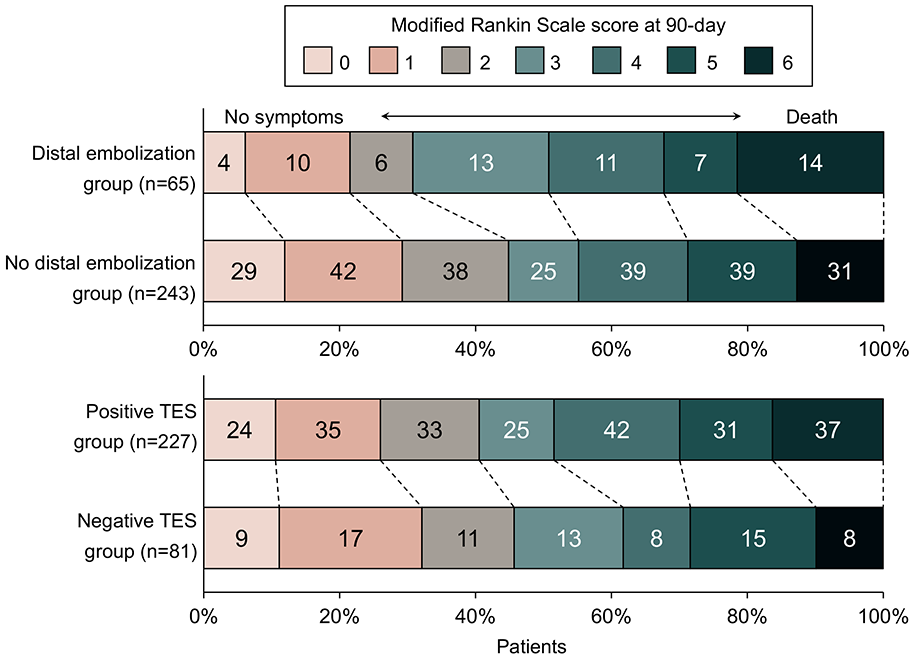
Distribution of modified Rankin Scale score at 90 days.
TES and thrombus characteristics
TES was detected in 73.7% (227 of 308) of cases using TS-MIP on CTA. Patients with positive TES had significantly higher collateral score (median 2 IQR [1–2] vs 1 IQR [1–2], p = 0.011) compared to those with negative TES. Atrial fibrillation was more frequent in patients with positive TES (62% [141/227]) than in patients with negative TES (33% [27/81], p < 0.001). The TES-positive group was significantly associated with a cardioembolic origin (61% [138/227] vs 27% [22/81], p < 0.001), whereas the TES-negative group had more patients with large artery atherosclerosis origin (10% [23/227] vs 57% [46/81], p < 0.001; Table 2 and Figure 3). No significant differences were observed between the groups in terms of thrombus density (median 47 IQR [42–49] vs 45 IQR [41–49], p = 0.163) or perviousness (median 15 IQR [9.6–21] vs 14 IQR [8.9–20], p = 0.272). Histopathological examination revealed that TES-positive thrombi had a higher F/P proportion (median 51% IQR [46%–58%] vs 48% IQR [43%-54%], p = 0.009), a lower RBC proportion (median 42% IQR [35%–47%] vs 44%, p = 0.043) and a similar WBC proportion (median 7.4% IQR [4.5%–9.5%] vs 6.9% IQR [4.3%–9.3%], p = 0.300) compared to TES-negative thrombi (Figure 4).
Comparison between patients with and without thrombus enhancement sign.

Data are expressed as n (%) or median and interquartile range as appropriate. ASPECTS: Alberta Stroke Program Early CT Score; ICA: internal carotid artery; IVT: intravenous thrombolysis; MCA: middle cerebral artery; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction; NIHSS: National Institutes of Health Stroke Scale; sICH: symptomatic intracerebral hemorrhage; TES: thrombus enhancement sign.
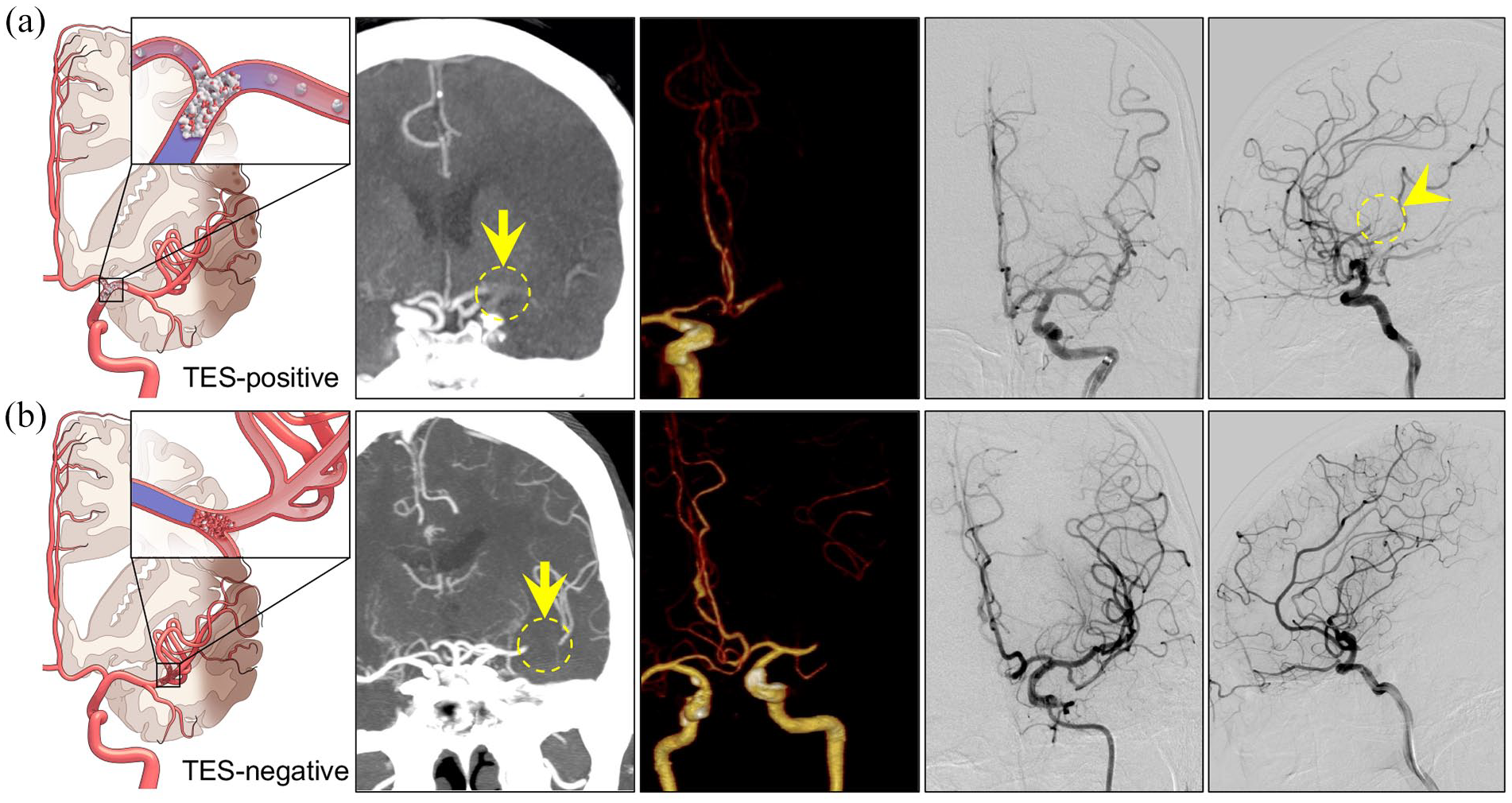
Example of the presence and absence of thrombus enhancement sign (TES) in patients with acute intracranial large vessel occlusion. (a) TES was clearly displaced on the thin-slab maximum intensity projection (TS-MIP) image (arrow) and was not detected on volume rendering (VR) image. Catheterized angiography after the final attempt of thrombectomy showed distal embolization in the downstream territory of the middle cerebral artery (arrowhead). (b) TES was not detected on TS-MIP (arrow), or VR images; catheterized angiography after a single attempt of thrombectomy revealed complete recanalization of the middle cerebral artery.
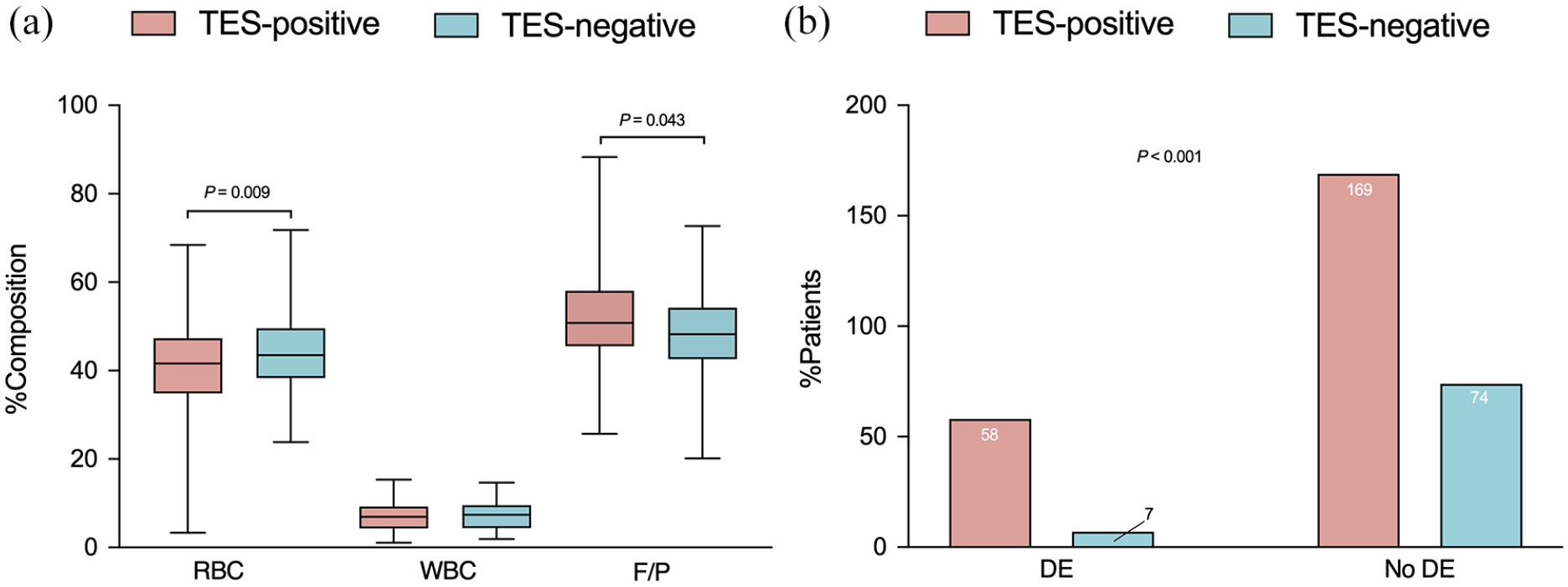
Comparisons of thrombus compositions (A) and occurrence of distal embolization in TES-positive and TES-negative patients.
For thrombus imaging characteristics, comparisons between the patients with DE and those without DE are shown in Table 3. Patients with confirmed DE had a significantly higher frequency of TES than those without DE (89% [58/65] vs 70% [169/243], p = 0.001). We also observed We observed a trend toward a shorter distance from the terminus of the intracranial carotid artery to the thrombus in patients with DE (median distance 9 mm, IQR [0–16 mm] vs 13 mm, IQR [0–20 mm] in patients without DE). However, this difference did not achieve statistical significance (p = 0.067). Other thrombus characteristics, such as length, density, perviousness, and clot burden score, did not show significant differences. Importantly, thrombi associated with DE had a lower RBC proportion (median 40% IQR [34%–44%] vs 43% IQR [36%-49%], p = 0.023), a higher F/P proportion (median 53% IQR [49%–59%] vs 49% IQR [43%–56%], p = 0.026) and a similar WBC proportion (median 7.6% IQR [5.4%–9.4%] vs 6.9% IQR [4.3%–9.3%], p = 0.339) compared to thrombi without DE.
Thrombus imaging characteristics for distal embolization patients at baseline CT imaging.

CI: confidence interval; DT: distance from terminus of intracranial carotid artery to the thrombus; F/P: fibrin/platelet; RBC: red blood cell; TES: thrombus enhancement sign; WBC: white blood cell.
The two readers demonstrated good interobserver agreement for measuring TES presence (ICC = 0.91, 95% CI 0.77–0.96), collateral score (ICC = 0.83, 95% CI 0.66–0.95), CBS (ICC = 0.79, 95% CI 0.60–0.92), thrombus density (ICC = 0.88, 95% CI 0.69–0.91), thrombus perviousness (ICC = 0.81, 95% CI 0.61–0.96), DT (ICC = 0.87, 95% CI 0.70–0.93) and thrombus length (ICC = 0.84, 95% CI 0.62–0.91).
Prediction of distal embolization
In the univariate analysis, the number of thrombectomy passes, puncture to recanalization time, successful reperfusion (mTICI 2b-3), TES and thrombus length significantly impacted DE. The multivariate analysis revealed that TES (odds ratio [OR], 2.76; 95% confidence interval [CI], 1.23–7.10; p = 0.022) was the only independent predictor of DE (Table 4). TES as a predictor of DE yielded a sensitivity of 0.89 (95% CI 0.82–0.97) and a specificity of 0.31 (95% CI 0.25–0.36). The positive predictive value was 0.26 (95% CI 0.20–0.31), the negative predictive value was 0.91 (95% CI 0.85–0.98), and the area under the curve (AUC) was 0.60 (95% CI 0.64–0.89; p < 0.001).
Logistic regression for distal embolization.

CI: confidence interval; DT: distance from terminus of intracranial carotid artery to the thrombus; IVT: intravenous thrombolysis; TES: thrombus enhancement sign.
Discussion
In this retrospective study, distal embolization occurred in 21.1% of cases. Patients with DE experienced longer recanalization times, lower rates of achieving successful reperfusion, and a decreased frequency of functional independence. Analysis of thrombi on CT imaging and histopathology revealed a strong correlation between TES and F/P-rich composition of the thrombus. Multivariate analysis demonstrated that TES was the only independent predictors of DE. Our findings highlight the significance of early identification of DE and suggest that TES could serve as a potential surrogate for assessing thrombectomy efficacy and predicting a reduced rate of full recanalization.
Stent retrieval and contact aspiration are two primary thrombectomy techniques commonly utilized in clinical settings.16,17 In the stent retrieval technique, a microcatheter is advanced beyond the thrombus to deploy the stent retriever. Accidental penetration of the thrombus by the microcatheter can increase the risk of thrombus fragmentation and migration. Once the thrombus is successfully engaged at the tip of the aspiration catheter, negative pressure suction is applied, which can lead to excessive elongation of the thrombus. This increased surface area of interaction between the thrombus and vessel wall may predispose to thrombus fragmentation. 18 Additionally, the abrupt decrease in suction force following thrombus engagement can generate shearing forces, causing the thrombus to fracture during contact aspiration or extend into multiple branches that may dislodge during thrombectomy. 19 Some studies suggested that applying additional proximal suction through a balloon guiding catheter (BGC) to temporarily halt arterial flow and decrease in the pressure gradient across the thrombus may reduce DE. However, this method is limited by the challenges of successful placing a BGC and may not be very effective in cases of medium vessel occlusion. 20
Thrombus imaging to predict the efficacy of recanalization therapy for acute LVO has been performed in a small number of studies.10,21,22 However, literature on the association between the occlusive lesion morphology on baseline imaging and EVT in relation to the occurrence of DE is scarce. Some studies have evaluated thrombus location, length, perviousness, and the incidence of DE during EVT, but findings are conflicting and with poor specificity and accuracy. 10 Certain imaging biomarkers, such as hyperdense artery sign, susceptibility vessel sign, and clot protrusion sign have been suggested to be associated with procedural distal emboli.23 –25 However, the diagnostic and prognostic significance of these signs was not firmly established. Our recent findings indicate that the TES can be consistently and accurately observed in the anterior circulation on TS-MIP CTA, and is significantly associated with first-pass failure of stent retriever thrombectomy. 12 In the present study, TES was identified in 73.7% (227 of 308) of enrolled patients and was accompanied by a higher prevalence of atrial fibrillation and cardioembolic stroke. This finding aligns with existing literature, which suggests that cardioembolic thrombi often present with a large thrombus burden, leading to a lower rate of complete recanalization, and an increased risk of thrombus fragmentation.26 –28 TES-positive thrombi usually exhibit a toughened texture and an irregular morphological shape, with little deformation in the artery. This allows the contrast agent to penetrate and delineate the thrombus profile and the length of the occluded vessel. Cardioembolic thrombi with a positive TES are typical older and less amenable, complicating their capture by stent retriever, which reduce the device’s effectiveness and increase the likelihood of secondary embolization events.
A comparative investigation was further conducted to analyze the histopathologic characteristics of the retrieved thrombi. It was observed that thrombi in distal embolization cases often displayed irregular boundaries, corrugation, and pores. These pathological features support the notion that the presence of pores or gaps facilitates the infiltration of contrast agents. Additionally, the histological examination of thrombi revealed that TES-positive thrombi had a higher proportion of fibrin/platelets (median 51% vs 48%, p = 0.009) and a lower proportion of RBC (median 42% vs 44%, p = 0.043) compared to TES-negative thrombi. Studies have shown that clots rich in RBCs are more easily recanalized than those rich in fibrin, possibly due to the greater deformability of RBC-rich thrombi, allowing stent struts to penetrate them more easily and optimize the retrieval process. 29 Furthermore, this study found that thrombi prone to distal embolization contained fewer RBC (40% vs 43%, p = 0.023) and more F/P (53% vs 49%, p = 0.026), consistent with previous research. 30 Fibrin/platelet-rich thrombi are harder and possess a higher static coefficient of friction.31,32 The higher friction may render them more challenging to remove during EVT, whereas multiple and prolonged thrombectomy procedures can easily cause the rupture and dislodgement of fragile thrombi, leading to DE.32,33 Thus, TES could serve as a reliable predictor for identifying high migration risk thrombi, providing important information about the requirement for additional thrombectomy attempts or maneuvers, or the use of proximal protection devices.
Previously published data suggest that the presence of DE during EVT for target LVO is associated with worse functional outcomes.3,34 The present study reported an incidence of DE at 21.1%, which is lower than that reported by Pilato et al. 8 (28.4%), Wong et al. 4 (42%) or Bala et al. 10 (50.6%). This difference may be due to the inclusion of patients from both anterior and posterior circulations in the previous studies, while our study focused solely on patients with anterior circulation. For analysis purpose, we excluded patients with unsuccessful reperfusion. Since the most treatment resistant thrombi, which are mostly F/P rich thrombi that are likely to cause DE, might not be available to provide a sufficient clot, the results are likely to underestimate the incidence of DE. Furthermore, thrombus length was identified as a potential predictor of DE. Our study showed that the lower rate of DE could be attributed to a shorter thrombus length (median 14 mm vs 28.5 mm in the Wong study).
Previous studies have established a link between DE and poorer clinical outcomes in patients with LVO undergoing thrombectomy. Our research extends this understanding by exploring the mechanisms connecting DE to adverse outcomes and identifying potential predictive imaging biomarkers. We found that DE correlates with specific thrombus characteristics, such as increased fibrin and platelet content, and procedural challenges, including more thrombectomy passes and longer recanalization times. These factors likely contribute to the detrimental impact of DE. Additionally, our study highlights the potential of the TES as a preprocedural biomarker for DE risk assessment. While the association between DE and poor outcomes is known, 10 our integrated analysis of imaging, histopathology, and procedural factors offers a more comprehensive understanding of DE’s pathophysiology, potentially guiding future therapeutic strategies.
In this case series, we report a cardioembolism rate of 55%, which exceeds previously reported rates in East Asian populations.35,36 Several factors may contribute to this observation. First, advancements in medical devices have enabled the treatment of an increasing number of M2 distal occlusions with thrombectomy at our center, and most of these cases are cardioembolization. Second, as a comprehensive stroke center, we may receive a higher proportion of cardioembolic cases, while cases of ICAS, which often present with milder symptoms, are less frequently referred to us for thrombectomy. Additionally, being a single-center retrospective study, selection bias is unavoidable. Consequently, the higher proportion of cardioembolic cases in present study might enhance the predictive value of TES as an imaging biomarker. This deficiency should be carefully considered in future clinical applications.
This study has several limitations. First, it was a single-center retrospective, observational study. While we controlled for measured covariates, there may be important unmeasured confounders. Second, we did not evaluate distal embolization in the follow-up imaging. Angiography is less sensitive in detecting small distal emboli and may not always reliably distinguish whether a vessel cutoff reflects a distal embolus, competitive hemodynamic flow patterns, or vasospasm. Third, the visualization of TES may be affected by image contrast enhancement and brightness adjustments, as well as potential misinterpretation as partial vessel occlusions. These factors could undermine the accuracy and consistency of TES assessments. Although our study demonstrated strong interobserver agreement between two independent readers in identifying TES presence (ICC = 0.91), greater variability might arise in multi-reader scenarios, potentially reducing reliability. 11 Fourth, thrombi are known to have heterogeneous structures, and the analysis of retrieved fragments may not fully reflect their overall composition. Repeated retrieval attempts can exacerbate this issue by increasing the risk of thrombus fragmentation, potentially altering its composition and making the analyzed samples less representative of the original clot. Despite using standardized sampling protocols to minimize bias, the sampling error remains a limitation of this study. Finally, although TES exhibits high sensitivity (89%) for predicting distal embolization, its low specificity (31%) restricts its effectiveness as a standalone predictor. Therefore, TES should be used alongside other imaging or clinical biomarkers to improve diagnostic accuracy. A strength of our study is the comparably large consecutive cohort with a homogenous histopathologic and imaging workup. To translate our findings into clinical practice, the results should be confirmed by prospective multicenter studies that account for different thrombectomy methods, such as the use of a balloon guiding catheter.
Conclusion
In conclusion, our study demonstrates that DE is a prevalent complication of endovascular therapy, which impedes full recanalization and negatively affects patient outcomes. The presence of TES on baseline CT imaging is associated with a high fibrin/platelet composition, suggesting increased thrombus fragility and could serve as a reliable radiologic predictor of DE. Further large-scale prospective studies are warranted to confirm our findings.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: (1) Shanghai Municipal Health Commission Clinical Research Project (No. 202140066). (2) New interdisciplinary research Project of Shanghai Municipal Health Commission (No. 2022JC022).
Informed consent
Informed consent was provided by patients or their family members.
Ethical approval
The ethics committee of Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine approved this study (No 2018-002-1).
Guarantor
Yi Gu and Yueqi Zhu
Contributorship
1) Guangchen He, Wenli Zhou: Acquisition of data, analysis and interpretation of data and drafting of the manuscript.
2) Yi Gu, Yueqi Zhu: Study concept and design, critical revision of the manuscript for important intellectual content, and study supervision.
3) Haitao Lu: acquisition of data, revision of the manuscript.
4) Guangchen He: statistical analysis.
Data availability
This study’s data are available from the corresponding author upon reasonable request.
