Abstract
Background:
Thrombus enhancement sign (TES) is associated with cardioembolic stroke and first-pass angiographic failure in anterior ischemic stroke. However, the relationship between TES and stroke subtype and recanalization status after endovascular treatment (EVT) in basilar artery occlusion (BAO) remains unknown.
Methods:
This retrospective study included consecutive patients with acute BAO who underwent EVT between January 2020 and September 2023. Each patient underwent baseline non-contrast computed tomography (CT) and CT angiography. Two independent readers assessed the presence of TES. Stroke types were classified according to the Trial of ORG 10172 for Acute Stroke Treatment. Successful recanalization was defined as a modified Thrombolysis in Cerebral Infarction score of 2b–3 after EVT. Clinical and interventional parameters, along with histopathological thrombi examination results, were compared between the TES-positive and TES-negative groups. The associations between TES and stroke subtype and recanalization status were analyzed using univariate and multivariate analyses.
Results:
A total of 151 patients were included in the analysis, among whom 116 (77%) exhibited TES. TES showed a significant correlation with cardioembolic and cryptogenic strokes (odds ratio [OR]: 8.56; 95% confidence interval: 3.49–22.4; p < 0.001), whereas the TES-positive thrombi were characterized by a higher fibrin/platelet proportion (p = 0.002) and lower erythrocyte proportion (p = 0.044). The TES-positive group demonstrated favorable outcomes compared to the TES-negative group, including a shorter procedure time (p < 0.001), lower number of thrombectomy attempts (p = 0.010), higher incidence of first pass success (p = 0.022), and lower rate of requiring rescue angioplasty and/or stenting (p < 0.001). In multivariate analysis, TES remained independently associated with successful recanalization (OR: 9.63; 95% CI: 2.33, 47.7; p = 0.003) after adjusting for baseline confounders.
Conclusions:
Visualization of TES serves as a reliable and easily accessible marker for identifying cardioembolic and cryptogenic strokes and predicting recanalization success in thrombectomy for basilar artery occlusion.
Introduction
Basilar artery occlusion (BAO) accounts for approximately 1% of all ischemic strokes and 10% of strokes due to large vessel occlusions (LVOs) with high mortality and morbidity rates without treatment.1,2 Although endovascular treatment (EVT) has become a standard approach for treating patients with LVO in the anterior circulation, limited and partially contradicting evidence was reported regarding BAO until recently.3,4 The ATTENTION (Endovascular Treatment for Acute Basilar Artery Occlusion) and BAOCHE (Basilar Artery Occlusion Chinese Endovascular Trial) trials indicated improved outcomes for BAO after EVT within 24 h of onset, with comparable treatment effects to those observed for LVO of the anterior circulation.5–7 However, the outcome of patients with BAO exhibits a considerable range of variability and heterogeneity, the influencing factors of which are not yet entirely understood.8,9
Recanalization is known to be a strong predictor of clinical outcome and infarct volume after EVT.10–12 Different pathologic mechanisms have been previously reported for BAO associated with successful recanalization.13,14 In cases of acute BAO caused by cardiac embolus, a simple thrombectomy is typically sufficient for successful retrieval of the thrombi. In contrast, large-artery atherosclerosis, which comprises a large proportion of local thrombosis due to underlying basilar stenosis (BS), commonly requires rescue therapy. Because angioplasty or stenting for underlying BS would affect perforating basilar arteries with worse clinical outcome due to ischemic lesions, there is a trend toward higher rates of futile recanalization and poorer clinical outcomes for the subtype of in situ atherosclerotic thrombosis.15,16 A better understanding of the mechanisms might help in planning therapeutic strategies, while identifying such cases at an early stage may be beneficial.
Computed tomography angiography (CTA) has been shown to be highly accurate in detecting acute BAO. 17 The thrombus enhancement sign (TES) visualized with thin slab maximum intensity projection (TS-MIP) image reconstruction of CTA has been proposed as a new imaging biomarker to predict stroke subtype and thrombus compositions in anterior circulation. 18 Considering the mechanism of TES formation, TES would be more pronounced in BAO for the following reasons: (a) the confluence of blood flow from bilateral vertebral arteries and potential improvements in the collateral connections of the vertebrobasilar system (e.g. a larger posterior communicating artery with an existing P1) enable more penetration of the contrast agent to outline the profile of the thrombus; and (b) the vertebrobasilar system exhibits elevated vascular tortuosity and diminished arterial diameter, which restricts the adaptive deformation of thrombus within the vessel lumen. This could result in the presence of additional gaps between the thrombus and the vessel wall, allowing for the penetration of contrast agents.
In this study, we aimed to investigate whether the presence of TES is affected by the underlying pathologic mechanism of BAO, thereby enabling the prediction of stroke subtype. We also evaluated the clinical significance of TES for the recanalization status in patients with acute BAO treated by EVT.
Methods
Study participants
This single-center retrospective study was conducted in accordance with the recommendations of the Declaration of Helsinki and was approved by the institutional review board. Patients who presented within 24 h of acute neurological symptoms attributable to BAO and underwent thrombectomy between January 2020 and September 2023 were consecutively included. These patients underwent non-contrast CT (NCCT) and CTA scans before receiving endovascular reperfusion therapy. Cases were excluded if NCCT or CTA was unavailable or if images were of insufficient quality. Cases of BAO with other causes, such as arterial dissection, or vasculitis, were also excluded. Data regarding patient demographics, imaging findings, time intervals, and angiographic and clinical outcomes were collected. The clinical outcome was assessed using the modified Rankin scale (mRS) score at 90 days.
Imaging Protocol
NCCT and CTA were performed using a 64-section CT scanner (Brilliance 64; Philips Healthcare, Best, Noord-Holland, the Nederland) with the following parameters: tube voltage, 120 kV; tube current, 333 mA; and rotation speed, 0.75 s or using a 640-section CT scanner (United Imaging Healthcare, Shanghai, China) with the following parameters: tube voltage, 120 kVp; tube current, 300 mAs; and rotation speed, 0.5 s. The intravenous contrast agent (60–100 mL of iopromide, 370 mg iodine per mL) (Ultravist 370; Bayer) was injected at a flow rate of 4 mL/s. Scanning was started when the CT attenuation in the ascending aorta reached 120 HU. Volume rendering and full-slab MIP image processing were performed at a multimodality workstation (Philips Medical Systems; Philips Healthcare) using 0.67 mm axial sections and were reconstructed at an increment of 0.8 mm. The acquired axial slice CT images were transferred to a dedicated workstation for TS-MIP construction (SyngoXWP; Siemens Healthcare). After the 0.67 mm axial images were loaded into the InSpace section at the workstation, the DSA Layout application (Siemens Healthcare) was used, and coronal MIP was applied for image reconstruction with a 10 mm-thick slab.
Endovascular treatment
Patients received either bridging therapy (EVT combined with IVT) or EVT alone. IVT was administered within a maximum of 4.5 h from symptom onset, according to the current guidelines. If a BAO corresponding to stroke symptoms was identified on CTA, the patient was brought to the angiography suite for EVT. A stent retriever (with a Solitaire stent system [Covidien, Irvine, CA, USA]) and/or a direct aspiration first-pass technique (with a Penumbra aspiration catheter [Penumbra, Alameda, CA, USA]) was used as the first-line EVT. If underlying basilar artery in situ atherosclerosis was suspected during thrombectomy and re-occlusion occurred, rescue treatment of balloon dilation or stent insertion was performed. Recanalization status was assessed on the final angiogram and was classified according to the modified thrombolysis in cerebral infarction (mTICI) scale, with successful recanalization defined as mTICI grade 2b–3. 19 A first pass effect (FPE) was defined as achieving complete recanalization (mTICI 3) with a single thrombectomy device pass. 20
Image analyses
Two interventional neuroradiologists who were blinded to the clinical information and the angiographic procedure results independently evaluated and determined the occurrence of TES using source axial section images and reconstructed volume rendering, MIP, and TS-MIP images. TES was defined as the contrast agent penetrating partially or completely into the thrombus or the space between the thrombus and arterial wall to make the thrombus visible or discriminable on CTA imaging according to our previous experience.18,21 The reviewers were allowed to adjust, rotate, and reformat the three-dimensional images and adjust the window setting or zoom in to optimally view the presence and location of the TES. In cases of disagreement between the readers, a third neuroradiologist joined, and a consensus was reached by the three readers in an additional joint review session that occurred less than 2 weeks after the individual reading sessions.
Stroke subtypes and histopathologic analyses
Stroke subtypes were determined according to the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) classification based on the patients’ medical histories, electrocardiograms, emergency laboratory reports, CT angiograms, and intraprocedure digital subtraction angiograms. 22 The stroke subtypes were then further determined using magnetic resonance imaging (MRI), duplex sonography of the cervical arteries, coagulation tests, long-term electrocardiography, and transthoracic or transesophageal echocardiography.
For histopathological analysis, the successfully retrieved thrombus was immersion fixed in 4% buffered formalin over 24 h, passed through a series of graded ethanol solutions (range: 70%–100%), and embedded in paraffin. The paraffin-embedded samples were sliced to a thickness of 4 μm and stained with hematoxylin–eosin, and the tissue sections were scanned with a high-definition scanner (Panoramic MIDI; 3DHISTECH). Three areas in each hematoxylin–eosin-stained image were randomly selected and photographed (original magnification: 340×) using image review software (Case Viewer; 3DHISTECH). Quantification and analyses of the erythrocyte, fibrin/platelet (F/P), and leukocyte proportions were manually performed using Orbit Image Analysis Software (www.Orbit.bio) by an experienced pathologist who was blinded to the imaging data and angiographic outcomes. The proportions of each component are expressed as a percentage of the respective areas. All chosen images were loaded into the software and split into three channels (red, green, and blue). The blue channels were used to measure the erythrocyte proportion, the inversion mode of the blue channel was used to measure the F/P proportion, and the red channel was used to measure the leukocyte proportion. After the threshold was set, the particles were analyzed automatically (areas covered by the respective cells [%] were measured). The mean values of the quantitative parameters measured in three areas of each hematoxylin–eosin-stained sample were used for analysis.
Statistical analysis
Data are presented as the mean ± standard deviation, median (interquartile range), or number and frequencies, as appropriate. The Kolmogorov–Smirnov test was used to verify the assumption of a normal distribution. Clinical and interventional parameters, along with histopathological thrombi examination results, were compared between the TES-positive and TES-negative groups using a χ2 test (for categorical variables) and Student’s t-test or Mann–Whitney U test (for noncategorical variables with or without normal distribution, respectively). Logistic regression analysis was performed to evaluate the independent variables for successful recanalization separately. All clinical and procedural variables with P-values < 0.1 in univariate analysis were included in a final multivariate logistic regression model. Inter-reader agreement for the presence or absence of TES was assessed using the Fleiss κ statistics, with a κ of 0.81–1.00 corresponding to excellent agreement; a κ of 0.61–0.80 corresponding to substantial agreement; a κ of 0.41–0.60 corresponding to moderate agreement; and a κ < 0.40 corresponding to poor agreement. All analyses were conducted using open-source R software (version 4.0.3; R Foundation for Statistical Computing, Vienna, Austria). Two-sided p-values < 0.05 were considered statistically significant.
Results
TES detection and patient characteristics
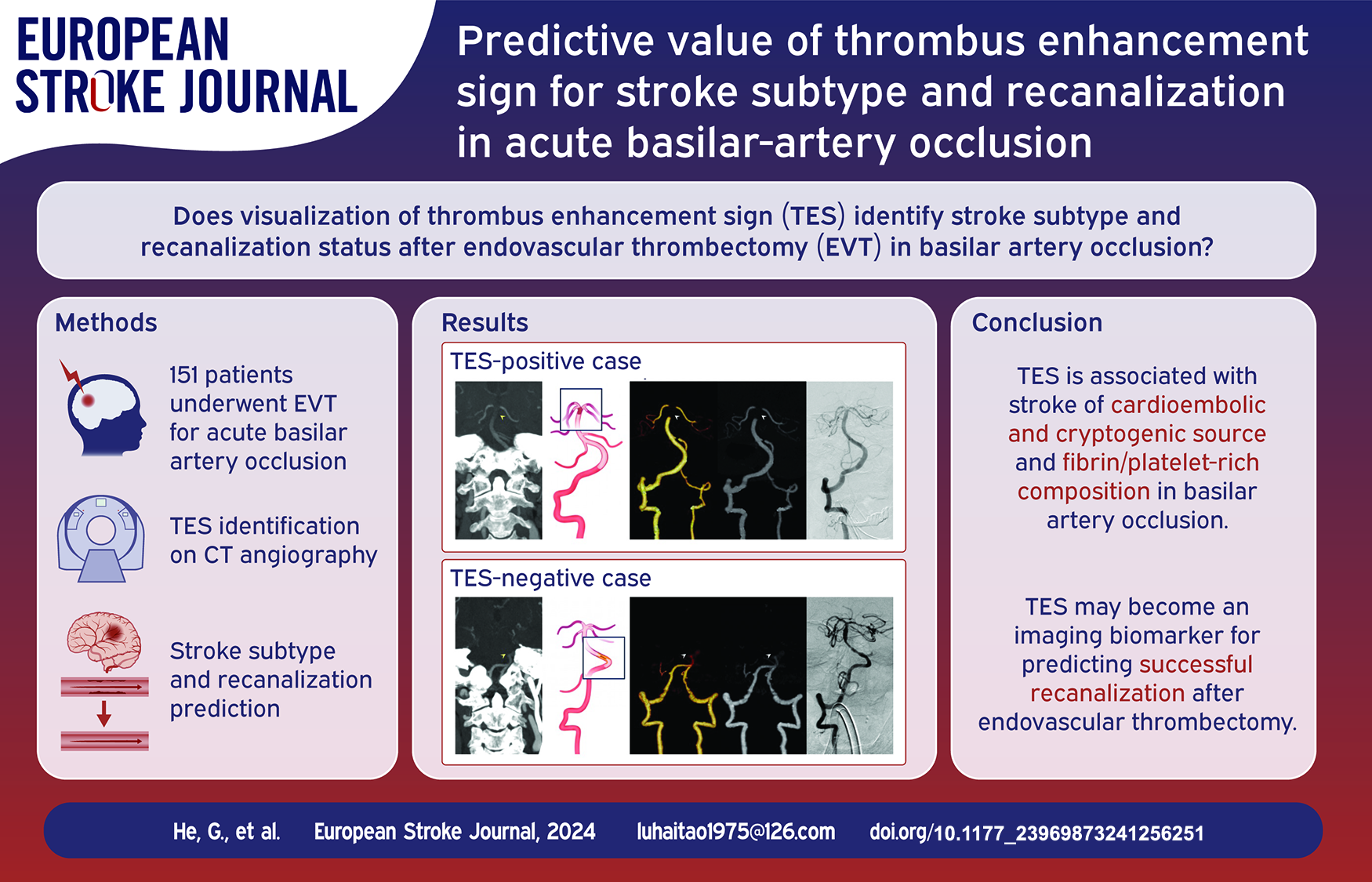
During the period from January 2020 to September 2023, 176 consecutive patients presented with acute neurological symptoms attributable to BAO and underwent thrombectomy at our comprehensive stroke center. After the exclusion of patients without pre-procedure CTA or with a suboptimal scan (n = 23) and those with stroke caused by other determined factors, such as dissection (n = 2), 151 patients qualified for this study (Figure 1). Among the included patients, TES was detected in 79% (119 of 151) of patients by reader 1 and in 72% (109 of 151) of patients by reader 2. After a consensus was reached on disagreements, TES was observed in 77% of enrolled patients (116 of 151). The two readers had excellent interobserver agreement for measuring the presence of TES (κ = 0.82, 95% CI: 0.72–0.93).
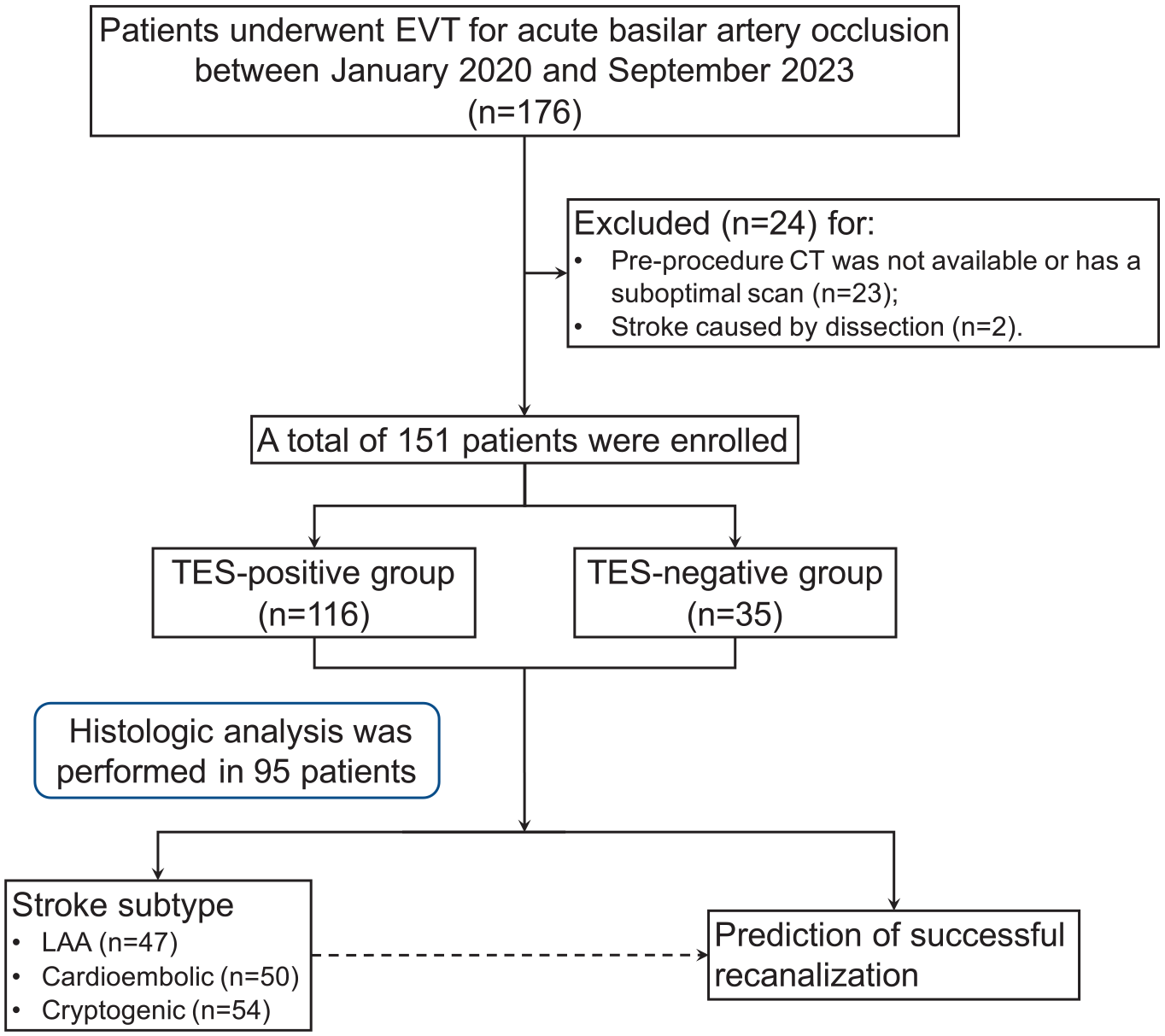
Flow diagram of the current study. EVT: Endovascular thrombectomy; LAA: Large-artery atherosclerotic stroke; TES: Thrombus enhancement sign.
On the basis of the presence of TES, 151 patients were divided into the TES-positive group (n = 116; median age: 72 years, interquartile range [IQR]: 65–80 years; 63 men [54%]) and the TES-negative group (n = 35; median age: 74 years, IQR: 68–79 years; 21 men [60%]). The baseline characteristics of all patients are summarized in Table 1. Atrial fibrillation was found in 43% (50 of 116) of patients in the TES-positive group and in 14% (5 of 35) of patients in the TES-negative group (p = 0.002). The TES-positive group exhibited a higher prevalence of distal BA occlusions (54% vs 29%) and a lower prevalence of proximal BA occlusions (22% vs 43%) compared to the TES-negative group (p = 0.014) (Figure 2).
Comparison between the TES-positive group and TES-negative group.
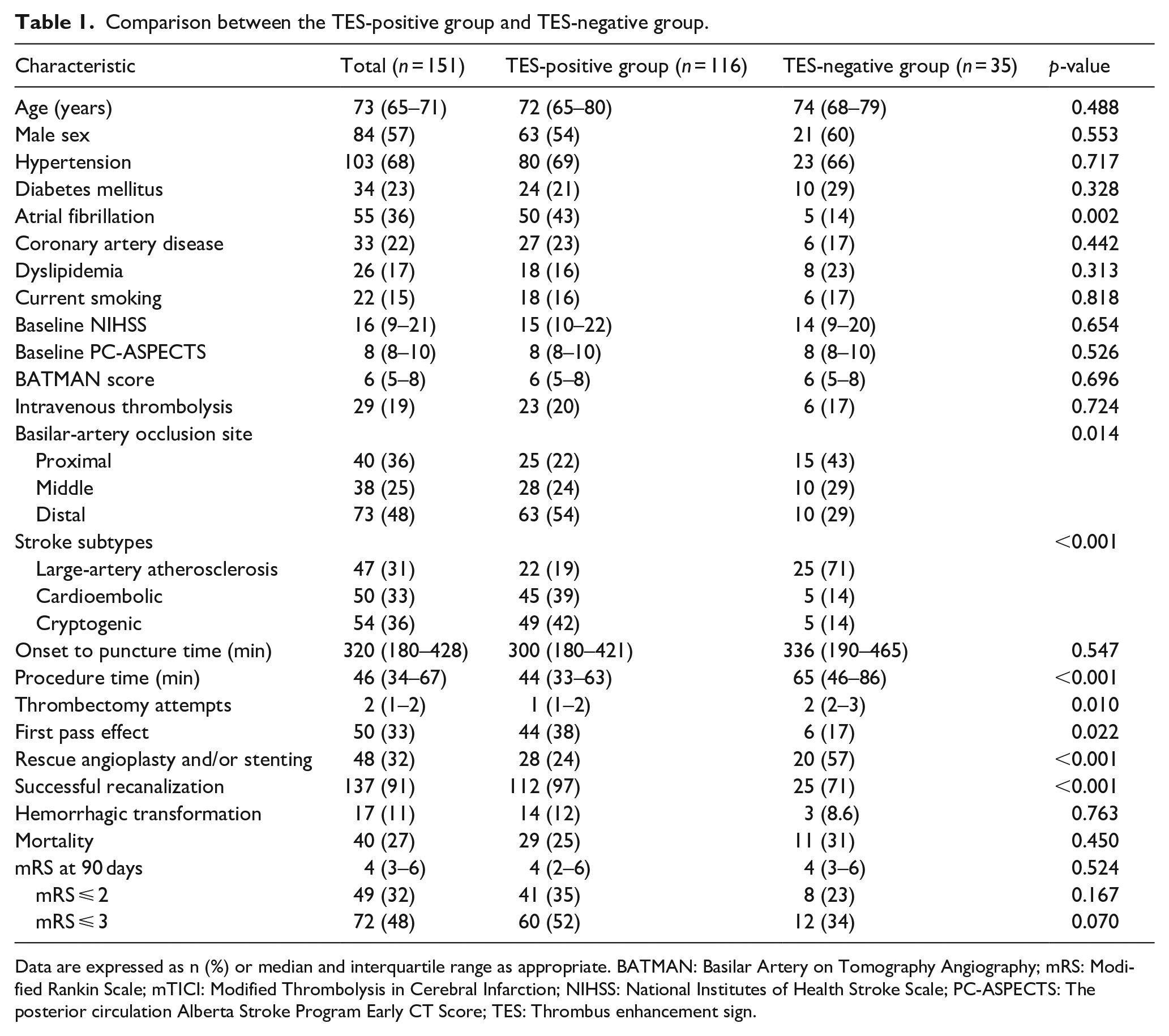
Data are expressed as n (%) or median and interquartile range as appropriate. BATMAN: Basilar Artery on Tomography Angiography; mRS: Modified Rankin Scale; mTICI: Modified Thrombolysis in Cerebral Infarction; NIHSS: National Institutes of Health Stroke Scale; PC-ASPECTS: The posterior circulation Alberta Stroke Program Early CT Score; TES: Thrombus enhancement sign.
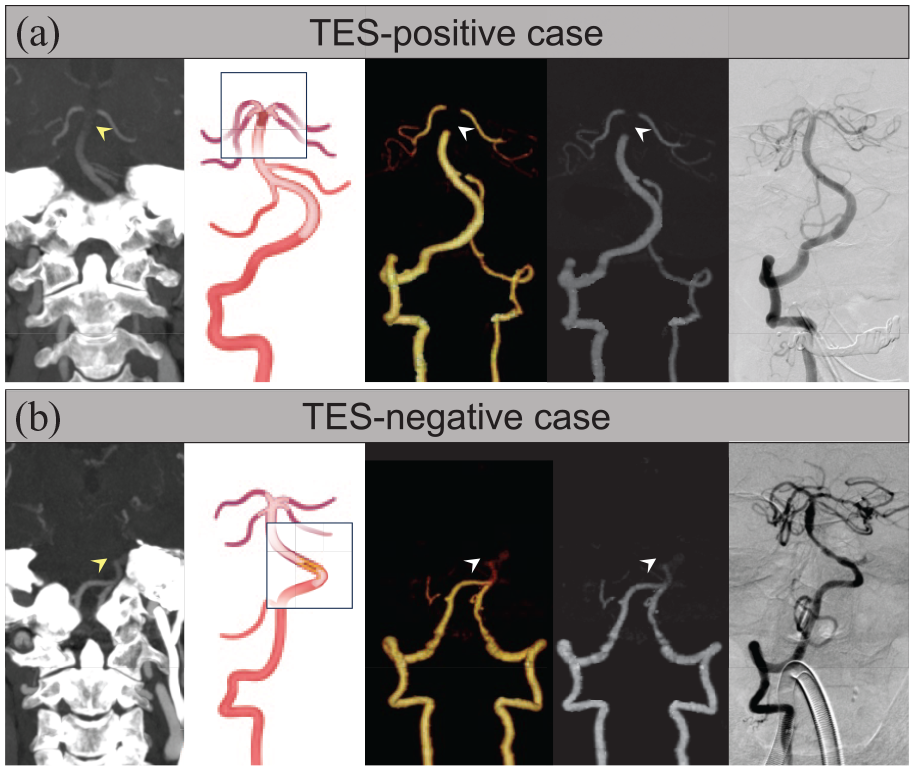
Example of the presence and absence of thrombus enhancement sign (TES) in patients with acute basilar-artery occlusion. (a) TES was clearly displaced on the thin-slab maximum intensity projection (TS-MIP) image and was not detected on full-slab maximum intensity projection (FS-MIP) or volume rendering (VR) images (arrowhead). Catheterized angiography after a single attempt of stent retrieval thrombectomy revealed complete recanalization of the basilar artery without residual stenosis. Schematic diagram of the pathogenesis indicates BAO of cardioembolism. (b) TES was not detected on TS-MIP, FS-MIP, or VR images (arrowhead); catheterized angiography after stent retrieval thrombectomy, rescued angioplasty, and stenting revealed residual stenosis and improved distal flow. Schematic diagram of the pathogenesis indicates BAO with in situ basilar stenosis. BAO: Basilar-artery occlusion.
Association between TES and stroke subtypes
On the basis of the TOAST classification, large-artery atherosclerosis strokes, cardioembolic strokes, and cryptogenic strokes accounted for 19% (22 of 116), 39% (45 of 116), and 42% (49 of 116) of patients in the TES-positive group, respectively, and for 71% (25 of 35), 14% (5 of 35), and 14% (5 of 35) of patients in the TES-negative group, respectively (p < 0.001). Of the 47 patients who suffered large-artery atherosclerotic strokes, 16 patients were considered embolic because of vertebral artery stenosis, while in the remaining 31 patients, the strokes were attributed to in situ BS. Positive TES occurred more frequently in embolic patients than in patients with in situ BS (75% [12 of 16] vs 16% [5 of 31], p = 0.003). After adjusting for BAO site and atrial fibrillation, the presence of TES was independently associated with the combined end point of cardioembolic and cryptogenic strokes (odds ratio [OR]: 8.56; 95% CI: 3.49, 22.4; p < 0.001).
Thrombi collected from 95 patients were included in the histological analysis. A comparison between the two groups revealed that the TES-positive thrombi consisted of a higher F/P proportion (50% ± 8.9% vs 38% ± 15%) in TES-negative thrombi (p = 0.002), a lower erythrocyte proportion (42% ± 9.0% vs 47% ± 8.9%) in TES-negative thrombi (p = 0.044), and a similar leukocyte proportion (7.3% ± 2.7% vs 7.0% ± 2.6%) in TES-negative thrombi (p = 0.745) (Figure 3).
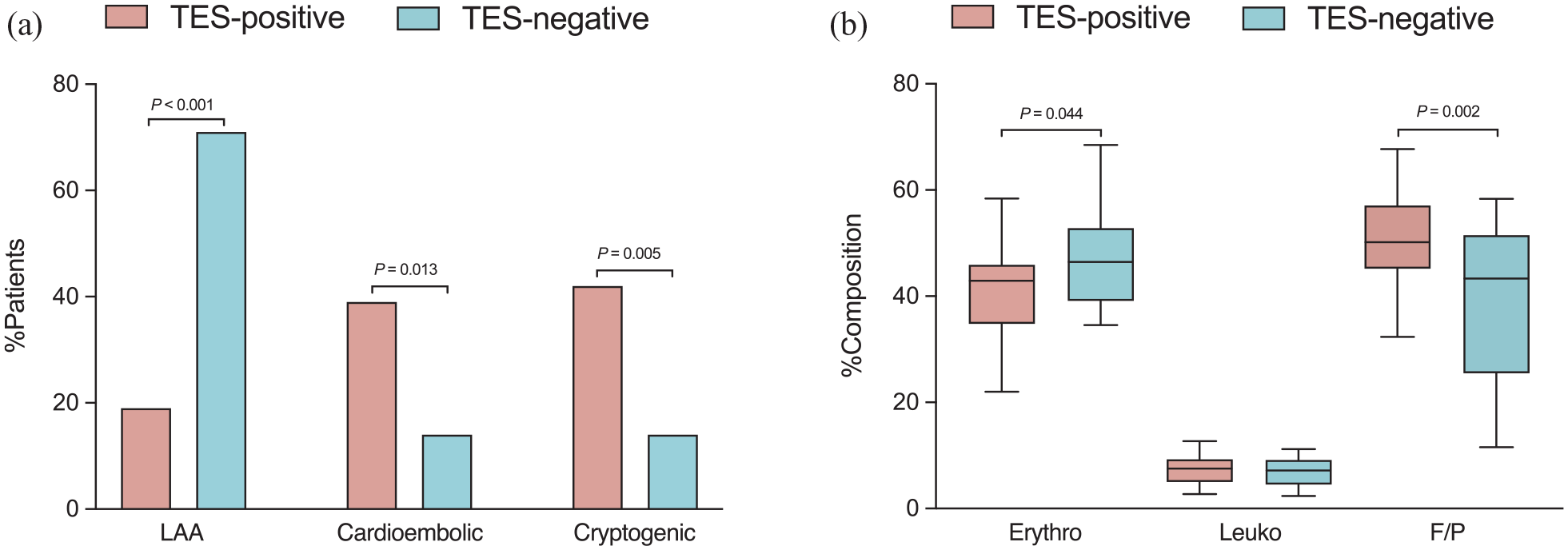
Comparisons of stroke subtype (a) and thrombus compositions (b) in TES-positive and TES-negative patients. F/P: Fibrin/platelet; LAA: Large-artery atherosclerotic stroke; TES: Thrombus enhancement sign.
Prediction of successful recanalization
Successful recanalization was achieved in 91% (137 of 151) of the patients. The TES-positive group showed a shorter procedure time (median 44 vs 65 min; p < 0.001), lower number of thrombectomy attempts (median 1 vs 2; p = 0.010), higher incidence of first pass success (38% vs 17%, p = 0.022), lower rate of requiring rescue angioplasty and/or stenting (24% vs 57%, p < 0.001), and higher rate of successful recanalization (97% vs 71%, p < 0.001) when compared to the TES-negative group (Figure 4).
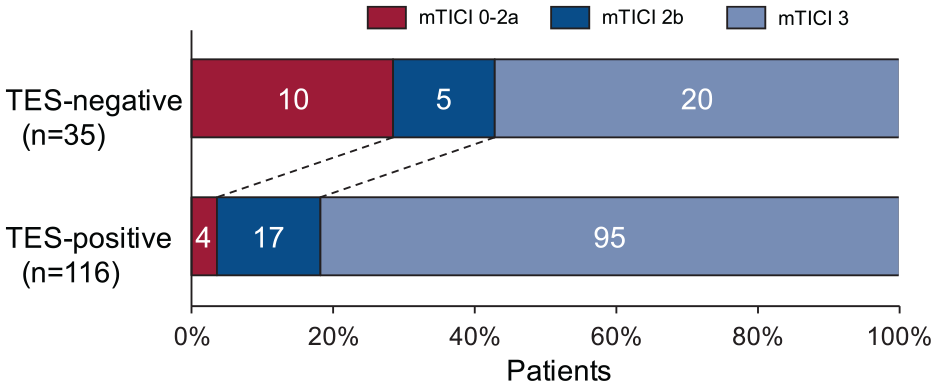
Distribution of modified thrombolysis in cerebral infarction (mTICI) score after EVT in patients with basilar-artery occlusion based on TES-positive and TES-negative groups. EVT: Endovascular thrombectomy; TES: Thrombus enhancement sign.
A favorable clinical outcome was more frequently achieved in the TES-positive group but did not reach statistical significance (mRS ⩽ 2 at 90 days, 35% vs 23%, p = 0.167; mRS ⩽ 3 at 90 days, 52% vs 34%, p = 0.070). Additionally, there was no significant difference in the incidence of mortality and hemorrhagic transformation between the groups.
Multivariate analysis revealed that the presence of TES was independently associated with successful recanalization (OR: 9.63; 95% CI: 2.33, 47.7; p = 0.003) after controlling for the effects of the BAO site and stroke subtypes (Table 2). The use of TES as a predictor of successful recanalization yielded a sensitivity of 0.82 (95% CI: 0.75, 0.88), specificity of 0.71 (95% CI: 0.48, 0.95), and area under the curve (AUC) of 0.77 (95% CI: 0.64, 0.89; p < 0.001).
Univariable and multivariable logistic regression analyses for successful recanalization.
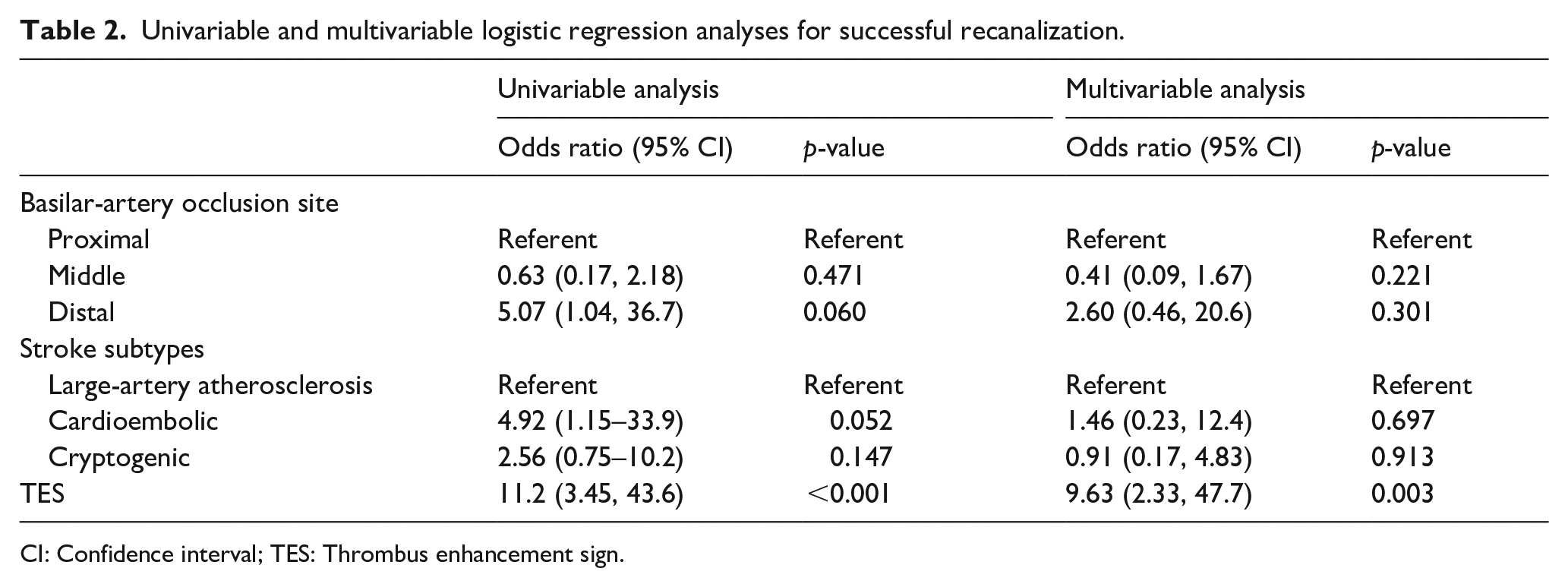
CI: Confidence interval; TES: Thrombus enhancement sign.
Discussion
The results of this study showed that TES could be well displayed and assessed in patients with acute BAO and serves as a discriminative imaging biomarker for predicting stroke of cardioembolic and cryptogenic source and fibrin/platelet-rich composition. Compared to the TES-negative group, patients with TES exhibited a higher rate of successful recanalization, shorter procedure time, lower number of thrombectomy attempts, higher incidence of first pass success, and lower rate of requiring rescue angioplasty and/or stenting after endovascular thrombectomy. Multivariate analysis confirmed that TES was independently associated with successful recanalization.
Some studies have conducted morphological analysis of the occlusive lesion on baseline imaging to predict the efficacy of recanalization therapy for BAO, such as hyperdense artery sign or susceptibility vessel sign.23,24 However, the diagnostic and prognostic value was less well-established. Recently, we reported that TES could be visualized reliably and reproducibly in anterior circulation on TS-MIP CTA and was considered to be associated with an F/P rich component. 18 In this study, TES was identified in 77% (116 of 151) of the enrolled patients with BAO, which was accompanied by a higher frequency of distal occlusion and stroke of cardioembolic and cryptogenic source. This finding is in agreement with the existing literature, which has demonstrated that embolic occlusions often manifest with distal BAO, whereas in situ BS occlusions often result in mid-BAO. 15 Embolic thrombus usually forms a toughened texture and an irregular morphologic shape and undergoes very little deformation adaptation in the basilar artery, enabling penetration of the contrast agent to outline the profile of the thrombus and the length of the occluded vessel to result in a TES. In BAO with chronic stenosis, the total basilar artery flow and caliber are reduced. 25 The poor and delayed contrast agent filling with thrombi or between the thrombus and the arterial wall causes the occlusive edge to be less distinct and the non-opacified segment to take longer to exhibit a negative TES. Because the incidence of underlying in situ intracranial atherosclerosis is higher in the posterior circulation than in the anterior circulation, reaching up to approximately 40%, 13 prompt knowledge about underlying stenosis in a case of acute BAO might lead to a more personalized selection process for thrombectomy.
For a better understanding of stroke subtype, we next performed histopathologic analysis of the retrieved thrombi. We found that the TES-positive thrombi consisted of a significantly higher F/P proportion (50% vs 38%) and a lower erythrocyte proportion (42% vs 47%) than the TES-negative thrombi. These results are in agreement with findings from previous studies, which suggested that TES-positive thrombi correlated with lower fractions of erythrocytes and more F/P conglomerations, concurrent with an association with cardioembolic origin. 26 Thrombus histology also revealed a strong overlap between cardioembolic and cryptogenic strokes, which further strengthens the theory that strokes in which the pathogenesis after complete diagnostic workup is classified as cryptogenic are mostly of cardiac origin. 27 TES could serve as a reliable predictor for identifying the combined etiology of cardioembolic and cryptogenic strokes in BAO. Thus, TES can aid in differentiating the etiology of stroke and selecting targeted examinations for diagnosis. For example, esophageal ultrasonography can be used to confirm the cardiac origin of emboli, and high-resolution magnetic resonance imaging can be used to identify culprit plaques. Additionally, targeted drug treatments can be administered for post-procedural secondary prevention of recurrent stroke. Positive TES results in patients with cardiac embolism indicate potential benefits from anticoagulation treatment, while negative TES results suggest suitability for antiplatelet therapy due to in situ intracranial atherosclerotic disease. 28 Additionally, the occurrence of large-artery atherosclerosis strokes involving distal embolization from the exfoliation of atherosclerotic plaques might also display TES (75% vs 16% for in situ BS).
Our study showed that the presence of TES was an independent factor for successful recanalization. Considering that the presence of TES-positive was associated with an F/P-rich component and indicated thrombus of cardioembolic source with irregular morphologic shape, the thrombus may have a greater available area for contact with the thrombectomy device and a reduced area making contact with the vessel wall. This may facilitate easier and broader engagement of the thrombectomy device, resulting in reduced friction and adhesion between the thrombus and the vessel wall. 29 Moreover, TES-positive thrombi were more likely to occlude at the basilar artery tip with a relatively lower clot burden (compared to proximal BAO), 13 enabling fast and efficient aspiration or the use of a guidewire to navigate through the block and correctly select into the true posterior artery lumen. In addition, studies on embolic thrombi in BAO have demonstrated a high technical success rate, whereas if significant in situ underlying stenosis is present, recanalization may not be achieved sufficiently even after repeated thrombectomy.15,30 Patients with underlying BS lesions often require more complex adjuvant therapy, resulting in longer procedural times, as observed in our series. In such cases, a TES-negative in BAO could indicate the presence of underlying BS. This information allows the interventionalist to make initial adjustments to the choice of access catheter and other devices, in preparation for a potential stenting procedure.
Although not statistically significant, there was a numerical difference in functional outcomes favoring TES-positive patients. 31 This may be attributed to TES serving as an imaging biomarker for occlusion and predicting a higher rate of recanalization. However, the involvement of perforating arteries and their compensation in inferior collateral circulation often results in poorer ischemic tolerance of brainstem cells following occlusion of the main trunk of the posterior circulation.32,33 As a result, the benefits of achieving better and faster vascular recanalization in TES-positive patients are somewhat diminished. Consequently, substantial improvement in clinical outcomes poses a challenge.
This study has several limitations, which are partly attributed to the single-center retrospective design and inherent selection bias. In addition, 56 patients were excluded from the histological analysis because of insufficient thrombus retrieval and thrombus dilution or separation in the formalin fixer. Moreover, patients with strokes with other determined causes were excluded from our study because of limited numbers (i.e. only two patients had strokes caused by arterial dissections). Furthermore, the underlying causes of cryptogenic stroke were not explored during the follow-up period, which restricts the effectiveness of TES in providing further clarification of the source in cryptogenic strokes. Therefore, future studies with a larger cohort are warranted.
Conclusion
In conclusion, we demonstrated that the TES is a reliable and easily accessible marker for identifying cardioembolic and cryptogenic strokes and represents an accurate predictor of recanalization success in thrombectomy for acute BAO. Further prospective studies with a larger sample size are warranted to confirm our findings.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:(1) Shanghai Municipal Health Commission Clinical Research Project (No.202140066)
(2) New interdisciplinary research Project of Shanghai Municipal Health Commission (No.2022JC022)
(3) Shanghai Jiao Tong University “Medical and Research” Program (ZH2018ZDA19)
Ethical approval
The ethics committee of Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine approved this study (No 2018-002-1).
Informed consent
Informed consent was provided by patients or their family members.
Guarantor
Haitao Lu and Yueqi Zhu
Contributorship
(1) Guangchen He, Sheng Guo: Acquisition of data, analysis and interpretation of data and drafting of the manuscript.
(2) Haitao Lu and Yueqi Zhu: Study concept and design, critical revision of the manuscript for important intellectual content, and study supervision.
(3) Hui Fang, Haoyang Xu, Runjianya Ling: acquisition of data, revision of the manuscript.
(4) Guangchen He: statistical analysis.
Data availability
This study’s data are available from the corresponding author upon reasonable request.
