Abstract
Introduction:
While adiposity is an established risk factor for incident ischemic stroke, its influence on functional recovery after stroke is uncertain. We leveraged Mendelian randomization (MR) to examine the causal effects of body mass index (BMI) and waist-to-hip ratio (WHR) on post-stroke motor, cognitive, and global recovery.
Materials and methods:
Genetic proxies for BMI and WHR were obtained from a genome-wide association study (GWAS) meta-analysis of the GIANT consortium and the UK Biobank (n = 806,834). The primary outcomes were longitudinal trajectories of three National Institutes of Health Stroke Scale (NIHSS) measures assessed over a 2-year period: motor function subscores, cognitive performance subscores, and total NIHSS scores (as a measure of global recovery). Genetic associations with these stroke recovery outcomes were obtained from a GWAS conducted within the VISP trial of mild ischemic stroke (n = 1270). MR effects were estimated using the inverse-variance weighted method.
Results:
A 1-standard deviation (SD) increase (~4.8 kg/m2) in genetically predicted BMI associated with lower odds of post-stroke motor improvement (OR = 0.37, 95% CI = 0.19-0.72; p = 0.003). Similarly, a genetically predicted increase in BMI was associated with worse cognitive (β = −0.12, 95% CI = −0.21, −0.03; p = 0.009) and global recovery (β = −0.36, 95% CI = −0.59, −0.13; p = 0.002). Associations between genetically predicted WHR and post-stroke recovery were directionally concordant but were not statistically significant (p > 0.05).
Discussions and conclusions:
Human genetic evidence suggests that elevated adiposity negatively impacts multiple post-stroke outcomes, including motor function, cognition, and overall recovery. The biological pathways underlying these associations warrant further investigation.
This is a visual representation of the abstract.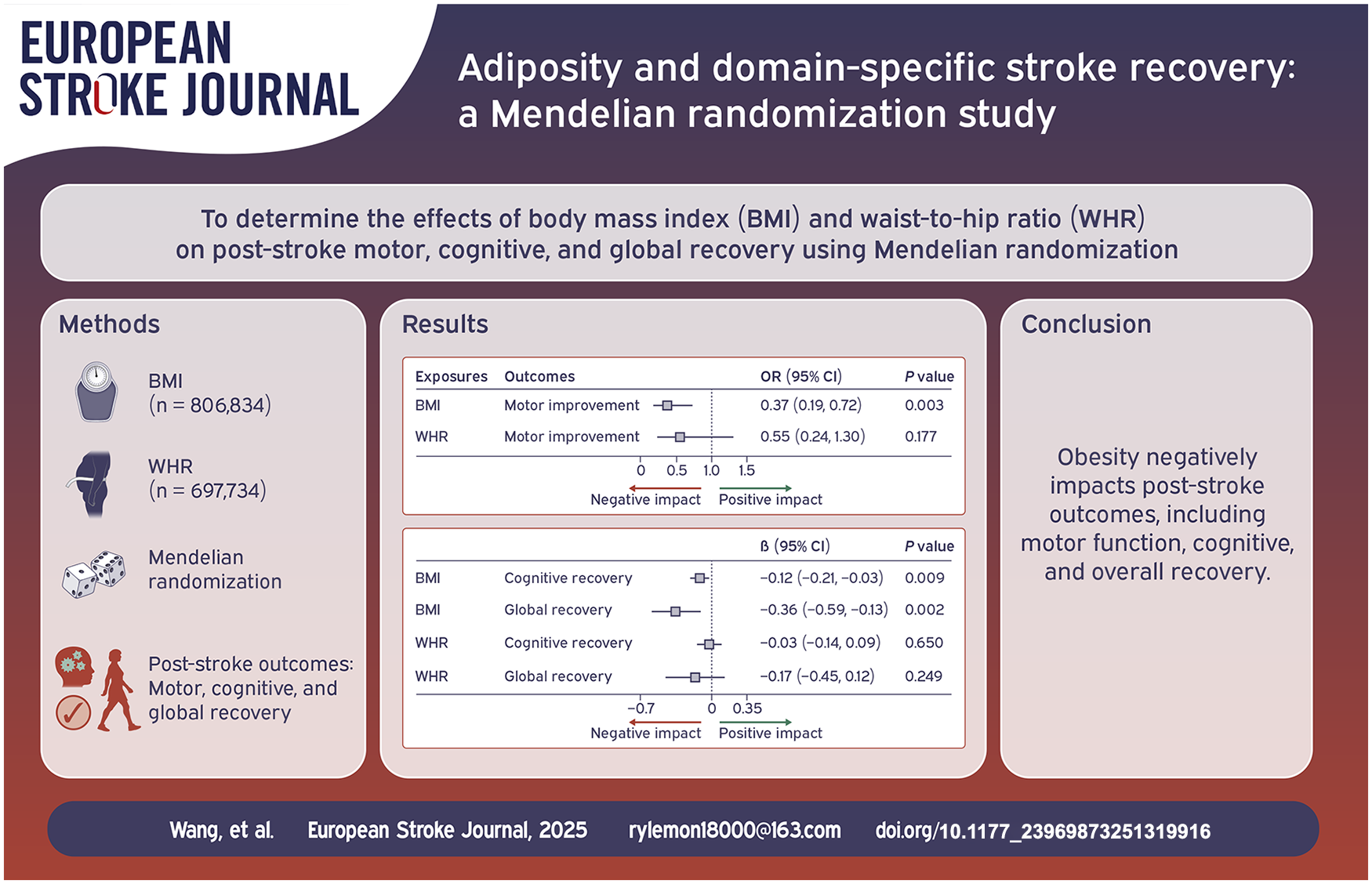
Introduction
Obesity is a well-established risk factor for ischemic stroke incidence. 1 However, its effect on post-stroke functional outcomes remains uncertain.2–6 Previous research has demonstrated a paradoxical relationship between obesity and post-stroke outcomes, whereby elevated body mass index (BMI) was associated with improved functional recovery.2–4 However, other studies have not replicated this finding.5,6 For example, a multicenter, prospective, hospital-registry study showed no association between obesity and post-stroke functional recovery. 5 In addition, our previous Mendelian randomization (MR) study 6 demonstrated that genetically predicted higher waist-to-hip ratio (WHR) was associated with poor post-stroke functional outcome (OR = 1.48;95% CI = 1.03–2.13; p = 0.033), while the association with BMI was null with inconclusive confidence intervals (OR = 1.01; 95% CI = 0.75–1.36; p = 0.937). This MR analysis investigated post-stroke functional recovery using the global endpoint of the modified Rankin scale (mRS). The mRS aggregates diverse neurological deficits into a composite measure, limiting its ability to differentiate recovery patterns across specific functional domains such as motor and cognitive systems. To date, no MR study has investigated the association of adiposity with specific domains of post-stroke recovery.
MR is an analytical method that uses genetic variants as instrumental variables to investigate causal relationships between exposures and outcomes. By employing genetic variants strongly associated with adiposity measures as instruments, MR can estimate the causal effects of adiposity on post-stroke recovery.7,8 This approach minimizes bias due to reverse causation and confounding since genetic variants are randomly allocated at conception. Here, we conduct the first MR study to examine the potential causal effects of BMI and WHR on specific domains of post-stroke recovery, including motor and cognitive outcomes.9–11
Methods
Data sources
All data sources used in this MR study are presented in Table 1. Genetic associations with BMI (n = 806,834) and WHR (n = 697,734) were obtained from a GWAS meta-analysis of the Genetic Investigation of ANthropometric Traits consortium and the UK Biobank. 9 Genetic estimates for BMI and WHR were scaled to standard deviation (SD) units (BMI ~ 4.8 kg/m2, WHR 0.09 ratio). Genetic associations were adjusted for age, sex, and principal components of ancestry. 9
Data sources used in this present study.

GIANT: Genetic Investigation of ANthropometric Traits; VISP: Vitamin Intervention for Stroke Prevention.
The study outcomes included post-stroke motor, cognitive, and global recovery measured over a two year period.10,11 Genetic associations with these phenotypes were obtained from GWAS that included up to 1270 participants with a non-disabling ischemic stroke (mRS ⩽3).10,11 These GWAS were performed using data from the Vitamin Intervention for Stroke Prevention (VISP) trial, which investigated whether high dose vitamin supplementation reduces the risk of recurrent ischemic stroke.10–12 VISP enrolled 3680 participants with a nondisabling ischemic stroke (mRS ⩽3) within 120 days from ischemic stroke onset (median 72 days; interquartile range 45.75–102), and these participants were randomized to a high or low-dose vitamin treatment arm. 12 The study did not find a significant effect of vitamin supplementation on stroke outcome. All participants were followed up every 3 months for 2 years after randomization. 12 The motor recovery GWAS included 448 VISP participants with genotyped data, motor impairment at randomization and without a recurrent stroke. 10 The motor drift subscores of the National Institutes of Health Stroke Scale (NIHSS; 5A/5B and 6A/6B) was used to evaluate the degree of motor weakness for the upper and lower limbs. Motor improvement was defined as a decrease in the initial motor drift subscores of the weakest limb from randomization to each follow up period. 10 Genetic associations with this phenotype were estimated using logistic regression, estimating the odds ratio of motor improvement over 2 years with adjustment for age, sex, race, initial motor score, treatment arm, and principal components. 10 The GWAS of cognitive and global recovery included 1270 VISP participants with genotyped data, an NIHSS score greater than zero at randomization, and without a recurrent stroke. 11 Cognitive impairment was defined as the change in the sum of the NIHSS scores for level of consciousness (LOC) questions, LOC commands, aphasia, and inattention/neglect. 11 Global impairment was defined as the change in the total NIHSS score. 11 For cognitive and global recovery, a linear mixed-effects model was used to estimate the change in NIHSS scores in each phenotype (delta = initial score − follow-up score) at each time point from randomization with adjustment for age, sex, treatment group, initial NIHSS score, and the first five principal components of ancestry. 11
Genetic instruments selection
To identify genetic instruments for BMI and WHR, we selected single nucleotide polymorphisms (SNPs) associated with BMI or WHR at genome-wide statistical significance (p < 5 × 10−8) and that were available in the outcome dataset. We then harmonized the effect alleles in the exposure and outcome datasets, and removed all palindromic variants. 13 Finally, we performed linkage disequilibrium clumping (r2 < 0.001, window 10,000 kb) using the 1000 Genomes European reference panel.13,14 The F-statistic for each SNP was calculated to assess instrument strength, 15 with an F-statistic greater than 10 indicating low risk for weak instrument bias. 16
MR analyses
The primary MR estimates were derived using the random-effects inverse-variance weighted (IVW) method. 17 In these primary analyses, we used a false discovery rate (FDR)-corrected p value <0.05 to declare statistical significance.
Heterogeneity among SNP-specific causal estimates was assessed using Cochran’s Q test. 18 In sensitivity analyses, we used four MR methods that are more robust to pleiotropic instruments: MR using Robust Adjusted Profile Score (MR-RAPS), 19 weighted median, 20 MR-Egger regression, 21 and weighted mode 22 methods. These analytical approaches employ distinct assumptions regarding genetic pleiotropy. Concordant effect estimates across multiple methods therefore provide more robust evidence supporting causal relationships.23,24 The intercept from the MR-Egger regression was used to assess the presence of directional horizontal pleiotropy with a p < 0.05 indicating significant pleiotropy. 21 MR-Pleiotropy Residual Sum and Outlier (MR-PRESSO) 25 method was used to detect any genetic variants with outlying causal estimates that may be influencing the outcome through pleiotropic pathways. When significant associations were observed, we also performed multivariable MR 26 adjusting for genetically predicted systolic blood pressure (SBP) 27 and type 2 diabetes (T2D). 28
MR analyses with correction for index event bias
A significant limitation of applying MR in case-only studies is the potential for index event bias.29,30 In the present study, given that BMI is a causal risk factor for ischemic stroke incidence, 1 the genetic instruments for BMI may be associated with other stroke risk factors, and so the association between the genetic instruments and post-stroke motor recovery outcomes may be susceptible to confounding by these factors. Accordingly, we repeated MR analyses using post-stroke motor recovery outcomes genetic effects adjusted for this bias using the methodology implemented in the indexevent R package.10,31,32 Given that genetic associations for cognitive and global recovery outcomes were not derived from logistic regression models we were unable to apply this analysis to the GWAS for post-stroke cognitive and global recovery.
All MR analyses were conducted using the TwoSampleMR, 13 MR-PRESSO, 25 MendelianRandomization, 33 and mr.raps 19 packages in R version 4.3.0.
Results
The genetic variants used as instruments for BMI and WHR are presented in Supplemental Tables 1–4. The F statistic for individual SNPs ranged from 28 to 1482, providing evidence against weak instrument bias.
A 1-SD increase (4.8 kg/m2) in genetically predicted BMI was associated with lower odds of motor improvement after non-disabling ischemic stroke (OR = 0.37, 95% CI = 0.19–0.72, p = 0.003, FDR-P = 0.009; Figure 1). Additionally, higher genetically predicted BMI was associated with poorer cognitive improvement (β = −0.12, 95% CI = −0.21, −0.03, p = 0.009, FDR-P = 0.018) and worse global recovery (β = −0.36, 95% CI = −0.59, −0.13; p = 0.002, FDR-P = 0.009; Figure 1). The results were directionally consistent across sensitivity analyses using alternative MR methods (Supplemental Table 5). The association of BMI with post-stroke motor and cognitive recovery was unchanged in the multivariable MR adjusting for SBP and T2D (Supplemental Table 6). The association between BMI and post-stroke motor recovery remained significant when adjusting for index event bias (IVW OR = 0.40, 95% CI = 0.21–0.77; p = 0.007; Supplemental Table 7). The association of genetically predicted WHR with post-stroke recovery outcomes was directionally concordant with BMI, but was not statistically significant (Figure 1, Supplemental Table 5).
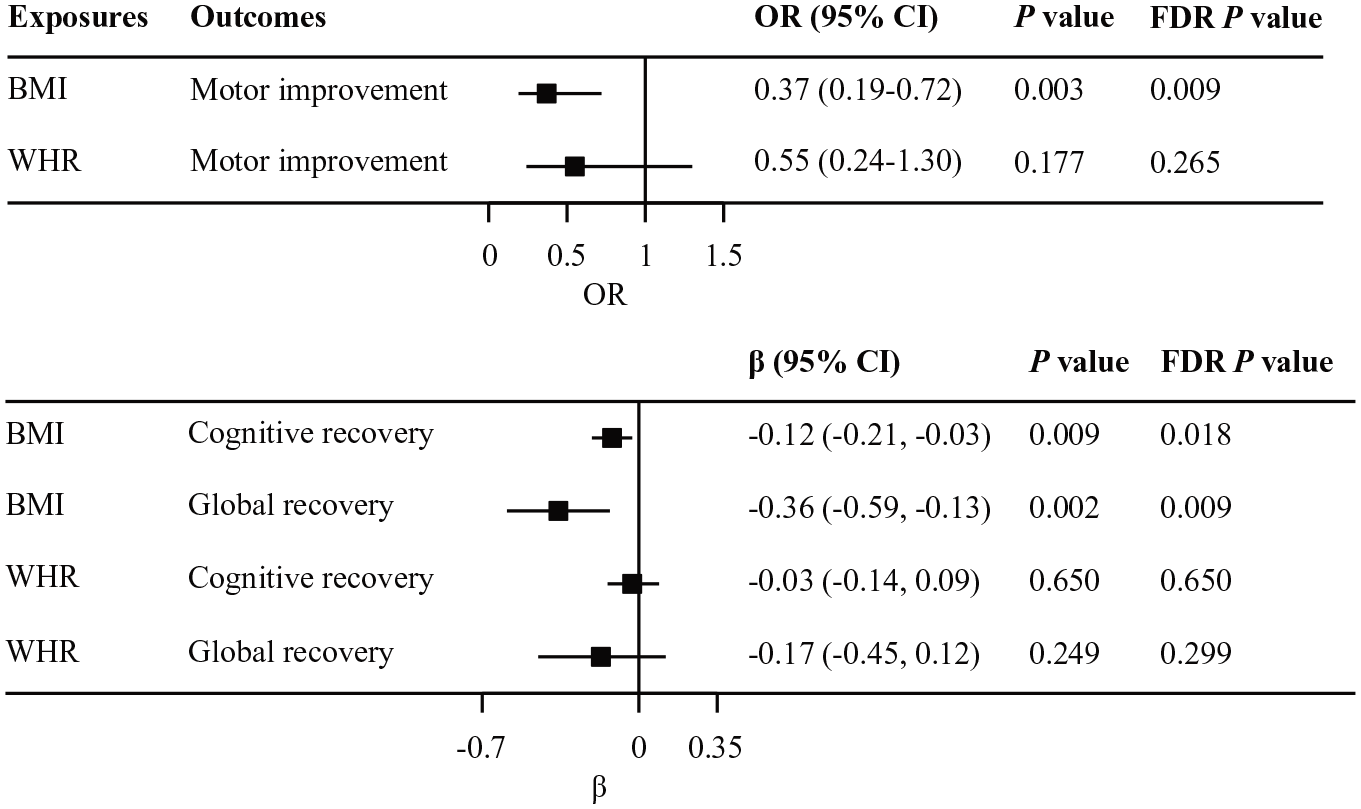
Mendelian randomization associations of genetically predicted body mass index (BMI) and waist-to-hip (WHR) with post-stroke recovery phenotypes; results derived from random-effects inverse-variance weighted analyses; OR are presented for motor improvement and β coefficient for cognitive and global recovery.
In all analyses, there was no evidence of heterogeneity (P for Cochran’s Q > 0.12) or directional pleiotropy (P for intercept >0.27; Supplemental Table 8). In addition, no pleiotropic outliers were identified by MR-PRESSO.
Discussion
This is the first MR study to investigate the causal effects of adiposity on specific subdomains of post-ischemic stroke recovery. We found that genetically predicted higher BMI reduced odds of motor improvement, impaired cognitive recovery, and poorer global recovery after ischemic stroke.
Previous observational studies2–6 have found inconsistent associations of obesity with post-stroke functional outcomes. For example, a post hoc analysis of the Telemedical Project for Integrative Stroke Care (TEMPiS) trial 2 showed that obese patients with stroke or transient ischemic attack had a lower risk of poor functional outcomes at 30 months, defined as mRS >3 or Barthel index <60. However, a multi-center, prospective, hospital-based registry study 5 including 4782 patients with ischemic stroke showed no association between obesity and post-stroke functional outcome at 3 and 12 months. In addition, our previous MR study 6 showed a positive association of genetically predicted higher WHR with poor functional outcome (mRS ⩾3, OR = 1.48; 95% CI = 1.03–2.13; p = 0.033), whereas no association was observed for BMI. A key limitation of previous studies was their reliance on global outcome measures like the mRS, rather than domain-specific assessments such as NIHSS subscores for motor and cognitive function.34,35 By contrast, we used NIHSS subscores to define motor and cognitive domain stroke recovery phenotype and investigated the effect of BMI and WHR on post-stroke motor and cognitive recovery, which was not investigated in our previous MR study. 6 The mechanisms underlying the association between BMI and post-stroke recovery remain speculative and may be attributable to adiposity-related alterations in neuroplasticity,36–38 low-grade neuro-inflammation,39,40 or to glycemic dysregulation. 41 Further studies are warranted to investigate these putative mechanisms.
This study has several strengths. The genetic instrumental variable approach helps protect against reverse causation and residual confounding that can bias traditional observational studies. In addition, the results of several sensitivity analyses, including adjustment for index event bias, were consistent, providing evidence to support a robust association between BMI and post-stroke motor, cognitive, and global recovery.
There are also several limitations to consider. First, the null associations of WHR with the recovery outcomes may be attributable to low statistical power. Second, the post-stroke recovery GWAS did not include stroke subtype data, and therefore we are unable to assess if the observed associations vary across ischemic stroke subtypes. Third, the motor recovery GWAS included a generalized estimating equation with the logistic regression to account for the correlation of motor impairment scores measured repeatedly over time. The use of the index event bias correcting method is not well-established in this setting. Fourth, despite consistent results in sensitivity analyses, we cannot exclude the possibility that the associations of BMI with post-stroke cognitive and global recovery may be due to index event bias. 1 Fifth, the two-sample MR study design assumes a linear relationship between exposure and disease, and thus we are unable to examine nonlinear relationships between BMI, WHR, and post-stroke recovery. Sixth, the VISP post-stroke recovery GWAS was performed in patients with non-disabling ischemic stroke (mRS ⩽3). 10 Therefore, it is uncertain whether these findings generalize to patients with more severe stroke. Independent replication of these findings in additional cohorts is warranted to increase confidence in these findings. Finally, the motor and cognitive NIHSS subscales used in the present analysis are limited as measures of their respective functional domains. Future analyses should incorporate more granular measures of these phenotypes, such as objective measures of muscle power, or more extensive cognitive testing such as with formal aphasia batteries.
In conclusion, our MR analyses provide evidence for an adverse effect of elevated adiposity on multiple domains of ischemic stroke recovery, including motor function, cognitive, and global outcomes. Investigation of the biological pathways underlying these associations may inform the development of novel therapeutic strategies to improve recovery after ischemic stroke.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251319916 – Supplemental material for Adiposity and domain-specific stroke recovery: A Mendelian randomization study
Supplemental material, sj-docx-1-eso-10.1177_23969873251319916 for Adiposity and domain-specific stroke recovery: A Mendelian randomization study by Mengmeng Wang, Iyas Daghlas, Chad M Aldridge, Zhizhong Zhang and Yi Ren in European Stroke Journal
Footnotes
Acknowledgements
We thank the Cerebrovascular Disease Knowledge Portal for providing summary data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent and ethical approval
This study used publicly available summary data from published GWAS studies. Ethical approval and informed consent were obtained from in original studies. This study was completed in accordance with the Helsinki Declaration as revised in 2013.
Trial registration
Not applicable.
Guarantor
Mengmeng Wang.
Contributorship
Mengmeng Wang, Iyas Daghlas, Chad M. Aldridge and Yi Ren conceived and designed the study; Mengmeng Wang drafted the manuscript and conducted the statistical analysis; Iyas Daghlas, Chad M. Aldridge, Zhizhong Zhang, and Yi Ren revised the manuscript; each author approved the version to be published.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
