Abstract
Background:
Frailty appears to be associated with unfavorable prognosis after stroke in observational studies, but the causality remains largely unknown.
Aims:
The aim of this study is to investigate the potential causal effect of frailty on functional outcome at 3 months after ischemic stroke using the Mendelian randomization (MR) framework.
Methods:
Genetic instruments for frailty index were identified in a genome-wide association study meta-analysis including 175,226 individuals of European descent. Corresponding genetic association estimates for functional outcome after ischemic stroke at 90 days were taken from the Genetic of Ischemic Stroke Functional Outcome (GISCOME) network of 6021 patients. We performed inverse-variance weighted MR as the main analyses, followed by several alternate methods and sensitivity analyses.
Results:
In univariable MR, we found evidence that genetically predicted higher frailty index (odds ratio (OR) = 5.12; 95% confidence interval (CI) = 1.31–20.09; p = 0.019) was associated with worse functional outcome (modified Rankin Scale score ⩾3) after ischemic stroke. In further multivariable MR adjusting for potential confounding traits including body mass index, C-reactive protein, inflammatory bowel disease, and smoking initiation, the overall patterns between genetic liability to frailty and poor functional outcome status remained. Sensitivity analyses with complementary methods and with model unadjusted for baseline stroke severity (OR = 4.19; 95% CI = 1.26–13.90; p = 0.019) yielded broadly concordant results.
Conclusions:
The present MR study suggested a possible causal effect of frailty on poor functional outcome after ischemic stroke. Frailty might represent a potential target for intervention to improve recovery after ischemic stroke.
Introduction
Frailty refers to a state of increased vulnerability to stressors due to decreased physiological reserves across multiple systems and thus leads to adverse clinical outcomes, including hospitalizations, complications, disability, shorter survival time, poorer quality of life, and other health problems. 1 In recent years, the role of frailty in stroke has received much attention. A recent meta-analysis with 48,009 participants reported the prevalence of frailty in stroke patients as 22%. The prevalence of frailty was twofold in patients with stroke compared to those without stroke. 2 Frailty might indicate dynamic imbalance and impaired physiological resilience to stroke events, resulting in the deterioration of the functional ability of frail persons rapidly, increasing the risk of poor outcomes. Notably, observational studies have suggested that frailty may affect stroke disease trajectory and prognosis.3–7 However, the causal link has not been fully established.
Mendelian randomization (MR) technique applies genetic variants as instruments to make a causal inference. This analytical approach leverages the random assortment of genetic variants at conception to minimize bias caused by confounding factors and reverse causation. Recently, two MR studies provide evidence of a causal relationship between frailty and increased risk of any stroke,8,9 while no causal link between frailty and risk of ischemic stroke was detected. 9 However, both previous MR studies focused on stroke risk. The causal role of frailty in poststroke outcomes is still largely unknown. Considering the predicted rise in the prevalence of frailty associated with changing demographics over the coming decades, establishing understanding of how frailty influences neurological recovery after stroke has substantial implications, as it may be possible to attenuate or reverse frailty trajectories by multifaceted intervention programs to protect against unfavorable prognosis after stroke. 10 Here, we designed a two-sample MR to explore whether genetic liability to frailty may be related to poor outcomes at 3 months after ischemic stroke.
Materials and methods
All summary-level genomic data adopted for this work are publicly available. All data sources were approved by relevant institutional review boards from original studies, and all participants were given informed consent. This study was reported according to the STROBE-MR statement. 11
Date source and single-nucleotide polymorphism selection
The data sources employed in the present study are detailed in Supplementary Table 1. We selected associated single-nucleotide polymorphisms (SNPs) for frailty, measured by frailty index (FI), in the large genome-wide association study (GWAS) meta-analysis in the UK Biobank and Swedish TwinGene, which included 175,226 individuals of European descent. 12 FI, which is widely accepted as one operationalization of frailty, 13 was derived based on the accumulation of a number of health deficits during the life course using self-reported questionnaire. The 49 or 44 self-reported components including physiological and mental health variables were used to calculate the FIs for UK Biobank and Swedish TwinGene, respectively. Both FIs are constructed using the Rockwood deficit accumulation model according to the standard procedure 14 and have been validated elsewhere.15,16 Details of the phenotype measure are described in Supplementary Tables 2 and 3. Comparability of the two sets of items has been previously validated. 12 Specifically, 29 of the 49 items used in UK Biobank have approximate counterparts in TwinGene (Supplementary Table 4). Besides, the UK Biobank’s subset of 29 items correlate well with the full set of 49 items (r2 = 0.85, p < 0.001). 12
We selected independent SNPs (r2 < 0.001) that achieved genome-wide significance (p < 5 × 10−8) upon adjustment for age, gender, and 10 principal components as instrumental variables. For each index SNP, we calculated F statistic and R2 value, which represented the strength and variance explained by an individual genetic instrument, respectively.
Outcome data sources
Participants were of European ancestry only to minimize potential bias from population stratification, except for the body mass index (BMI) dataset with a small portion of people of non-European ancestry. The GISCOME (Genetic of Ischemic Stroke Functional Outcome) GWAS meta-analysis was used to obtain genetic association estimates for functional outcome after ischemic stroke (6021 patients). 17 The GISCOME network included 12 studies from the United States, Europe, and Australia. Functional outcome was measured by modified Rankin Scale (mRS) evaluated as close as possible to 90 days poststroke. In most studies, mRS assessment was done by trained assessors at face-to-face or telephone follow-up. An mRS of 0–2 denoted good functional outcome (3741 patients), while mRS of 3–6 indicated poor functional outcome (2280 patients). The primary model was adjusted for age, sex, five principal components, and baseline stroke severity assessed by the National Institutes of Health Stroke Scale (NIHSS) at 0–10 days after stroke onset. Additionally, model without adjustment for baseline NIHSS was considered for sensitivity analysis.
We carefully reviewed the original cohorts in the exposure and outcome dataset, and found that sample overlap was negligible. When index SNPs for the exposure were not available in the outcome dataset, we replaced them with proxy SNPs (r2 > 0.8) defined using 1000 genomes European reference data. Then, we harmonized the effect sizes for the SNPs on FI and the outcome dataset. For palindromic SNPs, we used allele frequency information to resolve strand ambiguity and dropped those with minor allele frequency close to 0.50. One SNP (rs9275160) associated with FI was not available in the GISCOME dataset, and no suitable proxy was found.
Statistical analyses
We performed fixed effect inverse-variance weighted (IVW) MR as the main analyses unless significant heterogeneity was present. This approach can provide an accurate estimate in the absence of horizontal pleiotropy, or when horizontal pleiotropy is balanced. A number of complementary MR methods were conducted as complementary analyses, including the random-effects IVW, weighted median, 18 MR-Robust Adjusted Profile Score (MR-RAPS), 19 and MR-Pleiotropy Residual Sum and Outlier (MR-PRESSO) 20 approach. The weighted median estimate gives consistent estimates when at least half of the instrument variables are valid. The MR-RAPS method was used to provide unbiased estimates in the presence of many weak instruments. MR-PRESSO was used to identify potential outliers. These methods are generally robust to potential violations of instrumental variable assumptions.
Heterogeneity of each SNP was tested using Cochran’s Q statistic. Leave-one-out plot was created to estimate whether individual SNPs dominated the overall estimates. To assess the possibility of reverse causality, we made Steiger analyses 21 to verify the causal direction, as well as performed reverse MR analyses using genetic instruments with suggestive significant threshold (p < 5 × 10−6).
Beyond standard analyses, we also carried out additional sensitivity analyses for further validation and address the bias in studies of genetic associations with prognosis. First, we examined the relationship between genetically predicted fatigue, a characteristic of frailty, and 3-month stroke outcomes. Genetic variants associated with self-reported fatigue were ascertained from a UK Biobank GWAS comprising 449,019 individuals. 22 Fatigue was assessed based on the frequency of tiredness/lethargy in the last 2 weeks. UK Biobank’s participants were asked to answer the question, “Over the last two weeks, how often have you felt tired or had little energy?” Possible answers were “Not at all/Several days/More than half the days/Nearly every day/Do not know/Prefer not to answer.” Participants answering with “Do not know” or “Prefer not to answer” were excluded, leading to a four-category variable for fatigue. Second, a potential challenge to assess prognostic factors among disease patients is vulnerability to index event bias, a type of selection bias that happens when studying risk factors for subsequent events among disease cases. Thus, we estimated the causal association of FI with ischemic stroke risk to help to evaluate whether index event bias is likely to affect our results of poststroke outcomes. Summary statistics data for association of SNPs with ischemic stroke were obtained from MEGASTROKE consortium, including up to 34,217 ischemic stroke cases and 406,111 stroke-free controls. 23 We also performed a replication analysis in an independent European population using data from the FinnGen consortium (10,551 ischemic stroke cases and 202,223 controls). 24 Finally, we repeated MR analyses using stroke outcome genetic effects corrected for index event bias with a correction method, which was described previously. 25
Results were reported for each standard deviation increment in FI. Post hoc power was calculated at an alpha level of 0.05 by mRnd (http://cnsgenomics.com/shiny/mRnd/). 26 All statistical analyses were conducted with TwoSampleMR 27 and MR-PRESSO 20 packages in R version 4.2.1.
Assessment of pleiotropy
MR-PRESSO global test and MR-Egger intercept were used to evaluate the evidence of horizontal pleiotropy. Moreover, we performed multivariable MR (MVMR) analysis to explore the independent effects of FI on functional outcome. We considered four covariates associated with frailty, 28 including BMI, C-reactive protein (CRP), inflammatory bowel disease (IBD), and smoking initiation. Summary statistics for BMI were derived from the GIANT (Genetic Investigation of Anthropometric Traits) consortium. 29 The CIWG (Genomic Epidemiology Inflammation Working Group) was used to obtain genetic association estimates for CRP. 30 Genetic variants for IBD were from the IIBDGC (The International IBD Genetics Consortium). 31 SNPs for smoking initiation were taken from the GSCAN (Sequencing Consortium of Alcohol and Nicotine) consortium. 32
Results
Genetic instruments for frailty index
The 14 SNPs were used to instrument FI (Supplementary Table 5), explaining ≈0.32% phenotypic variation. F statistics of these instrumental variables ranged from 166 to 212, indicating a low risk of weak instrument bias. We had 100% statistical power in detecting causal effects of FI and functional outcome after ischemic stroke.
Functional outcome after ischemic stroke
We found MR evidence that genetic liability to frailty in relation to worse functional outcome after ischemic stroke in the primary analysis (fixed-effects IVW: odds ratio (OR) = 5.12; 95% confidence interval (CI) = 1.31–20.09; p = 0.019; Figure 1). Results from random-effects IVW (OR = 5.12; 95% CI = 1.24–21.13; p = 0.024), MR-RAPS (OR = 4.94; 95% CI = 1.10–22.31; p = 0.038), and weighted median (OR = 3.05; 95% CI = 0.45–20.78; p = 0.254) yielded similar estimates, although less precise. MR-PRESSO outlier test did not identify potential SNP outlier, and Cochran’s Q test suggested no heterogeneity among individual SNPs (Cochran’s Q = 13.98; p = 0.375; Table 1). The scatterplot, forest plot, and leave-one-out plot provided additional support that the link between FI and worse 3-month stroke outcomes was not driven by any single SNP (Supplementary Figures 1–3). MR-PRESSO global test (p = 0.399) and MR-Egger intercept (intercept = −0.096; p = 0.505) suggested that horizontal pleiotropy did not exist. Additional analyses examining reverse MR (Supplementary Figure 4) and Steiger directionality test (Supplementary Table 6) did not indicate genetic liability to functional outcome affected FI.
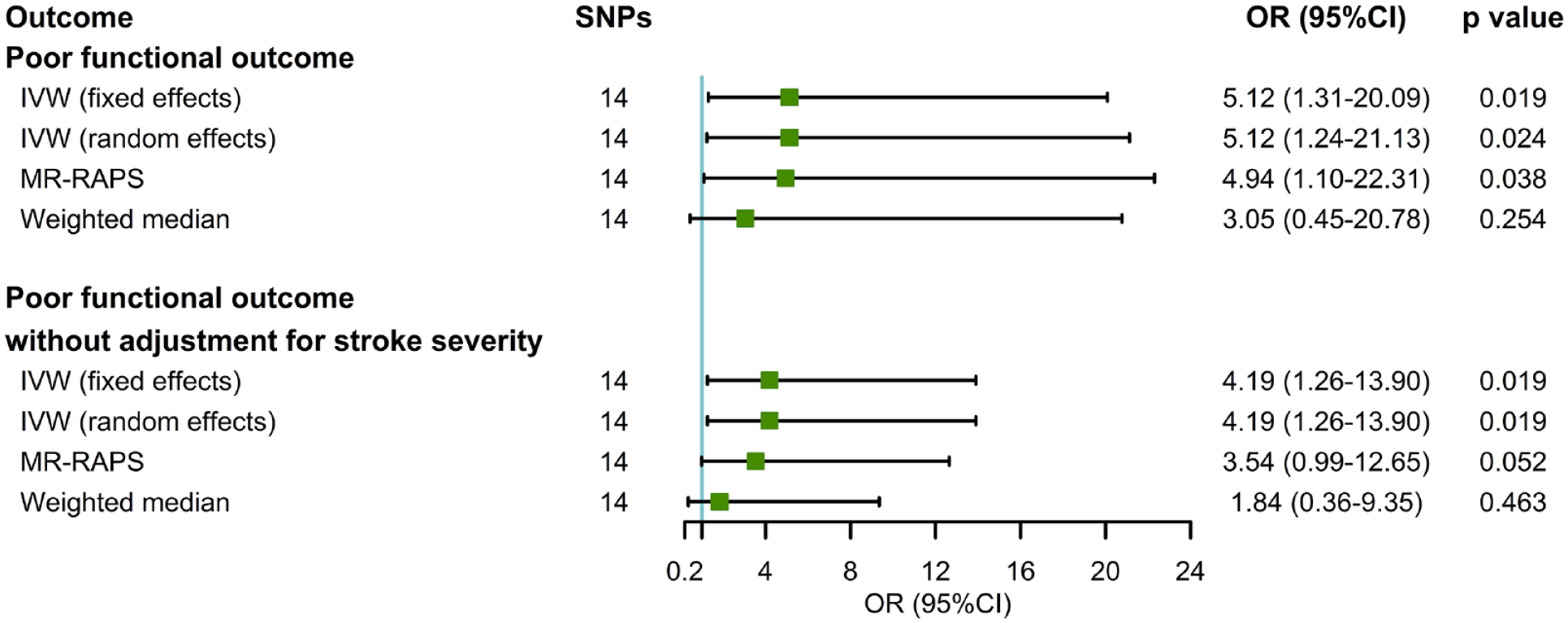
Complementary analyses of genetically determined frailty index (FI) with functional outcome after ischemic stroke. SNP: single-nucleotide polymorphism; OR: odds ratio; CI: confidence interval; IVW: inverse-variance weighted; MR-RAPS: Mendelian randomization-robust adjusted profile score.
Heterogeneity and horizontal pleiotropy tests of frailty index (FI) causally linked to functional outcome after ischemic stroke and ischemic stroke risk.
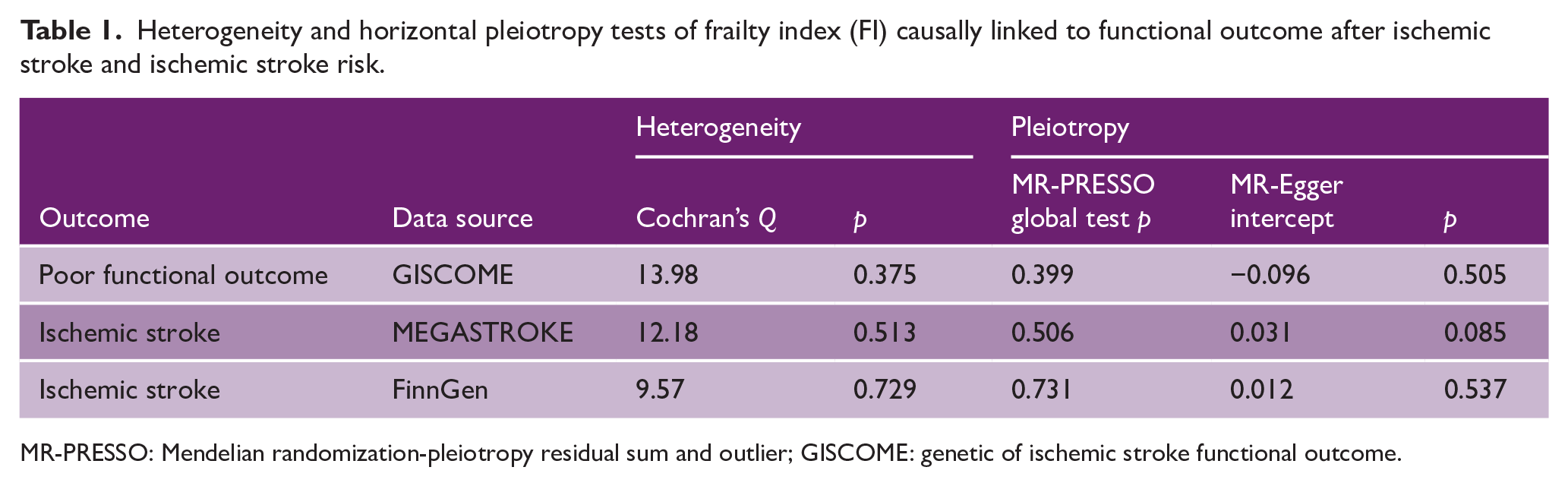
MR-PRESSO: Mendelian randomization-pleiotropy residual sum and outlier; GISCOME: genetic of ischemic stroke functional outcome.
Sensitivity analysis
We conducted several sensitivity analyses to assess the robustness of MR findings for poststroke outcomes. In models without adjustment for baseline stroke severity, genetic predisposition to frailty was associated with poor functional outcome after ischemic stroke (OR = 4.19; 95% CI = 1.26–13.90; p = 0.019; Figure 1). Complementary analyses including MR-RAPS and weighted median were in accordance with the prior result. In addition, because fatigue is known to be a characteristic of frailty, we also performed MR analyses for self-reported fatigue with 36 instruments (Supplementary Table 7). As expected, fatigue showed suggestive association with higher risk of an mRS score ⩾ 3 (OR = 4.37; 95% CI = 0.96–19.88; p = 0.056; Supplementary Figure 5). Results were directionally consistent when other MR methods (MR-RAPS and weighted median) were used and when stroke outcome unadjusted for baseline NIHSS.
To address the bias due to the prognostic nature of analyses, we first investigated the association of FI with ischemic stroke risk. Using the MEGASTROKE data, MR evidence suggested that genetically determined FI was not significantly associated with the risk of ischemic stroke (OR = 1.28; 95% CI = 0.98–1.67; p = 0.071; Supplementary Table 8). Results from other MR methods and replication analyses using FinnGen dataset further confirmed the lack of association with ischemic stroke risk, with little heterogeneity among SNPs (Table 1). MR-PRESSO global test (p > 0.05) and MR-Egger intercept (p for intercept > 0.05) also indicated no evidence of directional pleiotropy. Finally, the associations of genetically predicted FI and fatigue with poor functional outcome after ischemic stroke remained stable when stroke outcome effects adjusted for index event bias were used (Supplementary Table 9).
Multivariable Mendelian randomization analysis
In MVMR, the estimated effects of FI on functional outcome after ischemic stroke were still significant after adjusting for BMI (OR = 10.98; 95% CI = 2.48–47.84; p = 0.002), CRP (OR = 4.57; 95% CI = 1.01–20.74; p = 0.049), and smoking initiation (OR = 5.36; 95% CI = 1.33–21.55; p = 0.018), supporting an independent role of frailty in stroke prognosis (Figure 2). With the adjustment for IBD, the association was attenuated mildly, whereas the overall pattern of the association persisted (OR = 2.91; 95% CI = 0.72–11.79; p = 0.134).
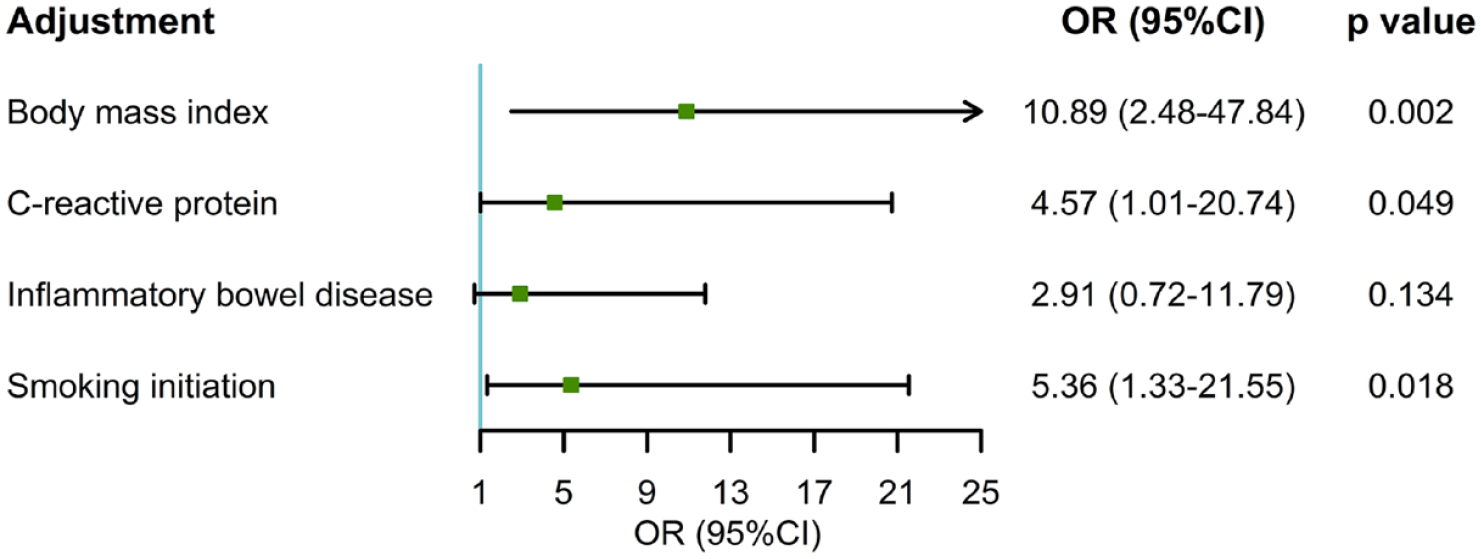
Multivariable Mendelian randomization analysis of the association between genetically predicted FI and poor functional outcome after ischemic stroke.
Discussion
We applied a two-sample MR approach to comprehensively evaluate the causal relationship between FI and functional outcome after ischemic stroke. The primary analysis suggested the genetic liability to frailty in relation to worse functional outcome at 3 months poststroke. The results were overall robust to sensitivity analyses and after adjustment for BMI, CRP, and smoking initiation, while the association was attenuated mildly when adjusting for IBD. Reverse direction analysis provided no evidence that genetic liability to poor functional outcome was associated with higher FI. What’s more, we did not identify a significant causal link between FI and ischemic stroke risk.
Frailty is a state of increased vulnerability to stressors due to the cumulative multisystem decline of physiological reserves to maintain homeostasis. Stroke represents a common stressor event; thus, it is possible that frailty affects clinical outcomes after stroke insults. Previous observational studies have reported the relationship between frailty and stroke prognosis. Clinical frailty was found to be an independent predictor of short-term and long-term adverse outcomes following ischemic stroke after adjustment for stroke severity.3–6 Similarly, a recent large-scale meta-analysis including 27,210 participants also showed that pre-stroke frailty is associated with poor outcomes, including death, disability, and increased length of stay. 7 Furthermore, observational evidence concluded that brain frailty on imaging (i.e. leukoaraiosis, atrophy, lacunes) is an important predictor for unfavorable mRS score at 90 days poststroke.33,34 However, the correlation between frailty and poor outcomes after stroke is not always consistent. Mediation analysis in a prospective longitudinal study suggested that pre-stroke frailty status is not related to worse functional outcome directly, but rather the effect is mediated by stroke severity. 35 The discrepancies may be partly explained by bias due to confounding and reverse causation from the limitations of observational epidemiological studies. Our present MR analysis can largely overcome such limitations and provided genetic evidence for the causal relationship between FI and poststroke outcomes. Notably, MVMR results demonstrated that functional outcome after ischemic stroke might be independently affected by the genetic liability to frailty.
The mechanisms by which frailty may increase the risk of poor outcome after stroke are largely complex. One explanation is that frailty is associated with a range of factors affecting stroke outcomes, including impaired physiological resilience, reduced neuroplasticity potential, 36 higher risk of poststroke delirium, 37 poststroke cognitive impairment, 38 and health care–associated infections. 39 Cumulative burden of risk factors, such as smoking, physical inactivity, poor diet, oxidative stress, and inflammation, are often shared in both cerebrovascular disease and frailty, which interact to further worsen the prognosis. 40 Besides, frailty is associated with impaired cerebral autoregulation and then further impedes functional recovery after ischemic stroke. 10 Frequently, because of a belief that patients perceived to be frail will not respond to treatments or have a higher risk of adverse events, frail individuals often receive reduced treatments, which may lead to greater occurrence of comorbidity and disability instead, further worsening their prognosis. 41 Moreover, several studies reported treatment-modifying effects of frailty upon hyperacute reperfusion therapies following stroke and consequently leading to poorer functional outcomes in frailty patients.42–44
Several limitations should be considered when interpreting our findings. First, causal effect estimates were imprecise resulting from the small fraction of the phenotypic variation explained. Second, residual pleiotropy may remain, despite a range of sensitivity analyses conducted. However, our MR results did not show significant evidence of heterogeneity, horizontal pleiotropy, or outlying effects. In addition, we implemented MVMR to adjust confounders, and the associations of FI with functional outcome persisted. Evaluating prognostic factors among disease patients may also introduce index event bias. Yet, we did not find that genetically predicted FI affects ischemic stroke risk. Therefore, it is unlikely that our analyses of functional outcome were severely affected by index event bias. When further correcting for index event bias, the effect estimates were generally unchanged. Third, functional outcome for ischemic stroke subtypes and hemorrhagic stroke outcomes could not be evaluated directly for the lack of available GWAS data. Fourth, measurement error may have been introduced due to the variation in the time that baseline NIHSS (0–10 days post stroke onset) 17 was measured, though the median time at NIHSS taken was 0 days after stroke, with interquartile range 0–1. What’s more, we lack data on other known determinants of outcomes such as reperfusion therapies and rehabilitation. 45 Fifth, a large proportion of the population used to obtain SNP effect estimates were obtained from the UK Biobank, which might induce selection bias due to low response rates. 22 Nevertheless, the FI for those in UK Biobank were not substantially different to corresponding values derived from health record data on the general population of the United Kingdom. 15 Moreover, due to the single question used to define fatigue, there may be some misclassification. Thus, additional researches are needed to assess whether our results replicate when better validated multi-item measures of fatigue are used. Similarly, future researches are required to confirm whether alternative methods to FI would be expected to give similar results. Finally, our analyses were restricted to individuals of European ancestry, so our findings may not be generalizable to other ethnic groups.
Conclusion
In conclusion, the present MR study suggested a possible causal effect of frailty on poor functional outcome after ischemic stroke. Future researches are required to investigate whether rational intervention on frailty may help to improve recovery after ischemic stroke.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930231194676 – Supplemental material for Genetic liability to frailty in relation to functional outcome after ischemic stroke
Supplemental material, sj-pdf-1-wso-10.1177_17474930231194676 for Genetic liability to frailty in relation to functional outcome after ischemic stroke by Huan Cai, Hao Zhang, Jialin Liang, Zhonghua Liu and Guozhi Huang in International Journal of Stroke
Footnotes
Acknowledgements
We gratefully thank the UK Biobank, TwinGene, GISCOME network, ISGC Cerebrovascular Disease Knowledge Portal, MEGASTROKE, FinnGen, GIANT, CIWG, IIBDGC, and GSCAN consortium for providing summary statistics data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (82072528) and Natural Science Foundation project of Guangdong Province (2022A1515012460).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
