Abstract
Introduction:
Contemporary estimates of the impact of type 2 diabetes (T2D) on stroke outcomes are important for care planning and resource allocation. This retrospective cohort study compared the incidence of stroke and subsequent clinical and economic outcomes following stroke among people with and without T2D.
Patients and methods:
Data were extracted from a subset of the French Système National des Données de Santé database. Stroke incidence as well as post-stroke clinical outcomes, healthcare resource utilization (HCRU), use of stroke preventative treatments, and costs were compared among people with and without T2D.
Results:
The study included 10,906 patients hospitalized for stroke including 2141 (19.6%) people with T2D. Age-standardized stroke incidence rates were 3.3 (95% confidence interval [CI] 3.1; 3.6) per 1000 person-years and 2.2 (2.1; 2.3) in people with and without T2D, respectively. Patients with T2D had significantly higher risk of stroke recurrence (adjusted hazard ratio [HR] 1.14, 95% CI: 1.01; 1.30) and mortality (HR 1.16, 95% CI: 1.08; 1.25) than patients without T2D. The proportion of patients with T2D treated with statins decreased from 27.3% pre-stroke to 20.6% post-stroke but remained relatively stable among patients without T2D (from 13.4% to 13.1%). The mean healthcare costs in the 12 months following stroke were higher among patients with T2D than those without (€22,635 vs €18,629).
Discussion and Conclusion:
The incidence and clinical and economic burden of stroke is considerably higher among people with T2D. Further efforts are needed to reduce this disparity.
This is a visual representation of the abstract.
Keywords
Introduction
Type 2 diabetes (T2D) is an independent risk factor for stroke, with people with T2D having an estimated two-fold increased risk of stroke as compared to the general population. 1 Modifiable risk factors for stroke including obesity, hypertension and dyslipidemia are prevalent in people with T2D, thereby increasing stroke risk further in T2D populations. Health outcomes, including mortality, stroke recurrence, and other cardiovascular disease (CVD) events following stroke are also worse among people with T2D.2,3 Less is known about the impact of T2D on post-stroke healthcare resource utilization (HCRU) and costs.
Clinical guidelines recommend aggressive management of modifiable risk factors through lifestyle modification strategies and prescription of pharmacological interventions, such as statins.2,3 This has yet to lead to reductions in the prevalence of diabetes in stroke populations. 4 With the advent of newer glucose-lowering agents such as glucagon-like peptide 1 receptor agonists (GLP-1 RA) with established CVD effects, the disparity in stroke incidence and adverse outcomes in people with and without T2D may be lessened.
Generating contemporary, benchmarking data relating to the impact of T2D on clinical and economic outcomes is important for informing stroke care planning, resource allocation and to address key evidence gaps. Using data from a nationally-representative administrative data source, this study compared the incidence of stroke and the incidence of clinical and economic outcomes following stroke among people with and without T2D.
Patients and methods
In this retrospective cohort study, data were obtained from a representative sample of the French national health database, the Système National des Données de Santé (SNDS). The SNDS collates health data from reimbursement claims and benefit data and so includes information on outpatient visits, hospitalizations and prescriptions. The representative sample, also known as the Echantillon Général des Bénéficiaires (EGB), includes data for 700,000 people.
The study population included adults (age ⩾ 18 years) who had an incident stroke requiring hospitalization between 1 January 2012 to 31 December 2018. Incident stroke events were identified using a previously described algorithm based upon hospitalizations with international classification of disease, version 10 (ICD-10) diagnostic codes for stroke (I60-I64, G45-G46). 5 Patients with any prior history of stroke were excluded. Index date was date of incident stroke hospitalization. Patients were followed from index date until the earliest of 31 December 2019 or date of death.
Patients were grouped according to T2D status at index date, ascertained using an algorithm which defined T2D people as those people with an ICD-10 diagnosis record of E11 in the long-term disease database or from a hospital discharge diagnosis, or using at least three records for reimbursements of oral antidiabetics or insulin prior to or at incident stroke hospitalization. Patients diagnosed with T2D following stroke were included in the without T2D patient group in the primary analyses. To account for the possibility that some of these patients were undiagnosed T2D patients at date of incident stroke and therefore at greater risk of adverse outcomes, sensitivity analyses were conducted whereby patients diagnosed with T2D in the 30 days following stroke were re-categorized as T2D patients.
Data on patient characteristics including age, sex and clinical characteristics were extracted. Comorbidities including previous history of CVD were assessed in the 24 months prior to index date to avoid misclassification, while use of stroke preventative treatments was assessed in the 12 months prior to index date to reflect treatments at time of incident stroke more accurately.
The clinical outcomes assessed during follow-up included stroke recurrence, hospitalizations for CVD, and all-cause mortality. Due to difficulties in distinguishing unique stroke events occurring in short time-frames, stroke recurrence was assessed from 21-days post stroke hospitalization discharge date until date of stroke recurrence, death, or end of follow-up. CVD hospitalizations from index date were defined as a hospitalization with an ICD-10 diagnosis code for coronary artery disease, cardiac arrhythmias, cerebrovascular disease (including stroke), peripheral arterial disease, heart failure or aortic disease, or thromboembolic disease. Treatment with stroke preventative treatments, defined as at least 1 reimbursement for a medication of interest in the 3- and 12 months following stroke was described among patients who were discharged alive from the index stroke hospitalization. The following HCRU was described: index stroke hospitalization length of stay, subsequent hospitalizations (day and overnight stay cases/overnight stays only/intensive care unit stays), outpatient physician visits according to specialty, physical rehabilitation therapy visits. Total annual healthcare costs were estimated for the 12 months post-stroke and full follow-up periods, including index date. Total annual healthcare costs included hospitalization, treatments, external consultations, and outpatient costs per patient and were estimated in Euros.
Crude incidence of stroke per 1000 person-years with corresponding 95% confidence intervals (CI) was estimated among people with and without T2D. Directly age-standardized incidence rates were calculated using the French population as the reference population.
Cox proportional hazards models were used to estimate adjusted hazard ratios (HRs) and 95% CIs for the impact of T2D on risk of clinical outcomes. To account for differences in the underlying CVD risk profiles of patients with and without T2DM, the Cox proportional Hazards models were adjusted for age, sex, baseline comorbidities (including previous CVD), and baseline CVD preventative treatments (e.g. statins). Death was treated as a competing risk for all non-mortality outcomes. In sensitivity analyses, patients diagnosed with T2D during follow-up were censored.
Results
The study cohort comprised of 10,906 patients hospitalized for incident stroke, including 2141 patients with T2D. Compared to stroke patients without T2D, patients with T2D were older (75.0 vs 71.5 years, p-value < 0.0001) and were more likely to be male (54.5% vs 47.4%, p-value < 0.0001; Table 1). There were statistically significant differences in history of cardiovascular disease and vascular risk factors in patients with T2D compared with patients without T2D. Specifically, patients with T2D were more likely to have a history of coronary artery disease (15.6% vs 7.1%, p-value < 0.0001), cardiac arrhythmia (20.0% vs 15.0, <0.0001), chronic heart failure (6.1% vs 12.2%, p-value < 0.0001), chronic kidney disease (3.7% vs 10.3%, p-value < 0.0001), hypertension (56.7% vs 82.9%, <0.0001) and dyslipidemia (32.1% vs 63.1%, p-value < 0.0001).
Baseline characteristics among stroke patients in France according to T2D status.

CCI: Charlson comorbidity index; DPP-4: Dipeptidyl peptidase-4; GLP-1 Ras: Glucagon-like peptide-1 receptor agonists; SD: standard deviation; SGLT2: Sodium-glucose cotransporter-2; T2D: type 2 diabetes.
Excludes stroke.
More patients with T2D received antiplatelets (46.6% vs 25.6%, p-value < 0.0001), statins (27.2% vs 13.2%, p-value < 0.0001) and anti-hypertensives (74.5% vs 48.7%, p-value < 0.0001) pre-stroke than patients without T2D. Use of stroke preventative treatments was highest among patients with previous CVD in both patients with and without T2D compared with patients with no previous CVD (Supplemental Table S1).
The overall crude stroke incidence rate (95% CI) was 2.5 (2.4; 2.5) per 1000 person-years, while the age-standardized incidence rates were 3.3 (3.1; 3.6) and 2.2 (2.1; 2.3) per 1000 person-years in people with and without T2D, respectively.
During a median (interquartile range, IQR) follow-up of 2.7 (1.0; 4.8) years, 14.2% of patients had a stroke recurrence and the risk of recurrence was significantly higher among T2D patients compared to those without T2D (adjusted HR: 1.14, 95% CI: 1.01; 1.30; Supplemental Table S2, Figure 1). The risk of any CVD outcome was significantly higher among patients with T2D than patients without T2D (adjusted HR: 1.19 [1.11; 1.28]). Patients with T2D had a significantly increased risk of all individual CVD outcomes, excluding aortic disease and cardiac arrhythmia disease compared with patients without T2D (Figure 1). Mortality risk was markedly higher among patients with T2D compared to patients without (adjusted HR: 1.16, 1.08; 1.25; Figure 1). Findings were similar after including patients diagnosed with T2D in the 30 days following stroke in the T2D group (n = 29) and on censoring at T2D diagnosis post-stroke (Supplemental Table S3).

Adjusted hazard ratios for clinical outcomes in patients with T2D compared to patients without T2D.
A greater proportion of patients who were discharged alive from incident stroke hospitalization (N = 9471, including 1800 T2D) were prescribed stroke preventative treatments in the 12 months following stroke than in the 12 months pre-stroke (Figure 2). However, the use of statins and antihypertensives declined in patients with T2D (statins: 27.3% to 20.6%, anti-hypertensives 74.8% vs 67.5%) but not among patients without T2D. Similar patterns were observed when the use of stroke preventative treatments in the 12 months pre-stroke was compared to their use 3-months post-stroke (Supplemental Figure S1).
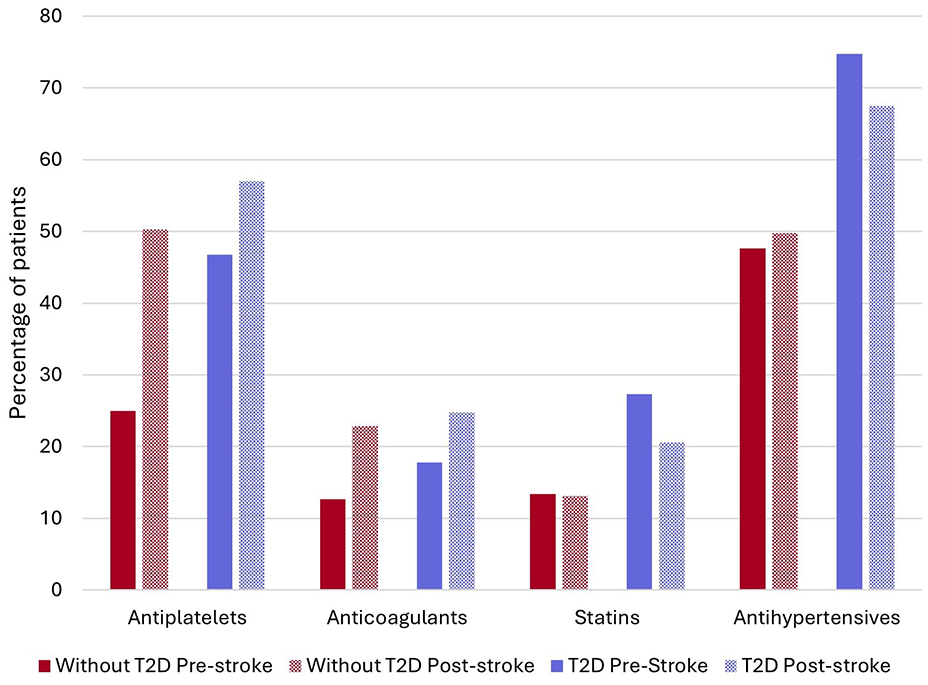
Stroke preventative treatments pre and 12 months post stroke according to T2D status.
Incidence rates of subsequent hospitalizations were almost two-times higher among patients with T2D than patients without T2D (2.51 vs 1.32 per person-year; Table 2).
HCRU and costs among stroke patients according to T2D status.

CI: confidence interval; HCRU: healthcare resource utilization; IQR: interquartile range; SD: standard deviation; T2D: type 2 diabetes.
Includes index admission.
The mean length of stay of subsequent hospitalizations was longer in patients with T2D than in patients without T2D (4.94 (6.64) vs 4.19 (7.2) days, p-value < 0.0001). The rate of neurologist visits following stroke was higher among patients without T2D than patients with T2D (0.22 vs 0.19 per person year, p-value < 0.0001). The mean (SD) total healthcare costs in the 12 months following stroke were €22,635 (25,154) in patients with T2D and €18,629 (24,180) in patients without T2D (p-value < 0.0001). The mean total yearly healthcare costs were €18,660 (21,115) in patients with T2D and €14,265 (19,583) in patients without T2D (p-value < 0.0001).
Discussion
Key findings
In this large cohort study, people with T2D had elevated prevalence of CVD risk factors such as hypertension, dyslipidemia and history of CVD in patients with T2D compared to patients without T2D. Even after adjusting for these differences, patients with T2D had higher risk of stroke and adverse stroke outcomes compared to people without T2D. As a result, the HCRU and economic implications of T2D following stroke are substantial, with hospitalizations, outpatient visits and total yearly costs higher among T2D patients than patients without T2D.
Despite the excess risk of stroke in the T2D population, the use of stroke preventative therapies, such as statins, was low pre and post-stroke highlighting the need to reinforce preventative strategies in this population.
Relation to other studies
That T2D confers an excess risk of stroke, particularly ischemic stroke, has been well established in population-based studies. 6 There are fewer data relating to the impact of T2D on stroke outcomes. Most previous studies have documented an excess risk of adverse outcomes, including stroke recurrence, long-term mortality, and CVD outcomes in patients with T2D following stroke.7–12
Our findings align closely with those from the Get With The Guidelines-Stroke, a US-based study which used a registry of over 1 million stroke patients from 2003 to 2011. 8 In that study, T2D conferred a 24% excess risk of mortality, as compared to 16% in the present study. Similarly, patients with T2D had a 17% excess risk of stroke readmission compared to patients without T2D, while the excess risk was 14% in the present study. In an Australia-based study of 27,802 unique ischemic stroke events, patients with diabetes had an excess risk of CVD (aHR: 1.14 [1.07; 1.21]) and stroke readmissions (aHR: 1.17 [1.06; 1.29]) compared to patients without diabetes. 12 In a meta-analysis including 27 studies, diabetes increased the risk of ischemic stroke recurrence by 50%. 7
Given the excess risk of stroke among people with T2D, American Diabetes Association (ADA)/European Association for the Study of Diabetes (EASD) and European Society of Cardiology guidelines recommend aggressive cardiovascular risk-reducing strategies in patients with T2D to prevent CVD events.3,13,14 ADA/EASD guidelines recommend that moderate-intensity statin therapy is initiated in all people with diabetes aged 40–75 years without previous CVD. 14 For people with diabetes and previous CVD, guidelines recommend high-intensity statin therapy for secondary prevention. Despite these recommendations, only 27.3% of patients with T2D and only 28.1% of patients with T2D and history of CVD had been prescribed statins in the 12 months prior to stroke. Only 20.6% of people with T2D were prescribed statins in the 3 months following stroke. The under-use of statin therapy in patients with and without T2D has been previously documented in France and elsewhere.15,16 There are several potential explanations for this observed finding. First, this observation may reflect significant clinical inertia in managing individuals with and without T2D, highlighting missed opportunities for stroke and other CVD secondary prevention. Second, given that 40.4% of patients were aged 80 years or older, clinicians may have opted against initiating statin therapy in these patients due to concerns regarding net benefits of statin treatment.17,18 Third, drugs prescribed in institutions such as hospices and rehabilitation centers may not be captured in the EGB resulting in an underestimation of treatment use, a problem likely exacerbated by the high proportion of elderly patients in the study population. Finally, misleading claims surrounding the safety of statins as well as concerns regarding the potential for statins to increase the risk of intracerebral hemorrhage and accelerate diabetes progression may have contributed to the under-use of these agents.19,20
Stroke is associated with substantial HCRU and costs, particularly in the 12 months post-stroke. Estimates derived from the present study align closely with those of previous studies in which the mean total healthcare costs in the 12 months following stroke was €16,686 in 2007 in France as compared to €19,416 estimated in the current study. 21 However, the full economic impact of stroke is likely considerably greater once costs associated with nursing care, private healthcare and productivity losses are taken into account. Our finding that T2D is associated with elevated HCRU and costs following stroke corroborates with findings from previous studies conducted in the US.22,23
Clinical guidelines also recommend the use of glucose-lowering therapies with established CVD benefits, such as SGLT2 inhibitors and GLP-1 RAs in patients with established CVD or at high risk of CVD, regardless of glycemic control .13,24 Evidence from a meta-analysis of clinical trials indicate that SGLT-2 inhibitors are associated with a reduced risk of major adverse CV events (HR: 0.90 [0.85; 0.95]) but are not associated with reduced risk of first stroke (HR: 0.96 [0.87; 01.07]). 25 A meta-analysis of GLP-1 RA clinical trials demonstrated that GLP-1 RAs can reduce the risk of all CVD outcomes in patients with T2D with the greatest impact on fatal and non-fatal stroke (HR: 0.83 [0.76–0.92]). 26 In addition, there was no evidence to indicate the effect of GLP-1 RAs on CVD risk varied among T2D patients with and without established CVD at baseline. GLP-1 RAs therefore offer the potential to reduce the excess risk of stroke in T2D and therefore reduce the considerable clinical and economic burden of stroke overall. Further studies are required to assess the extent to which the more widespread use of such therapies have modified the real-world excess risk of stroke among people with T2D.
Strengths and limitations
A key strength of this study is the use of a relatively large representative sample which enabled the comprehensive description of the disparities in outcomes following stroke according to T2D.
There are several important limitations of this study. First, data were unavailable for key confounders such as body mass index, blood pressure and lipid profiles and so differences in these characteristics according to T2D status could not be accounted for in multivariate analyses. Adjusted HRs may therefore be affected by residual confounding. Another key limitation of this study was the need to assign an arbitrary time frame of 21 days after index hospitalization discharge from which to identify recurrent stroke events. In addition, cases of some clinical outcomes may have been underestimated due to their identification using diagnostic codes alone. Lastly, this study focused on CVD outcomes and mortality and so did not capture differences in other post-stroke outcomes such as cognitive decline and patient quality of life according to T2D.
Conclusions
People with T2D are at increased risk of stroke and worse outcomes following stroke and subsequently higher HCRU and costs. Despite this, people with T2D do not appear to be receiving guideline-recommended treatments for optimal secondary prevention of stroke outcomes. Given the growing prevalence of T2D due to aging populations and increasing survival, efforts are required to reduce stroke incidence among people with T2D and reduce the number of patients experiencing adverse clinical outcomes.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251319172 – Supplemental material for Type 2 diabetes in stroke patients: Impact on outcomes, recurrence, resource use, and costs in France
Supplemental material, sj-docx-1-eso-10.1177_23969873251319172 for Type 2 diabetes in stroke patients: Impact on outcomes, recurrence, resource use, and costs in France by Igor Sibon, Kamel Mohammedi, Nadia Quignot, Stephanie Read, Raissa Kapnang, Artak Khachatryan, Kazue Kikuchi, Hongye Ren, Christine Massien, Lucile Vigié, Sara Larsen and Laurent Fauchier in European Stroke Journal
Footnotes
Acknowledgements
Data were provided by the French Caisse Nationale d’Assurance Maladie (CNAM) and its staff involved in the project, in particular the DEMEX team. Support during the data application process was provided by the Health Data Hub.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IS reports consultant activities for Bayer, BMS/Pfizer, Boehringer Ingelheim, Medtronic, Novartis, Novo Nordisk, Bioprojet, Astra-Zeneca, Alexion, and speaker activities for AstraZeneca, Bayer, BMS/Pfizer, Boehringer Ingelheim, Medtronic, Novartis, Novo Nordisk. KM reports consulting fees from Novo Nordisk; honoraria for lectures, presentations, or speaker bure aus from Novo Nordisk, Astra Zeneca, Boehringer-Ingelheim, Eli Lilly, Sanofi, Lifescan, Abbott, and Bayer; and participation to Advisory Board from Novo Nordisk, Sanofi and Amarin. NQ, SR, RK, AK, and KK are full-time employees at Certara, which received consulting fees to independently conduct this study. HR, CM, LV, and SL are all full-time employees of Novo Nordisk. LF reports consultant activities for Bayer, BMS/Pfizer, Boehringer Ingelheim, Medtronic, Novartis, Novo Nordisk and XO, and speaker activities for AstraZeneca, Bayer, BMS/Pfizer, Boehringer Ingelheim, Boston Scientific, Medtronic, Novartis, Novo Nordisk and Zoll.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by Novo Nordisk.
Ethical considerations
In accordance with the simplified procedure approved by the French data protection authority (Commission Nationale de l’Informatique et des Libertés, CNIL; deliberation 2020–072 of July 16, 2020), this study was endorsed by an independent scientific committee and approved by the Health Data Hub on April 19, 2022 (EGB-007).
Consent to participate
This study is a non-interventional study not involving the human being, leveraging only anonymized structured data, which according to applicable legal requirements do not contain data subject to privacy laws. Hence, obtaining informed consent from patients was not required.
Consent for publication
As this study involved anonymized structured data, which according to applicable legal requirements do not contain data subject to privacy laws, obtaining consent to publish from patients was not required.
Guarantor
Professor Igor Sibon (IS).
Contributorship
Protocol development: IS, KM, NQ, AK, KK, HR, CM, LV, SL, LF.
Study design and implementation: NQ, KK, HR, SL.
Data management/analysis: RK.
Results interpretation: IS, NQ, SR, HR, CM, LV, SL, LF.
Manuscript writing and/or editing: IS, KM, NQ, SR, AK, KK, HR, CM, LV, SL, LF.
Data availability
Data were shared by the French national health insurance: “Caisse Nationale d’Assurance Maladie” (CNAM). Data are available from The French National Healthcare Data System. Requests for data access should be made to the Health Data Hub: Contact | Health Data Hub; Plateforme de données de santé - Health Data Hub, 9 rue Georges Pitard, 75015 Paris’.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
