Abstract
Background:
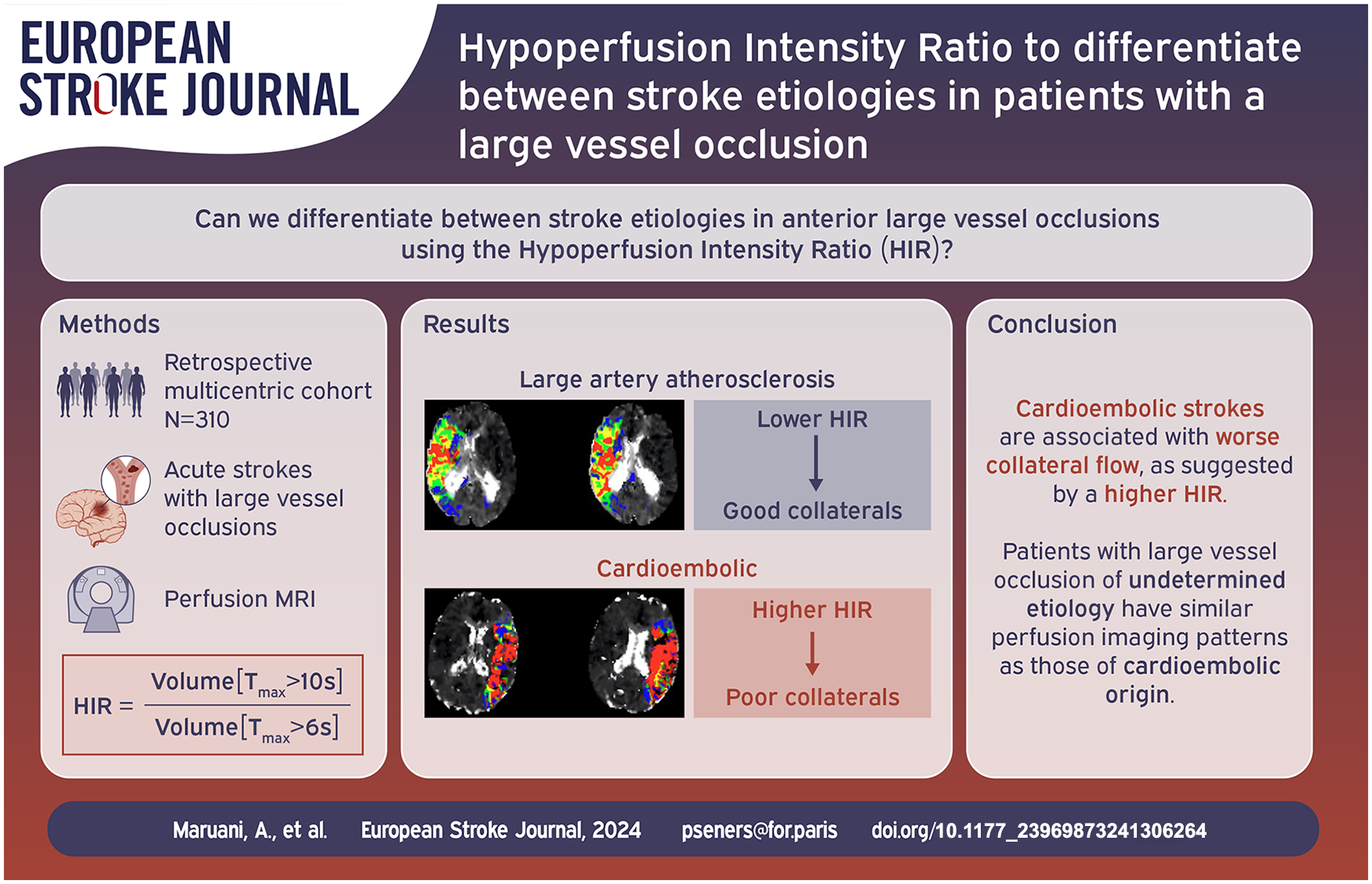
Collateral circulation plays a key role in acute ischemic stroke. We sought to determine the association between the arterial collateral status, estimated by the Hypoperfusion Intensity Ratio (HIR) on perfusion MRI, and stroke etiology in anterior circulation large vessel occlusion (LVO).
Methods:
We retrospectively analyzed anterior circulation LVO acute stroke patients with a baseline perfusion MRI performed within 24 h from symptom onset. To avoid selection bias, patients were chosen from (1) the prospective registry of one comprehensive stroke center that included both thrombectomy-treated and untreated patients with systematic use of perfusion MRI and (2) one prospective thrombectomy study where perfusion MRI was acquired per protocol, but treatment decisions were made blinded to the results. Stroke etiology was evaluated according to the TOAST classification. HIR, defined as the proportion of time-to-maximum (Tmax) > 6 s with Tmax > 10 s volume, was measured on perfusion imaging. The relationship between stroke etiology (large artery atherosclerosis [LAA]) versus cardioembolism [CE]) and HIR was assessed by bivariate then multivariable binary logistic regression analyses.
Results:
Among the 310 included patients, stroke etiology was CE in 178 (57%) and LAA in 51 (16%). Patients with CE stroke etiology had higher HIR (0.43 vs 0.31,
Conclusion:
CE etiology is associated with worse collateral circulation in LVO-related acute stroke patients.
Keywords
Introduction
In acute ischemic stroke with a large vessel occlusion (LVO), the leptomeningeal collaterals – that is, the alternative vascular network that provides residual blood flow to ischemic areas downstream of LVO – are of major prognostic importance. Poor collateral flow is associated with more severe baseline neurological symptoms, larger infarct size, subsequent infarct growth, resulting in worse 3-month functional outcome.1–3 At admission, leptomeningeal collaterals can be assessed either directly using CT-angiography or indirectly on CT- or MR- perfusion imaging with tools such as the hypoperfusion intensity ratio (HIR). The HIR reflects the percentage of the perfusion lesion that has severely delayed contrast arrival times and correlates with the arterial collateral status determined by conventional angiography4,5 and CT-angiography. 6
Several studies have described the factors associated with better collateral flow, such as younger age, female sex, statin use, low glucose levels, absence of history of hypertension, clot characteristics3,7–10. However, whether the etiology of stroke is related to the collateral circulation has not yet been adequately described. A few studies have shown an association between an atherosclerotic etiology and good collaterals, evaluated either on CT-angiography11,12 or MR-perfusion-imaging.13,14 Yet, these studies were limited by moderate sample sizes,11,13 the lack of comparison to another homogeneous etiology subgroup (e.g. cardio-embolism) 14 or inclusion limited to patients receiving thrombectomy,11–13 which may have biased the association observed because patients with poor collaterals are less frequently treated with thrombectomy.
Here, we aimed to describe the relationship between leptomeningeal collateral flow as estimated by the HIR and stroke etiology. To this end, we used a large multicenter cohort of unselected patients with LVO-related acute stroke with baseline perfusion imaging on MRI. Our hypothesis was that patients with LVO of cardioembolic (CE) origin had worse collaterals than strokes caused by large artery atherosclerosis (LAA).
Methods
Study design and data sources
We retrospectively analyzed data from patients with LVO-related acute ischemic stroke extracted from (1) the prospective registry of one comprehensive stroke center with systematic use of perfusion MRI for all acute stroke candidates, which included both thrombectomy-treated and untreated patients (Rothschild Foundation Hospital, Paris, France) and (2) a French early time-window thrombectomy study (FRAME) in which MR-perfusion imaging was acquired per protocol, but treatment decisions were made by experts blinded to the results (
Our research was approved by the Rothschild Foundation Hospital review board
Clinical data
The following variables were prospectively collected in each cohort: age, gender, vascular risk factors, National Institute of Health Stroke Scale (NIHSS) score on admission, baseline systolic blood pressure and baseline glycemia.
Stroke etiology evaluation
The clinical reports for at least 3-month following stroke onset and up to 3 years were carefully reviewed for each patient to determine stroke etiology. In the main analysis, stroke etiology was classified according to the TOAST classification. Patients in whom no stroke etiology was found despite complete work-up, those with incomplete work-up or in whom competing etiologies were found were classified as “Undetermined” (UD). According to international guidelines, atrial fibrillation detected after stroke was defined as: atrial fibrillation lasting more than 30 seconds on Holter or more than 6 minutes on cardiac monitor. In a sensitivity analysis, the stroke etiology was classified according to the atherosclerosis, small vessel disease, cardiac source, other cause, dissection (ASCOD) grading system. 18 Patients were classified as LAA if coded A1 or A2 and no other cause graded 1 or 2; and were classified as CE if coded C1 or C2 and no other cause graded 1 or 2. In both analyses, patients in whom no stroke etiology was found or in whom two etiologies were present simultaneously were classified as undetermined (UD).
Radiological data
All included patients underwent MRI on admission, which systematically included diffusion-weighted imaging, T2* gradient echo (FRAME) or susceptibility-weighted imaging (Paris), FLAIR, intracranial and cervical MR-angiography, and perfusion-weighted imaging. One stroke neurologist with 10 years of clinical expertise in stroke imaging reviewed all MRIs, blinded to the stroke etiology. The following variables were collected (1) infarct volume, defined as an apparent diffusion coefficient <620 × 10−6 mm2/s; (2) time-to-maximum (Tmax) > 6 s and >10 s volumes; (3) mismatch volume, defined as Tmax > 6 s volume − core volume; (4) leptomeningeal collateral status as assessed by HIR, defined as the proportion of Tmax > 6 s volume with Tmax > 10 s (i.e. Tmax > 10 s volume/ Tmax > 6 s volume), low HIR indicating milder hypoperfusion and better collaterals; (5) intracranial occlusion site on MR angiography, subdivided into intracranial ICA, M1, and proximal M2; the M1 segment being defined as the first part of the middle cerebral artery up to the main bifurcation; (6) cervical ICA occlusion or severe >70% stenosis and (7) susceptibility Vessel Sign (SVS) on the susceptibility-weighted imaging or T2*-weighted gradient echo, defined as dark blooming artifact visible at the site of occlusion with a diameter exceeding the diameter of the contralateral vessel. Both diffusion-weighted imaging and perfusion imaging were automatically processed by RAPID software (iSchemaView, MenloPark, CA). Core and Tmax maps were visually checked, and artifacts were manually removed using home-made software whenever necessary.
Statistical analysis
Continuous variables were described as median (interquartile range (IQR)) and categorical variables as numbers and percentages. For the main analysis, patients with CE and LAA etiology according to the TOAST classification scheme were compared (patients with undetermined etiology were excluded). Bivariate relationship between stroke etiology (CE vs LAA) and baseline clinical and radiological characteristics were assessed using the Mann-Whitney
Results
Baseline characteristics
Overall, 310 patients met study inclusion criteria (see flow chart shown on Figure 1). Median age was 74 years (IQR 62–83), and 50% were male. Median baseline NIHSS score was 16 (IQR 9–21). Baseline imaging was performed at a median delay of 162 min (IQR 102–282) following symptoms’ onset. Occlusion site was ICA in 24%, M1 in 50% and proximal M2 in 26%. Median core volume was 18 ml (IQR 6–50) and median HIR was 0.41 (IQR 0.27–0.54). According to the TOAST classification, stroke etiology was categorized as LAA in 16% (51/310) of patients, CE in 57% (178/310) and UD in 27% (81/310). The full baseline characteristics are presented in Supplemental Table 1.

Flow chart of included patients.
Bivariate comparison of patients with CE versus LAA etiology
Clinical characteristics
The baseline characteristics of patients stratified according to stroke etiology (CE vs LAA) are detailed in Table 1. CE patients were older, less frequently male and fewer current smokers. At baseline, they had a lower systolic BP and a higher NIHSS score.
Bivariate comparison of the clinical features of patients with CE vs. LAA etiology.

Abbreviations: LAA = Large Artery Atherosclerosis; CE = Cardio-Embolism; NIHSS = NIH Stroke Scale; BP = Blood Pressure.
2 missing values in each group.
1 missing value in LAA group, 2 missing values in CE group.
Categorical variables are expressed as n (%) and continuous variables as median [interquartile range].
Radiological characteristics
Imaging characteristics according to etiology subgroup are described in Table 2. Patients in the CE group had larger Tmax > 10 s volume, higher HIR (indicating worse collaterals) and less frequent cervical ICA occlusions as compared to those with LAA etiology. There was a trend toward a larger core volume in CE patients, although this did not reach statistical significance. Time from last-known-well to imaging was similar between the two groups. There was no difference in terms of SVS presence or site of intracranial occlusion. The different perfusion patterns between CE and LAA patients are illustrated in the Figure 2.
Bivariate comparison of imaging features of patients with CE vs. LAA etiology.

Abbreviations: LAA = Large Artery Atherosclerosis; CE = Cardio-Embolism; LKW = Last Known Well; ASPECTS = Alberta Stroke Program Early CT Score; Tmax = time to maximum; HIR = hypoperfusion intensity ratio; ICA = intracranial internal carotid artery; M1 and M2 = first and second segment of the middle cerebral artery; ECA = Extracranial Carotid Artery.
Categorical variables are expressed as n (%) and continuous variables as median [interquartile range].
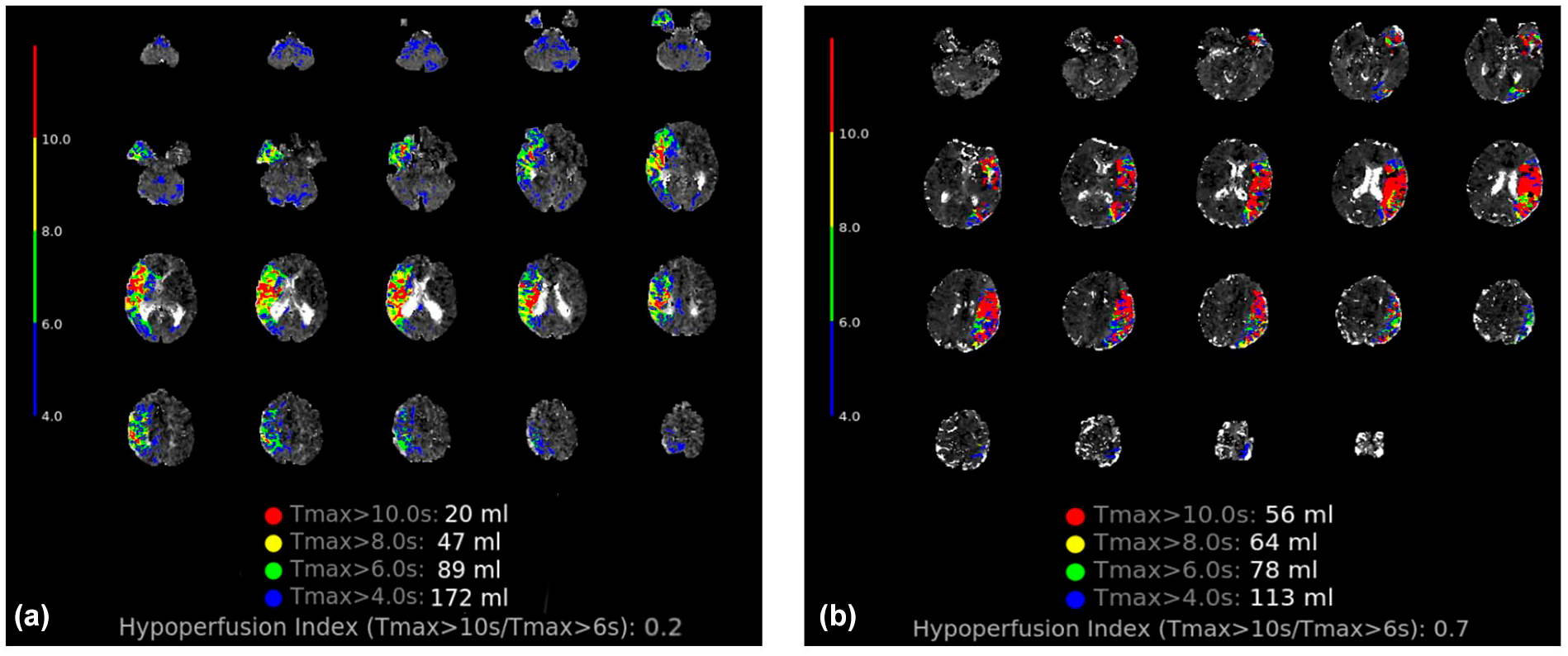
Example of perfusion characteristics from the two groups: (a) patient from the LAA group, NIHSS 8 at symptoms onset, perfusion MRI revealed a right MCA infarct with a M1 occlusion and a large mismatch between the Tmax > 10 s volume and Tmax > 6 s; HIR = 0.2 and (b) patient from the CE group, NIHSS 24 at symptoms onset, perfusion MRI revealed a left MCA infarct with a M1 occlusion and a small mismatch between the Tmax > 10 s volume and Tmax > 6 s volume; HIR = 0.7.
Association between HIR and stroke etiology in multivariable analysis
In multivariable analysis, higher HIR (indicating worse collaterals) was independently associated with CE etiology (OR = 1.5 per each 0.1 HIR increase; 95%CI [1.24–1.81];
Sensitivity analysis focusing on ICA/M1 occlusions
When focusing on patients with proximal occlusions (ICA or M1), the clinical and imaging differences according to stroke etiology were more pronounced (Supplemental Table 2). The core volume was significantly larger in the CE group (18 vs 11 ml,
Sensitivity analysis using ASCOD classification to classify CE versus LAA etiology
Using the ASCOD classification, 58 patients were classified as LAA etiology, and 186 as CE etiology, which was consistent with previous work.
20
The comparison of these two groups is presented in Supplemental Table 3. In multivariable analysis a similar independent association was found between the HIR and CE etiology (OR = 1.4 per each 0.1 HIR increase; 95%CI [1.18–1.61];
Comparison of HIR between patients with stroke of unknown etiology and those of LAA and CE etiology
81 patients suffered a stroke of unknown etiology (UD) based on the TOAST classification; their baseline characteristics can be found in Supplemental Table 4. There were no differences between the CE and UD subgroups. Compared to the LAA group, the UD patients had a significantly larger core volume (29 vs 12 ml,
Comparison of patients with stroke of unknwon etiology vs. those with LAA or CE etiology.
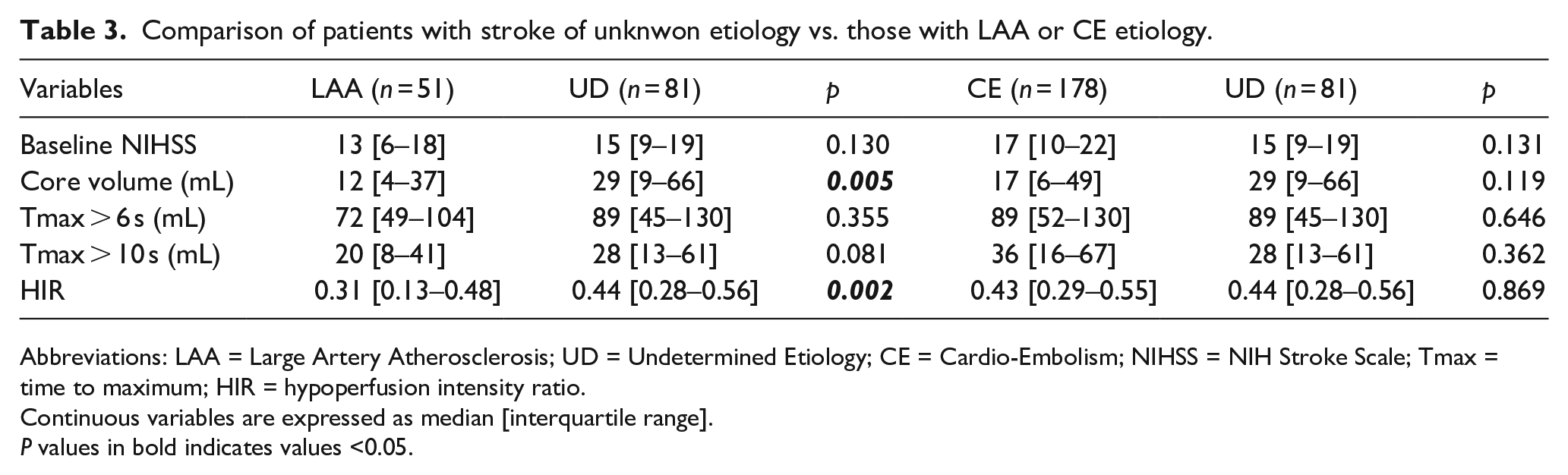
Abbreviations: LAA = Large Artery Atherosclerosis; UD = Undetermined Etiology; CE = Cardio-Embolism; NIHSS = NIH Stroke Scale; Tmax = time to maximum; HIR = hypoperfusion intensity ratio.
Continuous variables are expressed as median [interquartile range].
Discussion
In this large multicenter study of 310 unselected LVO-related acute stroke patients with baseline perfusion imaging, we found that strokes of cardioembolic origin had higher HIR, indicating less developed collateral vascularization, than those of atherosclerotic origin. We also found that strokes of undetermined etiology tended to have a similar radiological profile, including HIR, to cardioembolic strokes.
In our cohort, strokes of cardioembolic origin were most common, while large artery atherosclerosis accounted for approximately 15% of all patients. This is consistent with previous reports in similar LVO-population12,13,21 although the slightly higher prevalence of cardio-embolism in our study could be explained by a longer follow-up period. Concurring with the results of previous studies, patients in the CE group were older, less likely to be smokers and male, and they tended to have lower blood pressure at the time of stroke.12,13,22,23 Strokes were more severe in the CE group, both clinically with a higher NIHSS and radiologically with higher HIR, which is also consistent with previous reports.11–14
To our knowledge it is the first large, multicenter study to use HIR as a marker of collateral status to compare large vessel occlusions of cardiac or atherosclerotic origin, regardless of the treatment performed. The use of HIR as a collateral marker has several advantages. First, compared with subjective visual assessment of collaterals in CT-angiography or digital subtraction angiography, HIR can be generated automatically by post-processing imaging software, thereby limiting inter-observer variability, reported with other imaging modalities. Second, HIR has the advantage of measuring collateral status in a continuous manner. Third, it reflects the severity of hypoperfusion at the level of the brain parenchyma and thus assesses tissue perfusion through large collaterals pathways but also through smaller vessels not visible on standard arterial imaging, as well as cerebral venous outflow. Finally, HIR evaluation is very similar across imaging modalities (CT vs MRI). 24
In line with previous studies evaluating the collateral circulation on visual inspection on invasive or non-invasive arterial imaging, we observed an independent association between higher HIR, indicating poorer collateral circulation, and CE etiology. Most previous works12,13 comparing perfusion parameters between strokes of LAA and CE origins were based on endovascular treated patients. This may have biased the association since patients with poor collaterals are less frequently treated with thrombectomy.
The physiology behind collateral recruitment associated with large vessel atherosclerosis may result from several mechanisms. Chronic ischemia has been shown to be associated with neoangiogenesis in animal models. 25 In humans, stenosis of a large vessel leads to brain hypoxemia by reducing blood flow but also by repeated microembolism. Moreover, in patients with atherosclerosis of the carotid artery, the quality of collateral vascularization has been shown to be related to the degree of stenosis. 26 In contrast, the brutal onset of stroke of cardiac origin could explain the lack of acute collateral recruitment.27,28
We found the stroke of undetermined etiology had similar clinical and perfusion profiles than those of CE etiology, but different than those of LAA etiology. This may be explained by the predominance of cardioembolic sources in stroke of undetermined etiology, 29 or the lack of collateral development in patients with atherosclerotic lesions leading to mild stenosis.
Our study has several strengths. First, the multicenter design provided a large sample size that allowed us to reliably assess the association between HIR and the stroke etiology. Second, perfusion MRI was systematically performed in all participating centers, and patients with both treated and untreated LVO were included, reducing selection bias. Third, centralized imaging was performed for the whole dataset, ensuring a standardized assessment of key imaging variables. In particular, the same post-processing software was used to ensure uniform assessment of core volume and HIR. Lastly, stroke etiology was classified using two different classifications (TOAST and ASCOD), which provided consistent results.
This study also has limitations. First, we did not distinguish between stroke mechanisms (artery-to-artery embolism or in-situ thrombosis of a preceding stenosis) in the LAA subgroup, which could lead to different collateral patterns. Second, the FRAME dataset focused on patients treated with thrombectomy, which may have led to a selection bias. However, according to the FRAME design, patients were treated with thrombectomy regardless of the baseline imaging profile. Third, we did not retrieve data on Willis’ variants, multivessel occlusions, intracranial atherosclerosis, the quality of external carotid artery circulation or other mechanisms that could modulate collateral flow.30,31 However, such information is integrated in the HIR evaluation. Also, there might be residual potential confounders that were not included in the multivariable analyses. Fourth, stroke etiology classification is not always straightforward, and some patients might have been misclassified. Finally, we did not record the subtype of CE etiology and were therefore unable to provide subgroup analyses.
Conclusion
In this large multicenter study including acute stroke patients with anterior circulation LVO, a higher Hypoperfusion Intensity Ratio – indicating worse collateral flow – was independently associated with cardioembolic stroke etiology. Patients with LVO of undetermined etiology had similar imaging patterns as those of cardioembolic origin.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241306264 – Supplemental material for Hypoperfusion intensity ratio to differentiate between stroke etiologies in patients with a large vessel occlusion
Supplemental material, sj-docx-1-eso-10.1177_23969873241306264 for Hypoperfusion intensity ratio to differentiate between stroke etiologies in patients with a large vessel occlusion by Ari Maruani, Michael Obadia, Louis Fontaine, Julien Savatovsky, Jean-François Albucher, Lionel Calviere, Nicolas Raposo, Christophe Cognard, Alain Viguier, Gregory W. Albers, Candice Sabben, Igor Sibon, Jean-Marc Olivot and Pierre Seners in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FRAME (French Acute Multimodal Imaging Study to Select Patients for Mechanical Thrombectomy) was supported by a research grant from the French Ministry of Health, Clinical Research Hospital Program 2015 (PHRCI-15-076).
Consent to participate
Oral consent was obtained in the Paris’ cohort, written consent in the FRAME cohort by the patient or a relative.
Ethical considerations
Our research was approved by the Rothschild Foundation Hospital review board -IRB 00012801- under the study number CE_20240130_3_PSS.
Guarantor
Dr Pierre Seners.
Contributorship
AM, MO and PS researched literature and conceived the study. AM wrote the first draft of the manuscript. LF, JA, LC, NR, CC, AV, GA, IS, JO were part of the FRAME investigators. PS, JS, CS helped building the FOR database. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
