Abstract
Purpose:
Uncertainties remain on the optimal treatment for acute minor stroke with nondisabling symptoms. The two most common therapeutic approaches are intravenous thrombolysis (IVT) and antiplatelet therapy, notably dual antiplatelet therapy (DAPT). We synthesized data from the literature to compare IVT to DAPT and identify the best treatment for this population.
Method:
We systematically searched Pubmed, Web of Science and the Cochrane Library for randomized trials and observational studies comparing IVT, aspirin, and/or DAPT, started within 24 h of symptom onset in patients with minor stroke (NIHSS ⩽ 5) and nondisabling symptoms. Random-effects Bayesian network meta-analysis was conducted. The primary outcome was excellent functional outcome at 3 months (mRS 0–1). Secondary outcomes included mRS 0–2, symptomatic intracranial hemorrhage, mortality, and recurrent stroke.
Findings:
Four randomized trials and 2 observational studies (5897 patients for the analysis of the primary outcome) were included. Compared with IVT (alteplase), DAPT (clopidogrel + aspirin) was significantly associated with higher odds of mRS 0–1 (OR = 1.52, 95% CrI, 1.09–2.35), but aspirin alone was not (OR = 1.36, 95% CrI, 0.87–2.30). DAPT was also associated with lower odds of symptomatic intracranial hemorrhage than alteplase (OR = 0.14, 95% CrI, 0.03–0.91). There were no significant differences between treatment groups regarding the other outcomes. For each outcome, the ranking for the best treatment was DAPT, then aspirin, and then IVT.
Discussion/Conclusion:
This network meta-analysis suggests that DAPT may be the optimal treatment for acute nondisabling stroke, with higher odds of excellent functional outcome compared with IVT.
Registration: PROSPERO ID: CRD42024522038

Introduction
About half of all patients with acute ischemic stroke present with a mild deficit (i.e. “minor stroke,” often defined as a National Institutes of Health Stroke Scale [NIHSS] score ⩽ 5 on admission). 1 Uncertainties remain on the optimal treatment strategy for acute minor stroke with nondisabling symptoms. Two therapeutic approaches are commonly used in this scenario: reperfusion therapies and antiplatelet therapy. They have distinct goals: the former, which consists in intravenous thrombolysis (IVT) and sometimes endovascular thrombectomy (EVT), aims to avoid early neurological deterioration, while the latter aims to reduce the risk of early stroke recurrence. The use of IVT with alteplase in patients with minor stroke has raised controversy. An individual participant data meta-analysis of randomized controlled clinical trials (RCTs) of alteplase versus placebo or open control showed that the benefit of IVT did not significantly differ with neurological severity, and that patients with a baseline NIHSS score of 0–4 were at greater odds of excellent outcome (modified Rankin Scale [mRS] 0–1 at 3 months) with alteplase (OR 1.48, 95% CI 1.07–2.06). 2 However, all but one of the trials included in this analysis excluded patients with nondisabling neurological deficits. 3 In contrast, the PRISMS trial of alteplase versus aspirin in patients with an NIHSS score of 0–5 and symptoms considered to be nondisabling by a local investigator (i.e. that would not prevent the patient from performing basic activities of daily living) was neutral. 4 Importantly, this trial was underpowered owing to early study termination after slow recruitment. Notwithstanding, the optimal antiplatelet therapy for patients with minor stroke has since become short-term dual antiplatelet therapy (DAPT), which showed superiority over aspirin alone to prevent early stroke recurrence.5,6 US and European Guidelines now recommend aspirin plus clopidogrel as first-line secondary prevention therapy within 24 h of noncardioembolic minor stroke (operationally defined as an NIHSS score ⩽3).7,8 More recently, a non-inferiority RCT (ARAMIS) of alteplase versus short-term aspirin plus clopidogrel was conducted in acute minor stroke patients with nondisabling deficit, defined as an NIHSS score of 0–5 with unimpaired consciousness and ⩽1 point on single-item scores such as vision, language, neglect, or single limb weakness. 9 In ARAMIS, DAPT was considered non-inferior to alteplase with regards to the primary outcome (mRS 0–1 at 3 months) at a non-inferiority margin of −4.5%. Indeed, 93.8% and 91.4% of patients in the DAPT and alteplase groups had an excellent functional outcome (risk difference, 2.3%, 95% CI −1.5% to 6.2%), respectively. Although ARAMIS is the only RCT directly comparing DAPT and alteplase, several other studies provide a comparison between either DAPT or alteplase and aspirin, therefore paving the way for indirect comparisons between alteplase and DAPT in a network meta-analysis (NMA). We aimed to synthesize the literature data to compare IVT, aspirin, and DAPT in patients with minor nondisabling stroke to identify the best treatment for this population.
Methods
This systematic review follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses and Network Meta-Analysis (PRISMA NMA) statement. 10 Neither patient consent nor ethical approval was required as this work constitutes an aggregate data meta-analysis of previously published studies. Data will be made available upon reasonable request. The review protocol was registered in PROSPERO (CRD42024522038).
Eligibility criteria
Studies published in English were eligible if the following criteria were fulfilled: (1) Comparative studies (RCTs or observational studies) (2) of the efficacy or safety of at least two of the three following treatments, started no later than 24 h after symptom onset: intravenous thrombolysis with alteplase (0.9 mg/kg), aspirin alone, and DAPT with aspirin plus clopidogrel (irrespective of the dose); (3) in patients with acute minor stroke (NIHSS 0–5) considered nondisabling; and (4) report of 3-month mRS.
Studies were excluded in the presence of any of the following criteria: (1) single-arm studies, (2) case reports or case series, (3) potential overlap in participants with an already included study, (4) insufficient information about at least one of the treatment arms (e.g. mixture of single and dual antiplatelet therapy in one treatment group or lack of mention of the type of thrombolytic), (5) stroke with large vessel occlusion and/or treated with EVT, (6) insufficient detail about the index event (e.g. mixture of stroke and transient ischemic attack), and (7) other stroke subtypes (i.e. cerebral venous thrombosis).
Outcomes
The primary outcome was excellent functional outcome at 3 months, defined as a mRS score of 0–1. Secondary outcomes were good functional outcome (mRS 0–2 at 3 months), symptomatic intracranial hemorrhage (sICH), asymptomatic intracranial hemorrhage (aICH), all-cause mortality within 3 months, and recurrent stroke within 3 months. Major bleeding was considered but eventually not included as a secondary outcome because it was not reported in most included articles.
Information sources, literature search, data collection
We searched PubMed, Web of Science, the Cochrane Library, and reference lists of relevant papers, for articles published between each database’s inception and July 2024. We only considered articles published in English. The detailed search strategy is provided in Supplemental Table 1. Two authors (FL, G Turc) independently screened the titles and abstracts of all records, excluded irrelevant articles, and obtained the full texts of the remaining articles. Using standardized forms, relevant data for the meta-analysis was independently extracted by the same authors, in duplicate. Disagreement was resolved by consensus.
Assessment of the risk of bias
The risk of bias in RCTs and observational studies was assessed by two authors (FL, G Turc) using the Cochrane Risk of Bias tool for randomized trials version 2 (RoB2) and the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I tool), respectively. Raters independently assessed the risk of bias for each study, and disagreement was resolved by consensus.
Statistical analysis
Before conducting any NMA, we appraised the clinical and methodologic characteristics of the eligible studies to assess the appropriateness of the transitivity assumption. We decided a priori to also include data from non-randomized studies in the NMA because (1) direct evidence from RCTs is scarce; (2) observational studies provide direct comparisons between alteplase and DAPT; and (3) to improve connectivity in the network.
NMAs were conducted under a Bayesian framework with R version 4.4.1 (BUGSnet and MetaInsight packages). We decided a priori to use random-effects models to calculate pooled estimates because we assumed that the true effect size may differ across studies. Fixed-effects models were used for sensitivity analyses. The principal summary measure was the OR. A continuity correction was used for zero-event studies. Pooled ORs with 95% credible intervals (95% CrIs) were estimated using arm-based analyses. We also calculated secondary measures of treatment effect for each intervention with surface under the cumulative rank curve (SUCRA) probabilities and treatment rankings. All results were presented with higher SUCRA values indicating better performance (e.g. higher probability of achieving mRS 0–1; lower probability of death, etc.). Inconsistency tests were conducted with the nodesplit model method to assesses whether the direct and indirect evidence is consistent. Significance was set at p < 0.05 (two-tailed).
Results
Of 4042 records identified, we excluded 714 duplicates. 3244 records were then excluded after screening of titles and abstracts. We retrieved 78 potentially eligible studies for full-text screening. Reasons for exclusion are provided in Figure 1. A total of 8 articles reporting data from 6 studies (4 RCT and 2 observational studies) satisfied our inclusion criteria.4,9,11–16 The main characteristics of the included studies is presented in Table 1. Six articles were related to four RCTs (see the footnote of Table 1), including subgroup or post-hoc analyses.4,9,11,12,15,16 Two RCTs compared DAPT with aspirin (subgroup or post-hoc analyses of the CHANCE and POINT trials),11,12 one RCT compared aspirin with alteplase (PRISMS) 4 and the last RCT compared DAPT with alteplase (ARAMIS). 9 Two observational studies compared DAPT with alteplase.13,14 Of note, the article presenting the subgroup analysis of CHANCE also included observational data from patients that were treated with alteplase but not enrolled in the trial. 11 We decided to exclude these patients. Our analysis did not include any overlap of patients.
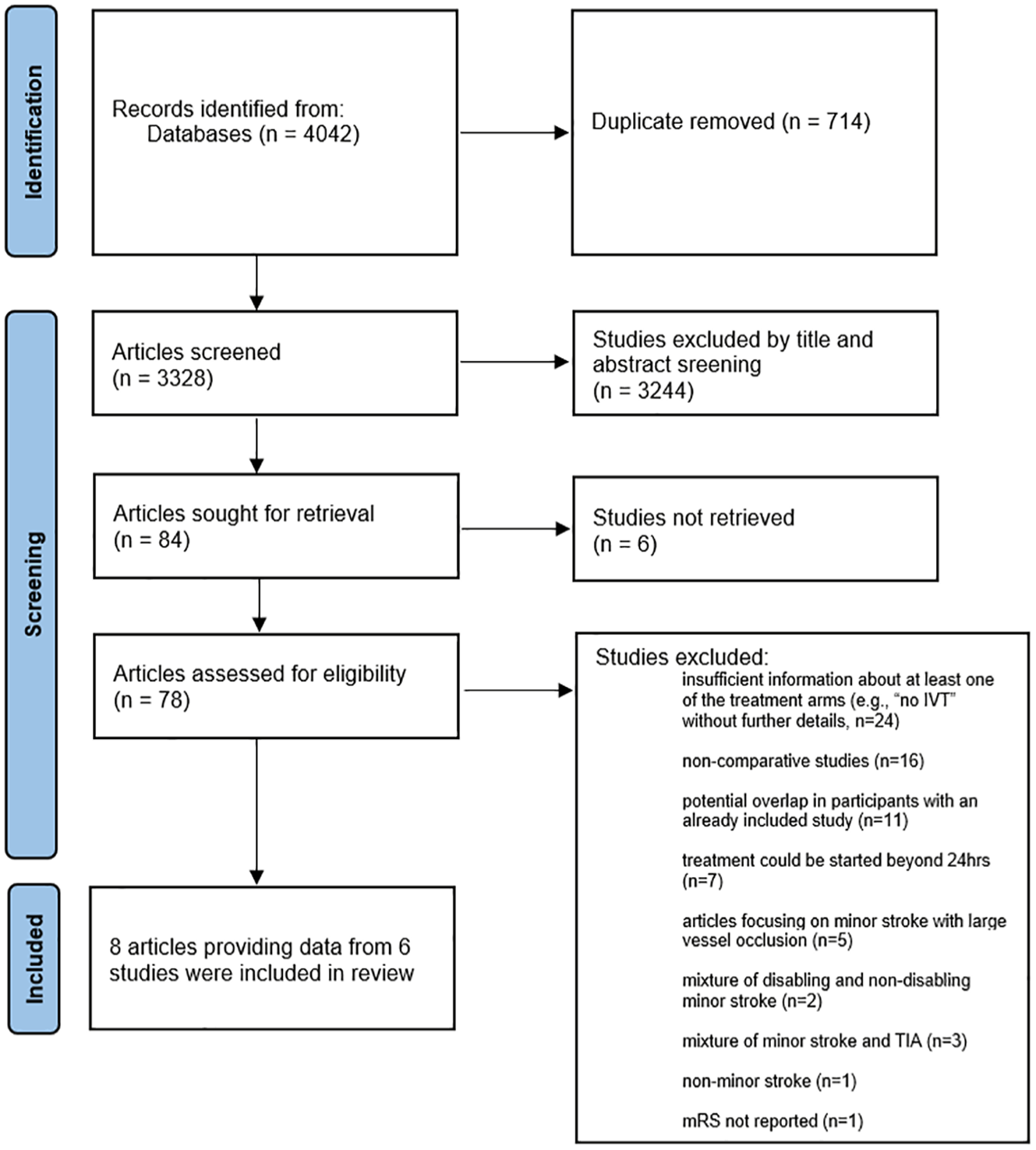
PRISMA diagram.
Characteristics of the included studies. a .
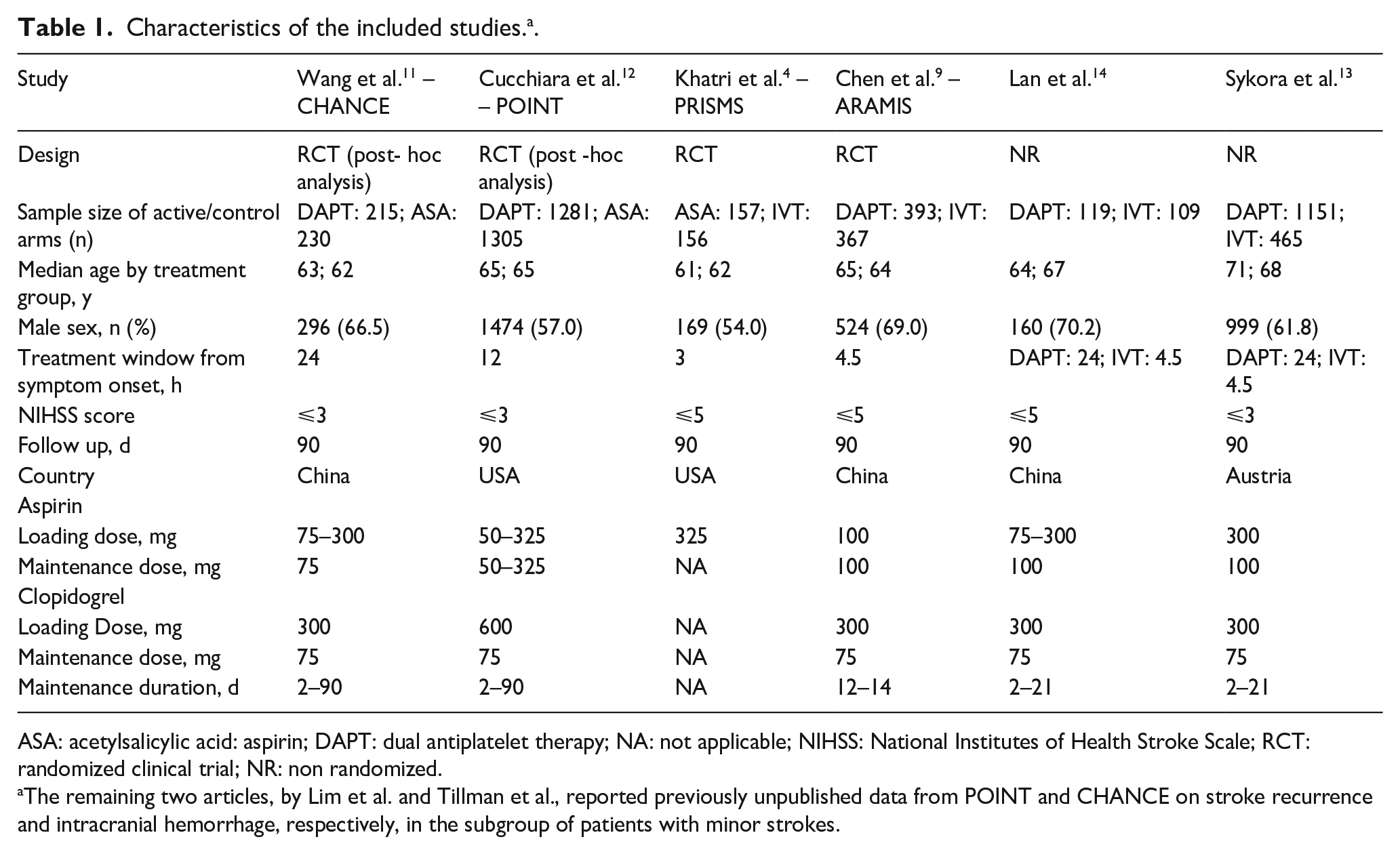
ASA: acetylsalicylic acid: aspirin; DAPT: dual antiplatelet therapy; NA: not applicable; NIHSS: National Institutes of Health Stroke Scale; RCT: randomized clinical trial; NR: non randomized.
The remaining two articles, by Lim et al. and Tillman et al., reported previously unpublished data from POINT and CHANCE on stroke recurrence and intracranial hemorrhage, respectively, in the subgroup of patients with minor strokes.
Risk of bias
The analysis of potential bias in the RCTs showed high quality of the source information with a low probability of bias across all RoB2 domains, except for the ARAMIS trial, which had some concerns (Supplemental Table 2). The two observational studies were at low and moderate risk of bias according to ROBINS-I, respectively (Supplemental Table 3).
Overview of studies and characteristics of the populations
The median age ranged from 62 to 71 years (Table 1). NIHSS scores ranged from 0 to 3 in three studies,11–13 and from 0 to 5 in the three remaining studies. The timing from symptom onset to antithrombotic medication initiation was within 24 h for all trials of DAPT versus aspirin, and within 3 or 4.5 h for studies including IVT.
Primary outcome
A total of 5897 patients from six studies were included in the analysis of excellent outcome at 3 months.4,9,11–14 The number and proportion of patients achieving excellent outcome stratified by treatment group were 872/1080 (80.7%) for alteplase, 1385/1692 (81.8%) for aspirin, and 2618/3125 (83.8%) for DAPT. The network diagram is presented in Figure 2.
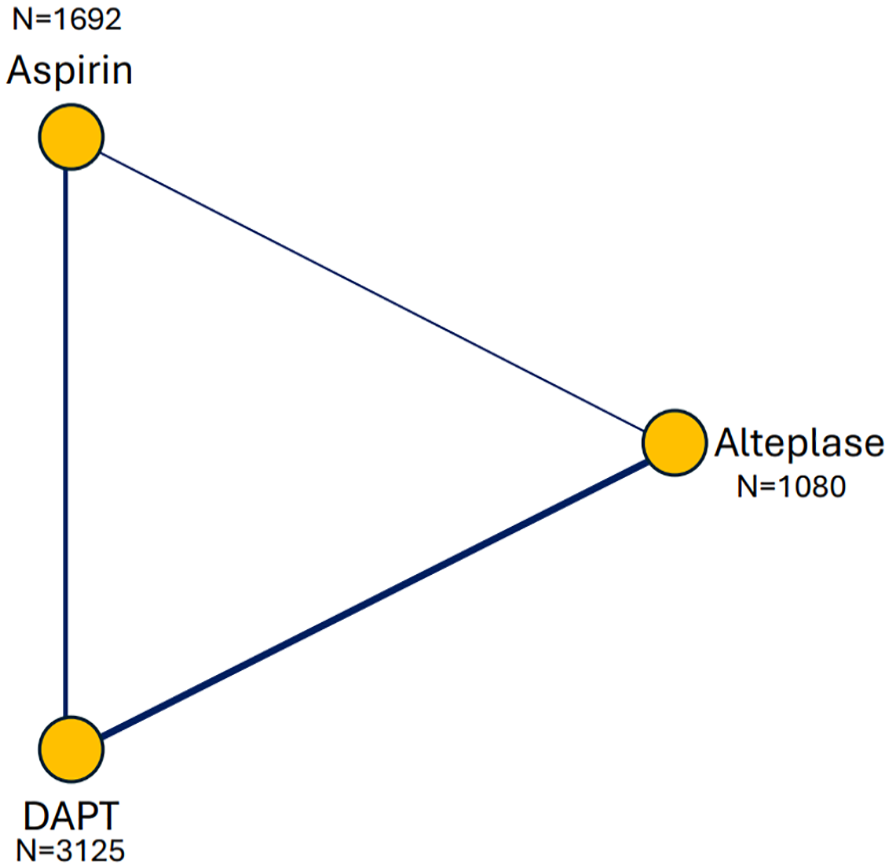
Network diagram of the total number of patients analyzed in each treatment arm for the primary outcome (mRS 0–1 at 3 months).
Compared with alteplase, DAPT was significantly associated with higher odds of excellent functional outcome (OR = 1.52, 95% CrI, 1.09–2.35), but aspirin alone was not (OR = 1.36, 95% CrI, 0.87–2.30; Figure 3). DAPT had the highest SUCRA value (88%), followed by aspirin (57%) and then by alteplase (5%; Figure 4). In sensitivity analysis, the fixed-effect model led to similar results, except that aspirin was significantly associated with excellent outcome (OR = 1.49, 95% CrI, 1.21–1.83 for DAPT and OR = 1.32 95% CrI, 1.02–1.71 for aspirin; Supplemental Figure 1).
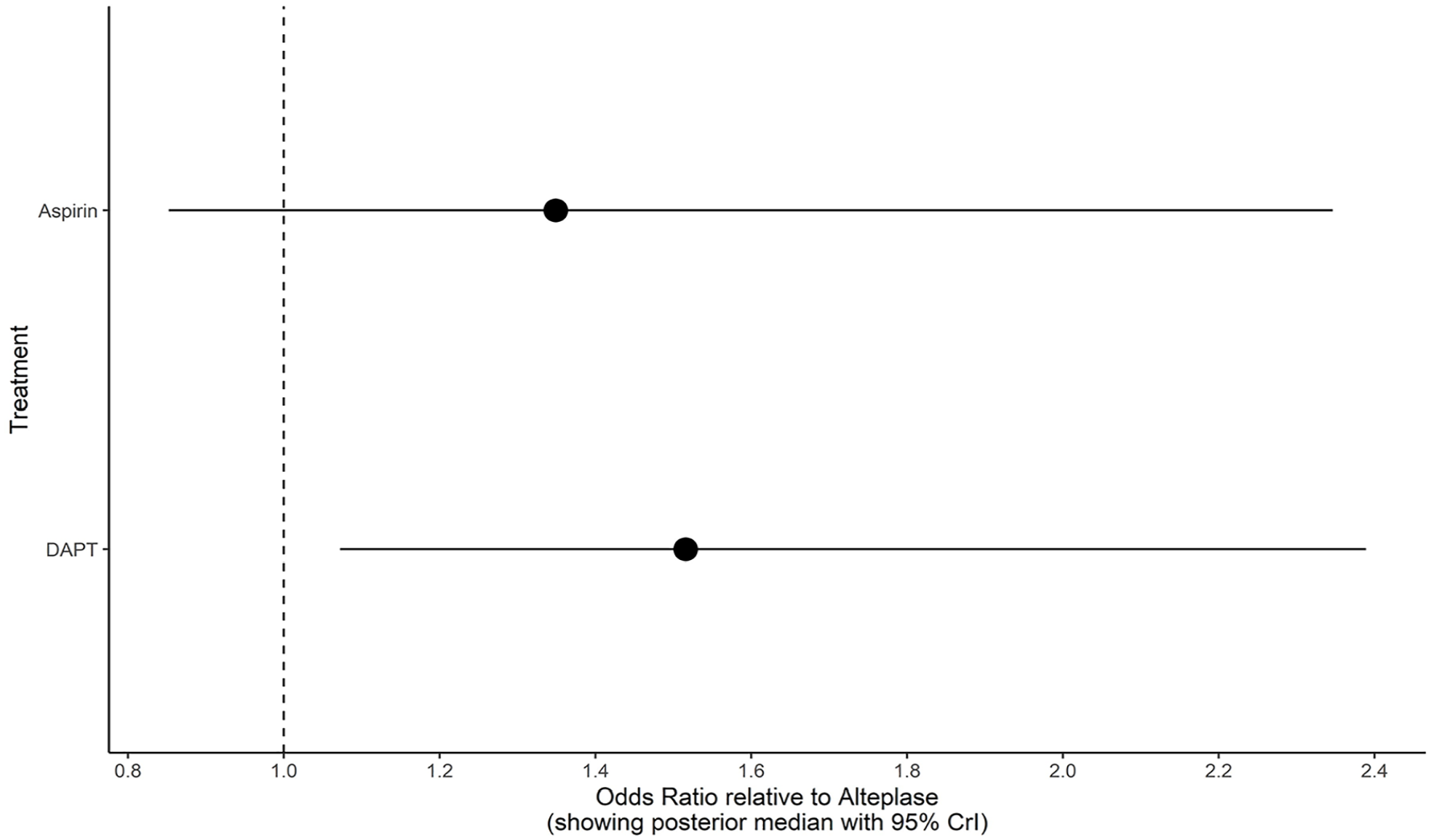
Odds ratios for the primary outcome (mRS 0–1 at 3 months) according to the treatment group, using IVT with alteplase as reference (random-effects network meta-analysis).
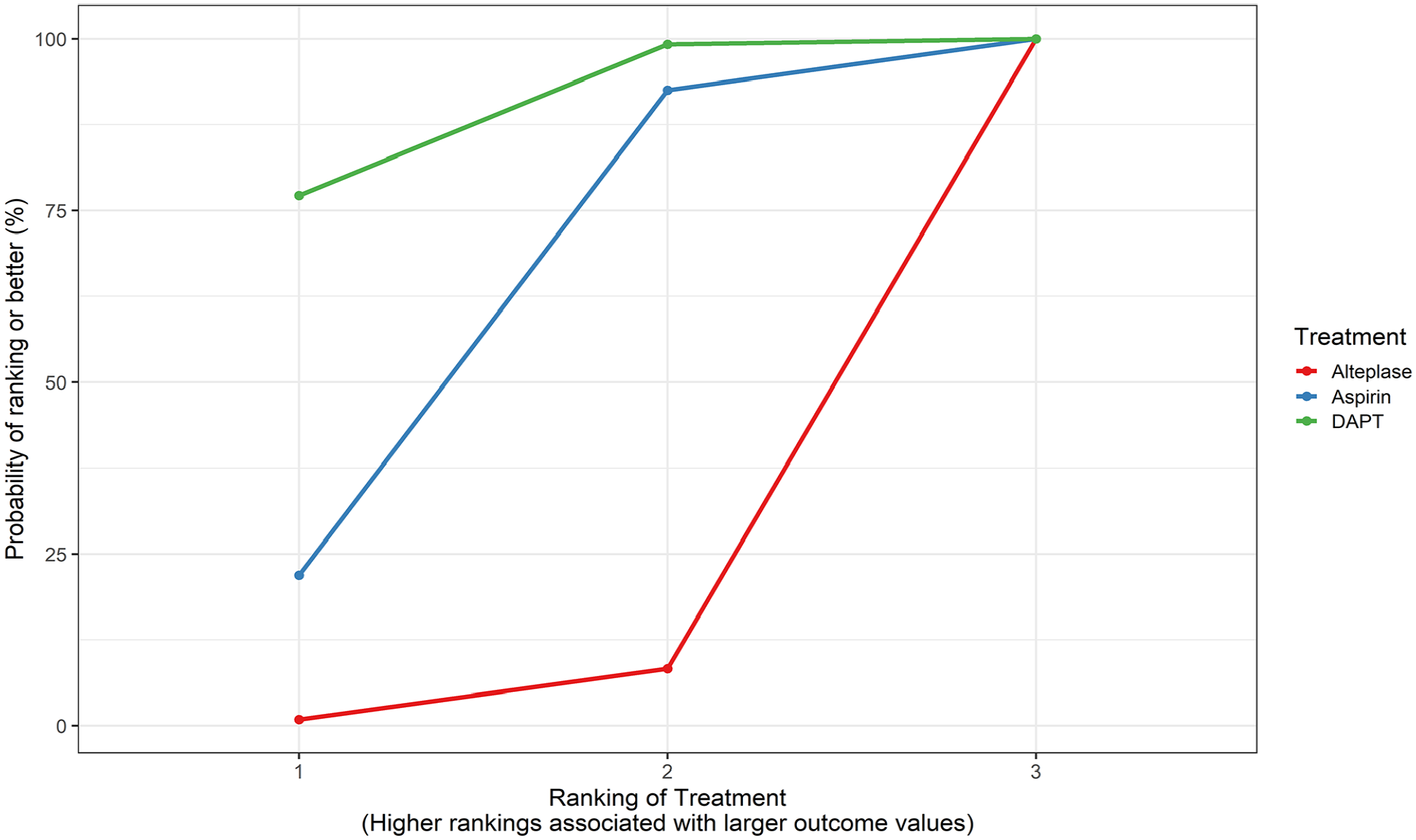
SUCRA plot for the primary outcome (mRS 0–1 at 3 months). Higher SUCRA (Surface Under the Cumulative Ranking Curve) values and cumulative ranking curves nearer the top left indicate better performance.
Secondary outcomes
Good functional outcome (mRS 0–2 at 3 months)
Good functional outcome was reported in four studies including 1695 patients.4,9,11,14 The number and proportion of patients achieving good functional outcome stratified by treatment group were 574/615 (93.3%) for alteplase, 359/387 (92.8%) for aspirin, and 659/693 (95.1%) for DAPT. Compared with alteplase, neither DAPT (OR = 1.60, 95% CrI, 0.67–4.29) nor aspirin alone (OR = 1.55, 95% CrI, 0.56–4.78) were significantly associated with higher odds of good functional outcome. DAPT had the highest SUCRA value (70%), followed by aspirin (66%) and alteplase (14%; Supplemental Figure 2). The sensitivity analysis based on a fixed effect model led to similar results (data not shown).
sICH
sICH was reported in six studies including 5515 patients.4,9,11,13,14,16 The number and proportion of patients experiencing sICH in each treatment group were 25/1810 (1.4%) for alteplase, 1/387 (0.03%) for aspirin, and 4/3318 (0.1%) for DAPT. Compared with alteplase, DAPT (OR = 0.14, 95% CrI, 0.03–0.91) was significantly associated with lower odds of sICH, while the association did not reach significance for aspirin alone (OR = 0.19, 95% CrI, 0.01–2.24). DAPT had the highest SUCRA value (79%), followed by aspirin (66%) and alteplase (5%; Supplemental Figure 3). The sensitivity analysis based on a fixed effect model showed a significant association for both DAPT (OR = 0.11, 95% CrI, 0.04–0.27) and aspirin (OR = 0.17, 95% CrI, 0.03–0.79), using alteplase as reference.
aICH
aICH was reported in four studies including 6073 patients.4,9,14,16 The number and proportion of patients with aICH in each treatment group were 11/617 (1.7%) for alteplase, 7/2578 (0.3%) for aspirin, and 5/2878 (0.2%) for DAPT. Compared with alteplase, neither DAPT (OR = 0.52, 95%CrI, 0.11–2.17) nor aspirin alone (OR = 0.52, 95% CrI,0.10–2.10) were significantly associated with lower odds of aICH. The SUCRA values were 66% for both DAPT and aspirin, and 18% for alteplase (Supplemental Figure 4). The sensitivity analysis based on a fixed effect model led to similar results (data not shown).
All-cause mortality within 3 months
All-cause mortality within 3 months was reported in three studies including 1477 participants.4,9,11 The number and proportion of deaths by treatment group were 4/506 (0.8%) for alteplase, 2/387 (0.5%) for aspirin, and 2/584 (0.3%) for DAPT. Compared with alteplase, neither DAPT (OR = 0.39 [95% CrI, 0.05–2.37]) nor aspirin alone (OR = 0.80, 95% CrI, 0.10–5.77) were significantly associated with all-cause mortality. DAPT had the highest SUCRA value (81%), followed by aspirin (41%) and alteplase (28%; Supplemental Figure 5). The sensitivity analysis based on a fixed effect model let to similar results (data not shown).
Recurrent stroke
Recurrent stroke within 3 months was reported in five studies (7748 patients).4,9,14,15 The number and proportion of recurrent strokes stratified by treatment group were 11/615 (1.8%) for alteplase, 326/3412 (9.6%) for aspirin, and 227/3721 (6.1%) for DAPT. Compared with alteplase, neither DAPT (OR = 0.56, 95% CrI, 0.15–1.65) nor aspirin alone (OR = 0.85, 95% CrI, 0.24–2.61) were significantly associated with stroke recurrence. DAPT had the highest SUCRA value (90%), followed by aspirin (33%), and alteplase (26%; Supplemental Figure 6). The sensitivity analysis based on a fixed effect model let to similar results (data not shown). In a post-hoc analysis, DAPT was associated with lower rates of recurrent stroke than aspirin in our fixed-effect NMA (OR = 0.67, 95% CrI, 0.56–0.80).
There was no evidence of inconsistency for all outcomes studied (p > 0.05 for differences between direct and indirect comparisons).
Discussion
This systematic review and network meta-analysis suggests that, in patients with acute nondisabling minor stroke, DAPT with aspirin plus clopidogrel is associated with significantly higher odds of excellent functional outcome at 3 months compared with alteplase, and a lower risk of symptomatic intracranial hemorrhage. For each outcome, the ranking for the best treatment was DAPT, then aspirin, and then IVT.
Despite its mild clinical presentation, minor stroke, which definition is usually solely based on the NIHSS score (0–5 points), results in long-term disability or death in approximately 25% of cases, 17 due to early neurological deterioration, stroke recurrence, or persistence of disabling symptoms. The contrast between the positive results of the subgroup analysis of the pivotal IVT trials and the neutral results of PRISMS has supported the concept of making a distinction between disabling and nondisabling minor strokes.2,4 Although such a dichotomy has sometimes been criticized, especially due to its subjective nature, it is now a pillar of both US and European Guidelines on IVT for minor stroke, which recommend IVT only in case of disabling symptoms.18,19 We therefore chose to focus on minor stroke with nondisabling symptoms. Although non-inferiority of DAPT over IVT was recently demonstrated in this population, neither the ARAMIS trial nor subsequent systematic reviews and pairwise meta-analyses were able to demonstrate a superiority of DAPT with regards to functional outcome. 20 In contrast, our NMA suggests a superiority of DAPT for mRS 0–1, which is the most frequently used primary outcome in IVT trials. A nominal but nonsignificant association was observed for mRS 0–2, which is a less relevant outcome for patients with minor strokes, 4 especially nondisabling. The superiority of DAPT regarding excellent functional outcome may be explained by a lower risk of sICH compared with IVT, as observed in our NMA. Another potential explanation could be that DAPT confers a lower risk of stroke recurrence, especially when started very early. 5 However, our NMA failed to demonstrate that DAPT is superior to IVT with regards to 3-month stroke recurrence, despite a favorable effect size (OR = 0.56). This finding might be explained by insufficient statistical power or by the lack of detailed information on the antithrombotics used for secondary prevention in the included IVT studies. Importantly, although IVT and DAPT are acute reperfusion and early secondary prevention strategies, respectively, these treatments are not mutually exclusive. Their sequential combination (i.e. IVT in case of a disabling minor stroke followed by initiation of DAPT at 24 h) has not been sufficiently explored. 21 Future studies are warranted to determine the risk-benefit profile of such combinations. If the safety of DAPT started 24 h after IVT is confirmed, a randomized controlled trial of IVT followed by DAPT after 24-h cerebral imaging versus DAPT (without IVT) started on hospital admission in patients with minor stroke will be needed. However, it seems unlikely that IVT will prove to be beneficial in patients with minor nondisabling strokes, as suggested by our SUCRA ranking of the best treatments for this condition, where IVT was consistently ranked lower than DAPT and aspirin for each outcome.
The treatments assessed in this NMA were restricted to aspirin, clopidogrel plus aspirin, and IVT with alteplase. Our analysis did not include tenecteplase, which has become a popular alternative to alteplase. However, our findings are in line with the neutral results of a recent RCT of tenecteplase versus non-thrombolytic standard care in patients with minor stroke and intracranial occlusion or focal perfusion lesion within 12 h of symptom onset. 22 Indeed, patients could not be enrolled in this trial if, in the judgment of the physician and the patient, routine IVT was warranted, which suggests that most patients had a nondisabling minor stroke. This trial also showed a numerically higher risk of sICH and a significantly higher risk of death with IVT. We decided not to include this trial in the NMA because the control group was heterogeneous (59% of patients treated with DAPT) and only selected patients, based on arterial and perfusion imaging, were enrolled. Importantly, our results do not apply to patients with minor stroke and large vessel occlusion, which corresponds to a subgroup where reperfusion therapies, notably endovascular treatment, are currently evaluated (NCT 03796468).
We did not include ticagrelor in this NMA because we could not identify any study reporting 3-month mRS in all enrolled patients treated with either ticagrelor alone or the combination of ticagrelor plus aspirin for acute ischemic stroke.
This systematic review and NMA has several strengths. First, we only selected studies with homogeneous treatment groups, excluding many studies with mixed or unclear non-IVT groups (Figure 1). Second, we only included articles focusing on nondisabling minor stroke rather than a mixture of TIA (for which IVT is almost never considered), disabling and nondisabling minor strokes. Third, the NMA methodology allows to produce rankings of treatments based on probabilities, which is novel for the present clinical question.
Several limitations need to be acknowledged. First, determining whether stroke symptoms are disabling or nondisabling is difficult and subjective. Although a strict definition was used in the ARAMIS trial, in other studies this process was not standardized and solely based on the judgment of each investigator. This may have limited the validity of the transitivity assumption. We could unfortunately not perform subgroup analyses stratified by NIHSS score because this data was only reported in the ARAMIS trial. However, stratification by NIHSS score also has potential limitations because patients with an identical NIHSS score may have very different deficits. An individual participant data meta-analysis would be needed to shed more light on this issue. Second, the secondary antithrombotic treatment after IVT was not detailed in included studies. It is likely that single antiplatelet therapy was used after IVT in most cases (in the most recent study, ARAMIS, only 9% of patients in the IVT group were treated with DAPT from hospital discharge to 90-day follow-up 9 ), but individual patient data would be needed to confirm this claim. Third, the assessment of our primary outcome was not blinded in all studies and the assessment and ascertainment of some secondary outcomes slightly varied across studies, notably for recurrent stroke and symptomatic intracranial hemorrhage. This heterogeneity was expected and, accordingly, random effects models were used for our main analyses. Fourth, the duration of the DAPT varied across studies, ranging from 21 to 90 days. Fifth, we may have lacked statistical power for rare events such as mortality. Sixth, detailed stroke etiology, such as the proportion of patients with intracranial atherosclerotic stenosis, was not reported in the included studies. Finally, assessment of publication bias is notoriously difficult in NMA due to the low number of studies for each comparison.
In summary, this systematic review and Bayesian network meta-analysis suggests that short-term DAPT may be the optimal treatment for acute nondisabling stroke, with higher odds of excellent functional outcome compared with IVT. Further research should provide insight on the usefulness of ticagrelor plus aspirin, and IVT with tenecteplase, in this population.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241293323 – Supplemental material for Intravenous thrombolysis or antiplatelet therapy for acute nondisabling ischemic stroke: A systematic review and network meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241293323 for Intravenous thrombolysis or antiplatelet therapy for acute nondisabling ischemic stroke: A systematic review and network meta-analysis by François Lun, Lina Palaiodimou, Aristeidis H Katsanos, Georgios Tsivgoulis and Guillaume Turc in European Stroke Journal
Footnotes
Acknowledgements
None.
Correction (May 2025):
Article Type updated from Review Article to Systematic Review
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AHK reports salary support from the Heart and Stroke Foundation Canada and consulting fees from DiaMedica Therapeutics Inc. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Neither patient consent nor ethical approval was required as this work constitutes an aggregate data meta-analysis of previously published studies.
Informed consent
Neither patient consent nor ethical approval was required as this work constitutes an aggregate data meta-analysis of previously published studies.
Guarantor
G Turc
Contributorship
FL and G Turc researched literature and conceived the study. FL and G Turc were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. FL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
