Abstract
Introduction:
Dual antiplatelet therapy (DAPT) is superior to single antiplatelet therapy (SAPT) for secondary prevention after minor, non-cardioembolic stroke. We aimed to assess whether DAPT efficacy is modified by large artery atherosclerotic (LAA) etiology, and DAPT safety by stroke size on MRI.
Patients and methods:
Post hoc analysis of the Phase 2 PACIFIC-STROKE randomized clinical trial, which enrolled patients with non-cardioembolic stroke, all with baseline MRI and compared the Factor XIa inhibitor asundexian with placebo on a background of DAPT or SAPT. We compared patients treated with DAPT versus SAPT. The efficacy endpoint was the rate of recurrent ischemic stroke, the safety endpoint was major or clinically relevant non-major bleeding during follow-up.
Results:
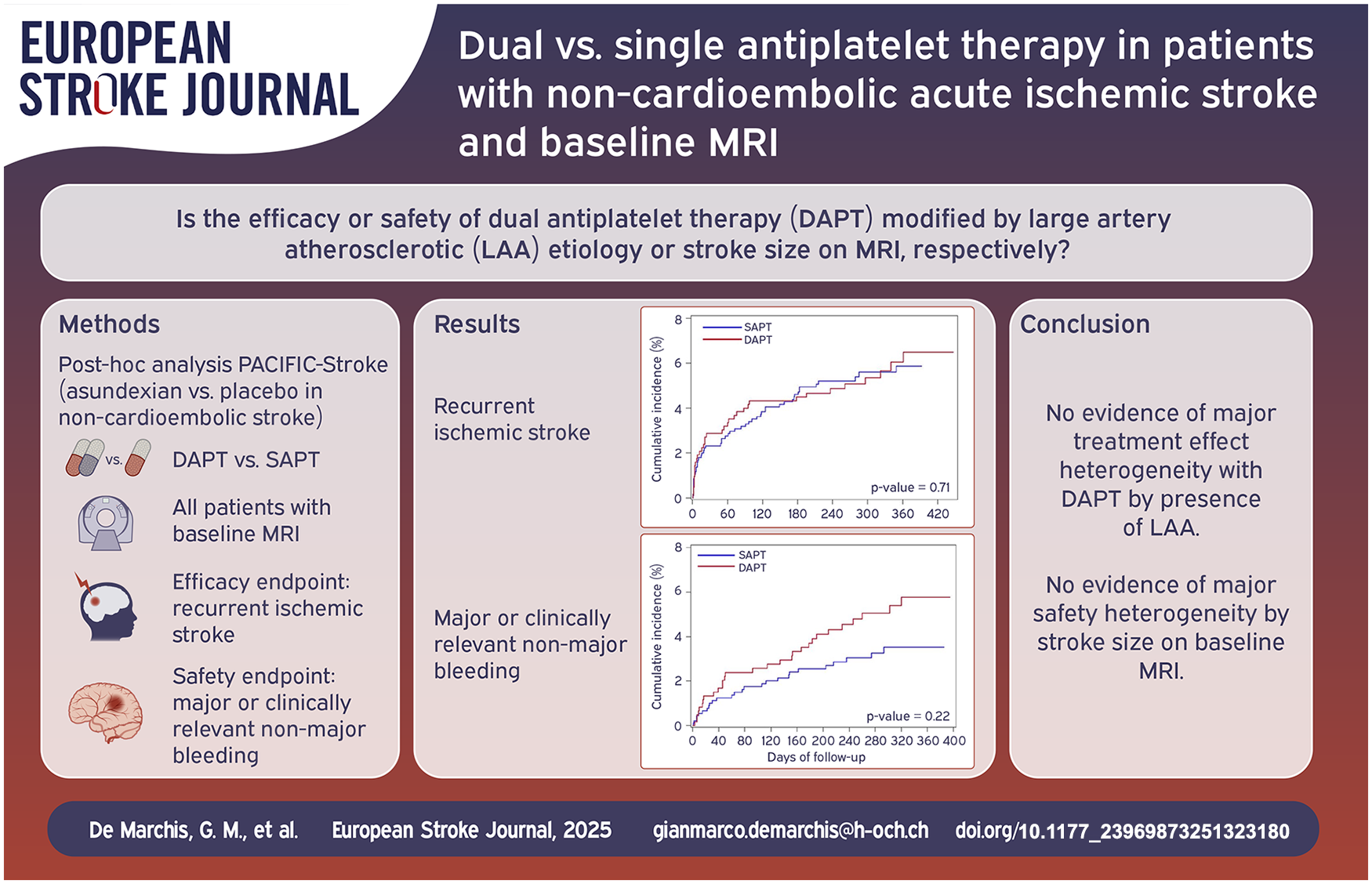
1590 patients were included, median NIHSS was 2 (interquartile range [IQR] 1–4), 40% received DAPT. Median follow-up was 11.5 months. The efficacy endpoint occurred in 4.4% and 4.8% in the DAPT group and SAPT group, respectively, with the strongest numerical benefit of DAPT over SAPT among patients with NIHSS ⩽ 3 not treated by intravenous thrombolysis. LAA index stroke etiology did not modify DAPT treatment effect. The safety endpoint occurred more often in the DAPT than in the SAPT group (4.6% vs 2.7%), with the numerically lowest risk among patients with NIHSS ⩽ 3 not treated by intravenous thrombolysis. Stroke size did not modify the effect of DAPT on the safety endpoint.
Discussion and conclusion:
We found no evidence of major treatment effect heterogeneity with DAPT compared with SAPT in patients with and without LAA or by stroke size on MR-DWI.
Introduction
The risk of recurrent stroke is highest during the first few weeks of the index stroke.1,2 Early dual antiplatelet therapy (DAPT) with aspirin and clopidogrel, compared with single antiplatelet therapy (SAPT), reduces the risk of recurrent stroke by 25% to 32% among patients with non-cardioembolic minor ischemic stroke, as shown in the POINT, CHANCE, and INSPIRES trials.3–5 The optimal duration of DAPT is 21 days of the index stroke, with the risk of symptomatic intracerebral hemorrhage (sICH) prevailing thereafter.6–8
Patients with large artery atherosclerotic (LAA) stroke have the highest early recurrence risk and may constitute the group with the largest risk reduction of DAPT over SAPT. 9 The focus on minor stroke in the trials comparing DAPT to SAPT aimed at limiting the risk of hemorrhagic transformation. The definition of minor stroke was based on clinical severity at baseline (NIHSS ⩽ 3 in CHANCE and POINT, and ⩽5 in INSPIRES). The NIHSS is not, however, a flawless surrogate of baseline infarct size. Large infarcts on MR-Diffusion Weighted Imaging (DWI) can be seen in patients with NIHSS ⩽ 5, particularly in the presence of visual field impairment, neglect, or aphasia. 10 In clinical practice, baseline infarct size on MR-DWI is often taken in consideration in the risk-benefit assessment of DAPT versus SAPT.
It is unclear whether DAPT efficacy is modified by large artery atherosclerotic (LAA) etiology, and DAPT safety by stroke size on baseline MRI of the index stroke. In a cohort of patients with acute non-cardioembolic ischemic stroke and available MR-Diffusion Weighed Imaging (DWI), culled from the PACIFIC-STROKE trial, we aimed at assessing the interaction between LAA and stroke recurrence risk, as well as baseline infarct size on MR-DWI and bleeding risk.
Methods
We conducted a post hoc analysis of the recently completed PACIFIC-STROKE trial. The study conduct was approved by local ethical boards, all participants provided informed consent. The trial is registered at https://clinicaltrials.gov (NCT04304508). The de-identified, individual participant-level data can be made available to investigators for secondary analyses after review of a submitted proposal by the PACIFIC-Stroke steering committee. 2
Study setting
The PACIFIC-STROKE trial was a randomized, double-blind, placebo-controlled, phase 2b dose-finding trial on the factor XIa inhibitor asundexian, conducted between 2021 and 2022; details have been described elsewhere. 2 Briefly, the PACIFIC-STROKE trial included – within 48 h of symptom onset – patients with acute non-cardioembolic ischemic stroke and baseline NIHSS of ⩽15. Background therapy consisted of DAPT or SAPT at the investigator’s discretion. While PACIFIC-STROKE allowed aspirin, clopidogrel, dipyridamole, or cilostazol as antiplatelet therapy, we restricted the present analysis to patients taking aspirin and clopidogrel. To be classified and analyzed as such, DAPT had to be started within 48 h of index stroke and pursued for at least 7 days. The overall DAPT duration was not available. All patients underwent a brain MRI at baseline.
Dual antiplatelet therapy
Dual antiplatelet therapy was defined as the combined use of aspirin and clopidogrel, provided that it was started within 48 h of the index event and was administered consecutively for at least 7 days.
Efficacy and safety endpoints for the current analysis
For this analysis, the efficacy endpoint was the time to recurrent symptomatic ischemic stroke within 26 weeks. The safety outcome was the time to major or clinically relevant non-major bleeding (as defined by the International Society of Thrombosis and Haemostasis criteria 11 ) within 26 weeks.
Statistical analysis
For statistical analysis SAS (release 9.4) was used. Continuous variables were summarized as medians with interquartile ranges, discrete variables as percentages. Logistic regression models were adjusted for baseline values that had a p-value < 0.05 in the comparison between DAPT and SAPT group. We analyzed the interaction between the type of antiplatelet therapy (DAPT vs SAPT) and the following variables: stroke severity (NIHSS ⩽ 3 or >3), eligibility upon POINT/CHANCE trial criteria (NIHSS ⩽ 3 and no thrombolysis or endovascular thrombectomy performed),3,4 stroke size (>15 mm or ⩽15 mm on baseline brain MRI), Large Artery Atherosclerotic stroke (LAA or non-LAA) as defined by ⩾50% stenosis of proximal carotid artery, and lacunar versus non-lacunar stroke (lacunar being defined as single subcortical infarct sized ⩽15 mm). To address potential selection bias, we performed inverse probability of treatment weighting using the propensity score for the efficacy outcome. Missing data were not imputed. Twenty patients were lost to follow-up; a missing follow-up visit value is replaced by that subject’s previously observed value, that is, the last observation was carried forward. p-Values < 0.05 were considered statistically significant.
Results
In PACIFIC-STROKE, 1808 patients underwent randomization, of which 218 were excluded from the present analysis (Figure 1). Baseline characteristics of the included 1590 patients are presented in Table 1. Overall, median NIHSS was 2.0 (IQR: 1.0–4.0). Among 1081 patients with NIHSS ⩽ 3, 818 (46%) had an infarct size >15 mm on MR-DWI. Eligibility according to POINT/CHANCE – that is, NIHSS< 3 and no acute recanalization therapy – was present in 59.6% of all patients, 70.7% in the DAPT and 52.3% in the SAPT group (p < 0.001). NIHSS at baseline was lower in the DAPT than SAPT group (2 [IQR: 1–3] vs 3 [IQR: 1–4], [p < 0.001]). In the DAPT group, compared with the SAPT group, an infarct size >15 mm on MR-DWI was less frequent, (46.4% vs 54.8%, p < 0.001). For raw numerical data and rate of the outcomes refer to Supplemental Table S1.

Flow chart.
Baseline characteristics stratified by antiplatelet treatment regimen.

CHANCE: clopidogrel in high-risk patients with acute non-disabling cerebrovascular events; DAPT: dual antiplatelet treatment; EVT: endovascular treatment; IQR: interquartile range; IVT: intravenous treatment; mRS: modified ranking scale; NIHSS: National Institutes of Health Stroke Scale; No.: number; POINT: platelet oriented inhibition in new transient ischemic attack and minor ischemic stroke; SAPT: single antiplatelet treatment; SD: standard deviation; TIA: transient ischemic attack.
Bold entries refer to statistically significant p-values.
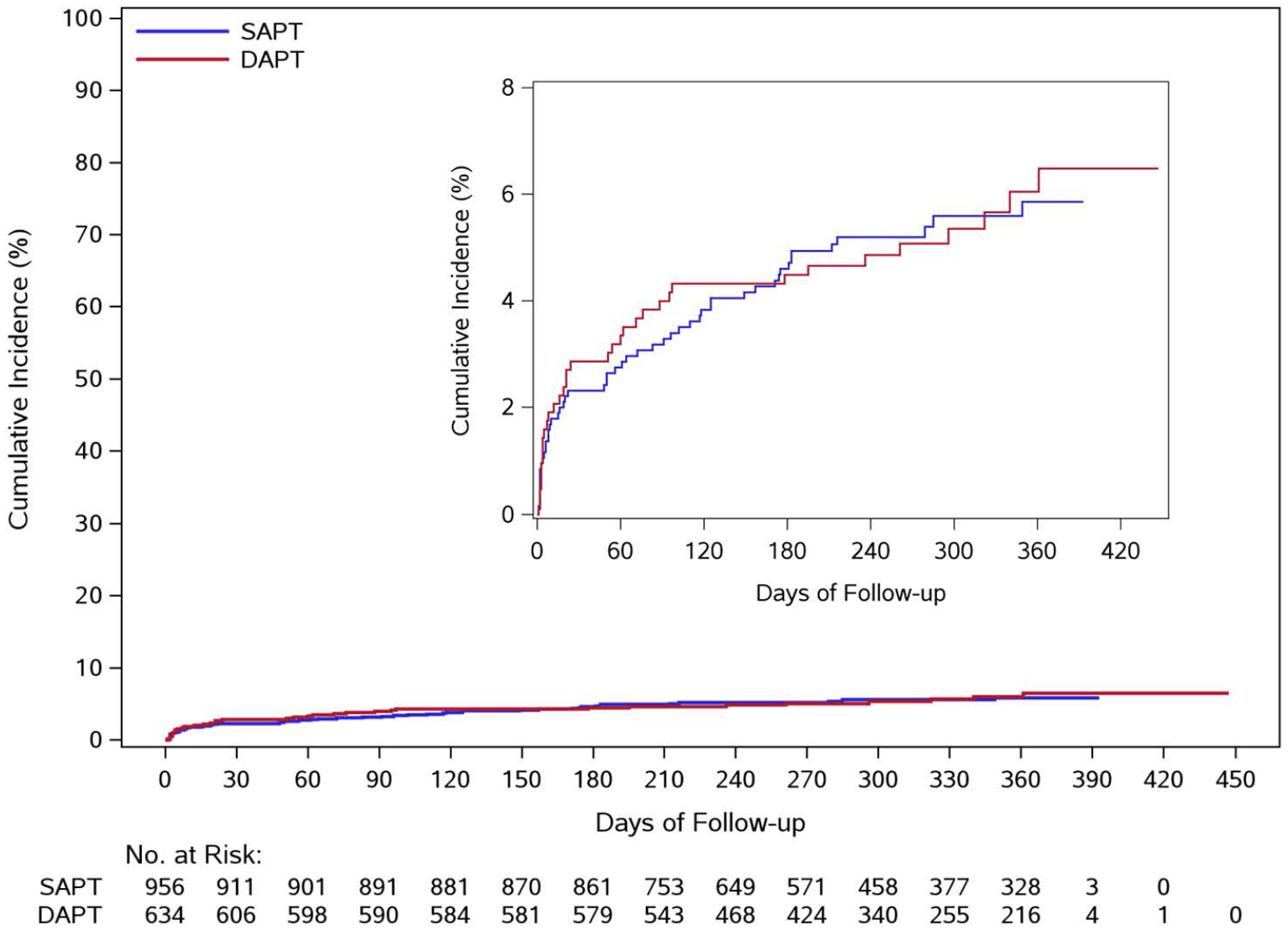
Kaplan-Meier curves for recurrent stroke, stratified by DAPT and SAPT group.
Outcome analysis for recurrent ischemic stroke using propensity scores and inverse probability treatment weighting.

CHANCE: clopidogrel in high-risk patients with acute non-disabling cerebrovascular events; CI: confidence interval, DAPT: dual antiplatelet treatment; LAA: large artery atherosclerosis; NIHSS: National Institutes of Health Stroke Scale; No.: number; POINT/CHANCE eligibility meant NIHSS ⩽ 3 and no intravenous thrombolysis nor endovascular treatment for the qualifying stroke (simplified eligibility criteria in the POINT/CHANCE trial); SAPT: single antiplatelet treatment; vs: versus.
p-Values were computed for interaction terms of the five models that include interactions.
Covariates for calculating propensity scores and inverse probability treatment weights include diabetes, congestive heart failure, coronary artery disease, geographic region, time from qualifying stroke to randomization in hours and IVT before randomization.
The program was developed based on the paper: Inverse Probability of Treatment Weighting (Propensity Score) using the Military Health System Data Repository and National Death Index.
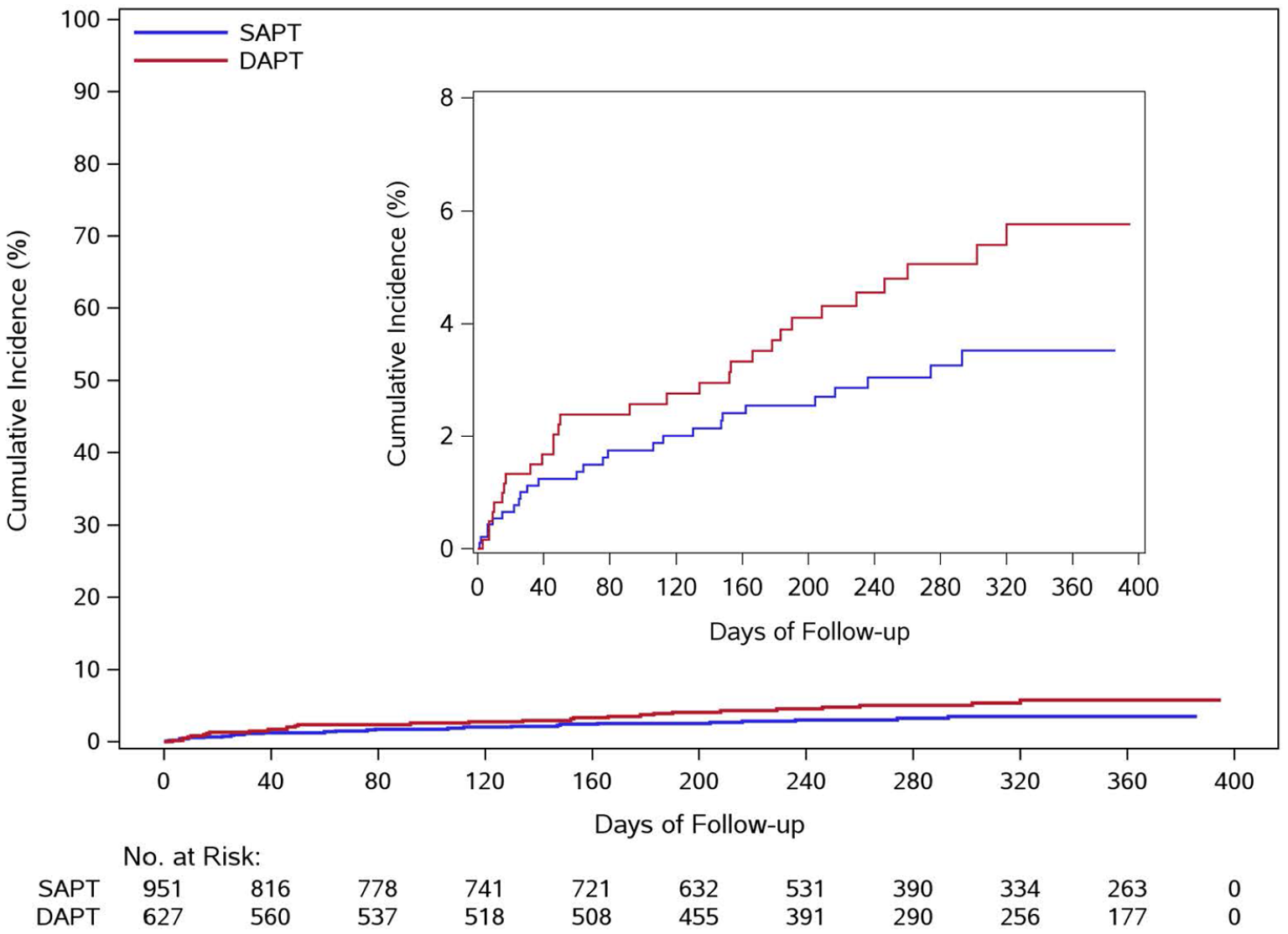
Kaplan-Meier curves for major or clinically relevant non-major bleeding, stratified by DAPT and SAPT group.
Outcome analysis for major or clinically relevant non-major bleeding. Adjusted model.

CHANCE: clopidogrel in high-risk patients with acute non-disabling cerebrovascular events; CI: confidence interval; DAPT: dual antiplatelet treatment; LAA: large artery atherosclerosis; NIHSS: National Institutes of Health Stroke Scale; No.: number; POINT/CHANCE eligibility meant NIHSS ⩽ 3 and no intravenous thrombolysis nor endovascular treatment for the qualifying stroke (simplified eligibility criteria in the POINT/CHANCE trial); SAPT: single antiplatelet treatment; vs: versus.
p-Values were computed for interaction terms of the four models that include interactions.
The variables included in this adjusted model are diabetes, congestive heart failure, coronary artery disease, geographic region, time from qualifying stroke to randomization in hours and IVT before randomization. In all other adjusted models, the included variables are diabetes, congestive heart failure, coronary artery disease, geographic region, NIHSS, time from qualifying stroke to randomization in hours and IVT before randomization.
Discussion
In this cohort of patients with non-cardioembolic ischemic stroke culled from the phase 2 PACIFIC-STROKE trial, all with available baseline MR-DWI, we did not find statistically significant effect modification by large artery atherosclerotic (LAA) etiology or stroke size on baseline MRI on DAPT versus SAPT efficacy or safety.
Forty-six percent of patients with NIHSS ⩽ 3 had an infarct size >15 mm on MR-DWI. Nearly the same number of participants from the DAPT as from the SAPT group received asundexian. Since we did not find a statistically significant difference between the asundexian assignment of the DAPT group versus SAPT group, we considered its potential impact on outcomes negligible. Additionally, asundexian was not associated with risk for recurrent stroke in the main trial analyses. Recurrent ischemic stroke occurred in 6% of placebo, 6% of asundexian 10 mg daily, 6% of axundexian 20 mg daily and 5% of asundexian 50 mg daily; the rate of recurrence did not differ from placebo for any group. 2
The numerically strongest, not statistically significant efficacy of DAPT (aspirin/clopidogrel) was in the subgroup of patients with NIHSS ⩽ 3 and no acute recanalization therapy for the qualifying stroke, that is, patients that would have been potentially eligible for the POINT/CHANCE trials. Presence of large-artery atherosclerosis did not seem to influence DAPT effectiveness. Concerning safety – that is, bleeding risk – the numerically lowest DAPT-associated risk was in the subgroup of patients with NIHSS ⩽ 3 and no acute recanalization therapy for the qualifying stroke, this effect was not statistically significant. The baseline infarct size on MR-DWI (>15 mm vs ⩽15 mm) did not appear to influence DAPT safety.
Regarding efficacy, the observed rate of recurrent symptomatic ischemic stroke in the DAPT versus SAPT groups (4.4%/4.8%) was lower than those observed in POINT (4.6%/6.3%), CHANCE (7.9%/11.4%) and INSPIRES (9.0%/6.8%), all comparing aspirin and clopidogrel to clopidogrel only. The latter two trials enrolled mainly Chinese patients, which have a high prevalence of intracranial arterial stenoses and are thus at higher risk of stroke recurrence. 12 While the strongest signal for DAPT efficacy was in the subgroup of patients who would have been eligible according to POINT/CHANCE, 52% of patients in the SAPT group would have met POINT/CHANCE criteria, suggesting real-world underuse of DAPT in at least some patients. In contrast, 29% of patients in the DAPT group did not meet the POINT/CHANCE criteria. A broader real-world use of DAPT has been documented. 13 In Western Europe and Australia, the proportion of DAPT use was higher than in Eastern Europe and Asia, possibly reflecting increased concerns on bleeding in the latter world regions. We found no significant interaction between the antiplatelet regimen and the cause of stroke. Specifically, there was no indication that DAPT effectiveness differed between patients with LAA and those with other causes of stroke. Prima vista, this is unexpected. CLAIR and CARESS – the two trials often cited as suggesting an even greater effectiveness of DAPT among patients with underlying symptomatic atherosclerotic stenosis – only included patients with symptomatic atherosclerotic stenosis, and their primary outcome was transcranial microembolic signals. No etiology by antiplatelet regimen interaction analysis is possible from CLAIR and CARESS. In these trials it was not possible to test the interaction between ischemic stroke etiology and antiplatelet regimen, since all patients had symptomatic atherosclerosis and outcomes were not reported separately in patients with lacunar and non-lacunar infarct.14,15 Moreover, post-hoc analyses from CHANCE and POINT found no increased DAPT-effectiveness among patients with either intracranial or cercival atherosclerotic disease.12,16 One reason is the likely high rate of misclassification into stroke of non-atherosclerotic versus atherosclerotic origin, since many strokes believed to be of small vessel disease or cryptogenic etiology arise from branch artery or non-stenotic atherosclerotic disease. 17
Concerning safety, a dilemma can arise when the NIHSS is low, but the corresponding infarct size on MR-DWI is larger that expected – a scenario frequent among patients with NIHSS ⩽ 5 presenting with visual field impairment (median DWI lesion volume 186 cm3), neglect (106 cm3) and aphasia (35 cm3), as shown in a post hoc analysis of the SWIFT trial. 10 The low NIHSS would, according to the POINT/CHANCE eligibility criteria, justify DAPT, but the large infarct size on MR-DWI would rise concerns of symptomatic intracranial hemorrhage (sICH) linked to DAPT. Our findings mitigate these concerns, suggesting that DAPT indication can rely upon the NIHSS score on admission. Indirect evidence for no increased sICH risk among patients with radiologically moderate or large strokes treated with early antithrombotic therapy comes from a post hoc analysis of the ELAN trial. ELAN showed that antithrombotic therapy with direct oral anticoagulants (DOAC) administered 6 to 7 days of a major stroke was not associated with an increased risk of sICH. 18 Admission MRI can also reveal the presence of hemorrhagic transformation (HT) of the ischemic lesion. While we did not assess for baseline HT, patients with baseline HT treated with early antithrombotic therapy with DOAC do not appear to have an increase sICH risk, compared with patients treated with late DOAC. 19
Strengths of this work are (1) the complete MRI availability among a contemporary cohort of patients, allowing to assess the implications of a discordance between NIHSS and MR-DWI infarct size; (2) central core MRI reading with independent interpretation by two radiologists masked to participant data and treatment assignment; (3) clinical endpoint definition (recurrent symptomatic stroke, clinically manifest or relevant bleeding).
Limitations are (1) the present study was a post hoc analysis of a phase-2 trial that was not designed nor powered to assess the superiority of DAPT versus SAPT; (2) the selection of DAPT versus SAPT was not randomized and prone to characteristic confounding found in observational analyses (3) the smaller sample size compared with CHANCE (n = 5170), POINT (n = 4881) and INSPIRES (n = 6100),3–5 reducing the power to detect a significant difference between DAPT and SAPT; (4) the potential for misclassification of stroke etiology, since it was not centralized and; (5) Analysis of stroke size on MRI was restricted to ⩽15 mm versus >15 mm in diameter; infarct volumes were not available.
In conclusion, we found no evidence of major treatment effect heterogeneity with DAPT compared with SAPT in patients with and without LAA. Patients whose baseline NIHSS ⩽ 3 appeared to have the greatest numerical, however not statistically significant, benefit of DAPT, with no evidence of major safety heterogeneity by stroke size on baseline MR-DWI.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251323180 – Supplemental material for Dual versus single antiplatelet therapy in patients with non-cardioembolic acute ischemic stroke and baseline MRI
Supplemental material, sj-docx-1-eso-10.1177_23969873251323180 for Dual versus single antiplatelet therapy in patients with non-cardioembolic acute ischemic stroke and baseline MRI by Gian Marco De Marchis, Anna Toebak, Tolga Dittrich, Dimitrios Vlachos, Angela Wang, Eric E Smith, Hardi Mundl, Pablo Colorado, Ashkan Shoamanesh and Robert G Hart in European Stroke Journal
Footnotes
List of abbreviations
aOR: adjusted odds ratio
DAPT: dual antiplatelet therapy
DOAC: direct oral anticoagulants
HT: hemorrhagic transformation
IQR: Interquartile range
LAA: large artery atherosclerotis
MRI-DWI: Magnet Resonance Imaging-Diffusion Weighted Imaging
NIHSS: National Institutes of Health Stroke Scale
SAPT: single antiplatelet therapy
sICH: symptomatic intracranial hemorrhage
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All coauthors or their institutions received financial support from Bayer for participation in the PACIFIC-Stroke trial except HM and PC.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and conduct was approved by local ethical boards.
Consent to participate
All participants or their legally authorized representative provided written informed consent.
Consent for publication
Not Applicable
Guarantor
Prof. G. M. De Marchis, corresponding author, is willing to take full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Contributorship
Authors contributed to the study as follows. The main idea was drafted by GMDM. The study design was set up by GMDM, AT, TD, AS, and RH. Data was pulled from the PACIFIC-Stroke trial, for which GMDM, TD, ES, HM, PC, AS, and RH collected data. Results were analyzed by AW and interpretated by GMDM, AT, and TD. The manuscript was drafted, written and finalized by GMDM, AT, and TD. All authors have reviewed the results and approved the final version of the manuscript.
Data availability
The de-identified, individual participant-level data can be made available to investigators for secondary analyses after review of a submitted proposal by the PACIFIC-Stroke steering committee.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
