Abstract
Introduction:
In Moyamoya angiopathy (MMA), mechanisms underlying cognitive impairment remain debated. We aimed to assess the association of cognitive impairment with the degree and the topography of cerebral hypoperfusion in MMA.
Methods:
A retrospective analysis of neuropsychological and perfusion MRI data from adults with MMA was performed. Ischemic and haemorrhagic lesion masks were created to account for cerebral lesions in the analysis of cerebral perfusion. Whole brain volume of hypoperfused parenchyma was outlined on perfusion maps using different Tmax thresholds from 4 to 12 s. Regional analysis produced mean Tmax values at different regions of interest. Analyses compared perfusion ratios in patients with and without cognitive impairment, with multivariable logistic regression analysis to identify predictive factors.
Results:
Cognitive impairment was found in 20/48 (41.7%) patients. Attention/processing speed and memory were equally impaired (24%) followed by executive domain (23%). After adjustment, especially for lesion volume, hypoperfused parenchyma volume outlined by Tmax > 4 s or Tmax > 5 s thresholds was an independent factor of cognitive impairment (OR for Tmax > 4 s = 1.06 [CI 95% 1.008–1.123]) as well as attention/processing speed (OR for Tmax > 4 s = 1.07 [CI 95% 1.003–1.133]) and executive domains (OR for Tmax > 5 s = 1.08 [CI 95% 1.004–1.158]). Regarding cognitive functions, patients with processing speed and flexibility impairment had higher frontal Tmax compared to other ROIs and to patients with normal test scores.
Discussion:
Cerebral hypoperfusion emerged as an independent factor of cognitive impairment in MMA particularly in attention/processing speed and executive domains, with a strong contribution of frontal areas.
Conclusion:
Considering this association, revascularization surgery could improve cognitive impairment.

Introduction
Moyamoya angiopathy (MMA) is characterized by progressive stenosis and occlusion of the terminal intracranial carotid artery and its main branches, with the development of a basal collateral network. These occlusive arterial lesions lead to chronic cerebral perfusion impairment, predominant in frontal areas, thus leading to stroke that is the main complication of the disease. 1
A few studies have suggested that cognitive impairment are common in adults with MMA.2–5 A high prevalence of these disorders (31%–42.5%) has been reported in a recent metanalysis by Kronenburg et al. 6 However, data is still limited due to the small sample size and the heterogeneity of the testing proposed. The mechanisms underlying cognitive impairment remain controversial. In addition to the impact of ischemic or hemorrhagic lesions on cognitive functioning, 7 little is known about the relationships between chronic cerebral hypoperfusion and the cognitive functioning of MMA patients. Emerging evidence suggests that a portion of cognitive impairment in MMA may be attributed to chronic cerebral hypoperfusion, especially in frontal areas.8–11 It has been observed that cognitive impairment can be present even in patients without apparent MRI lesions. 12 Some preliminary studies have found an association between frontal hemodynamic disturbances, their impact on white frontal matter diffusivity2,8,9 and executive dysfunction.8,9,13 However, the consideration of cerebral lesions was limited in these studies due to the small sample sizes, preventing proper statistical adjustment. Furthermore, small case series have suggested that revascularization surgeries, which aim to restore cerebral perfusion and reduce the risk of stroke may have a positive impact on cognitive outcome.3,14–16 These findings highlight the influence of cerebral hypoperfusion, both in terms of its degree and topography, on the cognitive performance of patients with MMA.
Cerebral hypoperfusion can be approached through the measurement of Tmax on perfusion-weighted magnetic resonance imaging (PWI-MRI). This parameter can be determined by automated software and is used in routine care in patients with acute stroke.17,18 In MMA and other patients experiencing chronic cerebral hypoperfusion, Tmax 4–6 s thresholds acquired on PWI MRI appears to be the most reliable parameters and thresholds to assess critical hypoperfusion lower than 20 ml/100 g/min on Xe CT.19–21 In addition, the results of a preliminary study from our group suggested that the volume of hypoperfusion lesions outlined by these thresholds could be relevant to determine the efficacy of surgery in MMA. 21
This study aimed to assess the relationship between cognitive impairment and cerebral hypoperfusion as measured with MRI Tmax in adult patients with MMA. We hypothesized that cognitive impairment may be independently associated with MRI hypoperfusion after adjustment on cerebral lesion volumes.
Methods
We conducted a retrospective single-center, hospital-based study of consecutive patients with MMA followed in our tertiary hospital (Toulouse University Hospital) between March 2008 and January 2019.
Population
Patients were eligible for the study if they met the following inclusion criteria: (1) adults (age ⩾ 18 years old) with MMA (Moyamoya disease or syndrome) according to the criteria of the research committee on the pathology and treatment of spontaneous occlusion of the Circle of Willis 22 ; (2) with neuropsychological assessment (NPA) performed before any surgery of revascularization; (3) interpretable perfusion-weighted imaging (PWI) MRI performed within the 6 months of the NPA and before any revascularization surgery; (4) no cerebrovascular event (cerebral infarction and/or brain hemorrhage) between MRI and NPA. Patients were excluded if they presented with all other non-vascular neurological and /or psychiatric disease potentially responsible for cognitive impairment (e.g. Parkinson disease, Alzheimer disease, Schizophrenia), and/or visual, auditory and oral or written expression skills that were insufficient for neuropsychological tests to be carried out properly. Patients with major cognitive impairment related to moyamoya angiopathy (i.e. dementia) according to the DSM-5 criteria were not included due to incomplete cognitive data. These patients are unable to perform a battery of tests sufficient to assess accurately cognitive domains or functions.
We recorded demographic, clinical and radiological characteristics of MMA on baseline: age at presentation (first symptoms) and at the moment of neuropsychological assessment, disease versus syndrome, vascular risk factors, family history, clinical presentation (first event): transient ischemic attack (TIA), ischemic stroke, intracerebral hemorrhage, other manifestations as headache, epilepsy, fortuitous discovery. The radiological data were coded for the presence or the absence of bilateral or unilateral angiopathy, posterior circulation involvement, MRI acute and chronic ischemic or hemorrhagic lesions, the Suzuki grading, and visible transdural anastomoses.
Neuropsychological assessment
Neuropsychological assessment is an integral part of the neurological follow-up protocol for MMA patients admitted to the stroke unit of the University Hospital of Toulouse. In the case of stroke, the NPA was performed at least 3 months after an acute symptomatic cerebrovascular event. Three cognitive domains were identified, including executive functioning, attention/processing speed and anterograde memory. Executive domain was explored with the Trail Making Test (TMT) for flexibility, the Stroop test for inhibition, the Digit Span test for working memory and with semantic (animal) and phonologic (P) verbal fluency tests for initiation. We used the score of the Code subtest (WAIS-4), the time variables of the Stroop naming and reading test and of the TMTA to characterize the processing speed functions. The D2 test and the Zazzo test were used to assess selective attention. Anterograde memory domain was explored with the Free and Cued Selective Reminding Test (FCSRT), and/or the 144 Memory Efficiency Battery (BEM 144), and/or the Brief Visuospatial Memory Revised Test (BVMT-R) and/or with the recall of the ROCF test (Rey-Osterrieth Complex Figure).
Due to the retrospective design of this study, cognitive raw scores were grouped according to the cognitive processes that they are supposed to measure followed by cognitive domain’s classifications. The percentage of patients who were tested for each cognitive function were detailed in Supplemental Table 1 (min-max bounds: 51%–96%).
Cognitive impairment was defined as impairment of at least one cognitive function with a score below 2 SD according to the clinical norms in at least two cognitive variables exploring the function.
Brain imaging
Magnetic Resonance Imaging acquisition
MRI studies were performed on a 1.5 (Gyroscan NT Intera; Philips, Best, the Netherlands) or a 3.0 T scanner (Skyra, Siemens, Germany). MRI protocol was standardized with DWI, T2-weighted imaging (T2), Fluid-Attenuated Inversion Recovery (FLAIR), T2 Gradient Echo (T2 GRE), two dimensions Gadolinium-enhanced T1-weighted imaging (T1 G), and Circle of Willis, Time Of Flight Magnetic Resonance Angiography (TOF) were acquired in axial plan. PWI was acquired using a gradient echo sequence with the following parameters: TR: 1710, TE 20 ms, FOV 100, matrix, “108 × 108”, flip angle “90°,” 4-mm-thick contiguous sections, NEX “1.” We measured cerebral perfusion using dynamic susceptibility contrast-enhanced MRI. Gadolinium (DOTAREM; Guerbet, Villepinte, France) was injected at a flow of 5 ml/s and a dose of 0.2 cc/kg.
Brain MRI analysis
As MMA usually spares the posterior fossa, we worked only on the brain hemispheres. For the different analyses, we manually excluded the cerebellum, the brainstem and the ventricular artifacts using the Olea Sphere software (version 3.0, Olea Medical, La Ciotat, France). We used a whole-hemisphere and a regional-based approaches to quantify cerebral perfusion, as described below.
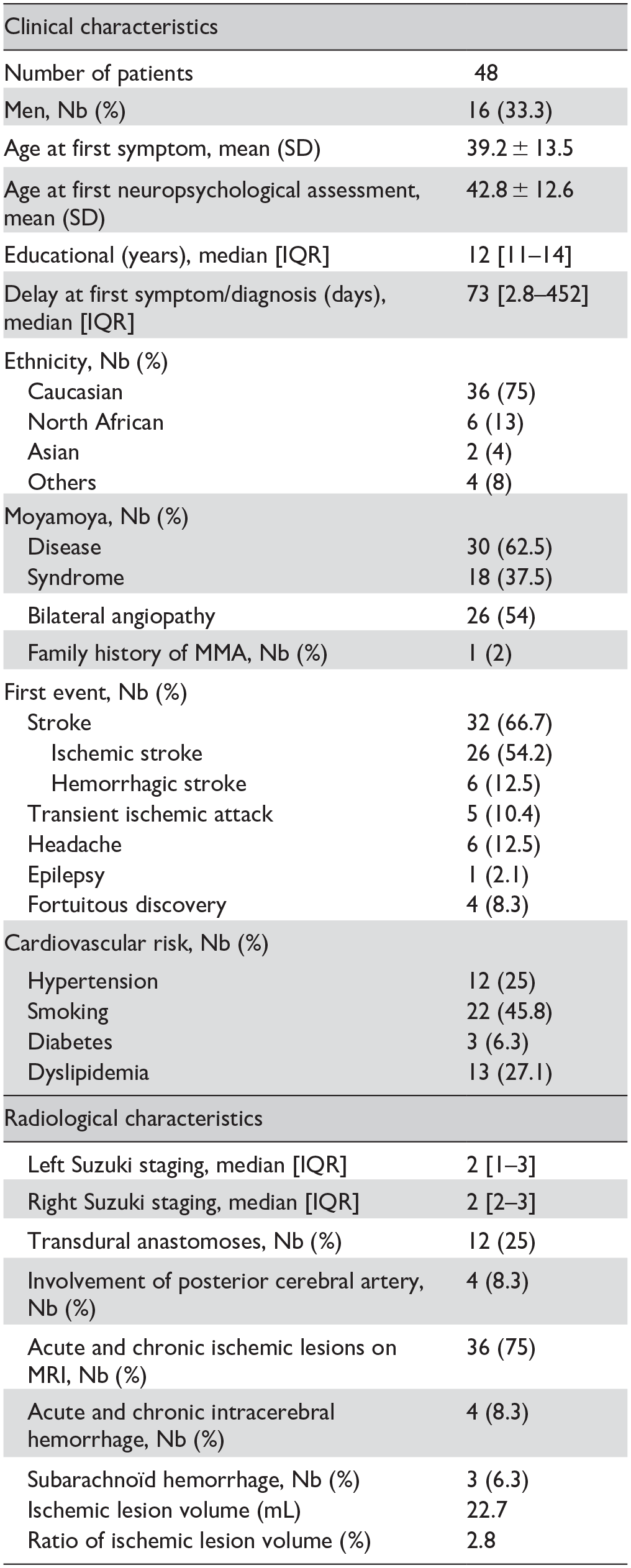
First, in order to quantify cerebral perfusion by controlling for the effect of cerebral lesions, we created a lesion mask for each participant that included the acute and chronic infarcts, the hemorrhagic lesions, and the white matter hyperintensities, using native FLAIR and T2 sequences. FLAIR was used for the infarcts and the white matter hyperintensities whereas T2 GRE was used for the hemorrhagic lesions. Lesions were manually segmented using MRIcron software (http://www.mccauslandcenter.sc.edu/mricro/npm/). Then, lesions were summed and were reported as a mask for each patient into the native cerebral perfusion sequence. The volumes of the brain lesions were converted into a ratio of the total brain volume obtained with Olea Sphere in order to homogenize the results and compare the patients who did not have the same total brain volume. Finally, to obtain a visual representation of lesion distribution, native lesion masks were binarized and spatially registered to T2 Montreal Neurological Institute template (2 × 2 × 2) to generate lesion maps for Moya patients using MRIcron software (Figure 1).
Lesion overlay maps for the two groups of patients with moyamoya: (a) simple overlay lesion map of the moyamoya group of patients with cognitive impairment and (b) overlay map of the moyamoya group of patients without cognitive impairment. The number of overlapping lesions, including ischemic, haemorrhagic and white matter hyperintensities, is illustrated by different colors coding increasing frequencies from sky blue (n = 3) to yellow (n = max (i.e. 24 in patients with cognitive impairment; 22 without cognitive impairment)). Lesions appear to be evenly distributed in both groups of moyamoya patients.
• Whole-hemisphere brain perfusion analysis
Perfusion brain volumes were estimated by Tmax maps calculated using Olea Sphere® software. For the chronic hypoperfusion measurement of MMA, we used different thresholds of Tmax: every second between 4 and 10 s, and every 2 s beyond. Volumes of cerebral hypoperfused parenchyma obtained for each threshold of Tmax of 1 s were also extracted. In the same way of brain lesion volumes, the distribution of hypoperfused volumes was converted to a ratio of the total hemisphere volume.
• Regional-based brain perfusion analysis
After a co-registration on native FLAIR sequences, we extracted Tmax values in four regions (Frontal, Parietal, Temporal and Basal ganglia) using ROIs extracted from the Automated Anatomical Labeling atlas (AAL https://doiorg.proxy.insermbiblio.inist.fr/10.1006/nimg.2001.0978). ROIs concerned territories of internal carotid arteries which are most concerned by MMA. Thanks to lesion masks, voxels inside lesion volumes were excluded for further analysis. For each patient, and in each region, a mean Tmax was calculated from the Olea Sphere® maps.
Standard protocol approvals, registrations, and patient consents
According to the French ethic and regulatory law, retrospective studies based on the exploitation of usual care data should not be submitted to an ethics committee but they must be declared or covered by reference methodology of the French National Commission for Informatics and Liberties (CNIL). Patients were informed that their codified data will be used for the study. Toulouse University Hospital signed a commitment of compliance to the reference methodology MR-004 of the French National Commission for Informatics and Liberties (CNIL). After evaluation and validation by the data protection officer and according to the General Data Protection Regulation, this study meets all the criteria. This study was registered in the register of retrospective studies of Toulouse University Hospital (register number: RnIPH 2021-65) and covered by the MR-004 (CNIL number: 2121529 v1).
Statistical analysis
Demographic data and clinical data for all patients were presented as counts and percentages, means and standard deviations (SD), and medians and interquartile ranges (IQR).
To investigate the impact of cerebral perfusion on cognition, we chose to compare the perfusion volume ratios of both hemispheres between patients with and without cognitive impairment (regardless of the nature of cognitive impairment) using the two quantitative approaches (whole-hemisphere and regions-based analyses). Demographic and clinical data of these two groups of MMA patients were also compared. To avoid potential artifacts and loss of information on extreme Tmax values, we have chosen to limit our analysis to Tmax < 12 s. We have presented the results as hypoperfused ratio volume of parenchyma (e.g. >4 s, >5 s, etc.) and comparisons were adjusted P-values for Bonferroni-Holm method. We used the χ2 test for categorical variables and the Mann-Whitney U test for continuous variables, where appropriate.
Given our hypothesis that cerebral hypoperfusion would be predominantly observed in the frontal areas of the brain, we performed additional analyses to compare brain perfusion volume ratios in relation to the presence of cognitive impairment within the three different cognitive domains. For the regional-based perfusion approach, we looked specifically for cognitive functions impairment in order to approach clinical-radiological correlations. We compared the mean values of Tmax between the different ROIs, in the patients with and without cognitive impairment, using the Wilcoxon signed-rank test. We then compared the mean values of Tmax in each ROI between the two groups using the Mann-Whitney U test.
Multivariable logistic regression analyses were also performed for all cognitive comparison groups using lesion volume and mean volume of hypoperfused parenchyma to assess the predictive factors of impairment. Other factors associated in the univariate analysis (p < 0.05) were added to the models, unless there was a strong interaction between two factors.
The significance level was set at p < 0.05. Statistical analysis was performed with SPSS 14.0 software (IBM corporation, 2016).
Results
During the study period, 79 patients were screened. Forty-eight were enrolled. Reasons for exclusion were as follows: three died within 3 months of an acute cerebrovascular event, nine patients had dementia, four had an insufficient level of French for neuropsychological testing, two had severe aphasia, six refused to undergo neuropsychological testing, three were lost to follow-up, and four had no PWI MRI available.
Population
The final cohort consisted of 48 patients with MMA (66.7% women, mean age at the first neuropsychological assessment 42.8 ± 12.6 years). For more details see Table 1. Eighteen (37.5%) had Moyamoya syndrome. Twenty-eight patients (58.3%) had a clinical history of stroke at presentation: 23 with an ischemic stroke (47.9%) and 5 with a hemorrhagic stroke (10.4%). Eight patients had a history of TIA (16.7%). The median duration between the first symptom and the diagnosis of MMA was 73 days (IQR 2.75–452). Brain lesions appear to be evenly distributed in both groups of moyamoya patients (Figure 1).
Clinical and radiological characteristics of MMA patients.
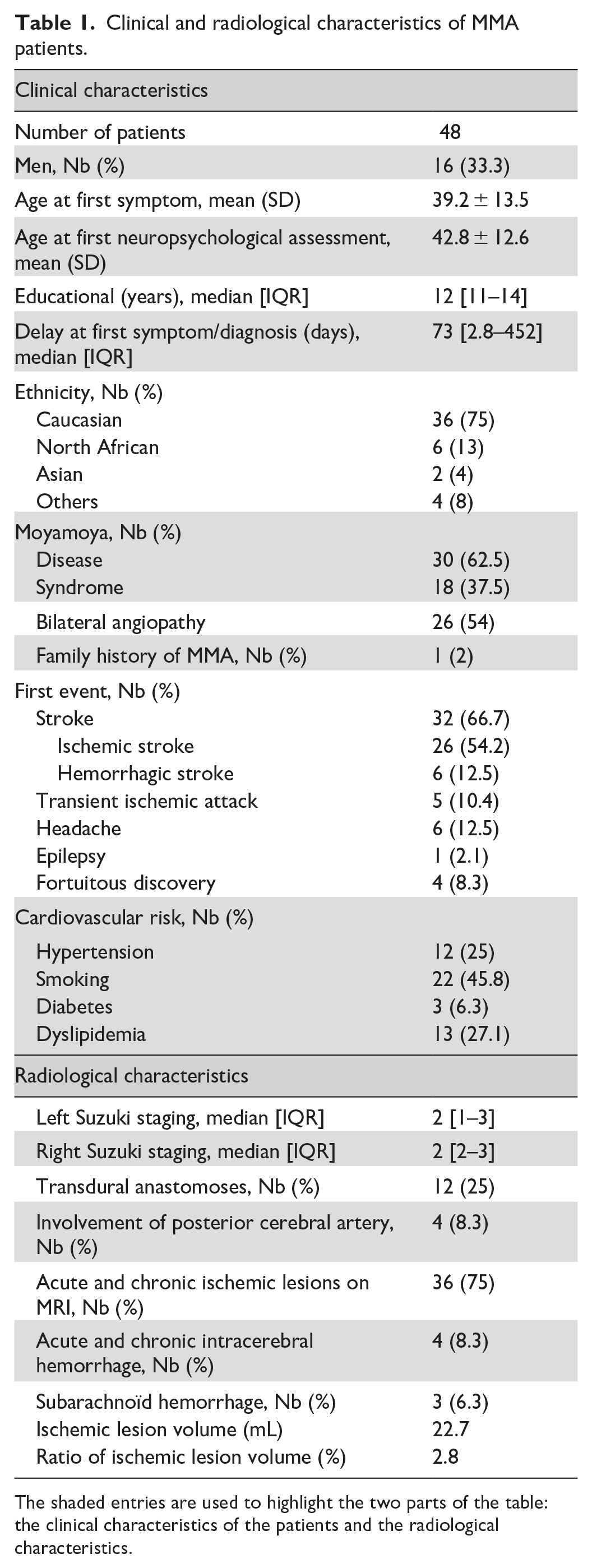
The shaded entries are used to highlight the two parts of the table: the clinical characteristics of the patients and the radiological characteristics.
Cognitive impairment
Twenty patients (41.7%) had a cognitive impairment. Attention/processing speed and memory domains were preferentially and equally impaired (11/46, 24%) followed by executive domain (11/48, 23%). More specifically, the most altered cognitive functions were flexibility (9/44, 20%), inhibition (5/25, 20%) and processing speed (8/45, 18%). Mean neuropsychological z-scores are detailed in Table 1.
Factors of MMA cognitive impairment
Patients with cognitive impairment [20/48 (41.7%)] had a larger ratio of lesion volume (p = 0.03) and parenchyma volume with Tmax > 4 s (p = 0.004), Tmax > 5 s (p = 0.006), and Tmax > 6 s (p = 0.004) (Table 2).
Clinical and radiological determinants of cognitive impairment.
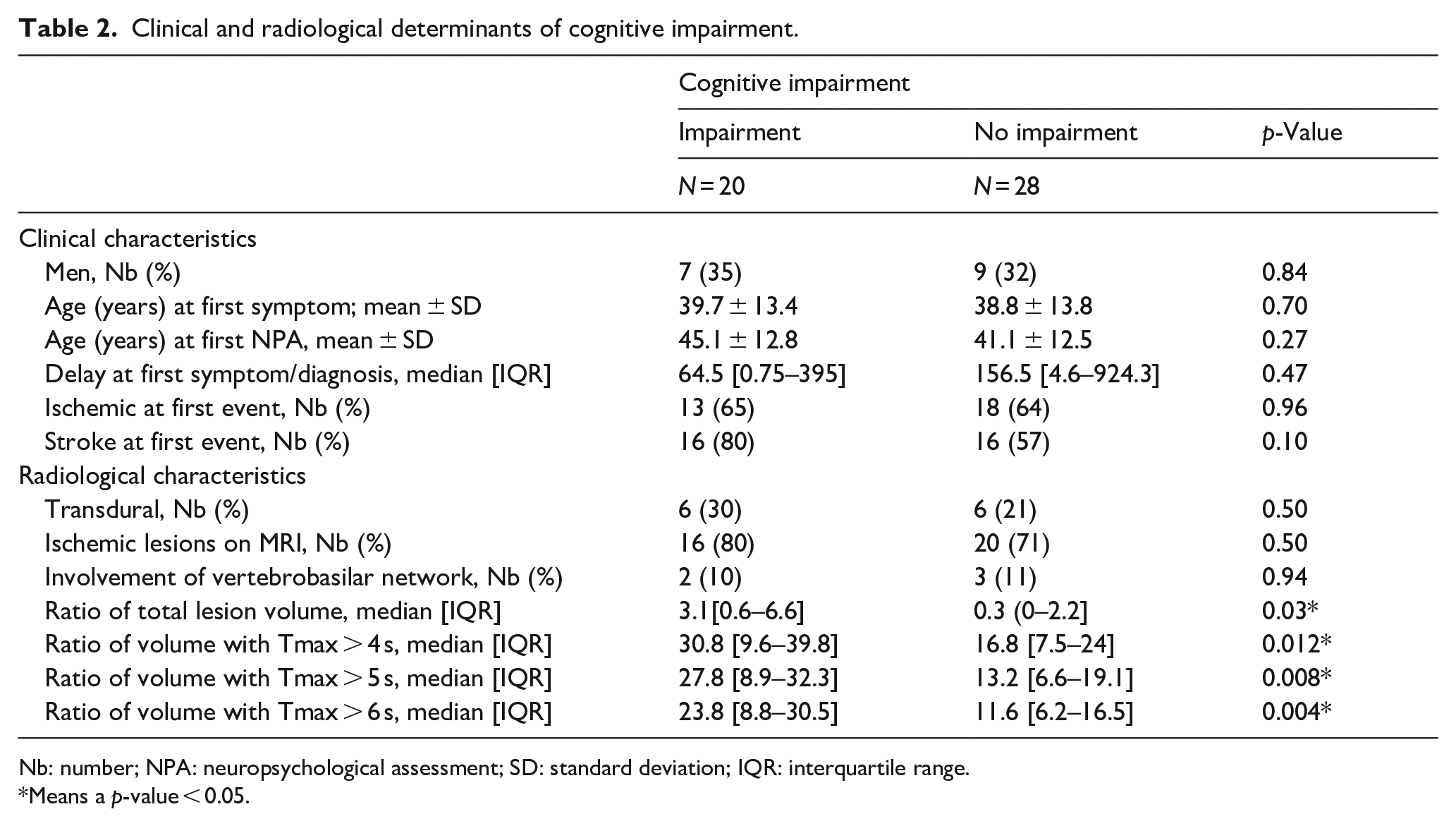
Nb: number; NPA: neuropsychological assessment; SD: standard deviation; IQR: interquartile range.
Means a p-value < 0.05.
In multivariable logistic regression analysis, a ratio of hypoperfused parenchyma with Tmax > 4 s was independently associated with cognitive impairment (OR = 1.06, CI 95% [1.008–1.123]). The independent association was also found using the thresholds Tmax > 5 s (OR = 1.08, CI 95% [1.012–1.145]) and Tmax > 6 s (OR = 1.08, CI 95% [1.011–1.16]) (Table 3).
Logistic regression.
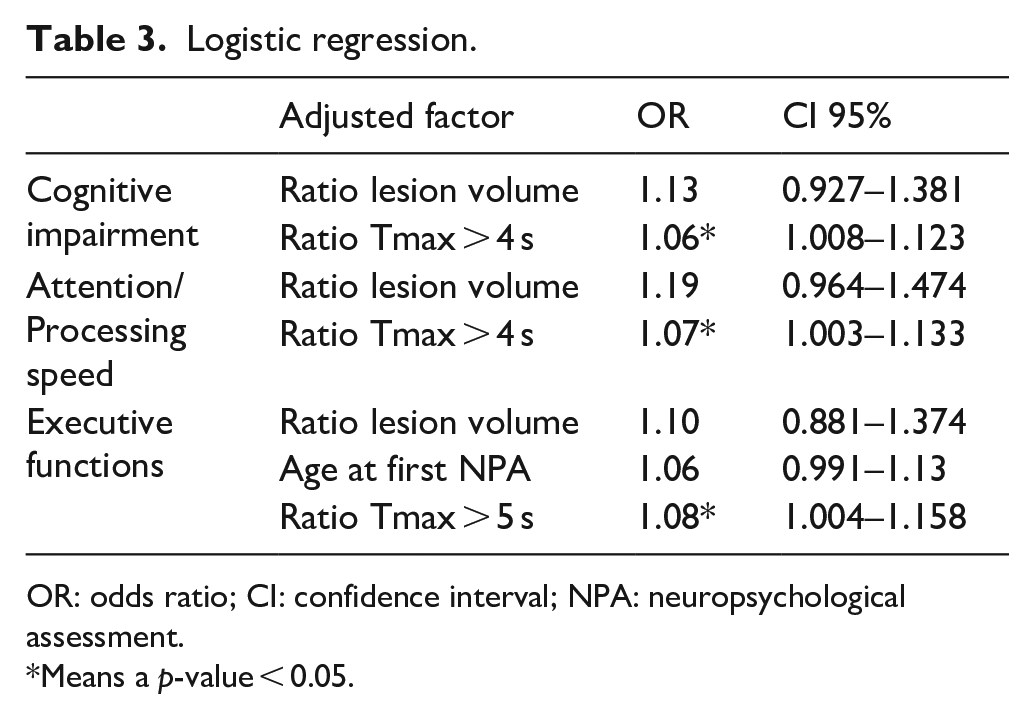
OR: odds ratio; CI: confidence interval; NPA: neuropsychological assessment.
Means a p-value < 0.05.
Determinants of specific cognitive domains impairment (tables in supplemental data)
• Attention/processing speed
Patients with attention/processing speed impairment were more likely to have at first stroke event (11/11 patients vs 19/35, p = 0.01), a greater ratio of lesion volume (p = 0.002)] and parenchyma volume with Tmax > 4 s (p = 0.002), Tmax > 5 s (p = 0.003), and Tmax > 6 s (p = 0.003).
In multivariable logistic regression analysis, a ratio of hypoperfused parenchyma with Tmax > 4 s was independently associated with impairment in the attention/processing speed domain (OR = 1.07 [CI 95% 1.003–1.133]). We did not include stroke at first event in the model due to a strong interaction between this factor and the lesion volume.
• Executive domain
Patients with executive domain involvement were older at first neuropsychological assessment (p = 0.039), had a greater ratio of lesion volume (p = 0.05) and parenchyma volume with Tmax > 4 s (p = 0.005), Tmax > 5 s (p = 0.003), and Tmax > 6 s (p = 0.001).
In multivariable logistic regression analysis, after adjusting the age at first neuropsychological assessment and the ratio of lesion volume, hypoperfused parenchyma with Tmax > 5 s (OR = 1.08 [CI 95% 1.004–1.158]) or Tmax > 6 s (OR = 1.1; CI 95% [1.011–1.191]) was independently associated with executive domain impairment.
• Anterograde memory
No correlation was found with impairment of anterograde memory.
Regional analysis
Mean frontal Tmax was longer in patients with impaired flexibility (5.35 ± 2.74 s vs 3.43 ± 1.49 s; p = 0.04) and processing speed (5.58 ± 1.93 s vs 3.40 ± 1.87 s; p = 0.004) compared to those with normal cognitive z-scores, whereas no difference was observed for parietal, temporal and basal ganglia areas (Figures 2 and 3).
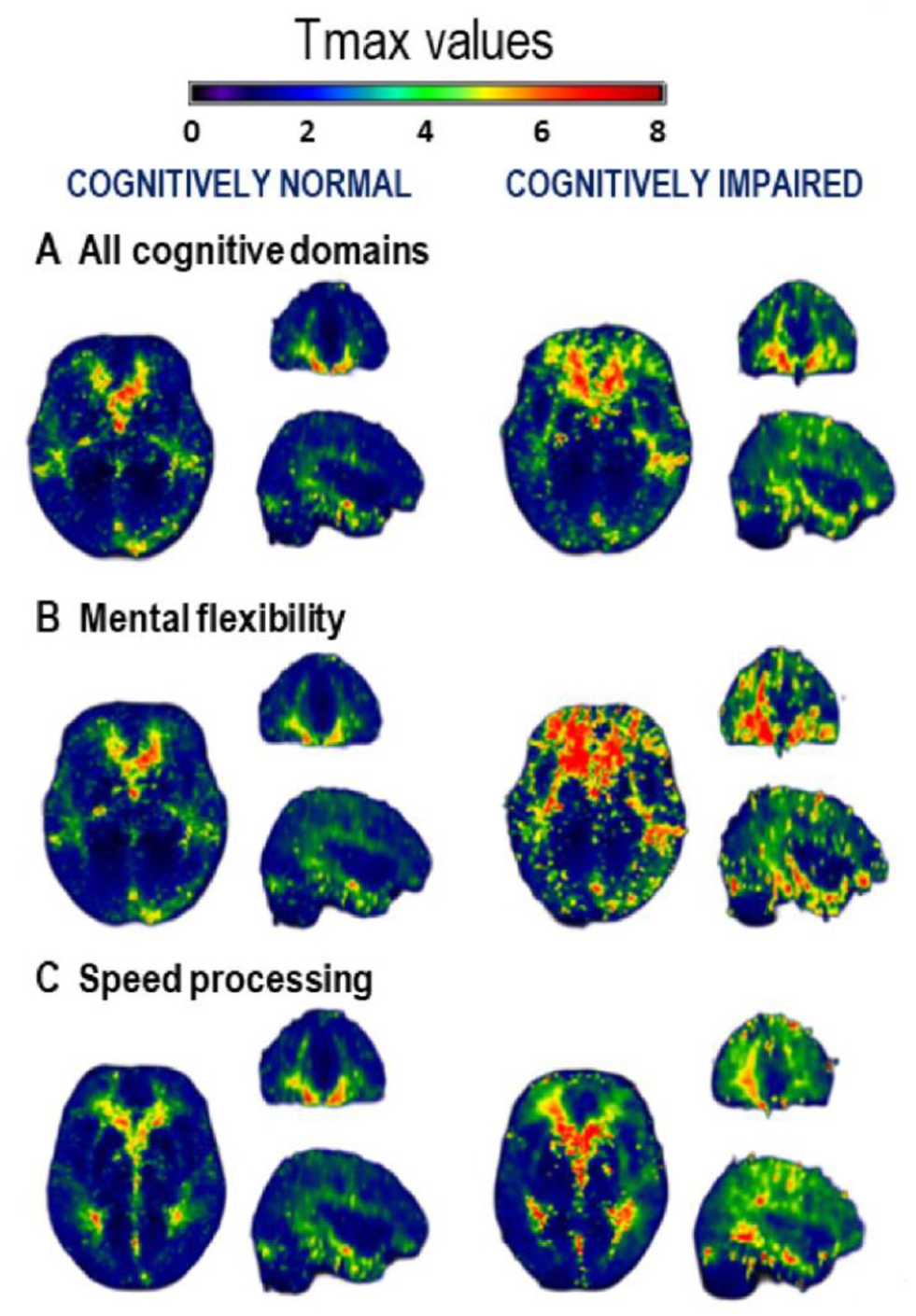
Representation of mean Tmax value among patients with or without impairment A. represents the Tmax values between cognitively normal versus cognitively impaired patients from all cognitive scores (MNI coordinates 66 84 31); from mental flexibility ((b) MNI coordinates 66 84 31); from processing speed ((c) MNI coordinates 66 84 36).
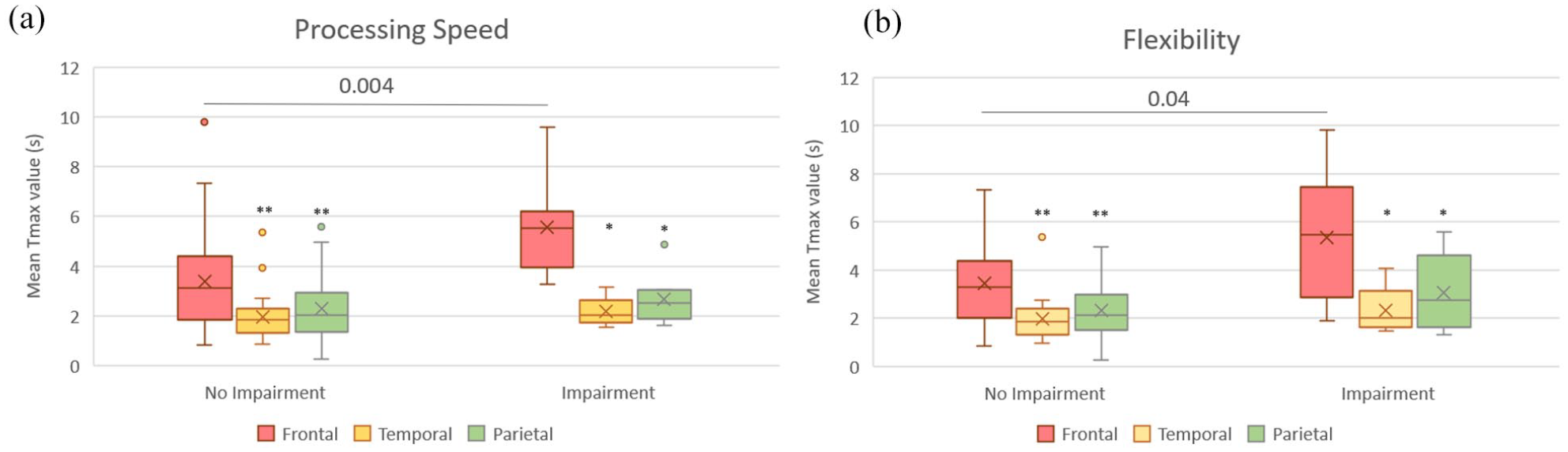
Box plot representing regional analysis of hypoperfusion using mean Tmax, among patients with and without impairment in processing speed (a) and mental flexibility (b).
Mean Tmax values did not differ between patients with and without impairment on inhibition and anterograde memory.
In patients with impaired flexibility and processing speed, mean frontal Tmax was higher than mean Tmax in temporal (p = 0.008; 0.012), parietal (p = 0.008; 0.012) and basal ganglia (p = 0.008; 0.012) areas. Frontal areas were the only ones with mean Tmax > 4 s (threshold for significant hypoperfusion in the literature) (Figure 3).
Frontal Tmax was still significantly longer than in other areas in MMA patients without cognitive impairment (flexibility p < 0.001; inhibition p < 0.01; processing speed p < 0.001; attention p < 0.001; memory p < 0.001) (Figure 3).
Discussion
We first documented in this cohort of 48 patients a high prevalence (41.7%) of cognitive impairment. Secondly, chronic cerebral hypoperfusion as measured with MRI Tmax was reported as an independent factor of cognitive impairment and mainly linked to attention/processing speed and executive domains.
Our study revealed that 41.7% of adult patients with MMA exhibited cognitive impairment, which aligns with the findings of a recent meta-analysis involving 127 adults with MMA. 6 However, it is important to acknowledge that the prevalence of vascular cognitive impairment in our study may have been underestimated due to the exclusion of patients with major cognitive impairment and/or moderate-to-severe aphasia. If language impairment had been included in our VCI calculation, then the prevalence of impairment would have been estimated to be 45.8%. The aim of the study was to assess the association of specific cognitive functions/domain impairment with perfusion.
The cognitive profile observed in our study encompassed multidomain impairment, including executive, attention, and processing speed functions. These findings align with numerous previous reports5,8,9,12,23,24 that have consistently highlighted these areas of cognitive dysfunction in MMA patients. We also identified higher prevalence of memory impairment. However, we assume that here this result may be overestimated and biased due to the limited number of memory variables included in the calculation of cognitive impairment. It is important to consider that different subgroups of memory processes, such as encoding and retrieval versus storage processes involving distinct brain areas, may contribute to the observed cognitive outcomes. 25 The absence of an association with imaging data may be attributed to this limitation. Further larger studies are needed to detail the subgroup of memory functioning and to confirm cognitive profile in adults with MMA.
The mechanisms of cognitive impairment in MMA are not fully understood. Regarding the literature of cerebrovascular pathologies, the role of stroke lesions is certain. However, 23% of patients without MRI stroke lesions had cognitive impairment in a previous cohort of adults with MMA 12 which was the first study to suggest the involvement of hypoperfusion. Over the last few years, increasing evidence supports that chronic cerebral hypoperfusion may be contributive to cognitive impairment.9–11,14,24,26 Our results showed that patients with cognitive impairment, especially those with impairment of processing speed/attention and executive domains, had larger hypoperfused volumes of parenchyma, using PWI-MRI hypoperfusion thresholds of stroke literature.17,19
After adjustment, particularly on the lesion volume, which was also strongly associated with univariate analysis, the ratio of parenchyma volume with Tmax > 4 or >5 s remained an independent factor of cognitive impairment. This independent association was found at the level of cognitive domains for attention/processing speed and executive functioning. Studies of hypoperfusion on animals also sustained this hypothesis. Rodents with carotid stenosis developed a progressive impairment of learning capacities in the Morris water maze. 27 In other human pathologies, several studies showed an association between mild cognitive impairment in Alzheimer’s disease and hemodynamic disturbances. 28 In MMA, very few previous studies have suggested that hypoperfusion could contribute to cognitive disorders, regardless of stroke lesions.12,29 In a series of patients revascularized or not, cognitive performance on the total Frontal Assessment Battery score was independent of the presence of frontal lobe infarction, whereas the authors found a significant link between frontal cognitive dysfunction and the value of cerebral blood flow in the anterior areas. 29 Recently, Shen et al., 30 found in Moyamoya patients without stroke significant association between mental flexibility and regional perfusion in bilateral lateral frontal area, temporal and semi-oval centrum regions. Our results, adjusted on ischemic burden are matching with these conclusions 31 and are confirmed by the regional-based analysis. In the whole population, we reported higher frontal Tmax values in comparison with parietal and temporal areas. Moreover, patients with impairment in flexibility and processing speed had a longer frontal Tmax than those with normal functions, with mean values greater than 4 s. These results suggested a critical role of frontal hypoperfusion in cognitive impairment of MMA, especially for processing speed and flexibility processes. 26 Chronic cerebral hypoperfusion may be responsible for subtle lesions of normal-appearing white matter considered as a “pre-leukoaraiosis state.” 32 These abnormalities can be approached indirectly in cerebrovascular diseases by measurement of MRI apparent diffusion or diffusivity/anisotropy on multimodal MRI.9,33–35 Pathologic studies have suggested microstructural changes with vasogenic edema, gliosis, demyelination and axonal loss.32,36 These lesions may disrupt connectivity in the cerebral network, particularly in frontal areas.2,37 Flexibility and processing speed requires these frontal–subcortical neuronal circuits38,39 and seems highly sensible to microstructural damages of white matter in MMA. 40
Our study has several limitations. Firstly, relationship between cognitive functioning and cerebral perfusion was studied only on patients able to complete neuropsychological assessment. This recruitment could have influenced the statistical association. Despite of heterogeneity of cognitive battery across MMA patients, we used cognitive z-score (calculated with clinical norms that are controlled for age and level and education) and grouped the tests according to the functions/domains that they are supposed to measure.
Attempting to evaluate the influence of strategic locations of brain lesions in the two groups of patients (cognitive/no cognitive impairment) would have been an interesting complementary approach. However, the lack of acquisition of 3D- T1 or Flair MRI sequences and the small size of our sample did not allow the use of Voxel-Based Lesion Symptom Mapping method. The overlap of lesions (Figure 1) between the two groups suggests that their distribution is quite similar. This means that the location of the lesion may not be the most important factor in cognitive impairment.
Regarding perfusion analysis, our results were obtained using a clinically available software but a specific post treatment to allow measurement of global and regional mean Tmax value. Validation with other techniques commonly used in MMA such as XeCT or TEP as well as other PWI software and modalities (perfusion CT) is required to determine the reproducibility of our findings. We decided to use thresholds of Tmax because it seemed to be more robust compared to CBF or CBV relative measurement obtained with gadolinium PWI MRI. Tmax thresholds have been validated and are now used in some conditions to select patients for thrombectomy. 18 Moreover, in patients with chronic hypoperfusion, including moyamoya, a 4–6 s threshold has been associated with chronic cerebral hypoperfusion lower than 20 mL/100 g/min on XeCT. In this study, the Tmax parameter appeared to be more reliable than MTT or CBF to assess hypoperfusion. 20 Other hemodynamic parameters can be used to study the influence of hypoperfusion on cognition. As our group and others have previously shown in small series, impairment of cerebrovascular reserve could be associated with cognitive dysfunction. 8 During the study period, this assessment was not performed systematically, and different measurement methods were used. Due to the lack of patients and this heterogeneity, we were not able to analyze this parameter.
We have analyzed patients with unilateral and bilateral angiopathy together. Tmax might depend on the compensatory mechanisms of the other hemisphere, especially in unilateral angiopathy. In univariate analysis, there was no association between cognitive impairment and unilateral/bilateral involvement, suggesting a weak influence of this parameter in our cohort. Due to the small number of patients in each subgroup (unilateral/bilateral), independent analyses with adjustment using logistic regression were not possible.
Finally, the independent association between Tmax volume and cognitive impairment did not mean a causal relationship. The Odds Ratio was significant but weak and close to 1, therefore our results must be interpreted with caution. Other prospective studies in multicentric cohorts are needed to confirm this result.
The potential relationship between frontal hypoperfusion and cognitive impairment has important clinical implications in MMA management. Revascularization surgeries aim to improve cerebral perfusion to prevent strokes. In a previous study, we described in 19 patients a decrease of slight (Tmax > 4 s) and severe (Tmax > 6 s) hypoperfused parenchyma volume after burr hole surgery.14,21 Some studies, using apparent diffusion coefficient or anisotropy fraction, have suggested that a part of subtle white matter lesions could be reversible by the improvement of perfusion.2,14,40 Thus, revascularization may improve some cognitive impairment, especially flexibility and processing speed that seemed to be the most associated with increased frontal Tmax. To the best of our knowledge, only case reports or small cohorts have previously suggested this fact.3,14,41,42 With these considerations, cognitive impairment in patients with significant hypoperfusion may be considered as an indication for surgery. Efficacy of surgery has to be confirmed in a large prospective cohort of patients with neuropsychological assessment before and several months after surgery. Indeed, the only study with a large sample size of 85 patients described rather a stabilization than an improvement of cognition. 43
Conclusions
Our study confirmed a high prevalence of cognitive impairment, around 40%, in a large cohort of adults with MMA. Flexibility, inhibition and processing speed were the most affected functions. Brain hypoperfusion volume as measured with Tmax was an independent factor of cognitive impairment, especially on the cognitive domains of attention/processing speed and executive functions. This impairment may be closely related to the degree of frontal hypoperfusion. Larger prospective studies are needed to assess whether revascularization surgeries, which aim to improve frontal perfusion, could be efficient to reverse a part of theses troubles.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241240829 – Supplemental material for MRI hypoperfusion as a determinant of cognitive impairment in adults with Moyamoya angiopathy
Supplemental material, sj-docx-1-eso-10.1177_23969873241240829 for MRI hypoperfusion as a determinant of cognitive impairment in adults with Moyamoya angiopathy by Marine Giroud, Mélanie Planton, Jean Darcourt, Nicolas Raposo, Pierre Brandicourt, Hélène Mirabel, Dominique Hervé, Alain Viguier, Jean-François Albucher, Jérémie Pariente, Jean Marc Olivot, Fabrice Bonneville, Patrice Péran and Lionel Calviere in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because, according to the French ethic and regulatory law, retrospective studies based on the exploitation of usual care data should not be submitted to an ethics committee but they must be declared or covered by reference methodology of the French National Commission for Informatics and Liberties (CNIL). This study was completed in accordance with the Helsinki Declaration as revised in 2013.
Informed consent
Verbal informed consent was obtained from all subjects before the study. Written informed consent was not obtained because it was a retrospective study.
Trial registration
This study was registered in the register of retrospective studies of Toulouse University Hospital (register number: RnIPH 2021-65) and covered by the MR-004 (CNIL number: 2121529 v1).
Guarantor
MG.
Contributorship
- MG, MP, JD, PP, LC: Drafting/revision of the manuscript for content, including medical writing for content; Major role in the acquisition of data; Study concept or design; Analysis or interpretation of data;
- NR: Drafting/revision of the manuscript for content, including medical writing for content; Analysis or interpretation of data;
- PB: Drafting/revision of the manuscript for content, including medical writing for content; Study concept or design; Analysis or interpretation of data;
- HM: Major role in the acquisition of data;
- DH, AV, JFA, JP, JMO, FB: Drafting/revision of the manuscript for content, including medical writing for content;
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.