Abstract
Introduction:
The diagnostic workup of stroke doesn’t identify an underlying cause in two-fifths of ischemic strokes. Intracranial arteriosclerosis is acknowledged as a cause of stroke in Asian and Black populations, but is underappreciated as such in whites. We explored the burden of Intracranial Artery Calcification (IAC), a marker of intracranial arteriosclerosis, as a potential cause of stroke among white patients with recent ischemic stroke or TIA.
Patients and methods:
Between December 2005 and October 2010, 943 patients (mean age 63.8 (SD ± 14.0) years, 47.9% female) were recruited, of whom 561 had ischemic stroke and 382 a TIA. CT-angiography was conducted according to stroke analysis protocols. The burden of IAC was quantified on these images, whereafter we assessed the presence of IAC per TOAST etiology underlying the stroke and assessed associations between IAC burden, symptom severity, and short-term functional outcome.
Results:
IAC was present in 62.4% of patients. Furthermore, IAC was seen in 84.8% of atherosclerotic strokes, and also in the majority of strokes with an undetermined etiology (58.5%). Additionally, patients with larger IAC burden presented with heavier symptoms (adjusted OR 1.56 (95% CI [1.06–2.29]), but there was no difference in short-term functional outcome (1.14 [0.80–1.61]).
Conclusion:
IAC is seen in the majority of white ischemic stroke patients, aligning with findings from patient studies in other ethnicities. Furthermore, over half of patients with a stroke of undetermined etiology presented with IAC. Assessing IAC burden may help identify the cause in ischemic stroke of undetermined etiology, and could offer important prognostic information.

Keywords
Introduction
Ischemic stroke is the second leading cause of death and disability and may reoccur in 20% of stroke patients. 1 Hence, the extensive diagnostic work-up of stroke patients is targeted at finding the cause of the stroke, in order to identify the optimal secondary prevention for a potential recurrent event. However, the underlying cause remains undetermined in 25%–40% of all ischemic strokes,2,3 complicating the selection of the most optimal strategy for secondary prevention.
Arteriosclerosis among the cerebropetal arteries is increasingly considered as an important risk factor for stroke. 4 Yet, the majority of studies focusing on the role of intracranial arteriosclerosis as a cause of ischemic stroke have been performed in Asian and Black populations, 5 where intracranial arterial calcifications (IAC), as a hallmark of intracranial arteriosclerosis, 6 are present in 72%–93% of ischemic stroke patients. 7 In contrast, there is a notable scarcity of data regarding IAC burden in white stroke patients, particularly regarding its associations with the underlying cause of ischemic stroke, in addition to key clinical parameters such as the severity of stroke symptoms and functional outcome. This scarcity of data may explain why intracranial arteriosclerosis is currently underrecognized as a cause of ischemic stroke in whites. 8 An overview of the IAC burden in white patients is required to determine whether IAC may explain those ischemic strokes with an undetermined cause in white patients.
To address these knowledge gaps, we assessed IAC presence, volume, and density as key characteristics of the IAC burden in a registry study of white ischemic stroke patients. We then investigated whether these IAC aspects are associated with the underlying cause of ischemic stroke, severity of stroke symptoms, and short-term functional outcomes.
Patients and methods
Study setting
We used data from the Erasmus Stroke Study (ESS). 9 The ESS is a clinical registry study of patients with neurovascular disease admitted to the academic Erasmus Medical Center in Rotterdam, the Netherlands. Patients included in the ESS underwent clinical evaluation at presentation in either the outpatient clinic, emergency care department, or neurology ward. In light of routine clinical care, these patients underwent blood sampling and CTA imaging. Written informed consent for participation in the study was obtained from all patients and the study was approved by the institutional ethics committee (Erasmus MC MERC, approval number MEC-2005-345). The data underlying this article cannot be shared publicly due to the privacy of participants in this study. However, anonymized data will be shared on reasonable request to the corresponding author.
Stroke assessment
Ischemic stroke was defined as a focal neurological deficit of presumed vascular origin lasting ⩾24 hours. Transient Ischemic Attack (TIA) was defined as a focal neurological deficit of presumed vascular origin lasting <24 hours. The most probable stroke pathogenesis was classified according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria. We further dichotomized “undetermined etiology” category into “several probable etiologies” or “unknown etiology” based on clinical records. Symptom severity was assessed at presentation using the National Institutes of Health Stroke Scale (NIHSS), and dichotomized to minor stroke or major stroke (NIHSS ⩽3 or >3). Event outcome was assessed at discharge using the modified Rankin Scale (mRS), with poor functional outcome defined as mRS >1 or death.
Assessment of intracranial arterial calcification
Imaging was performed using a 16-slice, 64-slice, or 128-slice MDCT system (Brilliance 64, Philips Healthcare Systems, Eindhoven, Netherlands; Sensation 16, Sensation 64, Definition, Definition AS+ or Definition Flash, Siemens Medical Solutions, Erlangen, Germany) with a standardized optimized contrast-enhanced protocol. The scans ranged from the ascending aorta to the intracranial circulation (3 cm above the sella turcica). Further details regarding scanning protocols are described elsewhere. 9 Image reconstructions were made with field of view of 120 mm, matrix size 512 × 512, slice thickness 1.0 or 0.75 mm, increment 0.6–0.4 mm, and an intermediate reconstruction algorithm.
We assessed IAC in the intracranial carotid arteries, the intracranial part of the vertebral arteries, and the basilar artery, and chose a threshold of 600 HU, based on recent literature at the onset of the study, to differentiate arterial calcification from luminal contrast. 10 Calcification in the intracranial carotid arteries was assessed from the segment within the carotid canal until the transition into the medial cerebral artery. Calcification in the vertebral arteries was assessed within the V4 segment, from the entrance within the foramen magnum until the merging into the basilar artery. Subsequently, basilar artery calcification was assessed from this merger until the top of the basilar artery. All raters were blinded for clinical data.
The assessment of IAC presence, volume and density, was performed using a custom-made plug-in for the freely available software ImageJ (version 1.46r, Rasband W.; National Institutes of Health, USA, MD). Regions of interest were drawn around hyperdense locations per investigated artery, on axial CTA slices. A detailed description on this evaluation method and its validity assessment has been published previously.11–13 Subsequently, a custom-made, python-based script (available from https://gitlab.com/radiology/aim/carotid-artery-image-analysis/kalk) automatically calculated the IAC volume and density using the pre-determined HU threshold and voxel dimensions, by applying the following method. After identifying those voxels within the region of interest with density above the HU threshold, the script calculated the calcification volume in millimeter cubed, and average HU density within the calcification by dividing the sum of selected voxel HU with the count of selected voxels. See Figure 1 for an example of the script output.
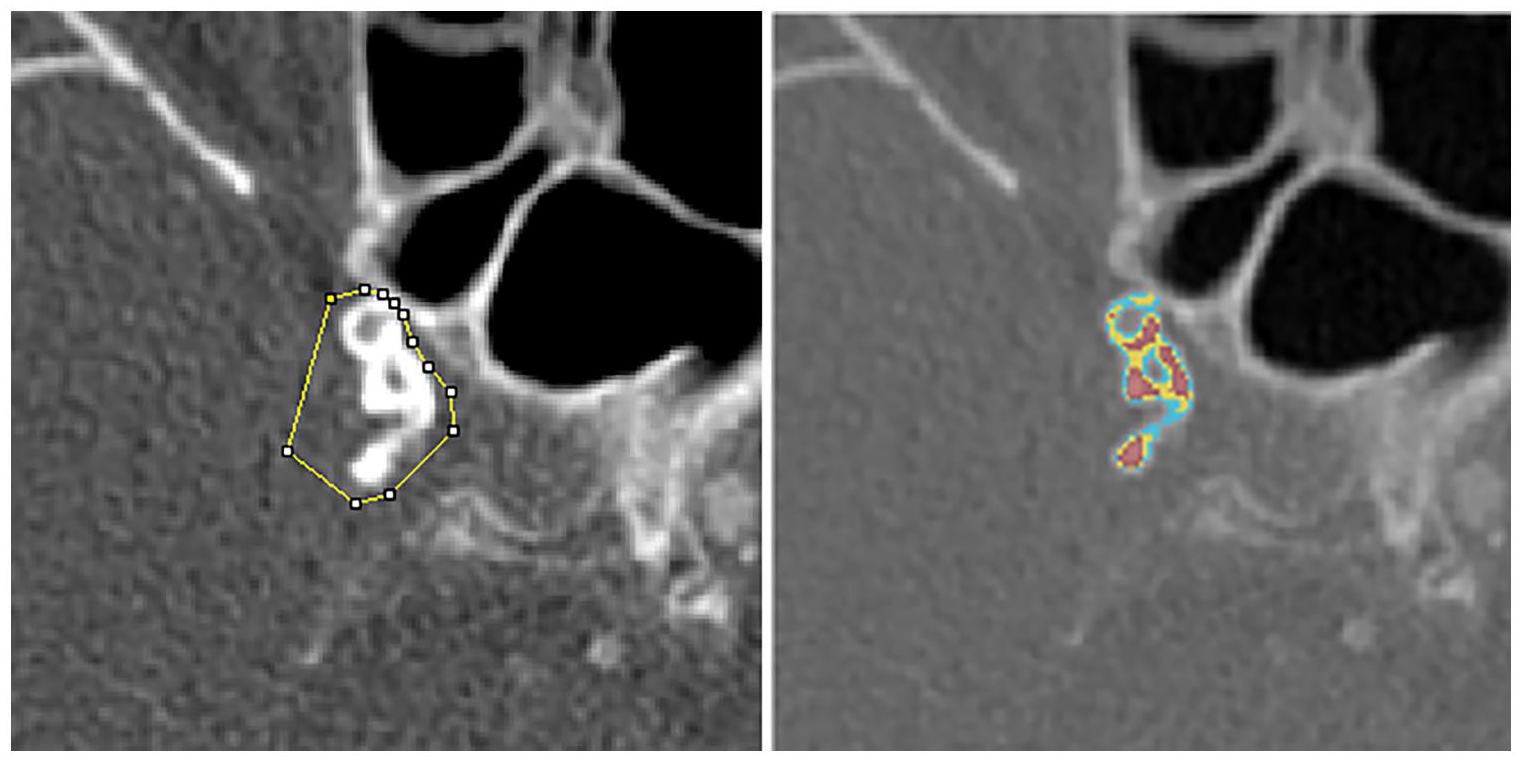
Example of a region of interest, drawn around a right intracranial carotid using ImageJ method (left) and corresponding Python script output (right). Cyan color denotes voxels with 600–800 HU, yellow 800–1000 HU and red >1000 HU.
This method yielded four distinct IAC characteristics for further analysis. First, prevalent IAC, defined by the presence of any volume of calcification above the threshold of 600 HU. Second, total IAC volume per patient, retrieved by summing calcification volumes per affected artery. Third, average IAC density per patient, retrieved by summing up the average calcification HU densities per affected artery divided by the number of affected arteries. Finally, using a HU threshold of 1000, we summed the volume of high-density calcification among the identified arterial calcifications into a total high-density IAC volume.
Covariate assessment
We retrieved the demographical, clinical characteristics and laboratory assessments including lipid profile of the patients from the medical records on admission, with data on ethnic background being collected by self-report. 9 Blood pressure was assessed during two episodes of 15 min of continuous noninvasive measurements. Presence of diabetes mellitus was defined as either a fasting plasma glucose level of >6.9 mmol/L, a 2-h post glucose loading level >11.0 mmol/L, or use of any diabetes medication. Kidney function impairment was assessed using the estimated glomerular filtration rate and defined as <60 mL/min. Smoking history was self-reported as never, previous, or current smoking.
Population for analyses
We included ischemic stroke and TIA patients, with available and adequately evaluable multi-detector computer tomography angiography imaging within 6 months after symptom onset, who presented for the first time to our medical institution between December 2005 and September 2010 and who reported themselves as having a white ethnicity. After the selection process illustrated in Supplement A, we analyzed imaging data from 943 patients.
Statistical analyses
Patient and IAC characteristics are reported as frequencies with percentages, mean and standard deviation (SD), or median and interquartile range (IQR) when skewed. Differences in IAC prevalence, volumes and densities between anterior and posterior arteries are initially explored using either McNemar test for nominal characteristics or Mann-Whitney U tests for skewed continuous characteristics. We also report differences in IAC prevalence between the five TOAST etiologies.
To investigate associations between IAC burden, stroke severity and functional outcome, we used the following strategy. First, odds ratios (OR) for IAC prevalence with major stroke and poor functional outcome were estimated using binomial logistic regression models, incorporating age at presentation, sex, blood pressure, serum lipid profile, comorbid diabetes, impaired kidney function, medication use, smoking history, and history of vascular diseases. Second, in order to deal with calcification values of 0 in line with previous studies using similar methods,11,13 we first performed a log-transformation of IAC volumes (ln[volume+1mm3]) and densities (ln[density+599HU]) to obtain a normal distribution of these continuous measures. We then repeated the above binomial regression analysis using these transformed variables. Lastly, we graphically described the distribution of mRS at discharge, stratified by IAC prevalence in a mRS shift analysis.
Apart from diabetes mellitus (18.9%) and blood pressure (11.5%), missing values in the covariates were less than 10%. Missing values were imputed using fivefold multiple imputation with 10 iterations, based on all variables in the adjusted model.
All statistical analyses were performed in “R for Windows” v4.2.2 (R Foundation for Statistical Computing, Vienna, Austria), using the packages “mice,” “UpSetR,” and “rankinPlot.”
Results
Table 1 shows an overview of the patient characteristics. The mean age of the study participants was 63.8 (14.0 SD) years, 452 (47.9%) were females and 382 (40.5%) patients had a TIA. We observed IAC among 588 patients, with a prevalence of 62.4%. Among these 588 patients, 218 (37.2%) were diagnosed with TIA while 370 (62.9%) were diagnosed with ischemic stroke. Supplement B shows patient characteristics stratified by diagnosis. Intracranial carotid artery calcification was present in 577 (61.2%) and vertebrobasilar artery calcification in 128 (13.6%) patients. Figure 2 provides an overview of the distribution of IAC among the five arteries investigated in this study.
Patient characteristics (N = 943).
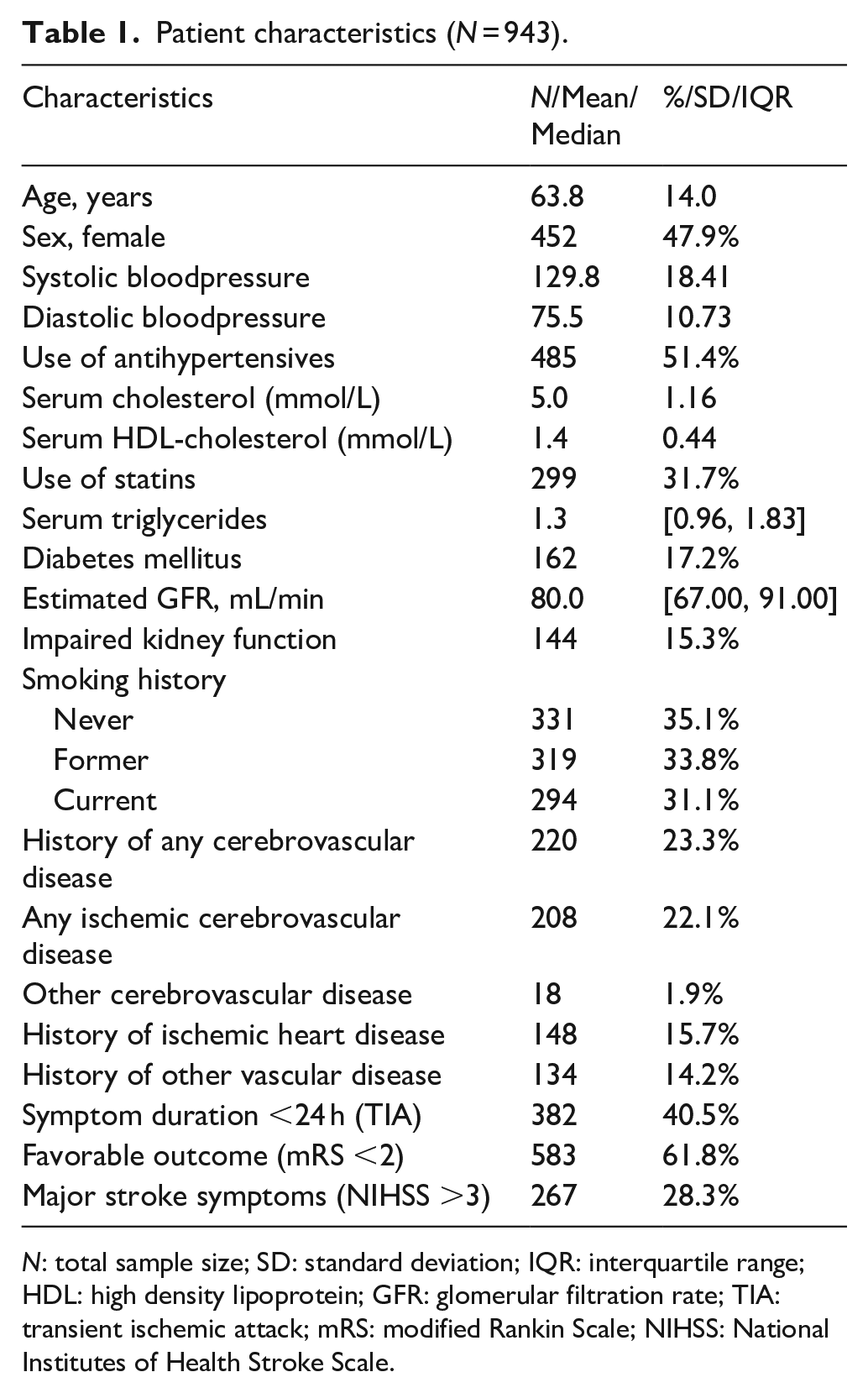
N: total sample size; SD: standard deviation; IQR: interquartile range; HDL: high density lipoprotein; GFR: glomerular filtration rate; TIA: transient ischemic attack; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale.
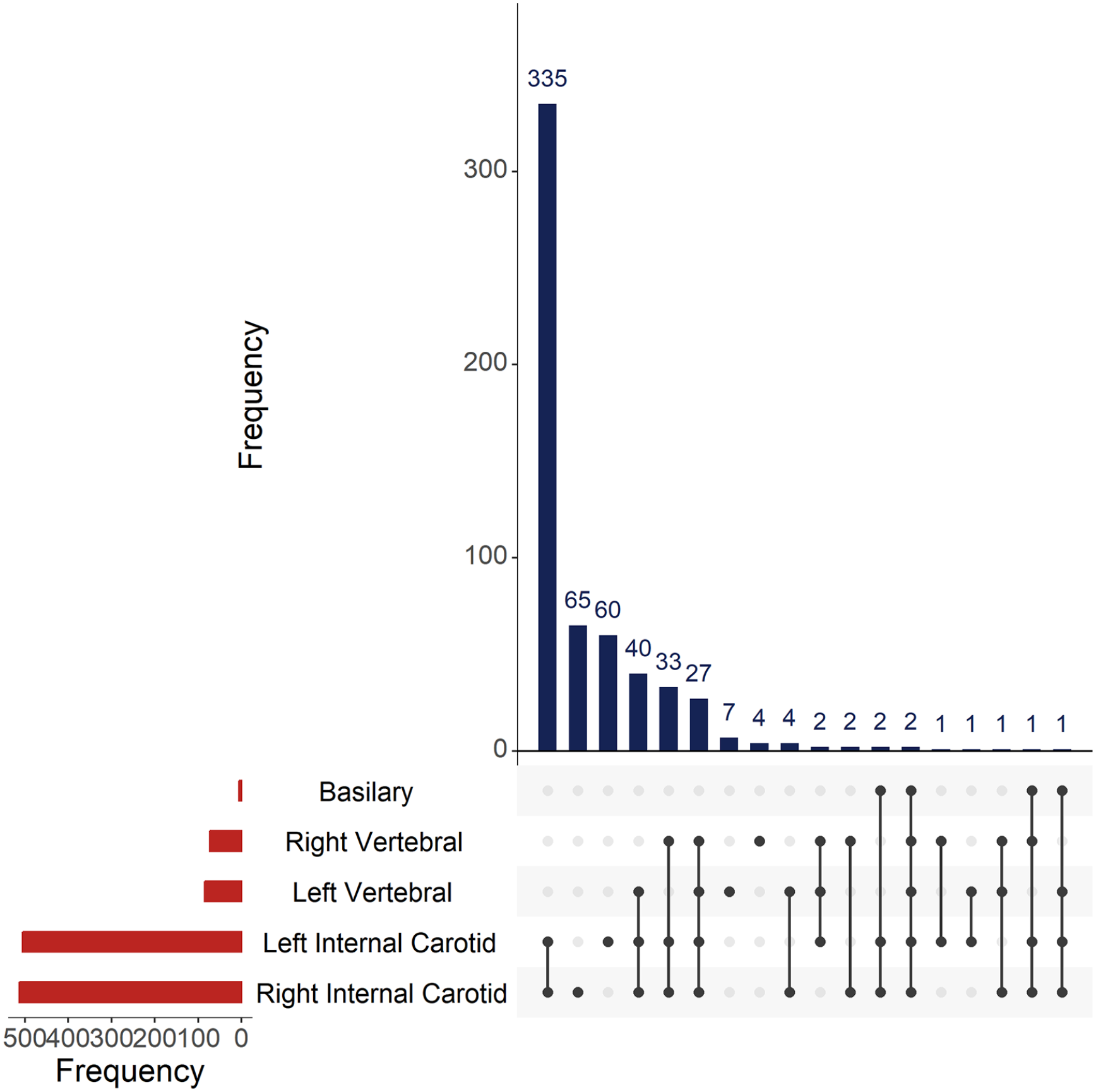
Combined histogram plot of the frequencies of arteries, and combinations of arteries, affected by IAC.
IAC burden by cause of ischemic stroke
Table 2 shows an overview of IAC characteristics by TOAST category. Notably, almost half of these patients presented with an undetermined etiology for their stroke, and the most prevalent causes were large-artery atherosclerosis and small-vessel occlusion. Importantly, IAC was present in 253 (58.6%) of all 432 patients with an undetermined etiology. This frequency remained high after restricting to those patients with an unknown etiology rather than several possible ones, where IAC was present in 226 (56.2%) versus absent in 176 (43.8%) patients. Furthermore, IAC was present among 134 (84.8%) of 158 patients with large-artery atherosclerosis, and in 96 (60.8%) of the 158 patients with small-vessel occlusion.
IAC presence among the different TOAST classification categories.
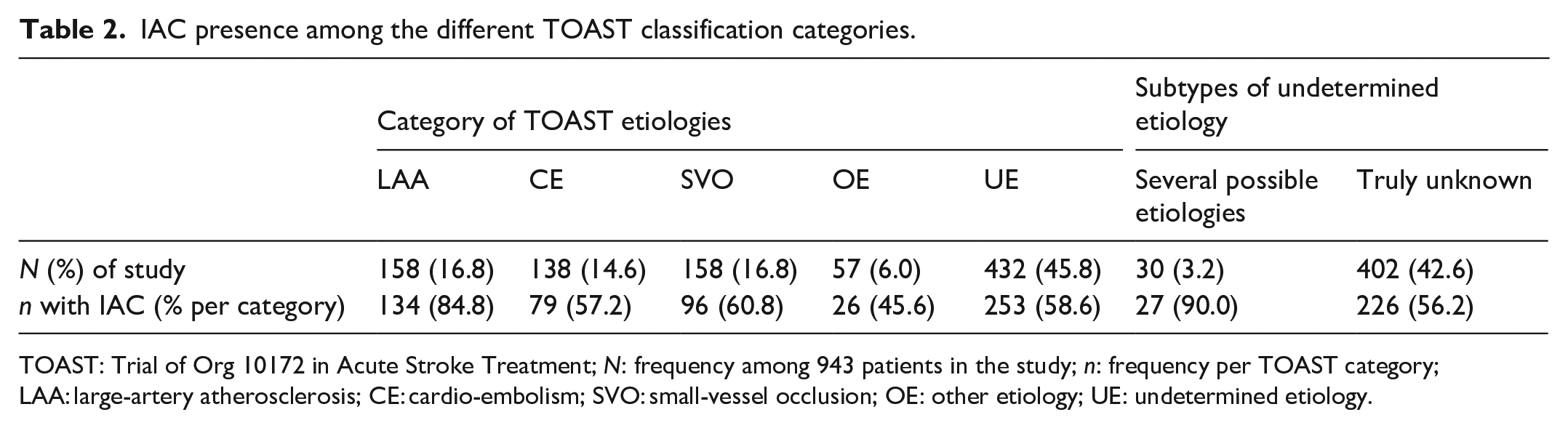
TOAST: Trial of Org 10172 in Acute Stroke Treatment; N: frequency among 943 patients in the study; n: frequency per TOAST category; LAA: large-artery atherosclerosis; CE: cardio-embolism; SVO: small-vessel occlusion; OE: other etiology; UE: undetermined etiology.
Quantification of IAC burden
In those with IAC, median overall IAC volume was 21.0 mm 3 [IQR 5.2–70.6]. The volumes of intracranial carotid artery calcification were larger than calcifications of the vertebrobasilar arteries (median 22.8 mm 3 [IQR 5.1–70.6] vs median 3.0 mm 3 [IQR 1.0–11.6], p < 0.05). The median averaged density of all IAC was 751.2 HU [IQR 704.1–805.4], with higher values among the intracranial carotid artery calcifications compared to calcifications of the vertebrobasilar arteries (median 707.0 HU [IQR 758.0–818.7] vs median 684.7 HU [IQR 654.7–735.6], p < 0.05). Additionally, >1000 HU components were more frequently seen among intracranial carotid artery calcifications compared to vertebrobasilar artery calcifications, 431 (73.3%) versus 42 (32.8%), p < 0.05. These components were also larger in intracranial carotid calcifications compared to vertebrobasilar calcifications (volume >1000 HU 4.7 mm 3 [IQR 1.2–15.8] vs, 2.4 mm 3 [IQR 0.7–4.3]), p < 0.05).
IAC burden and associations with ischemic stroke severity and outcome
Table 3 shows results of regression analyses of IAC characteristics on clinical aspects. Major stroke symptoms were seen at presentation in 267 (28.3%) patients and a favorable outcome of the event was seen in 583 (61.8%) patients. After full adjustment, IAC presence, volume and density were all associated with major stroke symptoms at presentation. Figure 3 demonstrates the shift in mRS per IAC prevalence, where patients with IAC appeared to more often have unfavorable functional status at discharge. However, IAC presence, volume and density were not associated with functional outcome in fully adjusted regression models, as seen in Table 3.
Associations between IAC characteristics and clinical aspects of stroke.
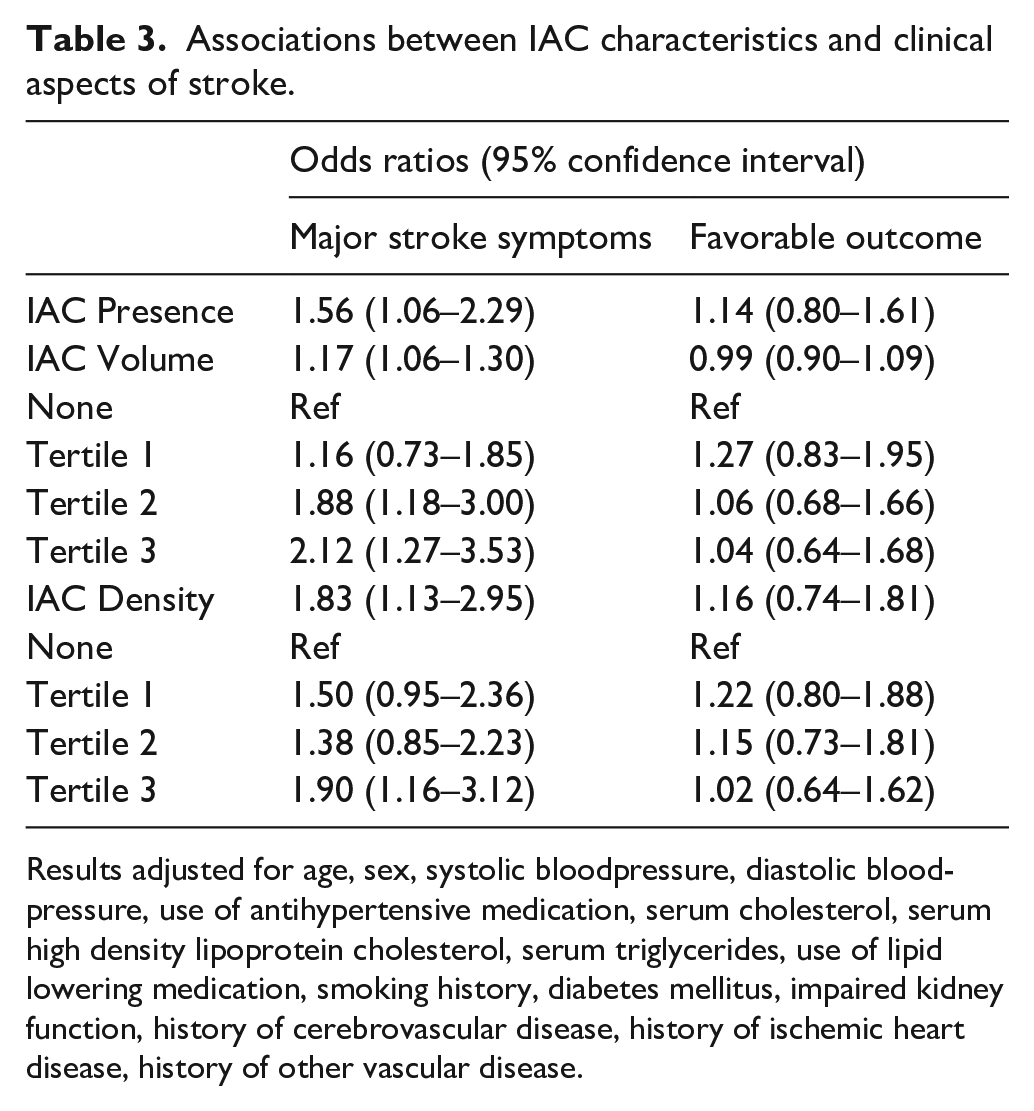
Results adjusted for age, sex, systolic bloodpressure, diastolic bloodpressure, use of antihypertensive medication, serum cholesterol, serum high density lipoprotein cholesterol, serum triglycerides, use of lipid lowering medication, smoking history, diabetes mellitus, impaired kidney function, history of cerebrovascular disease, history of ischemic heart disease, history of other vascular disease.
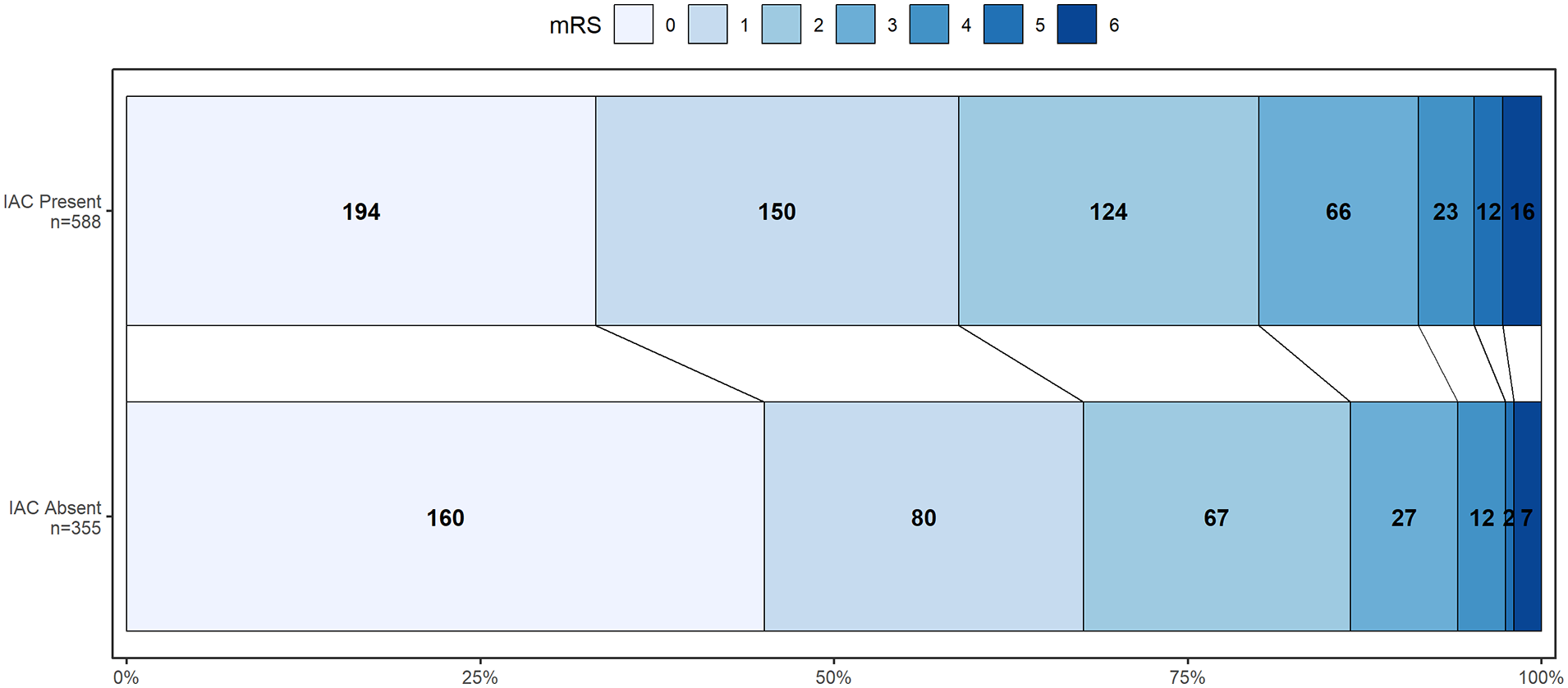
GrottaBar plot showing the distribution of modified Rankin Scale scores at discharge, by presence or absence of IAC.
Discussion
In this cross-sectional study among white patients with recent ischemic stroke or TIA, IAC was present in over half of patients where no cause could be found for their stroke. In clinical guidelines, 14 intracranial arteriosclerosis is persistently underappreciated as a cause of stroke in white populations, 15 whereas in Asian and Black populations it is acknowledged as an important cause of stroke. 4 Yet, our results perhaps suggest that intracranial arteriosclerosis could be a possible cause of the majority of strokes with an undetermined etiology in these white patients. Indeed, the prevalence of IAC in this study aligns with other reports of comparable studies across various other ethnicities, which range from 40.0% to 93.1%.16–18 And notably, in a white population-based study, four out of five elderly people had IAC and those with IAC developed stroke more often than those without IAC. 11 Our study therefore adds to a growing accumulation of evidence 19 indicating that intracranial arteriosclerosis is also an important factor in the pathophysiology of stroke in white populations.
Moreover, these results suggest a possible role for IAC burden within in the large-artery atherosclerosis etiological framework behind ischemic stroke. One potential implication following this, is that patients with ischemic stroke and concomitant IAC, but without a different determined cause, could possibly be allocated to the large-artery atherosclerosis category. Though ideally this idea requires further longitudinally designed studies to establish whether patients with IAC and with an undetermined etiology for their stroke may benefit from the use of double antiplatelet therapy and aggressive lipid management in the prevention of a recurrent stroke, similar to patients with a stroke caused by large-artery atherosclerosis. 20
Furthermore, CT based density of arterial calcification has previously been investigated as a potential risk modifier for the risk of acute coronary events, where high-density arterial calcifications are associated with a lower myocardial infarction risk.21,22 While clear reference values for arterial calcification in the heart-brain axis are lacking in the literature, it is assumed that a higher density of carotid artery calcification also reflects more stable atherosclerotic disease, with a subsequently lower risk of ipsilateral stroke. 23 Our study provides clear reference values for IAC density and volumes of high-density IAC, paving the way for further longitudinal studies on whether IAC density is a modifier of stroke risk in those with intracranial arteriosclerosis.
We additionally found that a larger burden of IAC, indicated by presence of IAC, larger IAC volumes and higher IAC densities, were all associated with worse stroke symptoms at presentation. This finding may reflect underlying vascular remodeling processes of both large and small intracranial arteries, where arterial elasticity declines due to the replacement of vascular smooth muscle cells with calcifying vascular smooth muscle cells. 24 A reduction in this arterial elasticity may result in hypertensive end-organ damage to the brain, and the presence of IAC may therefore signify a compromised status of the whole cerebrovasculature which is then reflected in worse symptoms once a stroke occurs. Patients with IAC in this study also more often experienced worse outcomes at discharge as illustrated by Figure 3, although this association was entirely attenuated by patient age in multivariate regression analyses. This finding aligns with recently published longitudinal data from a study among North-American stroke patients, 25 where a higher burden of IAC was associated with a higher post-stroke mortality. Future longitudinal data assessing metrics of functional outcome beyond mortality risk in stroke patients with IAC could provide valuable prognostic information for patients.
Strengths and limitations
Important strengths of this study are its use of a validated method11–13 to assess different aspects IAC in a large sample of patients, and its inclusion of many modifiable risk factors as covariates in regression analyses. Furthermore, we were able to include important clinical aspects, such as probable etiology, symptom severity and functional outcome, in one comprehensive overview of the IAC burden among white stroke patients. Nevertheless, several important limitations of this study should be discussed. First, while the use of contrast-enhanced imaging enables accurate discrimination of arterial calcification volume from other calcified intracranial structures, such as dural calcifications and bony protrusions often found in and around the carotid canal, it requires setting a high HU threshold to distinguish arterial calcification from luminal contrast. The use of contrast imaging is therefore a trade-off between more accurate assessment of arterial calcification and an underestimation of IAC prevalence, as it will not detect lower density calcifications. Although, lower density calcifications are likely to be smaller in size, meaning that they may also be missed in studies using non-contrast CT imaging where slice thickness is often considerably larger compared to contrast-enhanced imaging. Second, no follow-up information, such as survival time in either short- or long-term or subsequent events, was available in this study. Thus, we were unable to assess long-term consequences of IAC in these patients, such as the risk of recurrence or long-term mortality or differences in survival time. Third, this registry study was performed within a tertiary stroke center only, and our results may thus not be fully representative of non-tertiary stroke centers. Finally, while IAC prevalence, volume and density are all important facets to describe in an overview of IAC burden, we did not measure the IAC subtypes of intimal and medial calcification, as no validated method exists to score these subtypes on CTA imaging.
Conclusion
To conclude, intracranial arteriosclerosis may explain the majority of ischemic strokes with undetermined etiology in white populations. Our findings emphasize that IAC is also involved in the pathophysiology of stroke in whites, similarly to its role in Asian and Black populations. 15 Furthermore, IAC could provide important prognostic information for stroke survivors. Finally, this work offers an important basis for longitudinal studies on the role of IAC density in stroke.
Non-standard abbreviations and acronyms
IAC = Intracranial Artery Calcification, ESS = Erasmus Stroke Study, TOAST = Trial of Org 10172 in Acute Stroke Treatment, TIA = Transient Ischemic Attack, NIHSS = National Institutes of Health Stroke Scale, mRS = modified Rankin Scores, HU = Hounsfield units, HDL-C = High-Density Lipid Cholesterol, GFR = glomerular filtration rate.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241239787 – Supplemental material for Burden of intracranial artery calcification in white patients with ischemic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241239787 for Burden of intracranial artery calcification in white patients with ischemic stroke by Bernhard P Berghout, Robin YR Camarasa, Dianne HK Van Dam-Nolen, Aad van der Lugt, Marleen de Bruijne, Peter J Koudstaal, M Kamran Ikram and Daniel Bos in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: BPB, DB and MKI were supported by the Erasmus Medical Center MRACE grant (grant number 386070).
Informed consent and ethical approval
Written informed consent for participation in the study was obtained from all patients and the study was approved by the institutional ethics committee (Erasmus MC MERC, approval number MEC-2005-345).
Trial registration
Not applicable, as this work is not a clinical trial.
Guarantor
Daniel Bos
Contributorship
The authors have made a substantial intellectual contribution to the conception and design of the study (BPB, PJK, MKI, DB), acquisition of data (BPB, RYRC, HKVDN, DB), analysis and interpretation of data (all authors), drafting the article (BPB) or drafting a significant portion of the article or figures (BPB, RYRC, HKVDN, DB). BPB, HKDVN and DB had full access to the data in the study and take responsibility of data integrity and accuracy of data analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
