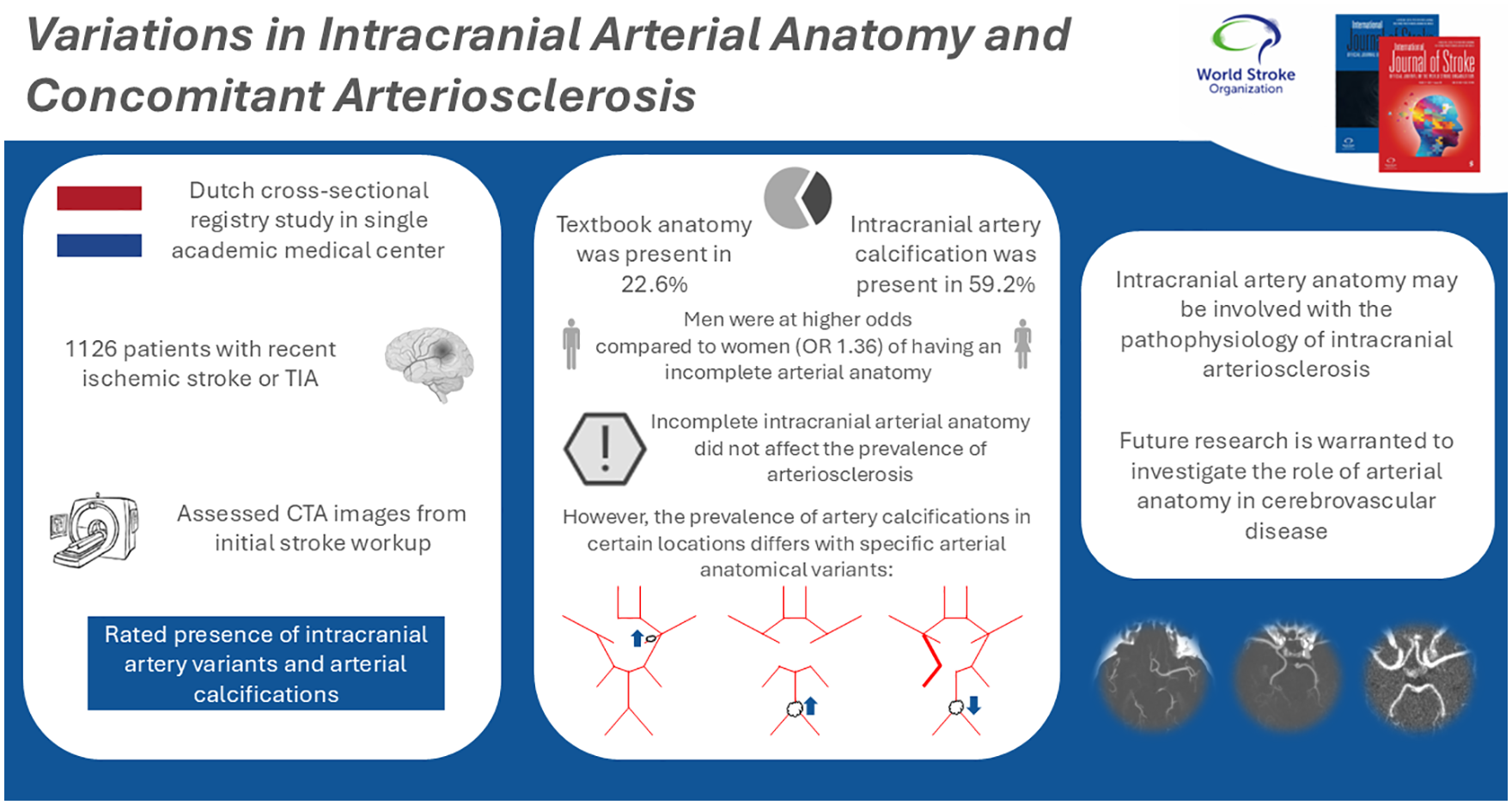
Abstract
Introduction:
An estimated 20–31% of all people are born with a textbook anatomical configuration of the intracranial arteries comprising the Circle of Willis. Individuals with specific anatomical variants may be at elevated risk of intracranial arteriosclerosis, and possibly its sequelae of stroke and dementia, as the distribution of blood flow and pressure is known to be different in variants with missing arteries or arterial segments. Therefore, we studied the association of anatomical variation of intracranial arteries with arteriosclerosis.
Methods:
Between December 2005 and October 2010, 1126 patients (mean age: 62.3 (SD: ±14.0) years, 48.0% female) were recruited, 59.9% of whom had ischemic stroke and 40.1% a transient ischemic attack (TIA). Within the routine diagnostic work-up for stroke, patients underwent cranial computed tomography (CT) angiography. These images enabled a detailed visualization of intracranial arteries, which allowed for the assessment of the anatomical configuration of the cerebral arteries, the anterior and posterior communicating arteries, the internal carotids, and the vertebrobasilar arteries. In addition, these images facilitated the identification of intracranial arterial calcifications, the defining feature of intracranial arteriosclerosis. Binomial logistic regression models adjusting for age, sex, and ethnicity were constructed to assess associations between intracranial artery variations and presence of intracranial arterial calcifications.
Results:
An incomplete Circle of Willis, defined by aplasia of any arterial segment, was present in 875 (77.7%) patients. The most common variation found was aplasia of the right posterior communicating artery, in 52.0% of patients. Men more often presented with an incomplete anatomy as compared to women (adjusted odds ratio: 1.36 (95% CI = 1.02–1.81)). Intracranial artery calcification was present in 59.2% of patients. Incompleteness of the intracranial arteries was not associated with the presence of any intracranial artery calcification (0.95 (0.68–1.34)). However, specific variants were associated with specific locations of intracranial artery calcification: The prevalence of vertebrobasilar artery calcification was lower among those with fetal-type posterior cerebral artery compared to individuals with a normal posterior cerebral artery (0.61 (0.38–0.99)). The prevalence of vertebrobasilar artery calcification was higher among those with a-/hypoplasia of both posterior communicating arteries as compared to those with normal posterior communicating arteries (1.63 (1.00-2.66)). Furthermore, patients with a-/hypoplastic left A1-segments had a higher prevalence of right internal carotid artery calcification as compared to people with a normal left A1-segment (2.30 (1.00-5.26)).
Conclusion:
The prevalence of arteriosclerosis in the intracranial arteries on CT imaging varies among patients with certain anatomical variants of the intracranial arterial system. Specifically, arteriosclerosis in the right internal carotid artery and the vertebrobasilar arteries was more frequently observed in patients who had an a-/hypoplastic left anterior cerebral artery or a-/hypoplasia of both posterior communicating arteries, respectively. In addition, arteriosclerosis was less frequently observed among vertebrobasilar arteries of patients with a fetal-type posterior cerebral artery. Future longitudinal research is warranted regarding the anatomical configuration of intracranial arteries and the development of intracranial arteriosclerosis, as this line of research may reveal a novel group of people at elevated risk of cerebrovascular disease.
Introduction
The anatomy of the intracranial arterial system is often subject to variation, and while the textbook standard taught to medical students depicts a complete circle of Willis, comprising a hexagonal shape formed by the posterior communicating arteries connecting the anterior and posterior cerebral circulations, only an estimated 20–31% of all people are born with this “standard” anatomy.1,2 Historically, such a complete anatomical configuration was assumed to reflect a compensatory mechanism in the event an arterial occlusion arises in one of the cerebropetal arteries, although more recent research suggests a more plausible role for this anatomical structure in facilitating adequate distribution of blood pressure across the intracranial arterial system.2–4 Indeed, specific anatomical variants of the intracranial arteries are reported to influence cerebral blood flow dynamics,3–5 and disturbed blood flow dynamics are known to promote arteriosclerotic processes.6,7 These findings imply a potential predisposition to the development of intracranial arteriosclerosis in people born with specific variants of the intracranial arterial system.
Patients with intracranial arteriosclerosis, characterized by the presence of intracranial artery calcifications (IAC) on cranial imaging exams, are at elevated risk of stroke8,9 and dementia. 10 The mechanisms behind this elevated risk involves blood pressure–mediated 11 changes to intracranial artery stiffness and a resulting loss in arterial distensibility, 12 leading to an increase in end-organ damage to the cerebral integrity and cerebral small vessel disease. 11 Additional modifiable cardiovascular risk factors, such as diabetes mellitus and dyslipidemia, have also been suggested to affect the pathophysiology of intracranial arteriosclerosis.13,14
Contemplating these associations reveals an interesting prospect. Early identification of these anatomical variants, either through routine assessments on cranial imaging performed for other indications or through future screening programs using neonatal cranial ultrasound techniques, 15 would provide clinicians the opportunity to initiate important cardiovascular risk management strategies. Such timely initiation could considerably reduce the risk of severe debilitating consequences associated with intracranial arteriosclerosis, including stroke and dementia, in individuals with precarious anatomical variants.
Whether the anatomical variation of intracranial arteries affects the development of intracranial arteriosclerosis is scarcely studied, yet seems plausible considering the differences in blood flow dynamics between patients with specific arterial variants. Therefore, we assessed whether anatomical variants of the intracranial arteries are associated with the presence of IAC, in a cross-sectional study of ischemic stroke and transient ischemic attack patients with available computed tomography angiography (CTA) imaging.
Methods
Study setting & population
This study was embedded within the Erasmus Stroke Study (ESS), a clinical registry study of patients with neurovascular diseases admitted to the Erasmus Medical Centre, a tertiary hospital in Rotterdam, the Netherlands. 16 All patients presenting between December 2005 and October 2010 to the outpatient clinic, emergency care department, or neurology ward were asked to have their routine clinical investigations, that is, acute diagnostic work-up, serum laboratory measurements, and imaging exams, used for research purposes. Written informed consent for participation in the ESS was obtained from all included patients, and the study was approved by the institutional ethics committee (Erasmus MC MERC, approval number MEC-2005-345). The data underlying this article cannot be shared publicly due to the privacy of participants in this study; however, anonymized data will be shared upon reasonable request to the corresponding author.
We selected all included ischemic stroke and transient ischemic attack (TIA) patients, with available and adequately evaluable multi-detector computed tomography angiography (MDCTA) imaging performed within six months after symptom onset, who presented for the first time to our medical institution. Ischemic stroke was defined as a focal neurological deficit of presumed vascular origin lasting ⩾24 h. TIA was defined as a focal neurological deficit of presumed vascular origin lasting <24 h.
Assessment of anatomical variation of the intracranial arteries
Clinical cranial MDCTA images were used to assess anatomical variation of the intracranial arteries and calcification of the cerebropetal arteries. A detailed description of MDCTA system settings is provided elsewhere. 16 In brief, MDCTA images acquisition consisted of using either a 16-slice, 64-slide, or 128-slice MDCT system (Sensation 16, Sensation 64, Definition, Definition AS+ or Definition Flash, Siemens Medical Solutions, Erlangen, Germany; Brilliance 64, Philips Healthcare Systems, Eindhoven, Netherlands), combined with a standardized and optimized contrast-enhanced protocol. MDCTA scanning ranged between the ascending aorta until 3 cm above the Sella Turcica. All MDCTA scans were assessed by raters who were blinded to clinical data. Patients were excluded from the study when an obstruction of blood flow or scan artifacts obscured the assessment of any intracranial structures of interest.
One author (R.F.S.) used axial MDCTA images to visually assess anatomical variation of the following intracranial arteries: anterior communicating artery, A1-segments of the anterior cerebral arteries, intracranial carotid arteries, M1-segments of the medial cerebral arteries, posterior communicating arteries, P1-segments of the posterior cerebral arteries, basilar arteries and the V4-segments of the vertebral arteries. The following strategy was applied to evaluate anatomical variants. First, when one of the arteries could not be detected, without evidence of intra-arterial or upstream occlusion, said artery was deemed to be aplastic. When an artery had a lumen diameter that was visibly clearly smaller than its contralateral counterpart, or smaller than adjacent arteries when no counterpart existed, said artery was deemed to be hypoplastic. Specific alternative variants were recorded for when a fetal-type posterior communicating artery was identified, when the basilar artery originated from an internal carotid artery, or when a vertebral artery terminated in a posterior inferior cerebellar artery rather than the basilar artery. Finally, other anatomical variants such as fenestrations or complex anterior communicating artery configurations were recorded separately in the “other” category.
We subsequently defined the following configurations of interest. A textbook anatomical configuration of the intracranial arterial system was defined by an absence of any aplastic arteries and an absence of disconnecting variants. An incomplete anterior circulation was defined by aplasia of the A1, anterior communicating, M1, internal carotid or posterior communicating arteries. An incomplete posterior circulation was defined by aplasia of the P1, a or an alternative variant of the vertebral arteries. Figure 1 illustrates some examples of particular variants of interest.
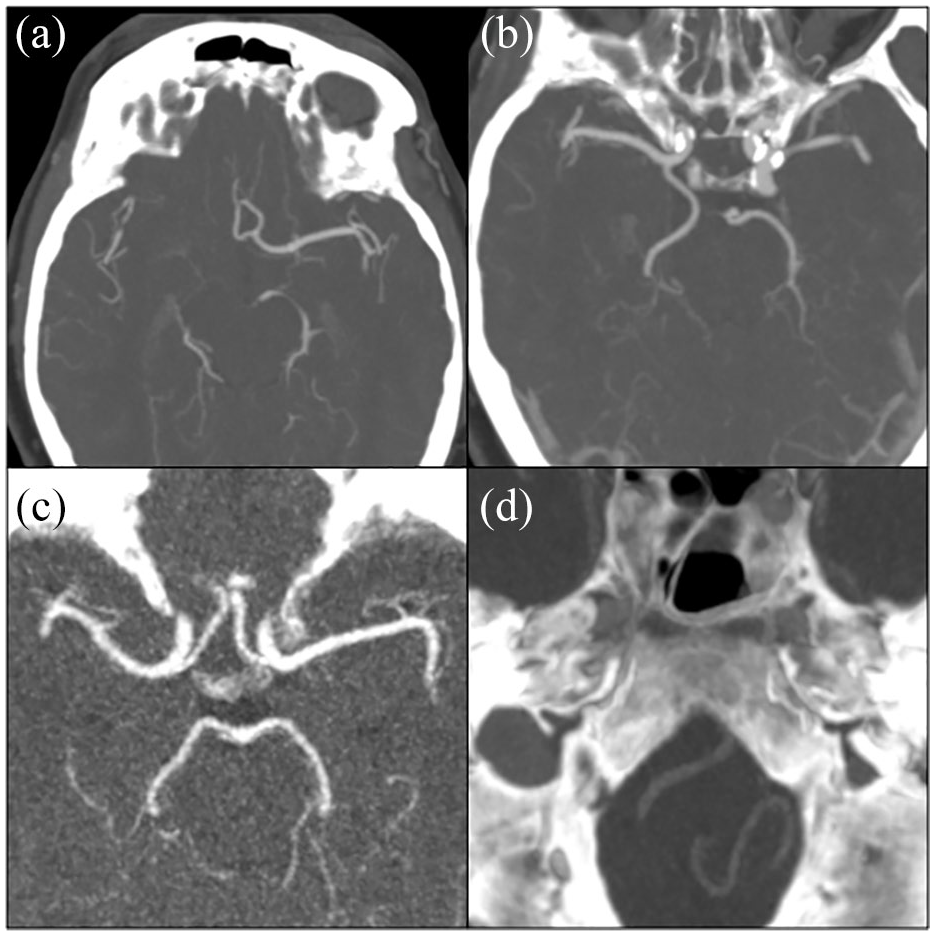
Axial maximum intensity projection examples of specific variants of the intracranial arteries. Panel (a) aplastic right A1 segment, (b) aplastic right P1 segment, (c) aplastic posterior communicating arteries, (d) left vertebral artery terminating in a posterior inferior cerebellar artery.
Any uncertainties arising during the assessment of these MDCTAs were discussed in a consensus meeting with a neurovascular physician-researcher (B.P.B., 6 years of experience in assessing intracranial arterial structures) and an expert in neurovascular imaging (D.B., 13 years of experience in assessing intracranial arterial structures). The interrater agreement for assessing the incompleteness of the intracranial arterial system was determined on a randomly chosen set of 40 patients, using ratings from authors R.F.S. and B.P.B., and had a Cohen’s Kappa of 0.84, 95% confidence interval (CI): 0.63–1.00.
Assessment of intracranial arterial calcification
The presence of IAC among the cerebropetal arteries was assessed through a well-established protocol.16–18 Using a custom-made plug-in for the freely available ImageJ software (version 1.46r), axial MDCTA slices were visually assessed for the presence of any hyperdensities bordering arterial lumens. For internal carotid arteries, this was done starting from the entrance in the petrous canal until the top of the Sella Turcica, while for vertebrobasilar arteries, from their entrance in the foramen magnum until the bifurcation into the P1-segments of the posterior cerebral arteries. Once a hyperdensity was identified, a region of interest was drawn closely surrounding the hyperdensity on each axial slice, and after applying scan-specific voxel dimensions and a predetermined Hounsfield unit (HU) threshold, a millimeter cubed volume of calcification per artery was automatically calculated. For this study, to distinguish arterial calcification from intraluminal contrast agent, we chose a HU threshold of 600 in-line with recent literature at the onset of the study. 19 The presence of arterial calcification was defined by an artery having any volume of calcification above zero millimeter cubed.
Covariate assessment
Demographical, clinical, and serological characteristics were retrieved from the medical records on admission, with data on ethnic background, history of cardiovascular disease, and smoking behavior being collected by self-report. 20 Blood pressure was assessed during two episodes of 15 min of continuous noninvasive measurements. The presence of hypertension was defined by a systolic blood pressure ⩾140 mmHg, or a diastolic blood pressure ⩾90 mmHg, or the use of anti-hypertensive medication. The presence of diabetes mellitus was defined as either a fasting plasma glucose level of >6.9 mmol/L, a 2-h post glucose loading level >11.0 mmol/L, or the use of any diabetes medication. Dyslipidemia was defined by either a serum total cholesterol ⩾6.2 mmol/L, a non-HDL cholesterol level of ⩾5.6 mmol/L, or the use of lipid control medication.
Statistical analyses
Data on patient characteristics, anatomical variants and calcification characteristics are presented through frequencies with percentages or means and standard deviations (SD). Normality of continuous data was assessed through inspection of their histogram distributions. Data were missing to some extent in 33.0% of patients, where the count of missing entries per participant had a 5th and 95th percentile of 1 and 3, respectively. There were <9% missing values among all variables, except for diabetes mellitus which had a missingness of 17.0%. Missing data were addressed using multiple imputation with 33 iterations and imputations, with results presented being based on pooled data.
The following strategy was used to analyze associations with anatomical variants. First, we hypothesized that detecting an incomplete intracranial arterial system could be attributed to one of two scenarios: (1) due to an anatomical variant present from birth, or (2) the structure is absent due to atherosclerosis, such as an upstream occlusion or dissection, which is associated with cardiovascular risk factors. To investigate this hypothesis, we performed a multivariable logistic regression analysis on the presence of an incomplete intracranial arterial system. This regression model adjusted for age at event, sex, White ethnicity, current smoking, hypertension, diabetes mellitus, dyslipidemia, history of cardiovascular disease, and use of any antithrombotic medication. Given our assumption that we were assessing anatomical variants rather than atherosclerotic arteriopathies, we did not expect to find any statistically significant associations with cardiovascular risk factors.
Second, we analyzed the association between having an incomplete intracranial arterial system and the presence of any IAC using a binomial logistic regression analysis while adjusting for age, sex and Caucasian ethnicity as these factors may reflect the genetic differences underlying both intracranial variants and intracranial arteriosclerosis. We then applied this regression model to assess associations between specific anatomical variants and different locations of IAC, based on literature suggesting significant effects from these variants on cerebral blood flow distribution.3,4
All statistical analyses were performed in “R for Windows” v4.2.2 (R Foundation for Statistical Computing, Vienna, Austria), using the packages “mice” (v3.16.0) and “vcd” (v1.4-10).
Results
Selection process and patient characteristics
In a previous ESS study, we assessed IAC burden in 1245 ischemic neurovascular disease patients. 16 From this previous sample, we excluded 119 patients for the following reasons: (1) 8 patients had imaging where the contrast timing was deemed insufficient to assess all intracranial structures of interest, and (2) 111 patients had noticeable arteriopathies such as occlusions and dissections which prevented the accurate assessment of the underlying arterial anatomy. Table 1 displays an overview of characteristics of the remaining 1126 patients that were rated for both anatomical variants and IAC burden. Patients were a mean 62.3 (SD 14.0) years old at study inclusion, with the majority being male (52.0%) and reported having a White ethnic background (79.9%). Two-fifths (40.1%) of patients experienced a TIA, and 27.5% of patients suffered severe stroke symptoms.
Patient characteristics (N = 1126).
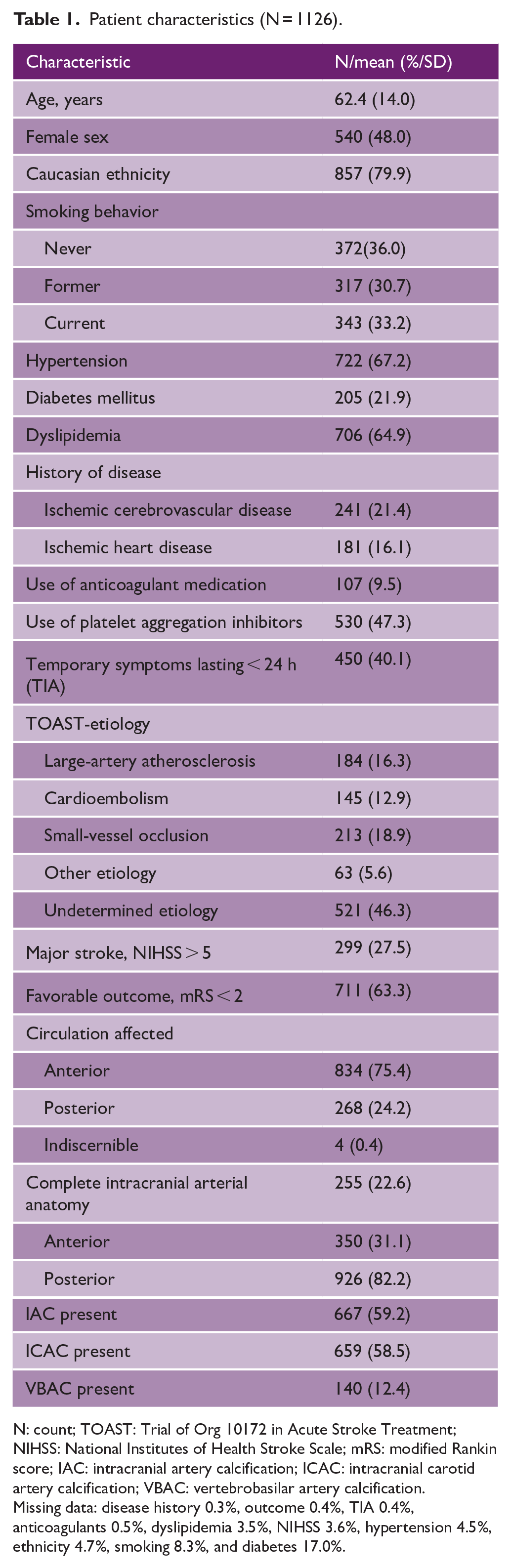
N: count; TOAST: Trial of Org 10172 in Acute Stroke Treatment; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin score; IAC: intracranial artery calcification; ICAC: intracranial carotid artery calcification; VBAC: vertebrobasilar artery calcification.
Missing data: disease history 0.3%, outcome 0.4%, TIA 0.4%, anticoagulants 0.5%, dyslipidemia 3.5%, NIHSS 3.6%, hypertension 4.5%, ethnicity 4.7%, smoking 8.3%, and diabetes 17.0%.
Anatomical incompleteness of the intracranial arteries
A textbook anatomical configuration of the intracranial arterial system was present in 255 (22.6%) patients. Table 2 displays an overview of the recorded anatomical variants of the intracranial arteries among patients. An incomplete anterior circulation was found in 776 (68.9%) of patients, most frequently due to aplasia of one or both posterior communicating arteries (754 patients, 67.0%). An incomplete posterior circulation was found in 200 (17.8%) patients, most frequently due to an aplastic right P1 segment (95 patients, 8.4%). Anatomical variants of more than one intracranial artery were seen in 869 (77.2%) patients and Figure 2 graphically displays the frequencies of combinations.
Overview of anatomical variants of the intracranial arteries.
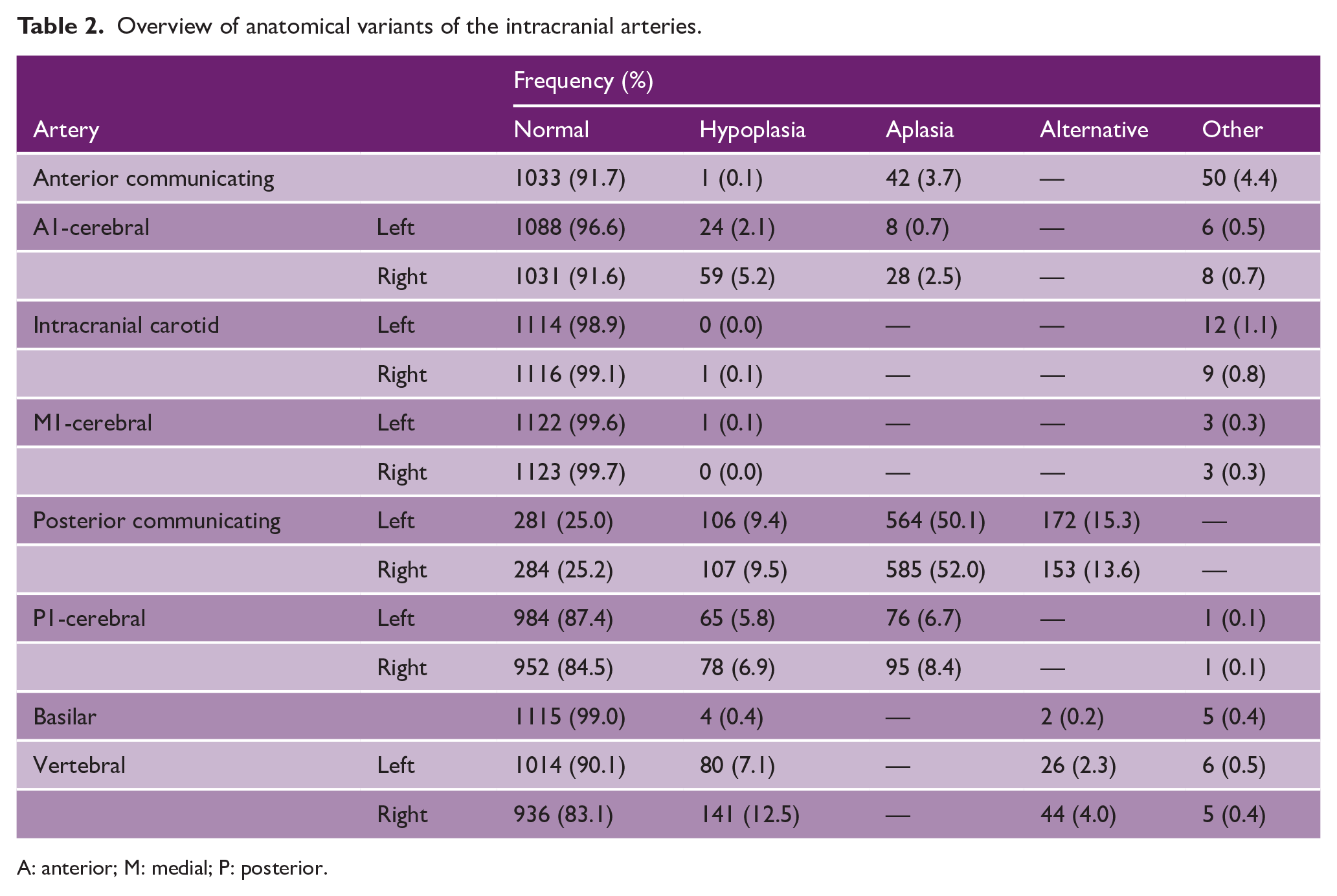
A: anterior; M: medial; P: posterior.
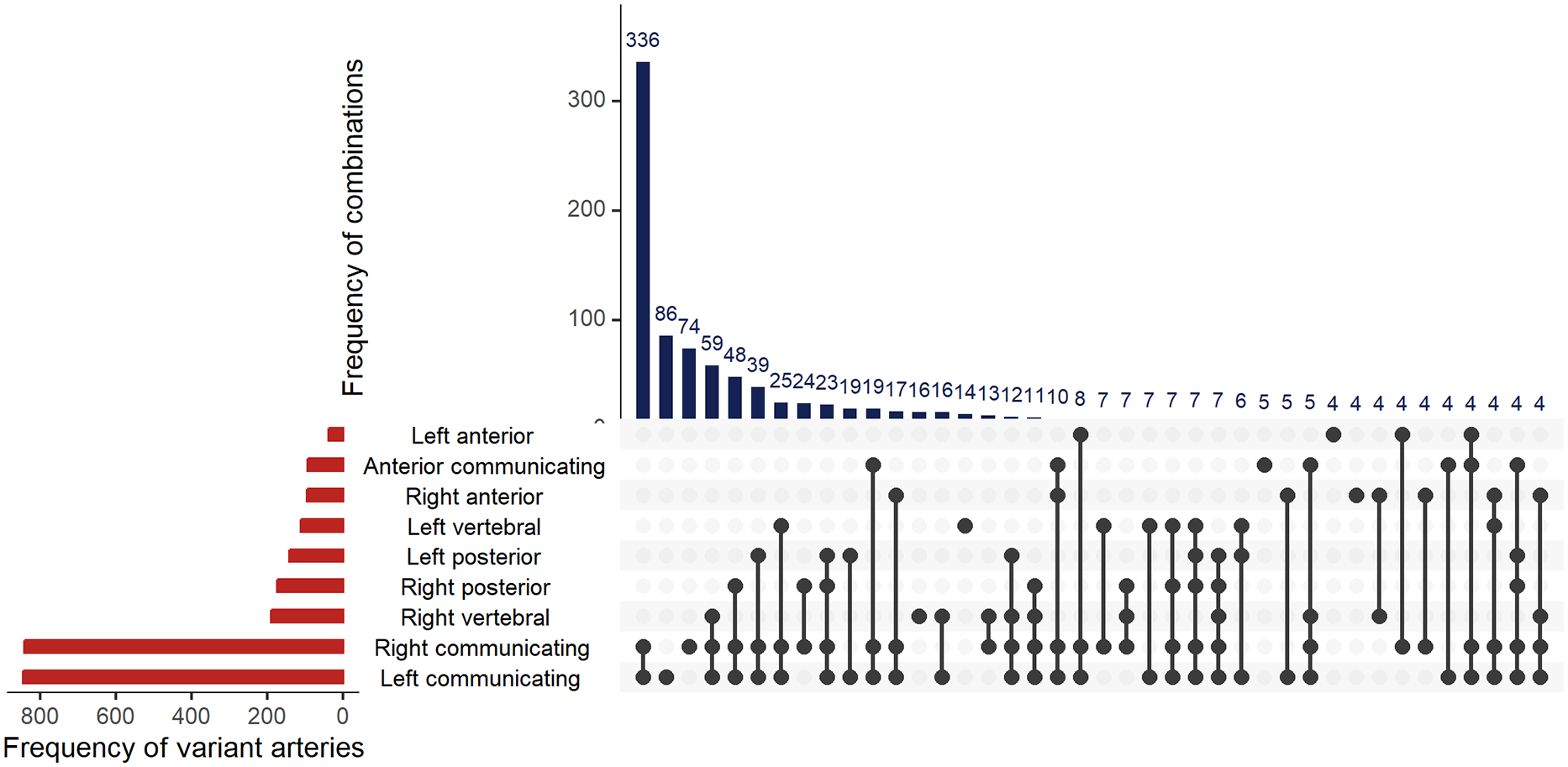
Combined histogram plot depicting frequency of combinations of variant arteries. Basilar, carotid and medial cerebral arteries left out due to low case numbers of variants.
Table 3 displays the results of binomial logistic regression analyses on incompleteness status of the intracranial arteries. After full adjustment, men more often presented with an incomplete intracranial arterial system compared to women (469 (80.0%) vs. 406 (75.2%), adjusted odds ratio (aOR): 1.36, 95% CI: 1.02–1.81), but no statistically significant association was found between any known cardiovascular risk factor and having an incomplete intracranial arterial system.
Results of regression analyses on having an incomplete intracranial arterial circulation.
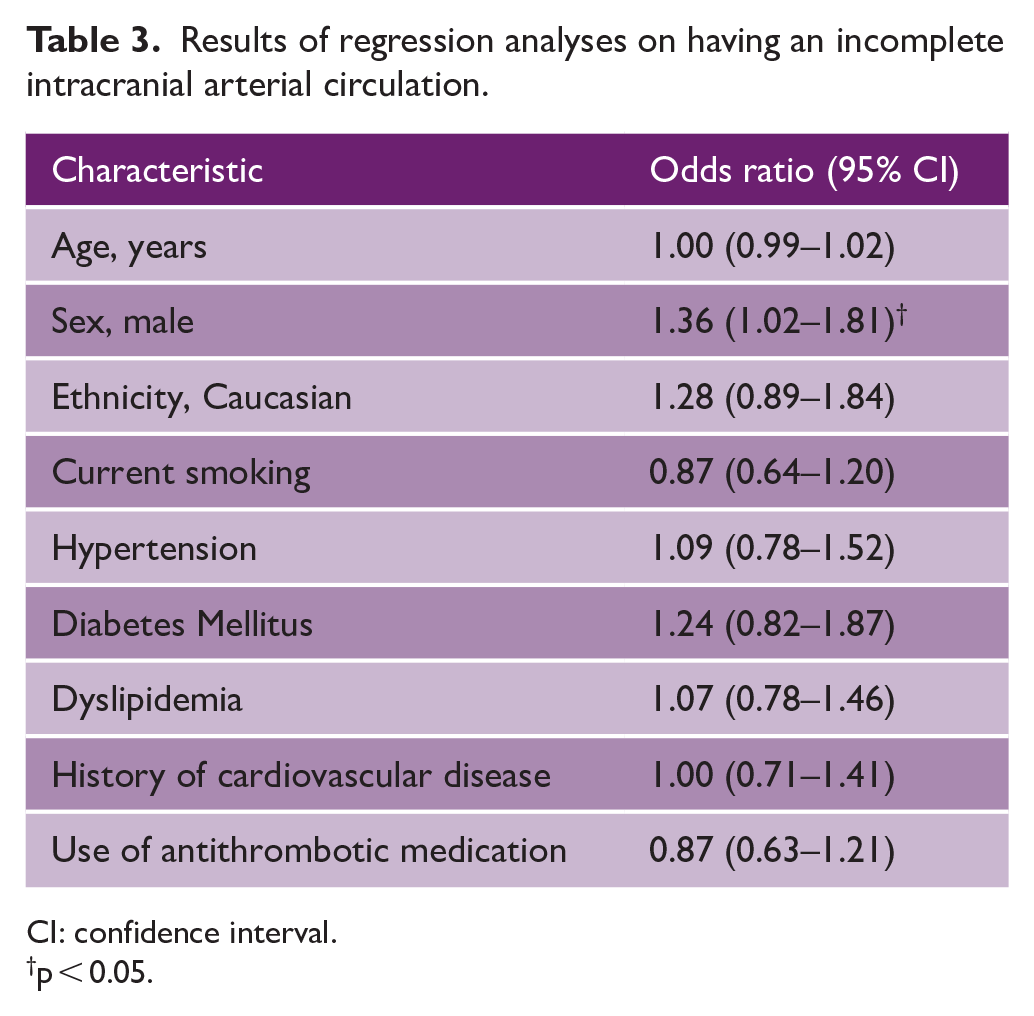
CI: confidence interval.
p < 0.05.
Association between incomplete intracranial arterial anatomy and calcification
IAC was present in 667 (59.2%) of patients, most often in the intracranial carotid arteries (ICAC) (58.5%). Table 4 displays the results of binomial logistic regression analyses between anatomical variation of the intracranial arterial system and IAC. Overall, patients with an incomplete intracranial arterial system had a prevalence of IAC similar to patients with a complete system (aOR: 0.95 (0.68–1.34)). However, specific anatomical variants of the intracranial arterial system were associated with the presence of IAC in particular cerebropetal arteries. Notably, a-/hypoplasia of the left A1 segment was associated with a higher prevalence of arterial calcification in the right internal carotid artery (aOR: 2.30, 95% CI: 1.00–5.26). An a-/hypoplastic right A1 segment was also associated with a higher prevalence of left internal carotid artery calcification, albeit statistically insignificant (aOR: 1.16, 95% CI: 0.71–1.90). Similarly, vertebrobasilar artery calcification (VBAC) was less often seen in those with a fetal-type posterior cerebral artery (PCA) (aOR: 0.61, 95% CI: 0.38–0.99). Finally, a-/hypoplasia of both posterior communicating arteries was associated with a higher prevalence of VBAC (aOR: 1.63, 95% CI: 1.00–2.66).
Results of adjusted regression analyses between intracranial arterial anatomy variants and intracranial arteriosclerosis status.
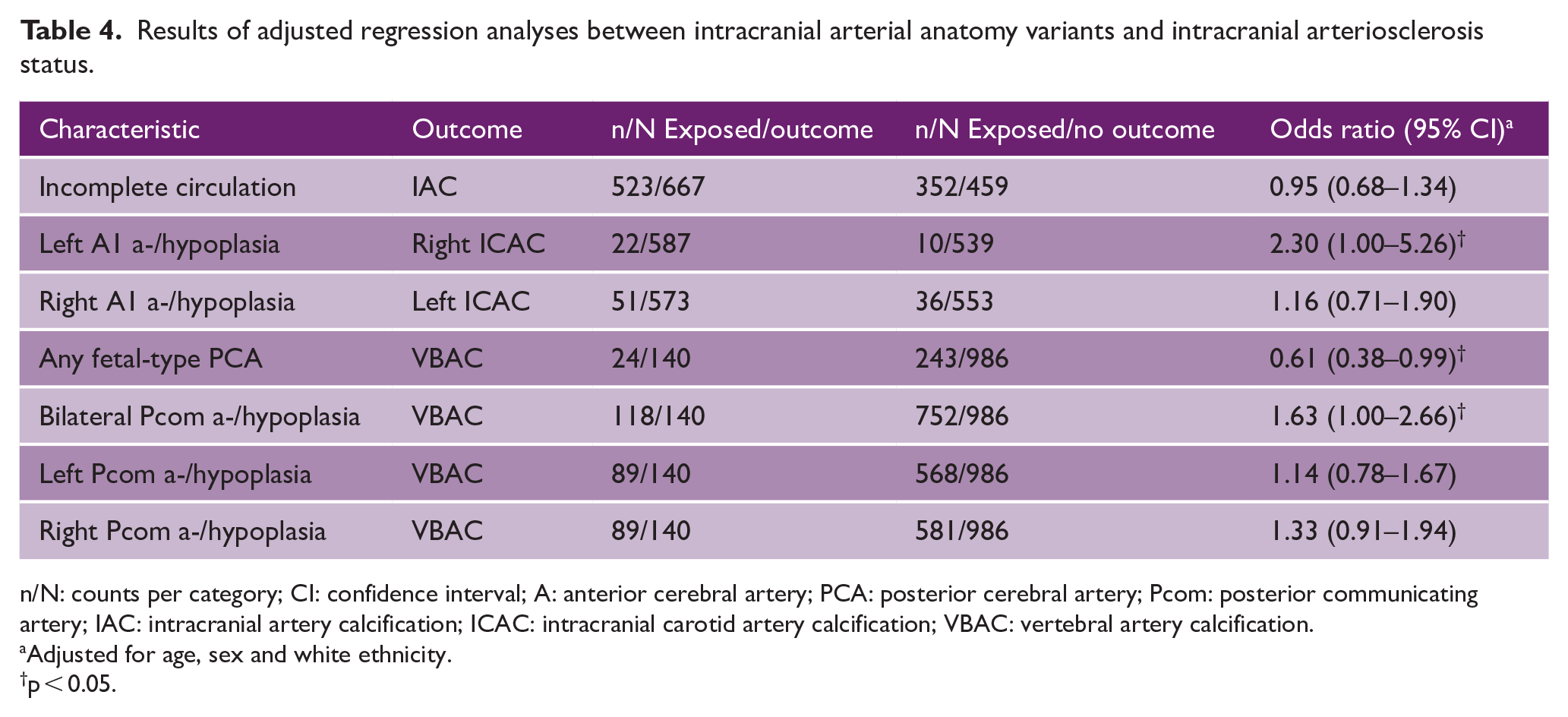
n/N: counts per category; CI: confidence interval; A: anterior cerebral artery; PCA: posterior cerebral artery; Pcom: posterior communicating artery; IAC: intracranial artery calcification; ICAC: intracranial carotid artery calcification; VBAC: vertebral artery calcification.
Adjusted for age, sex and white ethnicity.
p < 0.05.
Discussion
In this cross-sectional registry study, a textbook anatomical configuration of the intracranial arterial system was present in one-fifth of patients with recent ischemic stroke or TIA, and patients with this complete configuration did not show a differential prevalence of any intracranial arteriosclerosis when compared to patients with an incomplete configuration. However, specific anatomical variants of the intracranial arterial system were associated with IAC presence, particularly in the left internal carotid and the vertebrobasilar arteries.
Our findings are supported by previous literature on differences in cerebral blood flow dynamics among people with these same specific anatomical variants.3,4 A potential mechanism is that arteries with a high degree of curvature, such as the cerebropetal arteries, are predisposed to disturbed flow patterns, which become more pronounced with increased blood flow. 21 This increased blood flow will lead to higher levels of turbulent flow, which creates a local atherogenic environment through the following mechanisms: 7 (1) increased endothelial dysfunction due to irregular and oscillatory wall shear stress; (2) increased endothelial production of reactive oxygen species, leading to more oxidization of low-density lipoprotein cholesterol; and (3) increased luminal expression of adhesion molecules for macrophages, with a subsequent increased rate of foam cell development. And indeed, we more often found IAC of the right internal carotid artery among individuals with an a-/hypoplastic left A1 segment, which aligns with prior studies reporting increased cerebral blood flow in internal carotid arteries contralateral to the side with an a-/hypoplastic A1 segment.3,4 For those patients with an a-/hypoplastic right A1 segment, an increased OR for having IAC in the left internal carotid artery was also found, although this effect was not at all statistically significant.
Literature on the association between anatomical variance of the anterior cerebral arteries and ICAC is almost non-existent, with only one prior study reporting no association between any incompleteness of the circle of Willis and carotid siphon calcifications. 22 An important difference between this prior study and present study is that they investigated overall completeness of the anterior cerebral circulation, whereas we specifically investigated distinct anatomical variations per artery of the anterior cerebral circulation. Our study thus provides a more nuanced understanding of the hemodynamics underlying the association between anatomical variation and ICAC.
Our findings with regard to VBAC are also supported by literature reporting differences in cerebral blood flow among individuals with variants of the posterior cerebral circulation. Notably, prior studies report a diminished basilar artery blood flow in individuals with a fetal-type posterior communicating artery3,4—a finding which aligns with our study, where we find fewer VBAC in individuals with this anatomical variant. In addition, we found a higher prevalence of VBAC among individuals with a-/hypoplastic posterior communicating arteries, which also aligns with the literature reporting increased basilar artery blood flow in individuals with this variant anatomy.3,4
These results suggest that anatomical differences of the intracranial arterial circulation could play a role in the pathophysiology of intracranial arteriosclerosis, and perhaps even in its suggested sequelae of stroke 8 and dementia. 10 This hypothesis has also been alluded to previously in expert reviews.5,23 We thus believe that further longitudinal research is warranted into the role of intracranial artery anatomy and the development of intracranial arteriosclerosis, as this may reveal a novel risk factor for debilitating neurovascular diseases, which may be investigated early in life on non-invasive neuroimaging exams such as time-of-flight magnetic resonance angiography. 5
Some important limitations of this study need to be mentioned. First, rating of intracranial arterial variation and calcification occurred while blinded to clinical data. However, inherent to the cross-sectional design of this study, it may still be possible that arterial pathologies such as occlusions or dissections were rated instead of anatomical variation. We tried to assess this potential measurement error to the fullest extent possible within this study by performing a multivariable logistic regression analysis on incompleteness status, incorporating many cardiovascular risk factors in the adjustment model. Considering no significant associations were found in this control analysis between cardiovascular risk factors and an incomplete intracranial arterial system, we believe it is less likely that arteriopathies were mistakenly rated as anatomical variants. Moreover, we only found a higher prevalence of an incomplete intracranial arterial system among male patients compared to female patients, a finding which is in agreement with previous literature. 24 However, we acknowledge that establishing a temporal association is not feasible with a cross-sectional study design. Therefore, we recommend future longitudinal studies to confirm our findings and more accurately define the temporal relationship between intracranial arterial variants and arteriosclerosis. Furthermore, any possible arterial pathologies were discussed in consensus meetings with experienced raters of intracranial arteries. Despite these best efforts, given the large confidence intervals of the effects described here indicates the precision of our methods remains low, and some caution in their interpretation is warranted. Second, due to limitations in the resolution of early CT systems used in the study, and limitations in scanning windows of contrast-enhancement protocols at the time, we were unable to systematically gather data on the presence of collateral arteries with the main intracranial arteries of interest. These anastomoses may also be involved in a proper distribution of blood flow across the cerebrum, and could therefore provide important information in the association between anatomical variation and calcification of the intracranial arteries. A final noteworthy limitation is the cross-sectional nature of this study without long-term follow-up information, which hinders assessing any causal effects. As this study has likely recruited healthier patients with a smaller IAC burden, based on the relatively low percentage of major stroke in this study as compared to other cohorts of stroke patients, some survivor bias may have led to an underestimation of effects. Following this, as this is a sample of stroke patients, the true prevalence of anatomical variants of the intracranial arterial system is possibly lower in the healthy population.
To conclude, anatomical variation of the intracranial arterial system may be involved in the pathophysiology of intracranial arteriosclerosis and subsequent cerebrovascular diseases. Further research is warranted to assess whether people with specific intracranial artery variants may benefit from cardiovascular risk management strategies to prevent the development of intracranial arteriosclerosis, and to limit the risk of subsequent cerebrovascular diseases.
Footnotes
Acknowledgements
We thank Imke van Eert and Meike Brinkhuijsen for their aid in the feasibility study of attaining arterial anatomy data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B.P.B., D.B., and M.K.I. were supported by the Erasmus Medical Center MRACE grant (grant number 386070).
