Abstract
Background:
Intracerebral hemorrhage (ICH) is a manifestations of sporadic cerebral small vessel disease, and the survivors are predisposed to higher long-term risks of vascular events. Intracranial carotid artery calcification (ICAC), a potential marker for arteriosclerosis, is a risk factor for stroke but their roles in ICH is unknown. We aimed to investigate the prevalence and morphological subtypes of ICAC and their associations with long-term vascular events in ICH survivors.
Materials and methods:
Survivors of spontaneous ICH treated at a single center in Taiwan were included. ICAC was assessed by non-contrast CT; morphology was evaluated and categorized as intimal, internal elastic lamina (IEL), or mixed subtype. Patients were followed up for two years. Associations between calcification subtypes and follow-up events (stroke, cardiovascular event, death) were explored using multivariable Cox regression models.
Results:
Overall, 462 (80.1%) survivors of ICH had ICAC—223 (38.6%) were categorized as IEL calcification, 216 (37.4%) as intimal calcification, and 23 (4.0%) as mixed type calcification—and 115 patients (19.9%) had no calcification. Patients with IEL calcification were older than patients with intimal or no calcification (p < 0.001). Age (p < 0.001), diabetes (p = 0.010), and reduced renal function (p = 0.001) were independently associated with IEL calcification. During 2-years of follow-up, IEL calcification was not associated with a significant difference in the risk of recurrent ICH (HR=2.8 [0.8‒9.7]), but was associated with higher risks of incident ischemic stroke (HR = 9.0 [1.0‒77.4]), vascular mortality (HR = 13.6 [1.7‒1772.4], and all-cause mortality (HR = 13.9 [1.8‒105.8]).
Conclusions:
ICAC is common among ICH survivors and the subtype of IEL calcification may potentially have prognostic value for long-term vascular events.
Introduction
Spontaneous intracerebral hemorrhage (ICH) is a severe stroke caused by the rupture of small arteries or arterioles, typically due to cerebral small vessel disease (SVD), particularly hypertensive SVD (deep perforator arteriopathy) or cerebral amyloid angiopathy (CAA). 1 Intracranial arteriosclerosis is implicated in up to 75% of all strokes, and is increasingly recognized as a key risk factor within the complex landscape of SVD pathogenesis and mediates the association between hypertension and SVD.2–4 Intimal atherosclerosis, which is characterized by plaque formation due to the buildup of lipids and calcium in the intimal layer of an artery, is a frequent subtype of intracranial arteriosclerosis. Circular calcification of the internal elastic lamina (IEL) is another commonly observed characteristic manifestation of intracranial arteriosclerosis. 5 Clinically, these two distinct morphological subtypes of intracranial arteriosclerosis may have differing impacts on the risk of stroke due to their potentially varied effects on arterial wall structure and the associated hemodynamics.
A previous population-based study found that intracranial carotid artery calcification (ICAC), particularly the IEL calcification subtype, was associated with an increased risk of stroke. 3 However, there is a notable lack of research on the impact of ICAC in spontaneous ICH. In addition, the specific associations between these subtypes of ICAC and the future risk of cardiovascular and cerebrovascular events in survivors of ICH remain unclear. Survivors of spontaneous ICH are predisposed to the development of major adverse cardiovascular events;6,7 therefore, the identification of clinico-radiological risk factors could be clinically relevant and may inform long-term patient care and risk assessment for ICH survivors.
We conducted this study to evaluate the prevalence of different subtypes of ICAC, as proxies for intracranial arteriosclerosis, and assessed their association with the risk of stroke and vascular events in a cohort of consecutive survivors of spontaneous ICH treated a tertiary referral center. Our goal was to identify the clinical and neuroimaging risk factors associated with ICAC and determine whether specific subtypes of ICAC are related to a higher risk of future vascular events or mortality. Overall, we aimed to gain better insight into the pathophysiological role of ICAC in spontaneous ICH in order to potentially enhance stroke prevention strategies tailored to the ICH population.
Methods
Data availability
All data for this study are stored at the National Taiwan University Hospital (NTUH) and can be accessed upon request from qualified investigators.
Standard protocol approvals, registrations, and patient consents
The primary data used in this study were obtained from the NTUH Stroke Registry, which is a database that prospectively includes all patients with stroke from a tertiary referral center in Taiwan.8,9 The Research Ethnics Committee of the NTUH approved the registry. Informed consent was waived.
Patient enrollment
We enrolled consecutive patients with symptomatic ICH treated at NTUH between September 2014 and March 2022 (n = 1923) from a prospectively maintained stroke registry.8,9 Patients with potential causes of secondary hemorrhage—such as trauma, structural and vascular lesions, brain tumors, severe coagulopathy due to systemic disease or medication, or ischemic stroke with hemorrhagic transformation—were excluded (n = 652). Additionally, patients without comprehensive MRI studies (n = 684) or CT studies (n = 10) were excluded (Supplemental Figure).
The patient’s clinical data were collected systematically through standardized medical record reviews. Recorded clinical variables included age, sex, smoking history, chronic hypertension, classes of antihypertensive medication taken, diabetes mellitus, hypercholesterolemia, atrial fibrillation, and creatinine clearance (eGFR). Patients who were above 50-years-old were considered to have CAA if they had strictly lobar ICH, cerebral microbleeds (CMBs), and/or cSS, as per the Boston criteria. 10 We also defined deep perforator arteriopathy-related ICH if the patients presented with strictly deep ICH and/or CMBs, and mixed ICH if the patients presented with mixed lobar ICH and/or CMBs.8,11
CT imaging
Multiple CT scanners with detectors ranging from 64- to 320-slice were used for brain CT imaging. The scanners included LightSpeed VCT by GE Healthcare (Milwaukee, WI, USA), Brilliance 64 and Brilliance iCT 256 by Philips Healthcare (Best, The Netherlands), SOMATOM Sensation 64 and SOMATOM Definition AS by Siemens Healthineers (Erlangen, Germany), and Aquilion ONE by Toshiba Medical Systems (Tokyo, Japan). All scans were conducted at 120 kV and 300‒375 mAs. The imaging protocol involved scanning patients from the skull base to the vertex with a slice thickness ranging from 0.625 to 1 mm.
Classification of ICAC subtypes
We evaluated the presence and characteristics of ICAC bilaterally, extending from the horizontal part of the petrous segment to the convergence with other arteries forming the circle of Willis. To distinguish between different subtypes of calcification, we utilized a previously validated method that was cross-referenced with histological data. 12 This approach involved assigning a composite score based on attributes such as the circularity, thickness, and continuity of ICAC. Based on the scores, we subtyped both the left and right carotid arteries into three main groups: those primarily exhibiting atherosclerotic intimal ICAC (<7 points, indicating thick, small, and irregular calcifications), those predominantly showing IEL ICAC (⩾7 points, indicating elongated, circular, and thin calcifications), and those without any ICAC.
Subsequently, we categorized the participants into groups with different morphological subtypes of ICAC based on the observed combinations for the left and right arteries. Participants with bilateral predominantly intimal ICAC, intimal ICAC on one side, or with an absence of contralateral ICAC were classified into the intimal subtype group. Similarly, participants with bilateral predominant IEL ICAC or IEL ICAC on one side, in conjunction with the absence of contralateral ICAC, were categorized into the IEL subtype group. Individuals showing predominant intimal or IEL ICAC on one side and a different subtype on the contralateral side were classified as having mixed ICAC, and were excluded from the risk analysis. Participants without any ICAC were classified into the ICAC absent group. The classification of calcification subtypes was performed by a board-certified neuroradiologist with 11 years of experience (B.C.L.), who was blinded to the subjects’ clinical characteristics. Representative images of the IEL and intimal ICAC subtypes are shown in Figure 1.
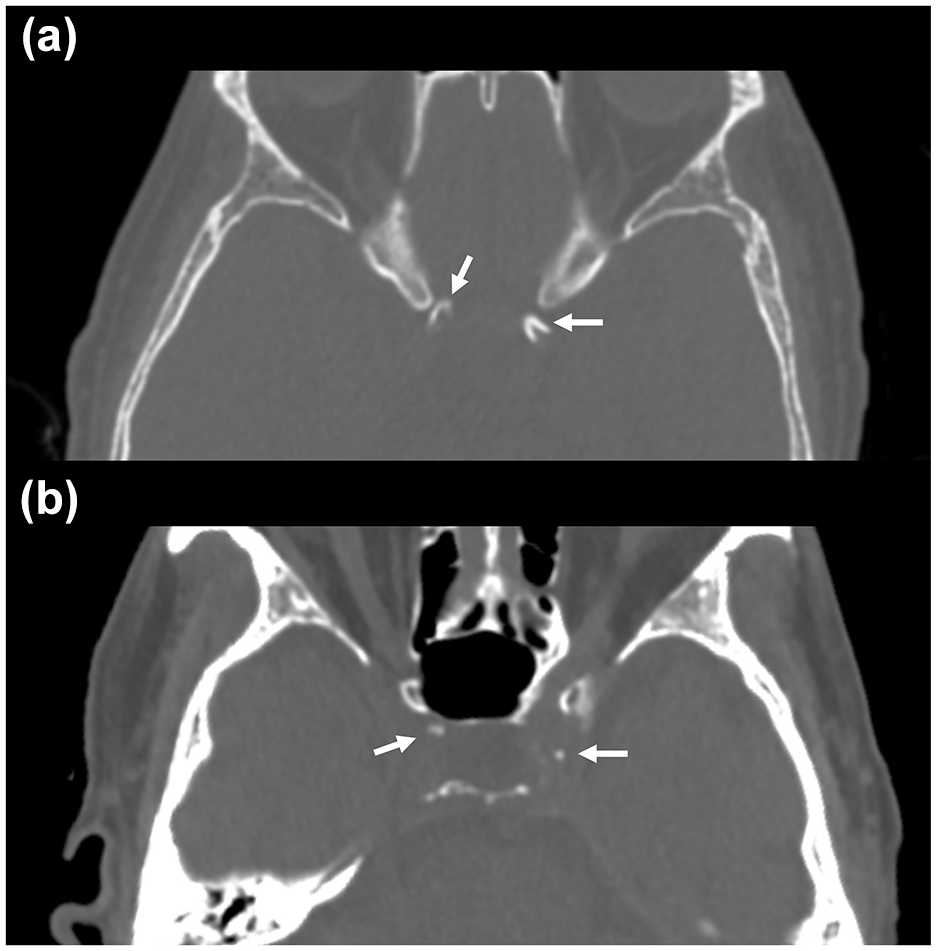
Representative axial CT images of the internal elastic lamina (IEL) (A) and intimal (B) subtypes of intracranial carotid artery calcification (ICAC).
MRI acquisition and analysis
MRIs were obtained using 1.5T or 3T MRI scanners (MAGNETOM Aera, MAGNETOM Verio, TIM or Biograph mMR, Siemens Medical Solutions, Malvern, PA, USA) using protocols that include T1-weighted, T2-weighted, diffusion-weighted, FLAIR, TOF angiography, and SWI. MRI SVD markers were assessed based on the STRIVE 2.0 criteria. 13 Briefly, CMBs and cortical superficial siderosis (cSS) were assessed using SWI, as we previously proposed.8,11 The location of CMBs was determined using the Microbleed Anatomical Rating Scale. 14 Lacunes were defined as round or ovoid subcortical fluid-filled cavities that are 3‒15 mm in diameter.15,16 White matter hyperintensity (WMH) volume was calculated from FLAIR imaging using a semi-automated measure for the ICH-free hemisphere and multiplied by two.17,18 MRI-visible enlarged perivascular spaces (PVS) were evaluated on T2-weighted imaging and defined as sharply delineated structures <3 mm. The number of enlarged PVS was counted in the centrum semiovale (CSO) and basal ganglion (BG), with severity rated on a 4-point scale (0 = none, 1 ⩽ 10, 2 = 11‒20, 3 = 21‒40, and 4 ⩾ 40, more severe side).19,20 A dichotomized classification of high (scale, 3 and 4) or low (scale, 0 to 2) severity was used. 19
Follow-up
We collected 2-year follow-up data through systematic reviews of multiple data sources, including telephone calls to the patients, medical records from outpatient clinics, and NTUH’s imaging databases, as previously described. 8 Index dates for outcomes were defined as the date of ICH, with patients followed-up until death, the last clinic visit for ICH, or last telephone call. Patients unreachable by phone were censored at their last clinic visit. Events including recurrent symptomatic ICH, incident symptomatic ischemic stroke, major cardiovascular events (admission for myocardial infarction, unstable angina or heart failure), and the occurrence and cause of death were carefully identified.
Statistical analysis
The demographic data collected for the patients who survived ICH included age, sex, hypertension, renal function (estimated glomerular filtration rate, eGFR), diabetes mellitus, hyperlipidemia, atrial fibrillation, 3-month modified Rankin Scale (mRS), and multiple neuroimaging markers. Continuous variables are reported as median with interquartile range (IQR) and categorical data, as frequencies and percentages. ICH patients were categorized into three groups based on the subtype of ICAC: ICAC absent, intimal ICAC, and IEL ICAC. The demographic and laboratory data of these groups were compared using Kruskal-Wallis test for non-normally distributed variables, followed by Dunn’s test for pairwise comparison. Proportional differences were estimated using chi-square tests with Bonferroni correction. The associations between the dominant ICAC subtypes (intimal or IEL) and the risk factors were analyzed via logistic regression (crude and adjusted). The results are presented as odds ratio (OR) with 95% confidence intervals (CI) adjusted for listed risk factors.
Kaplan Meier Curves were plotted to compare the survival curves between different ICAC subtypes for each follow-up outcomes. Cox proportional hazard models were used to estimate the hazard ratios (HR). If zero events were recorded in the comparator group, Firth's penalized partial likelihood correction was applied. We assessed the proportional hazard assumption by adding a time-dependent covariate for ICAC subtypes, and all time-dependent covariates were nonsignificant in our testing model. Analyses were conducted using SPSS 25 (IBM, Chicago, IL) and Firth’s penalized partial likelihood correction was performed using R version 4.3.1 with the “coxphf” package (version 1.13.4). Significance was set at p < 0.05 (two-tailed). There were no missing data of demographics in this study.
Results
Comparisons of characteristics between patients with and without MRI scan were shown in Supplemental Table 1. Patients of ICH with available MRI had lower National Institute of Health Stroke Scale (9 [4–15] vs. 18 [9–28], P < 0.001), higher Glasgow Coma Scale (15 [13–15] vs. 11 [8–15], P < 0.001) and lower mRS on discharge (3 [2–4] vs. 4 [3–5], P < 0.001) compared to patients without available MRI scan.
Among the 577 survivors of ICH with available MRI included in this study, 462 (80.1%) patients had ICAC—223 patients (38.6%) were classified into the IEL ICAC group, 216 patients (37.4%) were classified into the intimal ICAC group, 23 patients (4.0%) were classified into the mixed type calcification group—and 115 patients (19.9%) were classified into the ICAC absent group. Figure 2 showed the age-stratified distribution of ICAC subtypes. The clinical features of the patients without ICAC and the intimal and IEL ICAC groups are compared in Table 1. Patients with IEL ICAC were older and had significantly different demographics and neuroimaging variables compared to the other two groups.
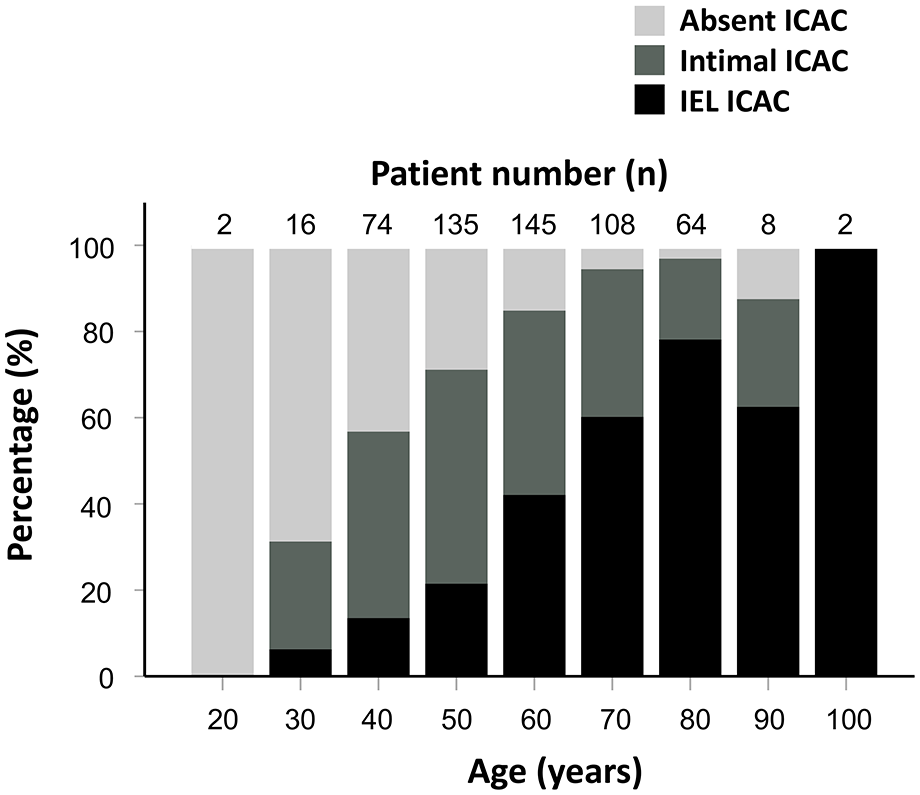
Age-stratified distributions of intracranial carotid artery calcification (ICAC) subtypes. Bar chart showing the distributions of internal elastic lamina (IEL), intimal and absent ICACs in spontaneous intracranial hemorrhage (ICH). Patients with mixed type ICAC were excluded.
Comparison of demographics in ICH patients with intimal or medial calcification in ICA.
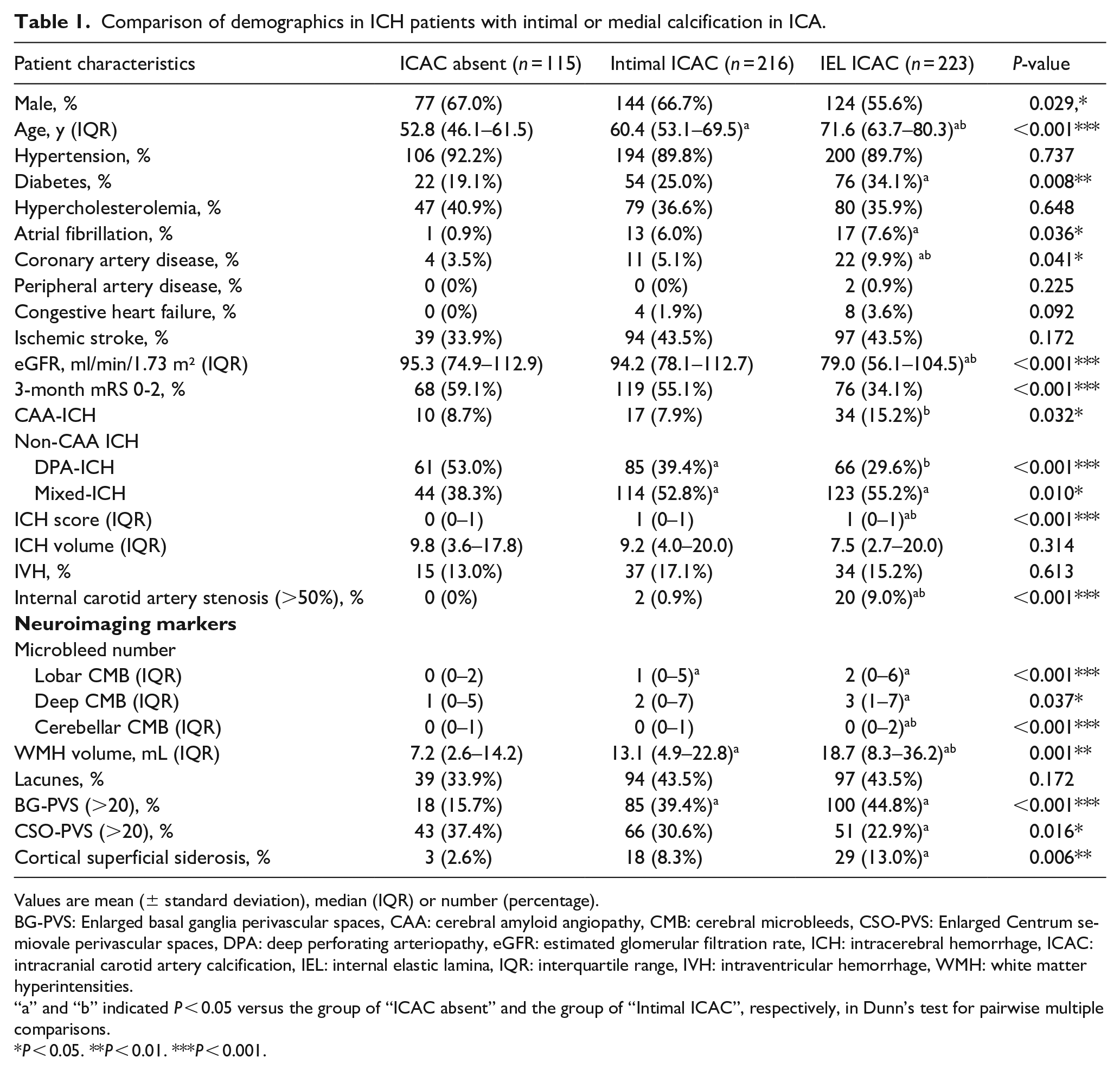
Values are mean (± standard deviation), median (IQR) or number (percentage).
BG-PVS: Enlarged basal ganglia perivascular spaces, CAA: cerebral amyloid angiopathy, CMB: cerebral microbleeds, CSO-PVS: Enlarged Centrum semiovale perivascular spaces, DPA: deep perforating arteriopathy, eGFR: estimated glomerular filtration rate, ICH: intracerebral hemorrhage, ICAC: intracranial carotid artery calcification, IEL: internal elastic lamina, IQR: interquartile range, IVH: intraventricular hemorrhage, WMH: white matter hyperintensities.
“a” and “b” indicated P < 0.05 versus the group of “ICAC absent” and the group of “Intimal ICAC”, respectively, in Dunn’s test for pairwise multiple comparisons.
P < 0.05. **P < 0.01. ***P < 0.001.
Compared to patients without ICAC, the patients with IEL ICAC were older (71.6 [63.7–80.3] vs. 52.8 [46.1–61.5], P < 0.001), more frequently had diabetes (34.1 vs. 19.1%, P = 0.008), atrial fibrillation (7.6 vs. 0.9%, P = 0.036) and coronary artery disease (9.9% vs. 3.5%, P = 0.041), had lower eGFR (79.0 [56.1–104.5] vs. 95.3 [74.9–112.9] mL/min/1.73 m², P < 0.001), more frequently had CAA (14.3 vs. 7.0%, P = 0.025) and significant (>50%) internal carotid artery stenosis (9.0% vs. 0%, P < 0.001). In terms of SVD neuroimaging markers, patients with IEL ICAC had a higher number of lobar CMBs (2 [0–6] vs. 0 [0–2], P < 0.001), deep CMBs (3 [1–7] vs. 1 [0–5], P = 0.037), cerebellar CMBs (0 [0–2] vs. 0 [0–1], P < 0.001), more frequently had cSS (13.0 vs. 2.6%, P = 0.006), had larger WMH volume (18.7 [8.3–36.2] vs. 7.2 [2.6–14.2] mL, P = 0.001), had more severe BG-PVS (44.8 vs. 5.7%, P < 0.001), and had less severe CSO-PVS (22.9 vs. 37.4%, P = 0.016) than patients who did not have ICAC (Table 1).
Compared to patients with intimal ICAC, patients with IEL ICAC were older (71.6 [63.7–80.3] vs. 60.4 [53.1–69.5], P < 0.001), had lower eGFR (79.0 [56.1-104.5] vs. 94.2 [78.1–112.7] mL/min/1.73 m², P < 0.001), more coronary artery disease (9.9% vs. 5.1%, p = 0.041), more frequently had CAA (14.3 vs. 7.4%, P = 0.025) and more frequently had significant internal carotid artery stenosis (9.0% vs. 0.9%, P < 0.001). In terms of SVD neuroimaging markers, patients with IEL ICAC had a higher number of cerebellar CMBs (0 [0–2] vs. 0 [0–1], P < 0.001) and had larger WMH volume (18.7 [8.3–36.2] vs. 13.1 [4.9–22.8] mL, P = 0.001) than patients with intimal ICAC.
Risk factors for morphological subtypes of ICAC
We next investigated the clinical and radiological risk factors associated with IEL ICAC in survivors of ICH compared to the patients without ICAC as the reference group using multivariable regression analyses (Table 2). The presence of IEL ICAC was independently associated with older age (per 10 years, OR [95% CI]: 2.843 [2.104–3.843], P < 0.001), diabetes (2.583 [1.255–5.314], P = 0.010), and lower eGFR (per 10 mL/min/1.73 m² eGFR, 0.869 [0.798–0.946], P = 0.001). IEL ICAC was also independently associated with more severe BG-PVS (3.255 [1.575–6.725], P = 0.001) and a trend of higher WMH volume (per mL, 1.015 [0.998–1.033], P = 0.091; Table 2).
Logistic regression model investigating the relationship between clinical characteristics and ICAC subtypes in patients with ICH, using patient without ICAC as reference group.
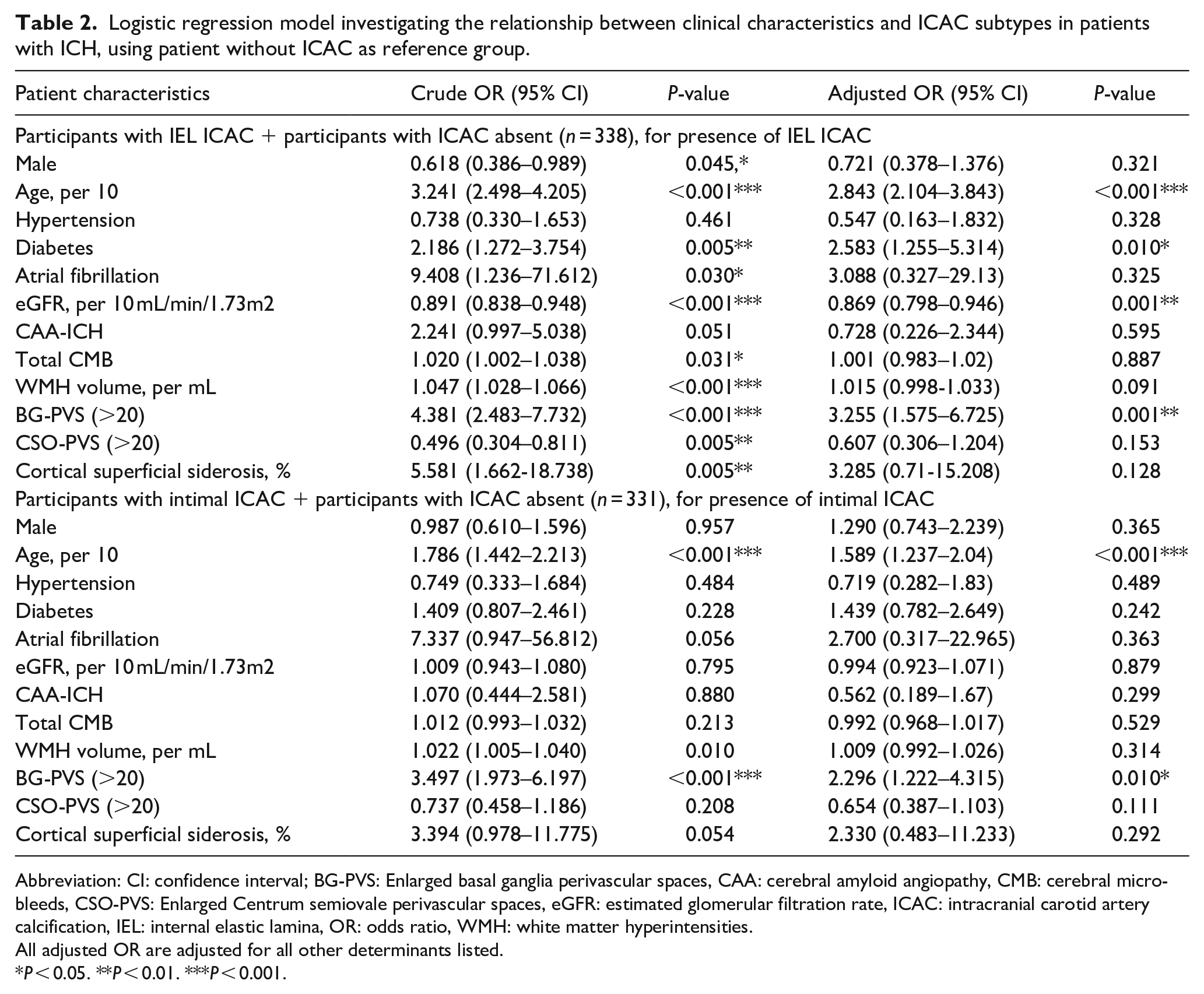
Abbreviation: CI: confidence interval; BG-PVS: Enlarged basal ganglia perivascular spaces, CAA: cerebral amyloid angiopathy, CMB: cerebral microbleeds, CSO-PVS: Enlarged Centrum semiovale perivascular spaces, eGFR: estimated glomerular filtration rate, ICAC: intracranial carotid artery calcification, IEL: internal elastic lamina, OR: odds ratio, WMH: white matter hyperintensities.
All adjusted OR are adjusted for all other determinants listed.
P < 0.05. **P < 0.01. ***P < 0.001.
Similarly, we investigated the risk factors associated with intimal ICAC compared to patients without ICAC as the reference group. Intimal ICAC was independently associated with older age (per 10 years, OR [95% CI]: 1.589 [1.237–2.04], P < 0.001) and more severe BG-PVS (2.296 [1.222–4.315], P = 0.010). However, no significant associations were observed between intimal ICAC and hypertension, diabetes, atrial fibrillation, renal function or CAA (all P > 0.05) in the multivariable regression analyses (Table 2).
ICAC subtypes and risk of events
Patients were followed up for a median of 2.0 years (IQR 1.8‒2.0 years). During follow-up, a total of 100 composite events were recorded, including 36 recurrent ICH, 29 ischemic strokes, and 12 major cardiovascular events. Additionally, 50 participants (9.0%) died during the follow-up period; 25 of these deaths (50%) were due to vascular causes. Of the composite events, 5.0% (5/100) occurred in the absent ICAC group, 31.0% (31/100) occurred in the intimal ICAC group, and 64.0% (64/100) occurred in the IEL ICAC group.
Overall, the cumulative incidence of vascular death, all-cause mortality, or any composite event was highest in the IEL ICAC group (Figure 3). Specifically, IEL ICAC was associated with a higher risk of ischemic stroke (adjusted HR [95% CI]: 9.0 [1.0‒77.4], P = 0.046), vascular death (13.6 [1.7‒1772.4], P = 0.009), all-cause mortality (13.9 [1.8‒105.8], P = 0.011), and any composite event (5.9 [2.2‒15.7], P < 0.001) compared the group without ICAC (Table 3).
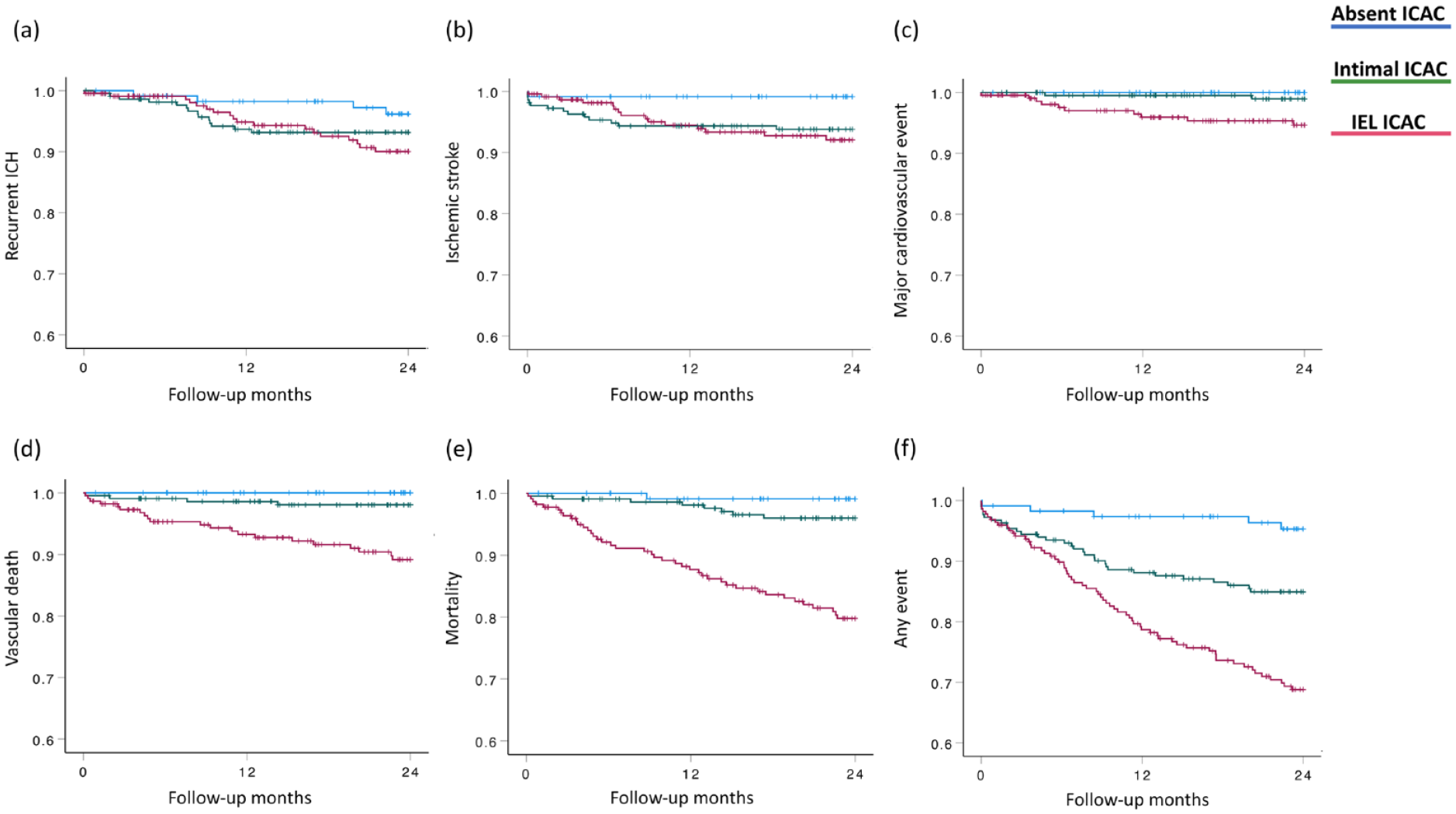
Kaplan-Meier analyses for the primary endpoints (a) recurrent intracerebral hemorrhage (ICH), (b) ischemic stroke, (c) major cardiovascular event, (d) vascular death, (e) mortality, and (f) any event for patients with ICH with different subtypes of intracranial carotid artery calcification (ICAC).
Time-to-event outcome analysis for patients with ICH with different morphological subtypes of ICAC.
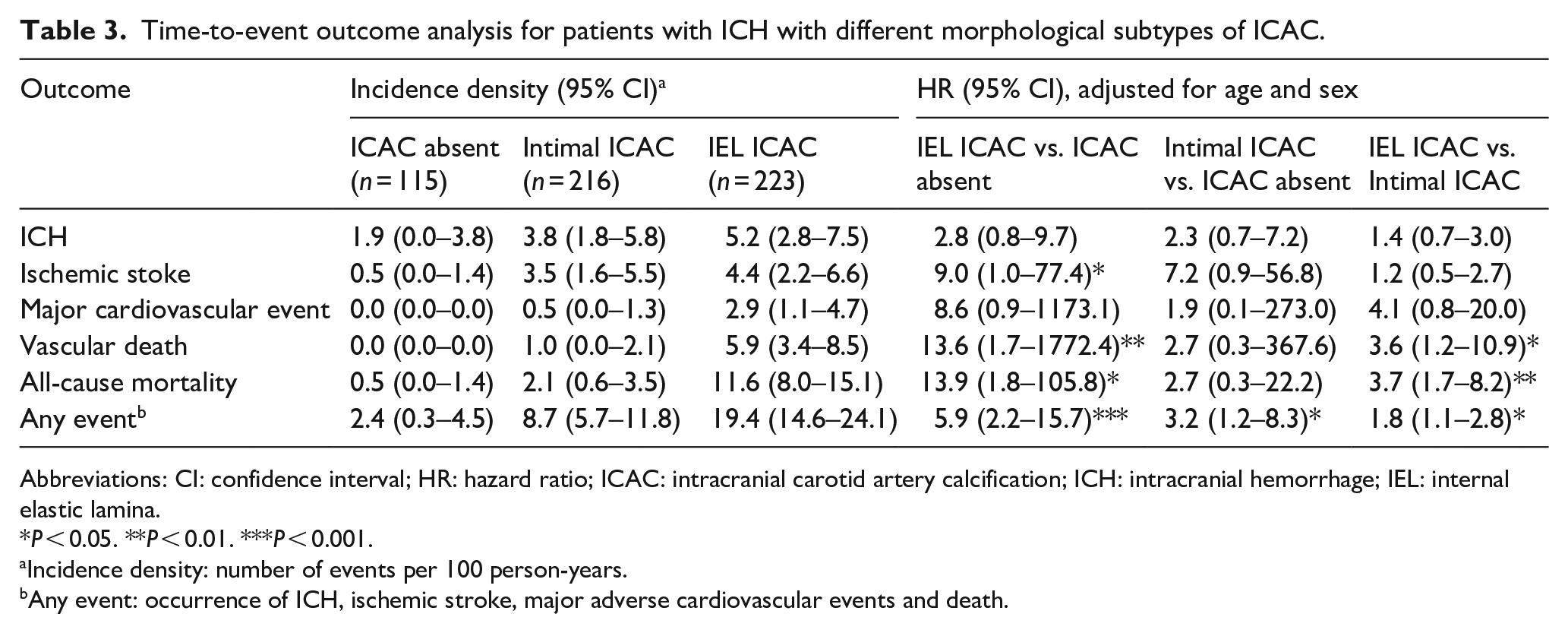
Abbreviations: CI: confidence interval; HR: hazard ratio; ICAC: intracranial carotid artery calcification; ICH: intracranial hemorrhage; IEL: internal elastic lamina.
P < 0.05. **P < 0.01. ***P < 0.001.
Incidence density: number of events per 100 person-years.
Any event: occurrence of ICH, ischemic stroke, major adverse cardiovascular events and death.
Patients with intimal ICAC had a higher risk of any composite event (adjusted HR 3.2, 95% CI 1.2‒8.3, P = 0.020), but similar risks for the individual events compared to patients without ICAC (Table 3). Furthermore, the IEL subtype group exhibited higher risks of vascular death (adjusted HR [95% CI]: 3.6 [1.2‒10.9], P = 0.025), all-cause mortality (3.7 [1.7‒8.2], P = 0.001), and any composite event (1.8 [1.1‒2.8], P = 0.012) compared to the group with intimal ICAC (Table 3).
Finally, we investigated the impact of IEL ICAC on the risk of the outcome events among survivors of deep ICH (n = 442) and lobar ICH (n = 112), respectively (Supplemental Table 2). In survivors of deep ICH, IEL ICAC was associated with higher risks of vascular death, all-cause mortality, and any composite event compared to patients without ICAC (all P < 0.05). IEL ICAC was also associated with higher risks of all-cause mortality and any composite event compared to intimal ICAC among the survivors of deep ICH (both P < 0.05). Among the survivors of lobar ICH, the risks of the outcome events were not significantly different between patients with IEL ICAC and patients without ICAC. All-cause mortality was the only event that had a significantly higher risk in patients with IEL ICAC compared to patients with intimal ICAC (P < 0.05).
Discussion
This study indicates that IEL ICAC is the most prevalent type of intracranial arteriosclerosis in patients who survive spontaneous ICH (38.6%), closely followed by intimal calcification (37.4%). The presence of IEL ICAC was independently associated with the clinical risk factors of older age, diabetes, and poor renal function, as well as small vessel neuroimaging variable of more severe BG-PVS enlargement. Notably, compared to patients without ICAC or with the intimal subtype of ICAC, IEL ICAC was associated with higher risks of the occurrence of any adverse event, vascular death, and all-cause mortality. In addition, patients with IEL ICAC exhibited a higher risk of incident ischemic stroke than patients without ICAC in long-term follow-up. Overall, our findings suggest that IEL ICAC is a vascular marker of a poor prognosis in ICH survivors.
The prevalence of ICAC in our study (80.1%) among ICH survivors (mean age: 63.7) aligns with previous Western studies, which reported rates of 76.9–79.6% in a slightly older population (mean age: 70.0–71.2).21,22 To our knowledge, this is the first study to examine the association between ICAC subtypes, its risk factors and implication on long-term prognosis in survivors of ICH. We found patients with the IEL subtype tended to be older, more frequently had diabetes, and had lower eGFR, which is consistent with prior studies of ischemic stroke cohorts and the general population.23–26 In contrast, age, but no other vascular risk factors, were associated with intimal calcification, which confirmed our hypothesis that these subtypes of ICAC are associated with distinct risk profiles and potentially have different pathophysiological impacts on patients who survive an ICH. This finding is supported by previous reports, including a study with histological data, showing that IEL calcification (also called medial calcification) involves the accumulation of calcium substrates in the tunica media of vessel walls, and is associated with arterial stiffness but is unrelated to atherosclerotic lesions.4,5,27–29 On the contrary, intimal calcification is more closely related to stable atherosclerotic plaques, and shows a distinct entity from IEL calcification.5,27,28
Interestingly, both IEL and intimal ICAC were independently related to important markers of SVD, including more severe BG-PVS and a trend toward increased WMH volume, which implies ICAC may potentially play a pathogenic role in the underlying cerebral microangiopathy that leads to spontaneous ICH. Intracranial arteriosclerosis has been recognized as a potential contributor to cerebral SVD, showing associations to neuroimaging markers such as WMH, lacunes and CMBs. 30 One study that investigated the relationship between blood pressure, ICAC volume and SVD suggested that intracranial arteriosclerosis, in particular IEL calcification, probably mediates the association between blood pressure and SVD change on MRI. 4 Together with our findings, intracranial arteriosclerosis may exert a pathophysiological effect and therefore increase the patient’s vulnerability to microvascular injuries.4,31
One important result of the current study is our observation of an association between ICAC and patient outcomes after spontaneous ICH, which implies that this CT marker could be considered as a potential prognostic marker for survivors of ICH. As expected, intimal ICAC was associated with a higher risk of composite events compared to patients without ICAC, which aligns with expectations based on atherosclerotic pathophysiology and the existing literature on intracranial atherosclerotic disease. 32 However, unlike prior studies in the general population, 3 we observed significantly stronger associations between IEL ICAC and higher risks of composite events, vascular death, and all-cause mortality in the ICH survivors, which indicates that IEL ICAC independently elevates the risk of vascular events and mortality in patients with severe SVD. This finding was in line with a previous study on ICAC subtypes, which reported a potential association between the presence of ICAC and in-hospital mortality in ICH patients. 22 Notably, our subgroup analysis showed that the elevated risks associated with IEL ICAC were mainly driven by deep ICH, in which hypertensive arteriolosclerosis is considered to be the predominant vascular pathology. Therefore, our results further confirm the detrimental role of large vessel pathology in patients with hypertension and the consequent deep perforator arteriopathy.
This study has several limitations. First, our CT methods for ICAC categorization may not be able to precisely estimate involvement of the IEL or intimal subtypes, which could lead to potential misclassification of the subtypes of ICAC. However, prior studies have suggested strong correlations between CT classification and histology. 12 However, our CT-based approach offers the advantages of enabling larger sample sizes and is more accessible and relevant to clinical practice. Future research could focus on developing a quantitative method to assess the proportions and coexistence of these subtypes of ICAC. Second, our cohort only included Asian patients, which could limit the generalizability of our findings to other ethnicities. The underlying subtypes of SVD and the severity of intracranial arteriosclerosis may vary between Eastern and Western populations;33,34 thus, our results need to be further validated in non-Asian ICH cohorts. Lastly, we included only patients with available MRI data, while the follow-up outcomes in patients without MRI study were unavailable. However, patients who did not receive MRI after ICH might have more neurologically severe disease, a larger hematoma volume, and/or a poorer prognosis in the early stage, while those who received MRI may have had specific clinical presentations prompting further investigation. These factors may introduce selection bias and limit the generalizability of our findings to the broader ICH population.
In conclusion, ICAC is frequently observed in patients who survive spontaneous ICH and the IEL ICAC subtype may have clinical relevance as a marker of poor prognosis. IEL ICAC was associated with age, diabetes, and impaired renal function and linked to neuroimaging SVD markers. IEL ICAC was identified as a prognostic indicator for any composite event, vascular death, and all-cause mortality among ICH survivors. Our findings clarify the role of large artery disease in the underlying microangiopathy and disease outcomes of patients SVD-related ICH.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241306576 – Supplemental material for Intracranial carotid arteriosclerosis and their prognosis in intracerebral hemorrhage
Supplemental material, sj-docx-1-eso-10.1177_23969873241306576 for Intracranial carotid arteriosclerosis and their prognosis in intracerebral hemorrhage by Bo-Ching Lee, Hsin-Hsi Tsai, Ya-Fang Chen, Jiann-Shing Jeng and Li-Kai Tsai in European Stroke Journal
Footnotes
Acknowledgements
We thank the staff of the Fifth Core Lab, Department of Medical Research, National Taiwan University Hospital for technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Taiwan University Hospital (Lee BC, 111-X0002 & 112-S0198) and National Science and Technology Council of Taiwan (Lee BC, 111-2314-B-002-250-MY2; Tsai HH, 112-2923-B-002-001-MY3).
Informed consent
Informed consent was waived.
Ethical approval
The primary data used in this study were obtained from the NTUH Stroke Registry, which is a database that prospectively includes all patients with stroke from a tertiary referral center in Taiwan. The Research Ethnics Committee of the NTUH approved the registry.
Guarantor
Hsin-His Tsai, MD, PhD
Contributorship
Bo-Ching Lee: study conceptualization, data collection and analyses, manuscript drafting
Hsin-Hsi Tsai: study conceptualization, data collection and analyses, manuscript drafting and revision
Ya-Fang Chen: data collection, critical revisions
Jiann-Shing Jeng: data collection, critical revisions
Li-Kai Tsai: data collection, critical revisions
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
