Abstract
Background:
Ischaemic stroke and coronary artery disease share risk factors and stroke survivors experience a high rate of cardiac events. Recent work suggests a high burden of asymptomatic coronary artery disease (CAD) in ischaemic stroke survivors. Thus, we performed this systematic review and meta-analysis to A) estimate the prevalence of CAD in ischaemic stroke survivors without known CAD and B) evaluate the association between coronary atherosclerosis and future major adverse cardiovascular events (MACE) in stroke survivors.
Patients and Methods:
We conducted a systematic review and meta-analysis according to the PRISMA statement. We included studies investigating acute ischaemic stroke or transient ischaemic attack where participants underwent anatomical assessment of all coronary arteries. For objective B) we included studies that reported an association between coronary atherosclerosis and MACE. Two reviewers used the Newcastle-Ottawa Scale to assess risk of bias. We used random-effects modelling for our analyses.
Results:
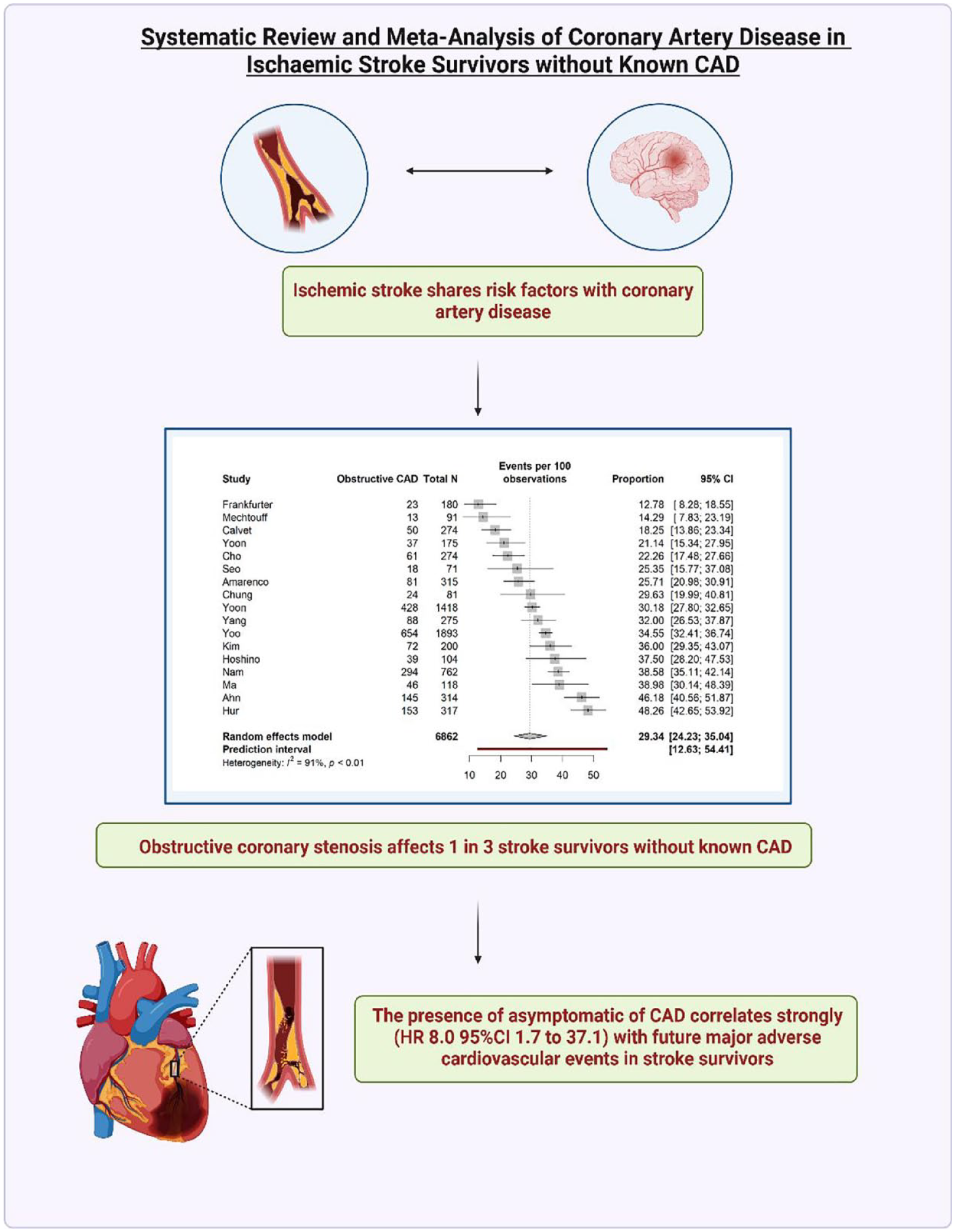
We identified 2983 studies of which 17 were included. These studies had a total of 6862 participants between 2008 and 2022. The pooled prevalence of any coronary atherosclerosis was 66.8% (95% CI 57.2%–75.1%) with substantial heterogeneity (I2 = 95.2%). The pooled prevalence of obstructive (>50%) stenosis was 29.3% with substantial heterogeneity (I2 = 91%). High-risk coronary anatomy (triple vessel disease or left main stenosis) was found in 7.0% (95% CI 4%–12%) with high heterogeneity I2 = 72%. One study examined high-risk plaques and found a prevalence of 5.9%. Five studies reported the association of coronary atherosclerosis with future MACE. The presence of obstructive CAD confers a HR of 8.0 (95% CI 1.7–37.1, p = 0.007) for future MACE.
Discussion and Conclusions:
Asymptomatic CAD is common in ischaemic stroke survivors. The presence and severity of asymptomatic CAD strongly associates with the risk of future MACE. Further evaluation of the benefits of routine coronary assessment in ischaemic stroke is warranted.

Keywords
Introduction
Ischaemic stroke and coronary artery disease represent two of the highest burden diseases in the world. The two conditions share common risk factors and thus, stroke survivors are likely to harbour asymptomatic coronary artery disease. 1 Indeed, the 1-year incidence of cardiac events in ischaemic stroke survivors approaches 10%, with an annual risk of non-stroke vascular death of 2.1% and annual myocardial infarction risk of 3%.2–4 The optimal strategy and clinical benefit of coronary risk assessment in ischaemic stroke survivors is uncertain. The most recent guidelines were published 20 years ago and suggest consideration of non-invasive coronary assessment in stroke survivors with a high cardiovascular risk profile or established carotid atherosclerosis. 5
These guidelines predate the widespread use of computed tomography coronary angiography (CTCA), as well as more recent insights into the limited role of revascularisation in chronic coronary syndromes.6,7 Additionally, there is a rapidly expanding range of disease-modifying therapies (e.g. potent lipid lowering agents such as PCSK-9 inhibitors and anti-inflammatory drugs such as colchicine) which may benefit from the identification of a polyvascular atherosclerotic phenotype to help achieve more aggressive prevention targets.8,9
Studies have demonstrated a high prevalence of coronary atherosclerosis and obstructive stenosis in stroke survivors using anatomical assessment (invasive angiography or CTCA). A previous meta-analysis recognised that 32% of stroke survivors have at least one >50% asymptomatic coronary stenosis. 4 Since then, multiple large studies have performed CTCA in stroke survivors to assess the prevalence of coronary disease and its prognostic implications. However, no systematic review to date has evaluated the association of asymptomatic coronary disease with cardiovascular outcomes in stroke survivors. The objectives of this systematic review and meta-analysis are to A) describe the prevalence and characteristics of coronary atherosclerosis in ischaemic stroke survivors without known coronary artery disease and B) define the association between coronary artery atherosclerosis characteristics at the time of ischaemic stroke with subsequent major adverse cardiovascular events (MACE).
Methods
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses statement. 10 Data not published in the article are available from the corresponding author on reasonable request. The study was listed on the PROSPERO register of systematic reviews (CRD 42023444306).
Information sources and search strategy
We systematically searched Medline (through Ovid), EMBase (through Ovid), the Cochrane Library and Scopus from the year 2000 to the 17th of April 2023 for published articles that reported an assessment of coronary atherosclerosis in adult ischaemic stroke survivors without a history of coronary artery disease (objective A) and articles that evaluated the association between coronary atherosclerosis and future MACE in the same population (objective B). The search strategy was executed after peer review by senior authors of the paper with expertise in meta-analysis (NN and SJN). The peer review used the PRESS checklist to ensure validity of the search strategy peer review. 11 The reference lists of all studies in the full text stage were searched for further references. The detailed search strategy is available in the Supplemental Material. We evaluated the success of our search strategy by checking against 11 relevant references identified in a meta-analysis performed 8 years ago in addition to a relevant study performed subsequent to that meta-analysis.4,12 Our search strategy identified 100% of the 12 references successfully.
Eligibility criteria
Studies were eligible for inclusion if they: (a) included adults aged 18 years and above with acute ischaemic stroke or transient ischaemic attack defined by clinical or radiological criteria without a known history of CAD, (b) attempted anatomical assessment of all participants’ coronary arteries with either invasive angiography or CTCA after acute stroke, and (c) were published in the year 2000 or onwards. Additionally, studies examining the effect of coronary atherosclerosis on outcomes were eligible for inclusion if they reported an association between the presence of coronary atherosclerosis and systematic follow-up for MACE. Exclusion criteria included the following: (a) relying only on self-reported coronary artery disease to define coronary atherosclerosis, (b) studies with no original data or reproducing data from a former study or registry, (c) isolated case reports, (d) incomplete data or conference abstracts and (e) studies with less than 50 participants.
Study selection
Two trained reviewers (RGM and TA) independently screened studies at abstract and full-text stages of the review with the use of an online review manager (Covidence systematic review software, Veritas Health Innovation). Disagreements were resolved by consensus with a third senior reviewer (NN). If multiple articles with the same patient cohort were identified, the article with the most participants or with follow-up data regarding MACE was included.
Data extraction
Data from each included article was independently extracted by two reviewers (RGM and TA) and discrepancies were resolved by consensus. The reviewers used an electronic spreadsheet to extract relevant data. They extracted study data regarding author, publication year, study design, traditional cardiovascular risk factors, types of strokes, type of coronary assessment (CTCA vs invasive angiography), number of participants with coronary plaque, obstructive stenosis, high risk coronary stenosis (triple vessel disease or left main coronary artery disease), high risk plaque features (defined as low attenuation plaque, spotty calcification, positive remodelling or the napkin ring sign) and follow-up MACE rates. If data were unclear or incomplete, we contacted corresponding authors by email for further information.
Risk of bias assessment
The quality of included studies was assessed independently by two reviewers using the Newcastle-Ottawa scale assessment tool for cohortstudies. 13 We modified this tool to make it relevant to this meta-analysis. The modified version is available in the Supplemental Materials. In brief, studies could be rated a maximum of 4 stars for objective A (assessment of coronary atherosclerosis) and a maximum of 3 stars for objective B (follow-up of MACE). For objective A, we classified 4 stars as low risk of bias, 3 stars as moderate risk and 1 or 2 stars as high risk of bias. For objective B, 3 stars was considered low risk of bias, 2 stars as moderate risk and 1 star as high risk of bias.
Statistical analysis
For objective A (assessment of the prevalence of coronary atherosclerosis in stroke survivors) we used the proportion (n/N) as the common measure of effect. For meta-analysis, the proportion of different coronary atherosclerosis features were first logit-transformed. The logit-transformed values were then pooled using a generalised linear mixed-effects model (GLMM) which fit a logistic regression model to the data including random effects. We used this method due to its described advantages over traditional arcsine transformations. 14 A maximum-likelihood estimator for τ2 was used. Heterogeneity was assessed using the I2 statistic, with a value of 0% indicating no heterogeneity, up to 25% low heterogeneity, 26%–50% moderate heterogeneity and over 50% as substantial heterogeneity.
We performed planned subgroup analyses for objective A, including analysis of studies using CTCA only for the presence or absence of coronary atherosclerosis given its higher sensitivity for a low burden of coronary plaque; analysis of studies with low risk of bias only; and analysis of only prospective studies. We also performed meta-regression using the weighted least squares method in the following analyses: year of publication to observe for changes in the prevalence of obstructive (>50%) stenosis over time; the proportion of vascular cardiovascular risk factors in each study for obstructive (>50%) stenosis; and stroke sub-types included in studies for obstructive (>50%) stenosis.
For objective B (assessment of the association between asymptomatic coronary artery disease and the risk of future MACE) we used log-hazard ratios (of the crude hazard ratio) as the common measure effect. The log-transformed hazard ratios were pooled using a random effects model with a maximum-likelihood estimator for τ2. We used the same definitions for heterogeneity as objective A with the I2 statistic.
All analyses were conducted using R software version 4.3.0 (R Foundation for Statistical Computing).
Results
Study selection
The systematic search identified 2983 records after exclusion of duplicate studies. After screening, 17 full text papers were included as demonstrated in the PRISMA flow diagram shown in Figure 1.12,15–30
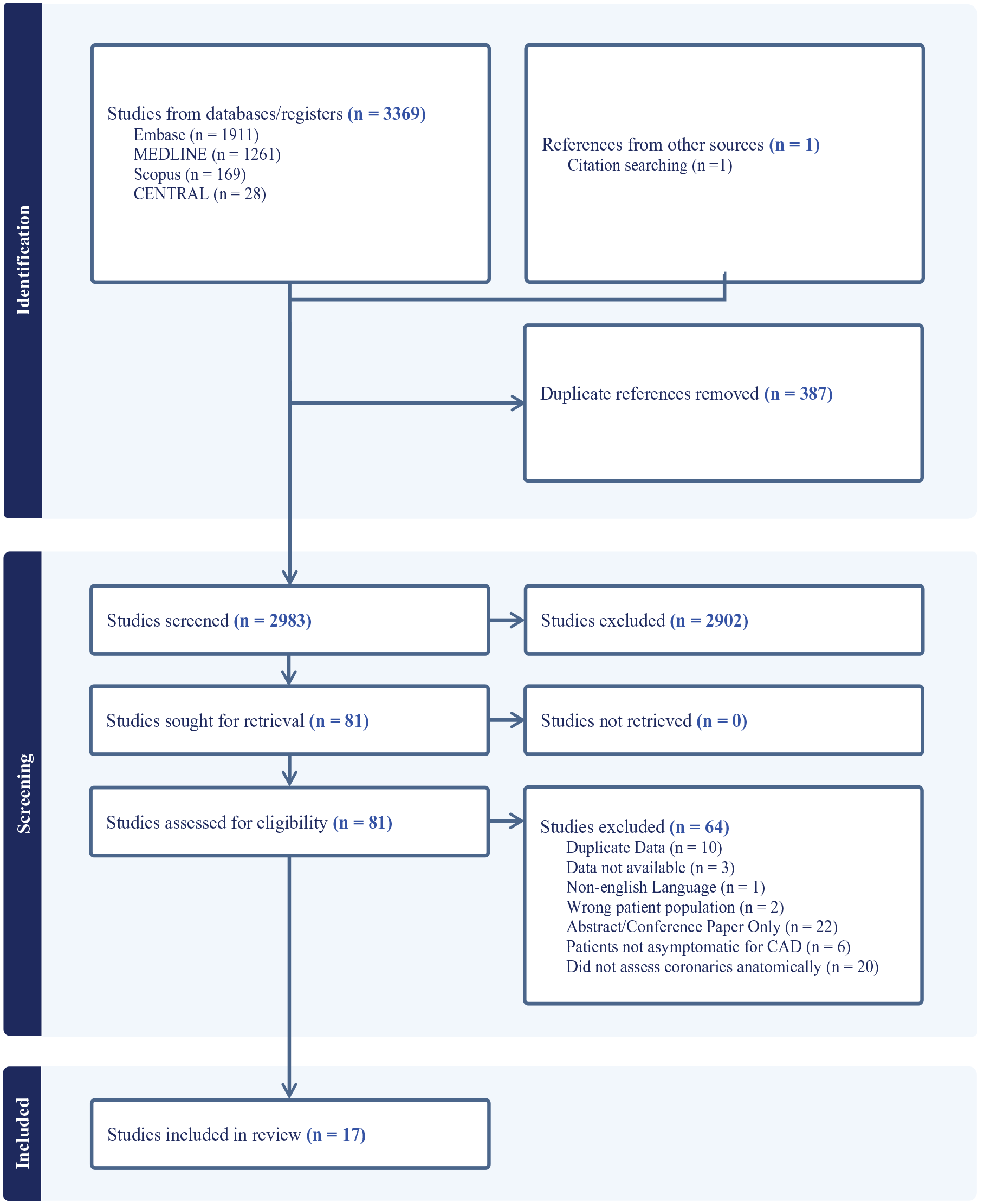
PRIMSA Flow Diagram.
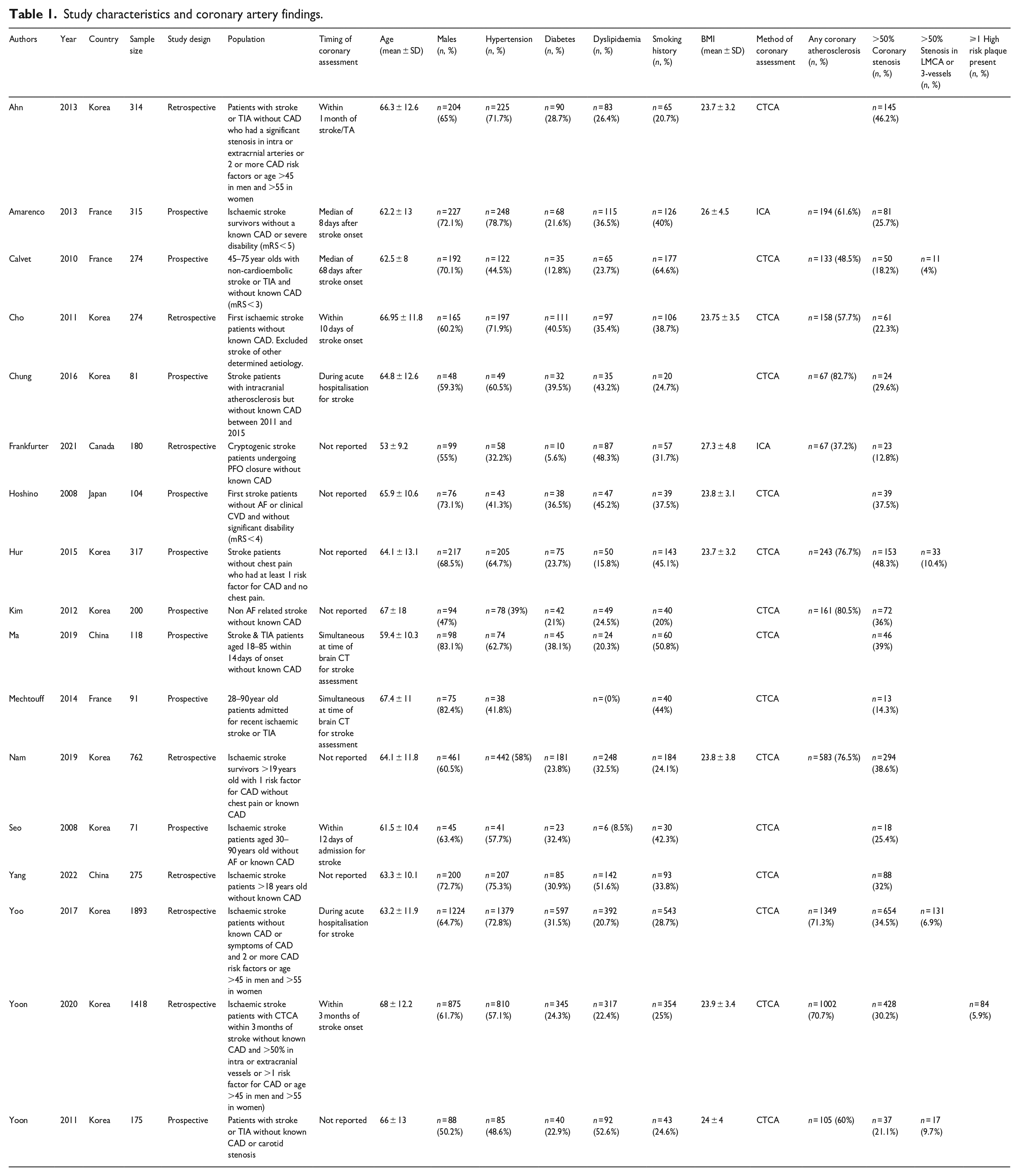
These studies included a total of n = 6862 participants and were published in years ranging from 2008 to 2022. There were 10 prospective studies and 7 retrospective studies. Most publications arose from Korea (n = 10). The pooled age of participants across all studies was 64.1 years (95% CI 63.8–64.3 years). Details regarding each study, including the breakdown of traditional vascular risk factors, are displayed in Table 1.
Study characteristics and coronary artery findings.
Risk of bias
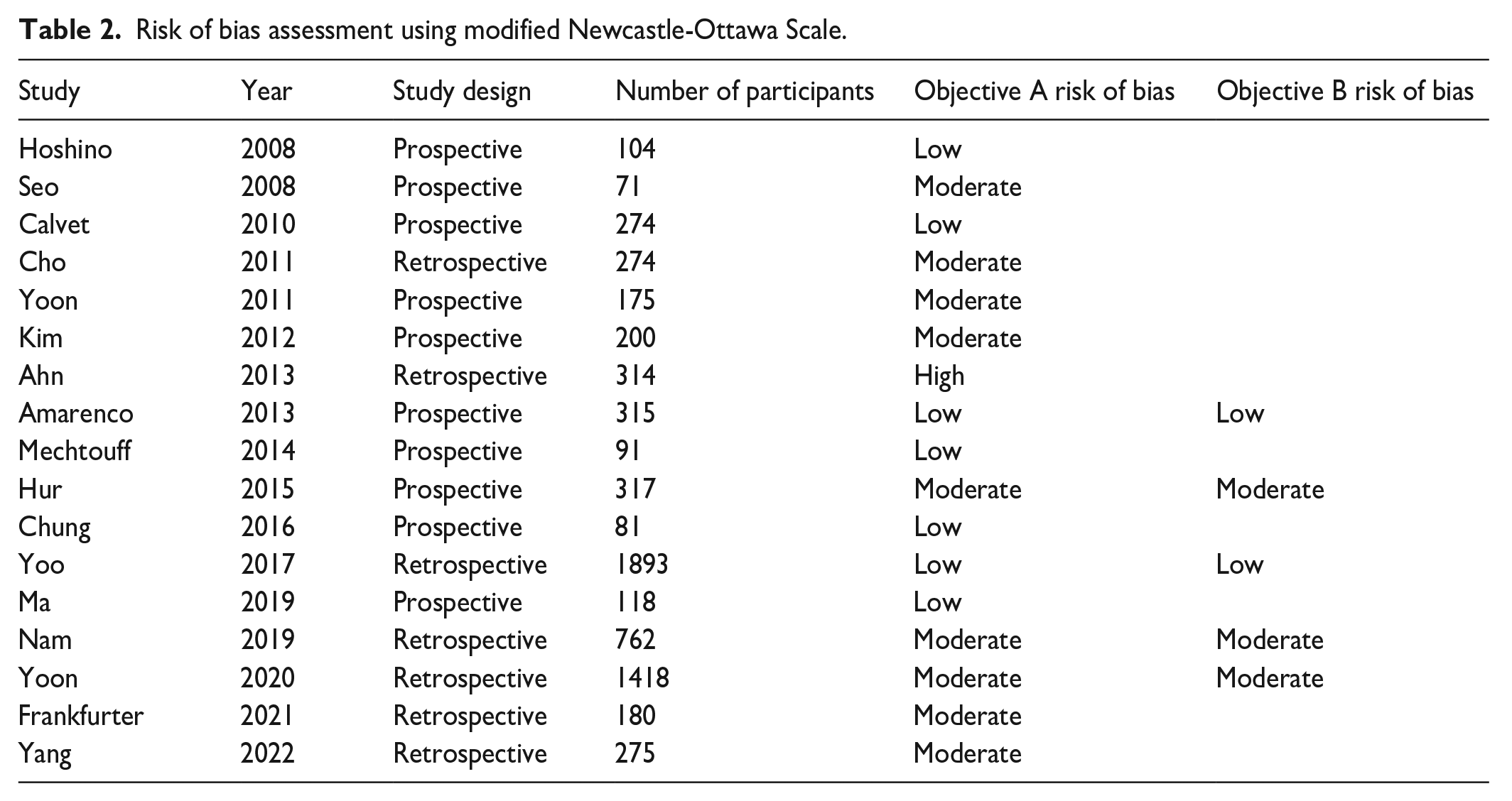
For objective A (examining the prevalence of coronary atherosclerosis in stroke survivors without CAD), most studies were of moderate (n = 9) or low (n = 7) risk of bias as shown in Table 2. There was one study with a high risk of bias due to recruiting only a low proportion of the eligible participants in a retrospective fashion. 15 For objective B (examining the association between coronary atherosclerosis and MACE in stroke survivors), all studies were either of low or moderate risk of bias as they followed up participants using direct methods at time periods long enough for outcomes to occur.
Risk of bias assessment using modified Newcastle-Ottawa Scale.
The prevalence of any coronary atherosclerosis
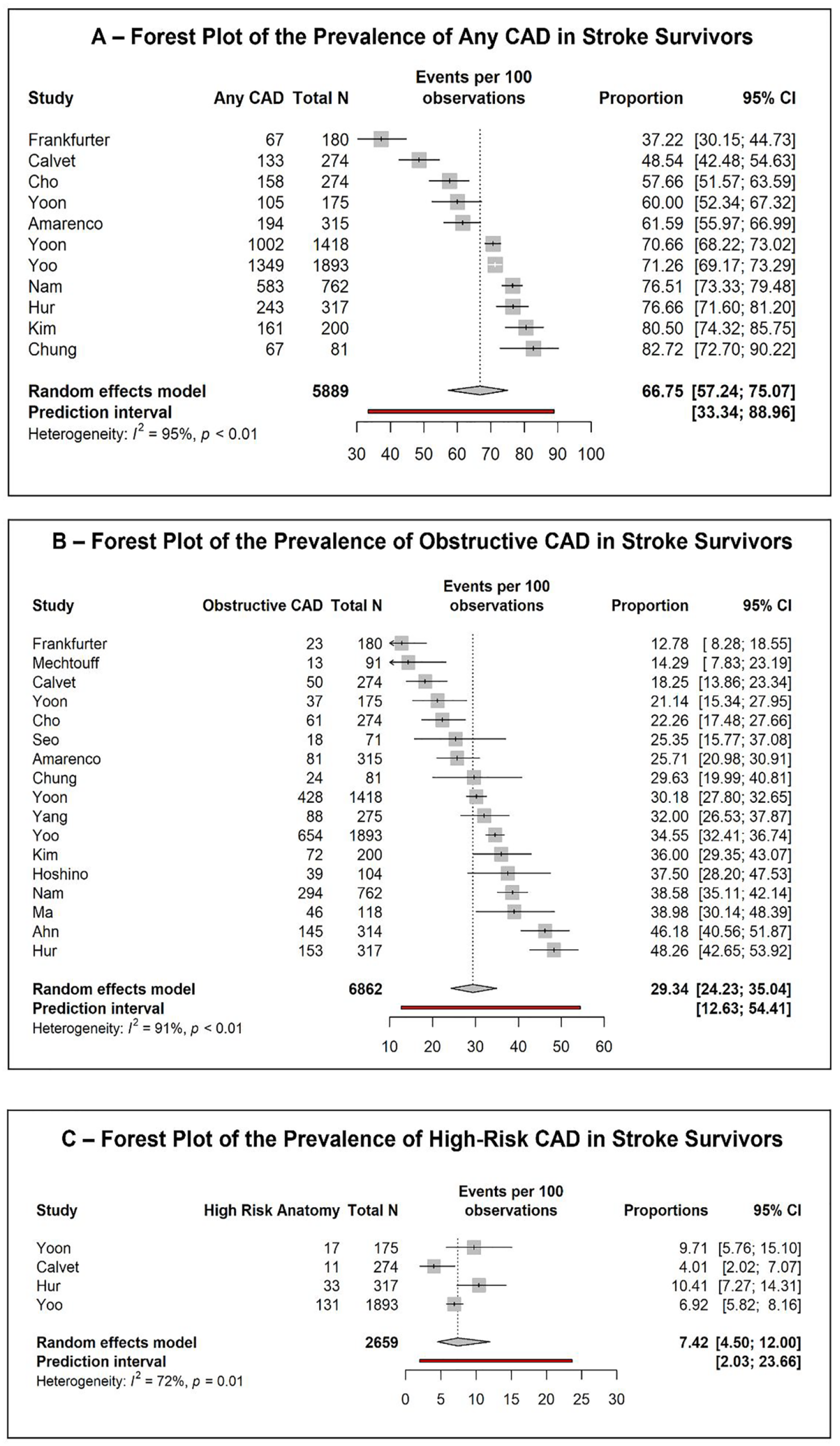
Eleven studies reported data regarding the prevalence of any coronary atherosclerosis in stroke survivors, representing n = 5889 participants. Nine of these used CTCA versus two using invasive coronary angiography. The pooled proportion of stroke survivors with any coronary artery plaque was 66.8% (95% CI 57.2%–75.1%) with substantial heterogeneity, I2 = 95.2% as shown in Figure 2. Baujat plots for influence analysis showed that the studies by Frankfurter et al. and Calvet et al. had outsized influence as shown in the Supplemental Material. Frankfurter et al. was a retrospective study of participants with invasive coronary angiography at the time of patent foramen ovale closure for cryptogenic stroke. 20 Sub-group analysis comparing studies using CTCA versus invasive angiography showed a higher prevalence of any coronary atherosclerosis with CTCA (70.1%, 95% CI 61.4%–77.5% compared to 49.6%, 95% CI 1.1%–98.9%, p = 0.02).
Central Illustration.
The prevalence of obstructive (>50%) stenoses
Seventeen studies reported the prevalence of obstructive (>50%) stenoses in stroke survivors across 6862 participants. Fifteen of these used CTCA compared to two using invasive angiography. The pooled prevalence of obstructive stenosis across all studies was 29.3% (95% CI 24.2%–35.0%) with substantial heterogeneity (I2 = 91%) as shown in Figure 2. Influence analysis demonstrated four studies with high influence, but there was no clear decrease in heterogeneity with leave-one-out analysis as shown in the Supplemental Material.
Comparing the prevalence of obstructive stenosis by risk of bias revealed a significantly higher prevalence in the one study with a high risk of bias (46.2%, 95% CI 40.1%–51.7%) compared to the moderate (28.9%, 95% CI 21.5%–37.6%) and the low (27.7%, 95% CI 21.5%–37.6%) risk of bias studies. There was no difference in the prevalence of obstructive stenosis when comparing retrospective (30.1%, 95% CI 21.3%–40.8%) and prospective (28.7%, 95% CI 21.9%–36.7%) studies.
Meta-regression of the prevalence of obstructive stenosis accounting for study year demonstrated no significant change in the period between 2008 and 2022 (p = 0.7) with no additional heterogeneity accounted for (I2 = 91%).
Six studies reported the proportion of participants with large artery atherosclerosis as the cause of stroke. Meta-regression for the proportion of strokes attributed to large artery atherosclerosis also demonstrated no effect on the prevalence of obstructive coronary stenosis (p = 0.9) and explained only 8% of heterogeneity as shown in Supplemental Figure 5. Additionally, meta-regression for the proportion of vascular risk factors (male sex, hypertension, diabetes, dyslipidaemia and smoking history) revealed that diabetes had a small effect (OR 1.02 95% CI 1.00–1.05 per % of diabetic participants). Vascular risk factors accounted for 26.8% of heterogeneity in the meta-regression. In the six studies that included only subjects with a vascular risk factor profile, the prevalence of obstructive CAD was higher than in those without risk factor profile as an inclusion criteria (37.9%, 95% CI 30.8%–45.5% vs 25.0%, 95% CI 19.5%–31.3% respectively, p = 0.001).
Subgroup analysis comparing studies that included only ischaemic stroke versus those that included both TIA and stroke did not reveal a significant difference in the prevalence of obstructive CAD (30.6%, 95% CI 25.0%–36.7% vs 26.4%, 95% CI 14.0%–44.2% respectively, p = 0.5).
There were significant differences in the prevalence of obstructive coronary stenosis by country of study, with Korea, China and Japan having significantly higher prevalence than France or Canada (p < 0.0001) as shown in Table 3 and Figure 3. However, there was only one study from Canada with a unique population of cryptogenic stroke survivors undergoing invasive angiography during patent foramen ovale closure.
Subgroup analysis of obstructive CAD by study country.

Overall p-value: <0.0001.
The prevalence of high-risk anatomy (Left main stenosis or triple vessel disease)
Four studies reported the prevalence of high-risk anatomy defined as left main significant stenosis or triple vessel disease representing 2659 participants.12,17,28,29 The pooled prevalence of high-risk anatomy across these four studies was 7.0% (95% CI 4%–12%) with substantial heterogeneity I2 = 72% as shown in Figure 2.
The prevalence of high-risk plaques
Only one study by Yoon et al. examined the frequency of high-risk plaques determined on CTCA (defined as >2 high risk features) in stroke survivors (n = 1418). In this study, 84 (5.9%) of participants were found to have high-risk plaques. 29
The association of coronary atherosclerosis features and MACE
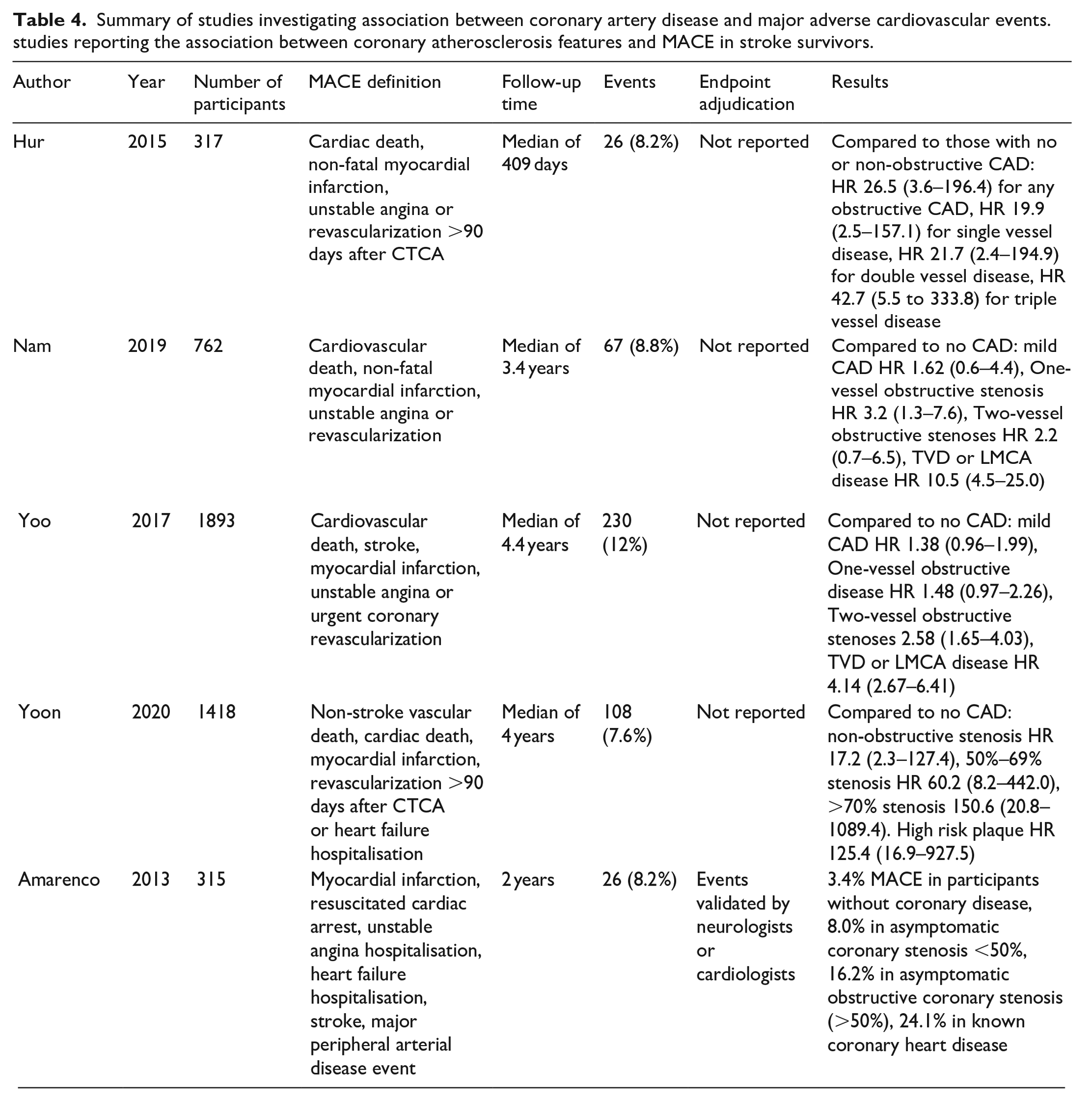
Five studies investigated the association between coronary atherosclerosis features and future MACE in ischaemic stroke survivors.12,16,25,28,29 Four studies reported the association in hazard ratio, while one study used proportion of participants with MACE at a 2 year time point. The studies differed in their definition of MACE and timeframe of follow-up as shown in Table 4. Only one study (Amarenco et al. 16 ) reported the adjudication of endpoints. The study by Amarenco et al. used invasive coronary angiography while the rest used CT coronary angiography to define coronary atherosclerosis. 16
Summary of studies investigating association between coronary artery disease and major adverse cardiovascular events. studies reporting the association between coronary atherosclerosis features and MACE in stroke survivors.
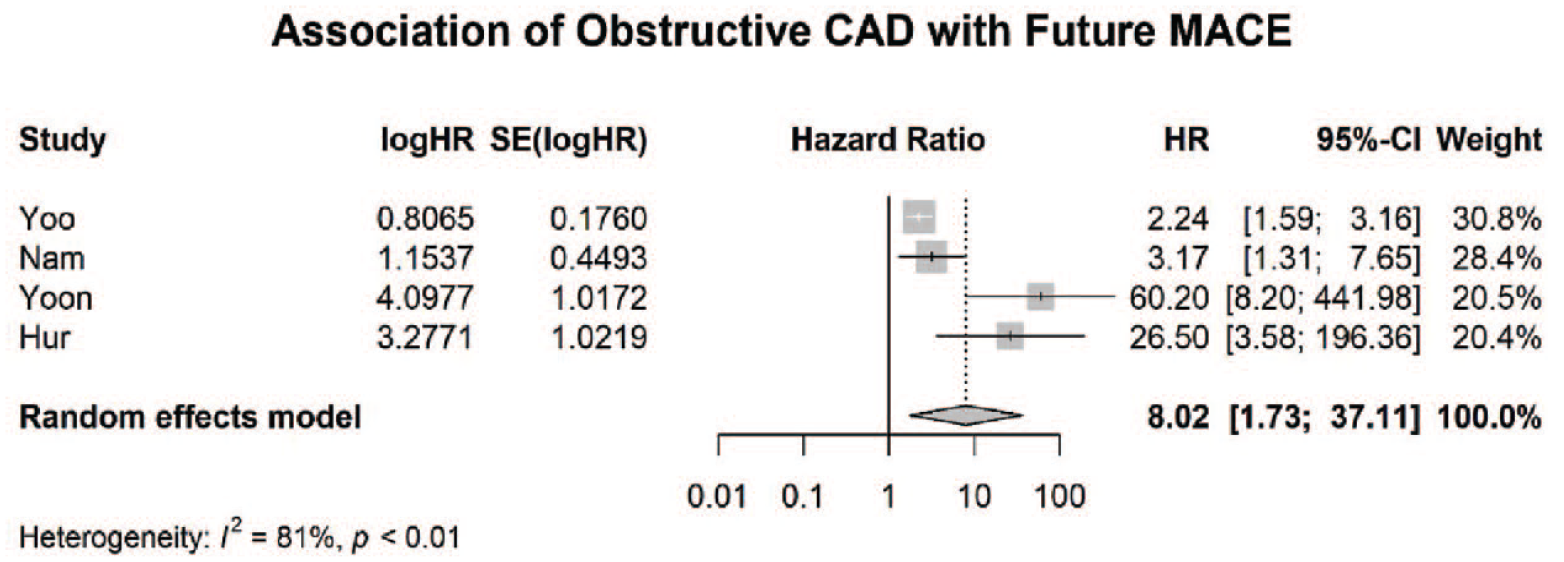
The pooled hazard ratio for future MACE in the presence of obstructive coronary artery disease was 8.0 (95% CI 1.7–37.1, p = 0.007) with substantial heterogeneity (I2 = 81%) as shown in Figure 4. Individual results from each study are shown in Table 4.
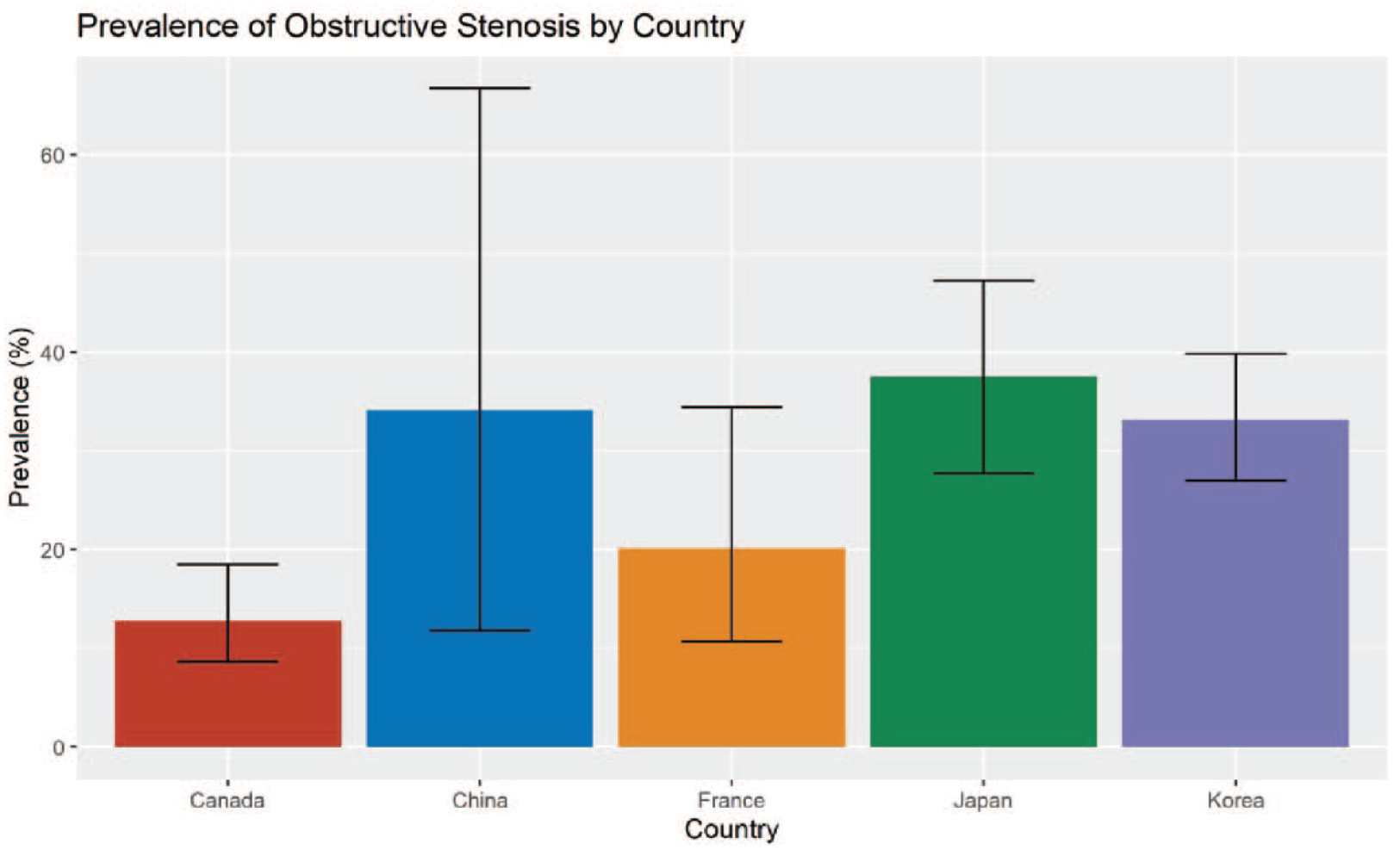
The Prevalence of Obstructive Coronary Stenosis in Ischaemic Stroke Survivors by Country of Study. Canada included only one study of individuals undergoing patent foramen ovale closure which likely explains the lower prevalence of obstructive stenosis.
MACE hazard ratios (HR) for any obstructive disease compared to those with no coronary disease ranged from 1.48 at 4.4 years in the study by Yoo et al. to a HR of 60.2 at 4 years in the study by Yoon et al.28,29 Triple vessel disease or left main coronary artery disease was associated with a hazard ratio ranging from 4.14 in Yoo et al. to 42.7 in the study by Hur et al.12,28 Only Yoon et al. examined events in patients with high-risk plaque features. 29 They found that the presence of high-risk plaque features conferred a hazard ratio of 125.4 (95% CI 16.9–927.5) when compared to no coronary atherosclerosis with notably wide confidence intervals.
Discussion
Our study presents several novel findings about coronary artery disease in ischaemic stroke survivors without a known history of cardiac disease. Firstly, we found that two in three asymptomatic ischaemic stroke survivors have coronary atherosclerosis. Secondly, approximately one in three are found to have asymptomatic obstructive stenosis when assessed with invasive or CT coronary angiography. Notably, one in 15 participants (7.4%) are found to have asymptomatic high risk or ‘surgical’ coronary anatomy defined as triple vessel disease or left main coronary artery stenosis. There was significant heterogeneity in these findings, which is at least partially reflective of the wide range of stroke aetiologies and the underlying risk factor profile of included participants. As an example, the study by Frankfurter et al. included only participants undergoing patent foramen ovale closure and thus had a unique population with a lower incidence of coronary artery disease as identified in influence analysis. 20 Additionally, we found that grades of asymptomatic coronary artery stenosis severity in stroke survivors, demonstrated by an increase in the number of vessels with obstructive CAD as shown in Table 4, are associated with an increase in future MACE risk with a pooled hazard ratio of 8.0 for the presence of obstructive CAD.
This systematic review represents a comprehensive update of the work done by Gunnoo with the addition of several large studies performed since then. 4 We also extend that work by presenting data regarding the association of coronary atherosclerosis features with future MACE. In their study, Gunnoo et al. included other forms of coronary assessment, such as autopsy findings and coronary artery calcium scoring. We limited our study to detailed anatomical assessment of the coronary arteries (with invasive angiography or CTCA) performed in stroke survivors. In comparison to our finding that 66.8% of stroke survivors have asymptomatic coronary atherosclerosis, they found a prevalence of 52%. This may relate to the inability of calcium scoring to detect non-calcified plaque. They also found a pooled prevalence for obstructive (>50%) stenosis of 32%, compared to our finding of 29.3%. The present study included many more participants even after excluding autopsy and coronary calcium scoring studies (6862vs 4869).
No meta-analysis to date has reported the association of asymptomatic coronary atherosclerosis to MACE in stroke survivors. We found wide-ranging effect sizes across studies from a hazard ratio of 1.5 to 150.28,29 These differences might partially relate to heterogeneity in study design, with altering definitions of MACE and follow-up periods as demonstrated in Table 4. Additionally, stroke is a heterogeneous condition affecting a wide variety of individuals – it is likely that individual characteristics strongly influence the risk of future MACE. Regardless, these findings are important. Current guidelines suggest coronary assessment in stroke survivors with intracranial or carotid stenosis or high risk by the Framingham algorithms (10-year MACE risk >20%). 5 These guidelines are now 20-years old and pre-date the existence of non-invasive and low-risk coronary assessment with CTCA. Our study demonstrates that asymptomatic coronary atherosclerosis does markedly increase the risk of future MACE. However, it is unclear whether the detection of such atherosclerosis leads to meaningful changes in therapy that justify the risks and costs of testing The SCOT-HEART trial of CTCA versus routine care for patients with stable chest pain demonstrated that CTCA resulted in the initiation of more preventative therapies than in those who did not have their coronary arteries anatomically assessed. 6 A recent observational study of over 90,000 older individuals with first-ever stroke demonstrated a 12-month incidence of cardiac events approaching 10%, highlighting the ongoing and unsolved importance of managing cardiac risk in stroke survivors. 2
Contemporary data from the ISCHEMIA trial suggest that revascularization for stable but severe ischaemic coronary lesions does not improve mortality, though extended follow-up does suggest that an invasive strategy reduces cardiovascular mortality.7,32 However, it is unclear whether recent acute stroke survivors should be considered in this same patient population given their high propensity for future vascular events. 33 Furthermore, ISCHEMIA excluded participants with left main coronary artery disease, which was prevalent in up to 7% of stroke survivors in our study. Moreover, determination of coronary atherosclerosis may lead to meaningful changes in medical therapy. There are an increasing number of anti-atherosclerotic therapies such as PCSK-9 inhibitors, GLP-1 receptor agonists and colchicine, which may be of greater clinical utility in stroke survivors with a demonstrated atherosclerotic phenotype. Indeed, the most recent European Society of Cardiology guidelines have suggested an aggressive LDL target of <1.4 mmol/L for those at very high risk of future events and a goal of <1.0 mmol/L who experience a second vascular event within 2 years of the first. 9 Our work suggests that coronary assessment may help further differentiate those at very high future MACE risk. The recent HUYGENS and PACMAN-AMI trials demonstrated that PCSK-9 inhibitors have stabilising effects on coronary plaque beyond statin therapy, with reduction in high risk plaque features and atheroma burden.34,35 An atherosclerotic phenotype may also help to drive the initiation of more aggressive antithrombotic therapy. Supporting this idea, a subgroup analysis of the SOCRATES trial of ticagrelor in stroke found the addition of ticagrelor was beneficial for participants with atherosclerosis despite having no effect in the wider study sample. 36 The COMPASS trial of low-dose rivaroxaban in addition to aspirin demonstrated a reduction in MACE at the cost of higher bleeding. 37 Targeting high atherosclerotic risk stroke survivors may help to improve the net clinical benefit of this therapy. GLP-1 receptor agonists have also been recently demonstrated to reduce the risk of MACE in individuals with a history of cardiovascular disease and may be of benefit to stroke survivors with an atherosclerotic phenotype. 38 Lastly, data suggests that demonstration of coronary atherosclerosis on a CTCA can improve patient adherence to preventative medications such as statin therapy. 39 These conjectures all remain to be tested through rigorous assessment of the effects of routine coronary assessment in ischaemic stroke survivors. One retrospective observational study has evaluated outcomes in stroke survivors who undergo CTCA compared to those who do not. They found that CTCA was associated with a much lower risk of mortality and cardiovascular events. However, this study is highly subject to selection bias and should serve only as hypothesis generating as to the benefits of routine coronary assessment in stroke survivors. 40
Novel coronary atherosclerosis features such as low attenuation plaque burden and high-risk plaque features on CTCA (such as spotty calcification, positive remodelling and low attenuation plaque) are potent predictors of future vascular events.41,42 Only one study examined the prevalence and prognostic impact of high-risk coronary plaque features. They found a prevalence of approximately 6% and that the presence of high-risk plaque conveys a hazard ratio of 125 for future MACE. The recent MIND THE HEART study demonstrated that CTCA also provides accurate diagnosis of intracardiac thrombus when performed at the time of ischaemic stroke. 43 These are exploratory findings but warrant more investigation into novel CTCA measures of future vascular risk in stroke survivors.
Limitations
Our meta-analysis is subject to several limitations. Firstly, there was substantial heterogeneity in the estimates across all types of coronary atherosclerosis. Influence and subgroup analysis did not identify any single explanation. However, differences between CT and invasive coronary angiography, country of origin, type of stroke and the vascular risk factor prevalence in studies may explain some heterogeneity. Notably, many of these studies had an enriched population which included only those with vascular risk factors which may limit the generalisability of findings. Secondly, there was a wide variation in the reporting of associations between coronary atherosclerosis and MACE. The wide confidence intervals around the measures of effect suggest that further study is needed into the associations between subclinical atherosclerosis and future MACE in stroke survivors.
Conclusion
Coronary atherosclerosis affects two in three ischaemic stroke survivors without a history of coronary artery disease. Moreover, one in three stroke survivors has evidence of obstructive (>50%) coronary stenosis and 7% have left main or triple vessel disease. The presence of asymptomatic obstructive coronary artery stenosis in stroke survivors is associated with an eightfold increase in the risk of future MACE. More work examining the role of routine coronary artery assessment after ischaemic stroke is warranted.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241231702 – Supplemental material for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241231702 for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis by Rahul G Muthalaly, Timothy B Abrahams, Nitesh Nerlekar, Adam J Nelson, Sean Tan, Jasmine Chan, Thanh Phan, Henry Ma and Stephen J Nicholls in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241231702 – Supplemental material for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis
Supplemental material, sj-docx-2-eso-10.1177_23969873241231702 for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis by Rahul G Muthalaly, Timothy B Abrahams, Nitesh Nerlekar, Adam J Nelson, Sean Tan, Jasmine Chan, Thanh Phan, Henry Ma and Stephen J Nicholls in European Stroke Journal
Supplemental Material
sj-tiff-3-eso-10.1177_23969873241231702 – Supplemental material for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis
Supplemental material, sj-tiff-3-eso-10.1177_23969873241231702 for Asymptomatic coronary artery disease in ischaemic stroke survivors: A systematic review and meta-analysis by Rahul G Muthalaly, Timothy B Abrahams, Nitesh Nerlekar, Adam J Nelson, Sean Tan, Jasmine Chan, Thanh Phan, Henry Ma and Stephen J Nicholls in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.J.N. has received research support from AstraZeneca, Amgen, Anthera, CSL Behring, Cerenis, Eli Lilly, Esperion, Resverlogix, Novartis, InfraReDx and Sanofi-Regeneron and is a consultant for Amgen, Akcea, AstraZeneca, Boehringer Ingelheim, CSL Behring, Eli Lilly, Esperion, Kowa, Merck, Takeda, Pfizer, Sanofi- Regeneron, CSL Sequiris, Cyclarity, Vaxxinity and Novo Nordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a PhD scholarship from the National Health and Medical Research Council and the National Heart Foundation.
Ethical approval
Ethics approval was not required as this is a meta-analysis.
Informed consent
Written informed consent was not required as this is a meta-analysis.
Guarantor
Rahul George Muthalaly
Contributorship
RGM, SJN, HM, TP and SN conceived the idea for the manuscript. RGM, TA and NN performed the literature search. AJN, JC, TP, HM, NN, ST and SJN all provided feedback and edits to the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
