Abstract
Introduction
Symptoms of occult brain cancer may mimic stroke. Misdiagnosis may lead to improper treatment and delayed diagnosis. We characterised strokes associated with occult primary brain cancer and determined risk that ischaemic and haemorrhagic strokes are associated with occult primary brain cancer.
Patients and methods
All patients with incident stroke in Denmark 2003–2015 were identified through the Danish Stroke Registry (n = 85,893) and matched 1:10 on age and sex to the Danish background population without a stroke history (n = 858,740). This cohort was linked to the Danish Cancer Registry and prevalence of occult primary brain cancer defined as the event of previously unknown primary brain cancer during a one-year follow-up was estimated. We used Cox regression models to study risk of occult primary brain cancer in comparison to the background population.
Results
Of 77,484 patients with ischaemic strokes, 39 (1 in 2000) were associated with primary brain cancer; of 8409 with haemorrhagic strokes, it was 126 (1 in 66). In the background cohort, 205 (1 in 4000) had occult primary brain cancer. The multivariate stroke risk factor analysis showed that patients with occult primary brain cancer differed significantly from those without occult primary brain cancer indicating they might have stroke mimics rather than true strokes.
Introduction
The first manifestation of previously unknown (occult) brain cancer may be indistinguishable from stroke.1–5 It may have serious consequences for the prognosis if relevant treatment is delayed because brain cancer is mistakenly believed to be stroke. Primary brain cancers (PBCs) are often fast-growing and volume doubling times as short as 1–2 months are not uncommon. 6 Earliest possible treatment is, therefore, crucial for the prognosis. Initiation of preventive and curative treatment with anticoagulants or fibrinolytics could cause serious bleeding complications in the event that a suspected stroke patient has an occult brain cancer.7,8
Little is known, however, about the extent of the problem in clinical practice, i.e. risk that stroke is associated with occult brain cancer. Studies are few and small and have almost casuistic character,2,9,10 and to our knowledge, there exists no data separately for ischaemic and haemorrhagic stroke.
Using Danish Stroke and Cancer Registries, the aim of this study was to characterise strokes with PBC and determine risk that stroke is associated with PBC in comparison to the risk of having occult PBC in the Danish background population without a history of stroke.
Methods
We studied the association between incident stroke and occult PBC by linking data from Danish registries. All Danish residents have a 10-digit personal identification number, enabling unambiguous identification and linkage of patients from national registers. 11 We identified all patients recorded with an incident stroke occurring in Denmark from 1 January 2003 to 12 December 2015 through the Danish Stroke Registry.12–14 We created a comparison cohort matched on age and sex and with no record of stroke at index date by linkage to the Danish Civil Registration System. 11 This cohort consisted of persons without a history of stroke at index date sampled 1:10 with replacement. Information on life-style and cardiovascular risk factors for the stroke population was from the Danish Stroke Registry.12–14 Linking to the Danish Cancer Registry,15,16 we identified all stroke patients and persons in the background population who became registered with previously unknown PBC (malignant brain neoplasm, ICD-10: C71) in the Danish Cancer Registry in the first year following the stroke/index date. These persons were defined as having occult PBC on the assumption that cancers diagnosed within that timeframe were also present, albeit occult, at the time of stroke/index date. Information on demographics and socio-economic status in both cohorts was from the National Patient Register 17 and Statistics Denmark. 18
The study protocol was approved by the board of the Danish Stroke Registry and the Danish Data Protection Agency (journal number 2012-41-0719). The need for obtaining written informed consent from the study subjects was waived as the study is based on administrative data available for research after the approval from the Danish Data Protection Agency.
The Danish Stroke Registry
The Danish Stroke Registry12–14 receives mandatory reports from all Danish hospitals on all patients hospitalised for acute stroke including information on age, sex, stroke severity, subtype (based on computed tomography (CT)/magnetic resonance (MR) scan) and cardiovascular profile. Stroke is defined according to the criteria of the World Health Organization 19 and stroke severity is measured by the Scandinavian Stroke Scale. 20 We included only incident hospital admissions for a first stroke (ischaemic or haemorrhagic). Transient ischaemic attacks, subarachnoid bleeding and patients for whom scanning were not performed (1.1%) were not included. Stroke diagnosis was based both on clinical history, symptoms and signs and on an initial (obligatory) CT/MR scan. In this study, infarcts with haemorrhagic transformation were considered as infarcts (ischaemic stroke). If brain cancer was identified on the initial CT/MR scan and the stroke-like symptoms were considered to be consequences of the brain tumour (i.e. stroke mimic), the patient would not be included in the Danish Stroke Register.
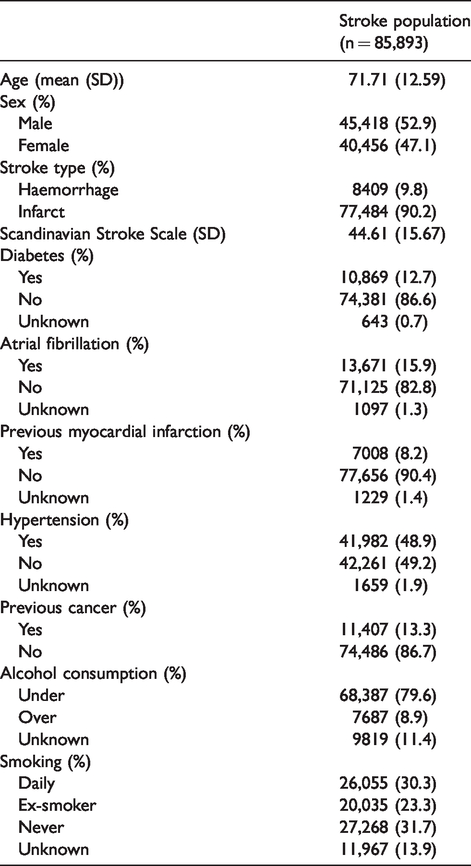
The cardiovascular profile includes information on alcohol consumption (≤14/21 and >14/21 drinks per week for women and men, respectively, corresponding to under/over the limit set by the National Board of Health), smoking, diabetes mellitus, atrial fibrillation (chronic or paroxysmal), arterial hypertension, previous myocardial infarction and previous cancer. Data on treatment such as antineoplastics, anticoagulants and fibrinolytics were not included in the data set. In March 2012, the recommended limit for alcohol intake was lowered to 7/14 drinks per week for women/men. The registry has high validity and covers now > 89% of all admissions for stroke in Denmark.13,14
Danish Cancer Registry
Information on patients with incident PBC (ICD-10: C71) was from the Danish Cancer Registry,15,16 which includes data on the topography, morphology and date of diagnosis of virtually all cancers diagnosed in Danish residents. Cancer diagnoses are recorded according to the International Classification of Diseases 10th revision.15,16 We included only PBC (malignant brain neoplasm ICD-10: C71). As this study focuses exclusively on previously unrecognised (occult) brain cancer, metastases were not included as the presence of an active primary tumour is often known prior to hospitalisation in these patients. Benign intracranial tumours including meningeomas were not included in the study.
Danish National Patient Register
The Danish National Patient Register contains information on all discharges from Danish hospitals. It is mandatory for the hospitals to report to the registry. 17 We used the registry to obtain information on comorbidities prior to the stroke and index date using the Charlson Comorbidity Index 21 divided into groups 0, 1, 2 or 3+ describing the number and severity of the comorbidities at index date. A score of zero indicates that no comorbidities were found.
Statistics Denmark
We obtained information on socio-economic factors on the entire cohort at an individual level from Statistics Denmark. 18 This included information on income, education, ethnicity and civil status. Income was defined as disposable income and was categorised into five groups based on the age- and sex-adjusted percentiles. We divided education into three categories: basic/high school (7–12 years of primary, secondary and grammar school), vocational (10–12 years, including vocational training) and higher education (≥13 years). Ethnicity was divided into ethnic Danes, immigrants from developed countries and immigrants from developing countries. Civil status was divided into married, unmarried, divorced and widowed.
Definition of events and time at risk
The event of occult PBC was defined when a person was registered with a previously unknown PBC (ICD-10: C71) within the first year following stroke/index date in the Danish Cancer Registry. Individuals were at risk until time of cancer diagnosis (event), death, emigration or until the end of the one-year follow-up, whichever came first.
Inclusion in the Danish Stroke Register was always preceded by a primary CT/MR scan. If this CT/MR scan identified a previously unknown PBC, the person would be registered in the Danish Cancer Registry as diagnosed with PBC at the same date as the incident stroke. In accordance with our definition of occult PBC, those patients would not be counted in the group of patients with stroke and occult PBC; only patients diagnosed with PBC at dates after the date of stroke were included.
Rates of detection of PBC within the first year after the stroke/index date were estimated and considered as proxies of prevalence as occult PBC detected within this time frame was assumed to be prevalent also at the time of stroke/index date.
Some patients/persons die before the first post stroke year/year after index date has expired so that follow-up time may vary between the two populations. For this reason, we present prevalence of occult PBC based both on the number of stroke patients/persons in the background population as well as the number of person-years of follow-up in the stroke patients/persons in the background population, thus also accounting for variations in follow-up time due to differences in death rate between the two populations.
Statistical methods
The characteristics of the stroke patients were compared to that of the background population and presented by means for continuous variables and proportions for counts. The stroke population was further described in relation to the cardiovascular risk factor profile, while these variables were not available for the background population.
We used Cox regression models to estimate the relative risk of occult PBC in stroke patients in comparison to the background population. The analyses were adjusted by age, sex, year of diagnosis, Charlson Comorbidity Index, attained education, income, civil status, ethnicity and previous cancer. Separately, for the stroke population, we undertook a within-cohort analysis to evaluate the association with cardiovascular factors and the risk of occult PBC. All risk estimates are presented as hazard ratios (HR) with 95% confidence intervals.
Results
In total, 85,893 incident stroke patients > 40 years of age were registered between 1 January 2003 and 12 December 2015 and included in the study. Of these, 77,484 (90.2%) patients had ischaemic stroke; 8409 (9.8%) patients had haemorrhagic stroke. In total, the background population consisted of 858,740 persons. Descriptive statistics on the stroke patients and the age- and sex-matched background population are presented in Tables 1 and 2.
Descriptive characteristics and cardiovascular risk factor profile of the stroke population.
Attained education, income, ethnicity, civil status, Charlson Comorbidity index and previous cancer in the stroke and the background population without history of stroke.
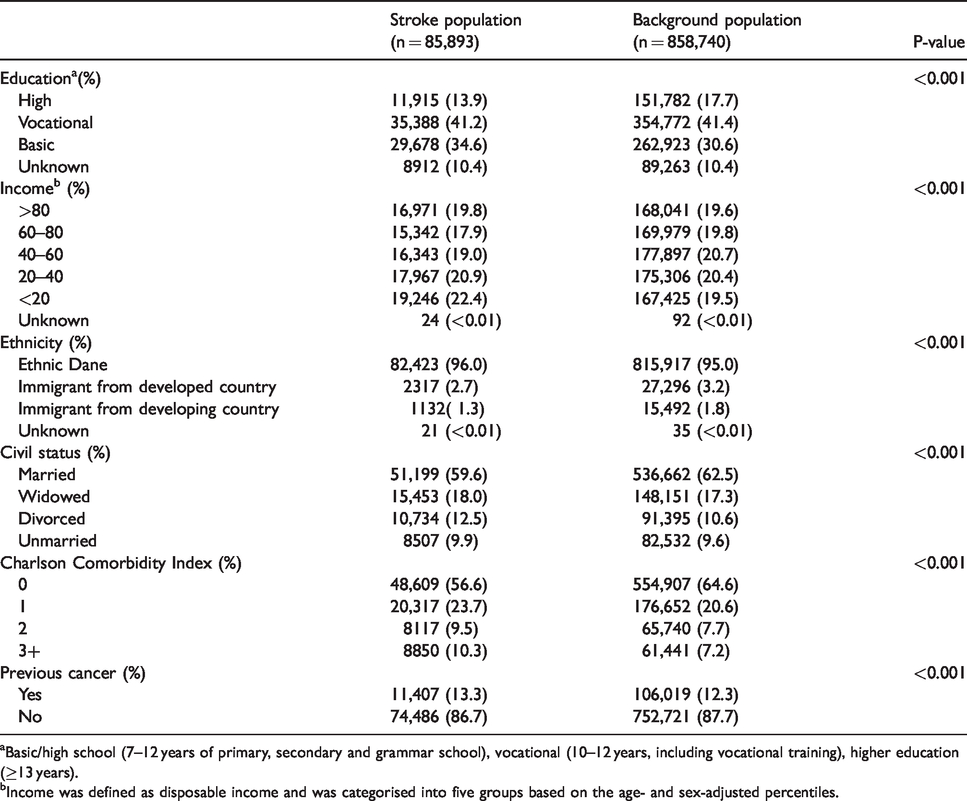
aBasic/high school (7–12 years of primary, secondary and grammar school), vocational (10–12 years, including vocational training), higher education (≥13 years).
bIncome was defined as disposable income and was categorised into five groups based on the age- and sex-adjusted percentiles.
Total follow-up time within the first year after stroke/index date was 73,117 person-years in the stroke population and 830,632 person-years in the background population.
Prevalence
Among the 77,484 patients with ischaemic stroke, 39 (1 in 2000) had occult PBC and among the 8409 patients with haemorrhagic stroke, it was 126 (1 in 66). Thus, strokes associated with occult PBC were mainly haemorrhagic (76%). In the sex- and age-matched background cohort without a history of stroke, 205 (1 in 4000) of 858,740 persons had occult PBC (Table 3).
Occult primary brain cancers in the stroke and the background population.
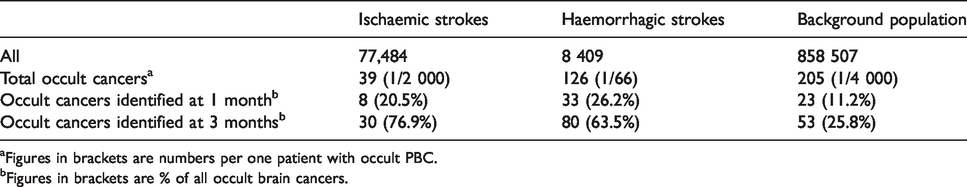
aFigures in brackets are numbers per one patient with occult PBC.
bFigures in brackets are % of all occult brain cancers.
If prevalence is calculated based on person-years of follow-up instead of numbers of patients followed within the first year, prevalence would be 1 in 1700 for ischaemic stroke and 1 in 56 for haemorrhagic stroke. For the background population, prevalence would not change ∼1 in 4000.
Risk
Adjusting for age, sex, income, length of education, marital status, ethnicity, co-morbidity and previous manifest cancer (Table 2), risk of occult PBC was increased; HR 2.33 (CI 1.65–3.29) for ischaemic stroke patients and HR 7.35 (CI 5.87–9.19) for haemorrhagic stroke patients when compared to the background population.
Stroke risk factor profile
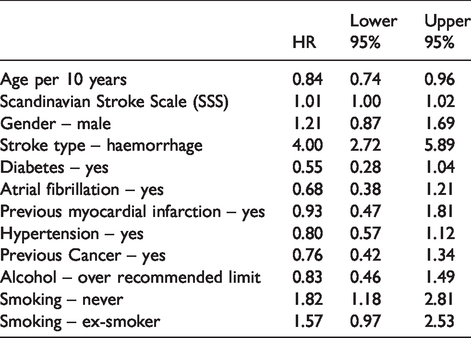
In the stroke population, we evaluated the risk factor profile associated with the risk of occult PBC. Adjusting for age, sex, stroke severity, type of stroke and the cardiovascular risk factor profile shown in Table 1, we found that patients with occult PBC (n = 165) differed in important aspects from a typical stroke population. They were significantly younger (HR 0.84 (per 10 years)), they had significantly more often intracerebral haematomas (HR 4.00) and they were significantly more often never-smokers (HR 1.82) than the stroke patients without occult PBC (n = 85,728). Moreover, they also tended to be less often diabetics (HR 0.55), and they tended to have less often atrial fibrillation (HR 0.68), previous myocardial infarction (HR 0.93), hypertension (HR 0.80) and previous cancer other than PBC (HR 0.76), all suggesting they were stroke mimics rather than true strokes (Table 4).
Risk of occult primary brain cancer (within stroke cohort analysis).
Time to identification of cancers
Table 3 shows the numbers of occult PBC identified at one and three months, respectively, after the stroke/index date. Identification rates did not differ significantly between the ischaemic and haemorrhagic stroke populations, but the identifications rates were significantly higher in the stroke population than in the background population; at three months, 76.9% and 63.5% in the ischaemic and haemorrhagic populations, respectively, vs. 25.9% in the background population (p < 0.01).
Discussion
For the patients included in the Danish Stroke Register, the risk that an ischaemic stroke was associated with PBC was very low (1 in 2000). Among patients with haemorrhagic stroke, risk was much higher (1 in 66). Prevalence would be slightly higher if person-years of follow-up were used for estimation, instead of number of patients followed within one year. However, since mortality in the stroke population is substantially greater than in the background population (for this study 11.6% vs. 1.3% within three months), prevalence in the stroke population would be overestimated relative to the background population, since most deaths in the stroke population have no relation to the relatively few PBC events. We therefore consider estimates based on the number of patients followed in the study period as the most valid.
Previous research
We are not aware of comparable population-based studies on the prevalence of occult brain cancer in stroke, nor of studies on the prevalence separately for ischaemic and haemorrhagic strokes. Regarding the risk of occult PBC, results from smaller studies varied. In a study of 2449 patients initially considered to have transient ischemic attacks, five (1 in 500) were subsequently diagnosed with PBC. 9 In 352 patients admitted in a stroke unit, all with CT-scan completed, two patients (1 in 175) were subsequently diagnosed with glioma, 2 and among 405 patients with suspected stroke in an emergency department, two (1 in 200) were discharged with a glioma. 10 These studies did not differentiate between ischaemic and haemorrhagic stroke. For all strokes in our study, ischaemic and haemorrhagic taken together, risk that a stroke was associated with PBC was 1 in 520.
Strokes vs. stroke mimics
The precondition for being registered in the Danish Stroke Register was to be diagnosed with stroke on the basis of both a clinical and an imaging diagnostic evaluation. However, our study suggests that at least a larger part of strokes associated with occult brain cancer might not be (as believed when included in the Danish Stroke Registry) true strokes but rather stroke mimics.4,22 According to the multivariate comparison of the 165 patients with occult brain cancer and the 85,728 patients without, those with occult PBC were younger, had more often intracerebral haematomas and they were more often non-smokers than the 85,728 patients without occult brain cancer. Moreover, they tended not to have diabetes, atrial fibrillation, myocardial infarction and hypertension. This is totally opposite to what characterises the traditional stroke population. So, the symptoms that led to hospitalisation for stroke could be more often expressions of occult brain tumour than of stroke. We cannot rule out that some strokes have occurred independently of an existing occult cancer (i.e. patients with atrial fibrillation or severe carotid stenosis, etc.), but they are probably in the minority given the considerations above. The tumours have most likely been hiding within the haemorrhages or hypodense tumours have been mistaken to represent infarcts.
The risk rates presented in this study should be seen in this light. Our multivariate analysis revealed a two- and seven-fold increased risk of occult brain cancer in patients with ischaemic or haemorrhagic stroke, respectively, when compared to occult PBC in the background population. This estimate would have been trustworthy if the stroke patients with occult brain cancer all had true strokes. But, as shown, that is not the case. For a majority of these patients, strokes appear not to be true strokes but rather stroke mimics. Thus, the estimate is misleading brought about by unintended misclassification of stroke mimics as being true strokes. Our study, therefore, cannot be taken to indicate higher risk of brain cancer in patients with stroke.
Prevalence of occult PBC in the background population was 1 in 4000 as compared to 1 in 2000 in the ischaemic stroke population and 1 in 66 in the haemorrhagic stroke population. As we have no reason to believe that patients with haemorrhagic stroke have higher risk for brain cancer than those with ischaemic stroke, the risk of occult brain cancer in patients with true stroke should be expected to be in the interval 1 in 2000–4000; in any case very low as in the background population.
Clinical implications
The very rare occurrence of occult brain cancer in patients with ischaemic stroke (1 of 2000) suggests that the first symptoms of brain tumours in the form of stroke mimics are very rarely misinterpreted as expressing ischaemic stroke. Therefore, the risk that a patient (considered having ischaemic stroke) may be exposed to a non-intended treatment-induced bleeding complication related to an overlooked occult brain cancer is extremely small.
On the other hand, it is not uncommon for patients believed to have haemorrhagic strokes that strokes are associated with occult brain cancer (1 of 66) and it is therefore important to have the possibility in mind especially when the haematomas are located atypically. Two-thirds of the occult cancers in patients with intracerebral haematomas were actually identified within three months after the stroke indicating awareness of the risk among Danish doctors. Nevertheless, there is still a need to emphasise that this risk exists since one-third of the occult cancers were not identified three months after the stroke. Given that PBCs are often fast-growing, 6 such a delay in diagnosis can cause a significant deterioration in prognosis.
Strengths and weaknesses
A major strength of this study was the large number of stroke patients and the population-based design, with low risk of information bias as data come from high-quality Danish registries with almost complete nationwide coverage. A weakness is that cardiovascular risk factors are not available for the background population. However, the increased prevalence of PBC in the age- and sex-adjusted stroke populations was still there after adjustment for co-morbidity, income, length of education, marital status, ethnicity and previous manifest cancer. In the current study, 11.6% of the stroke patients died within the first three month compared to 1.3% in the background cohort. Therefore, occult PBC in the stroke population may be underestimated since patients who die within a short time of the stroke diagnosis may not have been examined for occult cancer. It is a limitation that our dataset did not include more information on tumour sub-types and location in relation to infarcts and haematomas, interesting information from a clinical point of view. Likewise, due to small numbers of events in ischaemic stroke, it was not possible to determine if the predictors differ for ischaemic and haemorrhagic stroke. Furthermore, it is important to keep in mind that the ‘event’ (PBC within one year) will underestimate the actual number of people having a cancer because some will not be discovered and thus not being registered in the Danish Cancer Registry within the first year after stroke/index date.
It should be emphasised that numbers relate only to patients in whom PBC were not identified on primary diagnostic imaging. The proportion of PBC that clinically were mimicking stroke but were discovered through diagnostic imaging is larger than might be apparent from our study.
Summary
The risk of a patient with ischaemic stroke having an occult as yet unrecognised PBC is extremely small. The same should be expected for the risk of a potential cancer-associated bleeding complication when attempting preventative or curative treatment with antiplatelets, anticoagulants or fibrinolytics in patients believed to have stroke. Occult PBC occurs mainly and is not uncommon in patients with intracerebal haematomas, and the possibility of this should always be kept in mind in order not to risk delaying of relevant tumour treatment.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Jascha Foundation provided funding for this study (grant no. 4921). The Jascha Foundation had no influence on the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review or approval of the article and decision to submit the article for publication.
Ethical approval
The study protocol was approved by the board of the Danish Stroke Registry and the Danish Data Protection Agency (journal number 2012-41-0719).
Informed consent
The need for obtaining written informed consent from the study subjects was waived as the study is based on administrative data available for research after the approval from the Danish Data Protection Agency.
Guarantor
TSO.
Contributorship
Study concept and design: Andersen, Olsen. Acquisition, analysis or interpretation of data: All authors. Drafting of the article: Olsen. Critical revision of the article for important intellectual content: All authors. Statistical analysis: Andersen, Tybjerg.
